Abstract
Fullerenes are carbon molecules that are found in nature in various forms. They are composed of hexagonal and pentagonal rings that create closed structures. Almost 4 decades ago, fullerenes were identified in the form of C60 and C70, and following the award of the Nobel Prize in Chemistry for this discovery in 1996, many laboratories started working on their water-soluble derivatives that could be used in different industries, including pharmaceutical industries. One of the first fullerene forms that was the focus of different research groups was fullerenol, C60(OH) n (n = 2-44). Both in-vitro and in-vivo studies have shown that polyhydroxylate fullerene derivatives can potentially be used as either antioxidative agents or cytostatics (depending on their co-administration, forms, and concentration/dose) in biological systems. The current review aimed to present a critical view of the potential applications and limitations of fullerenols in oncology, as understood from the past 2 decades of research.
Introduction
Fullerenols (FRLs), C60(OH) n (n = 2-44), which are polyhydroxylated derivatives of the fullerene C60, have been extensively investigated over the past 2 decades, mostly due to their great potential as antioxidants. The first paper on FRLs was published in 1994, 1 and a few years later, Tsai et al 2 reported, for the first time, that FRLs are novel free-radical trappers that prevent hydrogen peroxide- and cumene hydroperoxide-elicited changes in rat hippocampus in vitro. This opened new opportunities for future investigations, both in vitro and in vivo, focusing on medical research. According to the Web of Science, 3 approximately 600 articles have been published on FRLs between 1994 and the beginning of 2023, of which only 32 were published in the 1990s. One of the major limitations for future investigations on FRLs is their extremely difficult synthesis, which results in a mixture of different FRLs containing mostly 18 to 24 hydroxyl groups. 4 Lai et al5,6 first reported the use of FRLs as antioxidants in vivo. Free radical scavenging by FRL was demonstrated in the ischemia-reperfusion-injured intestines of dogs 5 and in grafts after small bowel transplantation in dogs. 6 Many research groups have attempted different approaches in their laboratories to narrow down the potential applications of FRLs in medical treatments. In addition to their applicability as oxidative agents, several other medical applications have been investigated, such as anti-inflammatory, 7 antiproliferative, 8 or cytotoxic anticancer chemotherapeutic. 9 In addition, FRL showed scavenging activity toward superoxide anions (O2−) in the xanthine/xanthine oxidase system. The results obtained by Mirkov et al 10 confirmed the free radical-scavenging activity of FRL, which was the first evidence of the direct NO-quenching activity of hydroxylated C60 derivatives in different environments. Trajkovic et al 11 presented the radio-protective efficiency of FRLs in irradiated mice. The FRL C60(OH)24 was administered intraperitoneally, at doses of 10 and 100 mg/kg, 30 min before irradiation. The results showed that FRL, at an i.p. dose of 100 mg/kg, prolonged the LT50 of irradiated mice (7 and 8 Gy of X-rays). This was the first in-vivo study to be used by our research group as a basis for developing new potential applications of FRLs as organ-protectors against doxorubicin (DOX)-induced toxicity. Our first study on the influence of FRL C60(OH)24 on DOX-induced cardiotoxicity in rats was published in 2006, followed by 16 additional papers related to the idea of protecting (as antioxidants) different organs in vivo after the administration of DOX.12-27
In this review, we summarized the developments made over the past 2 decades, regarding the use of FRLs for medical purposes, with a focus on their applicability in oncology treatments, both as a co-treatment drug in chemotherapy and radiation owing to the antioxidant properties and as a cytostatic drug for major cancer treatment. The available data were reviewed and presented objectively, with all benefits and limitations related to future medical use, providing guidance and recommendations for future investigations.
Characteristics of FRL
Several articles were published in the late 1990s to determine the optimal process for the synthesis of FRLs and FRL-derived elastomers.28-33 Initially, the major concern was regarding 2 key processes. The first was related to synthesis, to obtain as pure as possible a form of the FRL C60(OH) n (eg, > 98% of C60(OH)24). Secondly, the synthesized FRL needed to be soluble enough in water to cause sufficient in-vivo effects in body fluids as well as at the cellular level following administration. Soon after, Wang and Cheng 34 introduced the factor of stability related to different FRL forms. Their investigation focused on FRLs with 6 and 12 hydroxyl groups, and the major stability conclusion was related to both the position of the hydroxyl groups in the C60 form as well as the synthesis method employed. Therefore, some FRLs were considered to possibly have a super-stable structure that could be used for different purposes, such as a starting point for polymer synthesis (so-called star-like polymers), whereas others could be extremely unstable and change both their structure and properties depending on the environment in which they are kept or used.
A key paper that supported the use of pure FRLs, without any additional chemical changes, was published by Mohan et al. 35 Dynamic light scattering of FRL solutions of C60(OH)18 revealed the formation of fullerene aggregates at high solute concentrations (up to 3.85 × 10−2 M), which would be ∼ 40 mg/mL. From the USP point of view, any molecule with solubility requiring 10 to 30 parts of solvent per part of the solute can be defined as soluble. 36 In this case, 1 part of the solvent was sufficient for 40 parts of the solute (FRL), which suggested the latter as a highly soluble form of hydroxylated fullerene. Photolysis of C60(OH)18 in aqueous solution yielded the immediate formation of a transient singlet excited state, which was related to the low stability profile of the highly soluble form. The energetically higher-lying singlet excited state transformed via intersystem crossing to the broadly absorbing triplet-excited state, resulting in a very dynamic solution that could have different in-vitro/in-vivo properties depending on the environment. The π-radical anion of FRL [C60(OH)18]−, generated by electron transfer from hydrated electrons and (CH3)2C(OH) radicals, was very active and played a key role in the oxidative capacity of the FRL. Based on electron transfer studies with suitable electron donor/acceptor substrates, the reduction potential of the C60(OH)18/[C60(OH)18] couple was estimated to be significant compared to that of other well-known antioxidants. 35
In the first 2 publications from our research group, FRL was dissolved in a sterilized and apyrogenic 0.9% NaCl water-based solution.12,13 However, within one hour of solution preparation, some particles settled down, creating a sediment that was difficult to resuspend. In order to increase their solubility, several agents were selected, which could be used for s.c., i.m., i.p., or even p.o. administration. In most of our subsequent studies on mice and smaller rats, Tween 80 was used to increase the solubility of FRL and obtain a stable solution. Normally, the solution is a combination of sterilized and apyrogenic 0.9% NaCl water-based medium with Tween 80 (80:20, w/w) containing 20 mg/mL FRL C60(OH)24. For larger rats, in which the volume of the solution administered could be higher, we used DMSO as a co-solvent. Sterilized and apyrogenic 0.9% NaCl water was mixed with DMSO (80:20, w/w) such that the final solution contained 10 mg/mL of FRL C60(OH)24. In both cases (with Tween and DMSO), FRL created a solution that was stable over a longer period and could be easily reconstituted by placing it on a vortex mixer for a few minutes.14-27 Using this approach, FRL was found to be highly soluble (still below the value published by Mohan et al 35 ), creating a dark brown solution that could be used for in-vivo administration. In one of our papers, 19 the differences across powder, saline water solution, and solution with co-solvent in water base were reported at the microscopic level. Additives significantly improved the stability and quality of water-soluble FRL, which are prerequisites for its in-vivo use. Further complications in vivo are described in the following sections.
Over the past decade, many research groups have investigated the properties of FRLs and the different options for improving their solubility in water.37-47 Although different FRLs showed great solubility in water and other aqueous solutions,37,39,41,43,46,47 using additives at room temperature 42 as well as at other temperature conditions, 39 laboratory results have not confirmed the potential behavior of FRLs under in-vivo conditions. After the in-vivo administration of clear FRL solution, small-to-medium particles were visibly observed, which could damage the treatment site (Figure 1). This phenomenon explains why FRLs are not yet in any regular human treatment, despite their potential antioxidant properties and organ-protection in oncotherapy.
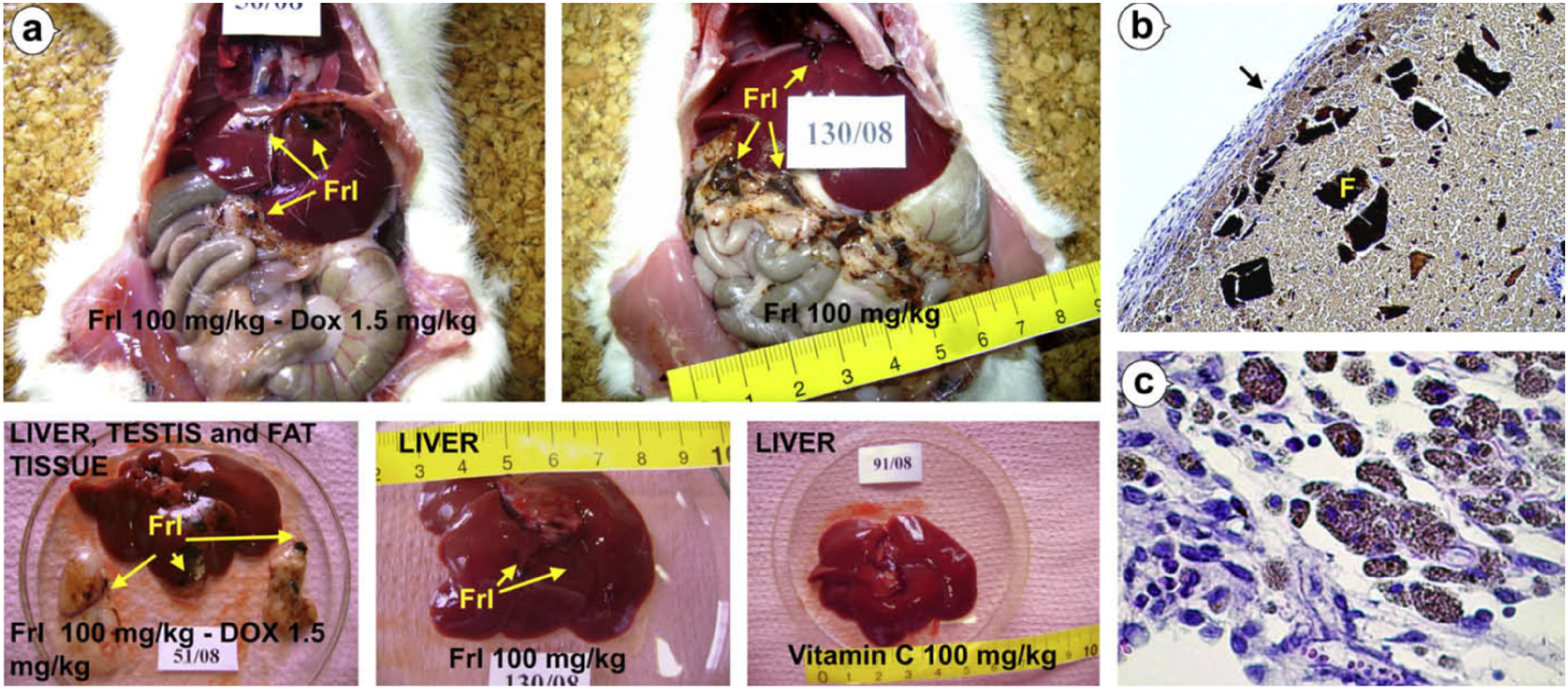
Macroscopic and microscopic changes in the abdominal cavity of rats treated with FRL. (a) Black and brown particles of FRL are seen in the abdomen, particularly on the surface of the liver and the mesenteric fat surrounding the liver. (b) Foreign body granuloma covered with thick peritoneum contains inflammation cells and fibroblasts (black arrow). Granulomatous tissue is predominantly composed of FRL composites in crystal form (F) and macrophages filled with FRL; H&E, magnification 100 ×. (c) A large number of macrophages with numerous brown granules of FRL in the cytoplasm; H&E, magnification 400 19 (with permission).
Potential Medical Use of FRL
From the very beginning of their discovery, different structures of C60 and C70 have been investigated for their use in medicine and nanotechnology. The first investigation led to the creation of FRLs containing different number of hydroxyl groups. An overview of the most significant investigations related to the potential medical use of FRLs is shown in Table 1.2,5,10,48-74 In the first decade of FRL research, only few studies focused on the biomedical applicability of FRLs.2,5,10,48-52,55 Most researchers have investigated the antioxidant properties of polyhydroxyl fullerene forms in acute toxicity using various in-vitro models. All the papers reviewed in this section are related to other medical uses of FRLs, except in oncology as organ-protectors or cytostatic. These 2 aspects are discussed in the following sections.
Research Investigation Related to Potential Medical Use of Fullerenol (FRL).

After the limited number of studies conducted in the first decade of FRL research, the next 2 decades saw more progress in new investigations of FRLs. Half of all the published studies are related to the antioxidative properties of FRL used in different systems, where free radical creation was detected and cell damage was observed.48-55,61-73 Every study confirmed the powerful antioxidant activity and cell protection in case of an oxidative imbalance. Therefore, future investigations should focus on diseases in which oxidative stress might be one of the key causes or consequences. The past 2 decades have confirmed the potential use of FRL in some CNS diseases,54,55,68,72 infections,57,65,73,74 ophthalmology, 63 dermatology,66,69 respiratory, 58 gynecology, 59 osteoporosis,60,71 alcoholism, 62 thrombosis, 64 and diabetes. 67
Several studies have focused on the central nervous system (CNS), where the antioxidative properties of FRL may have a positive influence on patients with depression, 54 Alzheimer’s disease,55,72 and brain stroke. 68
Depression is a common mental disease that is highly prevalent in all populations these days, especially after the major global crisis of the COVID-19 pandemic. Depression is related to neuroinflammation and the concomitant activation of microglia. Results of the presented study 54 indicated that FRL exerts anti-neuroinflammatory effects and indirectly protects against neurological diseases. Kong et al 54 evaluated the effects of FRL on lipopolysaccharide (LPS)-induced depression-like behavior in mice. FRL treatment showed an antidepressant effect, as indicated by the prevention of LPS-induced reduction in sucrose preference. The results indicated a shortening of the immobility duration in both the tail suspension test (additional weight fixed on the mouse tail in water, forcing it to use additional strength to swim) and the forced swim test (simple swimming test in water without any additional obstacle). Under different test conditions, different levels of “happiness” were observed in the mouse model after the tests were performed. According to the data presented in the study, 54 FRL exerts potential antidepressant effects, which may be due to its beneficial functions in reducing neuroinflammatory processes (as an antioxidant) and/or due to the promotion of neurogenesis in the hippocampus. 54
Following the same principle of antioxidative properties of FRL, 2 additional studies were published that tested the efficacy of FRL in Alzheimer's disease.55,72 Alzheimer's disease is a complex pathological status, and the complete mechanism of disease development is still unknown; therefore, it remains difficult to cure. From this point of view, most of the daily medications for Alzheimer's disease are focused on the symptoms and not the real cause. Huang et al 55 investigated the blockade of amyloid beta peptide-induced cytosolic free calcium by FRL in PC12 cells. Both amyloid beta and bradykinin elevated calcium ion levels in PC12 cells in a dose-dependent manner. FRL at a concentration range of 100 nM to 1 μM reduced the amyloid beta-induced [Ca2+] response in a dose-dependent manner. However, this did not influence subsequent bradykinin-mediated processes. The results collectively revealed that amyloid beta-stimulated [Ca2+] increase was due to Ca2+ influx from an extracellular source. Modification of the membrane lipid structure and permeability via free radicals generated by amyloid beta could be a major cause of Ca2+ influx, and the antioxidative potential of FRL might have a positive effect in this regard. In another study, 72 FRL C60(OH)30 samples were analyzed. Slobodina et al 72 used Drosophila melanogaster as a model to explore the applicability of FRL in Alzheimer's disease in relation to life expectancy, level of geotaxis, learning, memory, and levels of neurodegeneration and reactive oxygen species in the animal brain. The antioxidant properties of FRLs played a key role in this investigation, as seen in all previous studies. Unfortunately, both the papers provided hypotheses that were never investigated further, and accordingly, drawing any conclusion on their applicability was not possible. The same applied to a model of brain stroke. In a published work 68 using a rat model, a dose of 10 mg/kg FRL was administered 30 min before middle cerebral artery occlusion. Twenty-four hours after middle cerebral artery occlusion infarction, brain edema, malondialdehyde (MDA) and nitrate contents, and mRNA level of AQP-1 were determined. FRL was found to reduce ischemia-induced brain injuries and edema, possibly through the inhibition of oxidative damage and AQP-1 expression in case of brain stroke. 68 Considering the complexity of different CNS diseases, future investigations, including in-vivo studies, would be required.
Microbiological testing using FRL has been reported in very few papers.57,65,73,74 Only one publication covered the antibacterial effect of FRL itself, 57 whereas 2 focused on conjugates with better efficacy and fewer side effects.73,74 In one study, FRL was used as an adjuvant in the development of vaccines for HIV prevention. 65 Aoshima et al 57 evaluated the antimicrobial activities of FRL against 6 types of bacteria and 2 types of fungi. Three forms of FRL were used, namely C60(OH)12, C60(OH)36 × 8H2O, and C60(OH)44 × 8H2O. FRLs demonstrated good antimicrobial activity against Propionibacterium acnes, Staphylococcus epidermidis, Candida albicans, and Malassezia spp. In particular, C60(OH)44 exhibited a strong and wide-ranging antimicrobial activity, comparable to that of catechin. 57
A few years later, the first nanoadjuvant of FRL, which morphologically resembled a virus, was used as an HIV-1 DNA vaccine. 65 A dual function was proposed for the mechanism of action related to the nanoadjuvant, which acts as a plasmid DNA carrier and an activator of host immunity. However, this study, like many other clinical trials performed afterward, did not provide sufficient efficacy of the vaccine for approving its use in the prevention of HIV transmission in humans.
The FRL complex with Gd and its conjugate with gentamicin have shown great antimicrobial 73 and synergetic 74 effects. The Gd–FRL complex was found to inhibit bacterial and enzymatic bioluminescence at high concentrations (>2 × 10−1 g/L). The activation processes were accompanied by the consumption of reactive oxygen species (related to oxidative stress and the antioxidative properties of FRL). The bacteria effectively mitigated the increase in reactive oxygen species content induced by the Gd–FRL complex in aqueous solutions. 73 However, conjugation of FRL with gentamicin enabled the desired antibacterial properties without any unfavorable cytotoxic effect towards eukaryotic cells in vitro. 74 The obtained FRL may be presumed to be an effective carrier of gentamicin, and the developed conjugate may be applied locally at the site of skin injury. However, for validation of the same, proper clinical studies on animals and humans would be required, covering all aspects of antimicrobial usage and dermatology standards.
In addition to the previous study, 74 which was potentially used for dermatological purposes, 2 other studies have been reported in the same category, covering skin anti-aging therapy 66 and the inhibition of sebum as a therapeutic agent for acne. 69 In the present study, 66 the anti-aging effects of FRL on skin-derived stem cells were assessed in a mouse model. Mice were treated with the d-galactose-induced aging model, and after 2 weeks of acclimation, the FRL group was administered a subcutaneous injection of 100 mg/kg FRL. After 3 weeks of treatment, FRL effectively inhibited the retention rate of transplanted adipose-derived stem cells. At the same time, it increased the thickness of the dermal portion of the skin and the collagen ratio in mice. The results established that the anti-aging effect of FRL on the skin through the derived stem cells may be mediated in mice via the peroxisome proliferator-activated receptor-gamma/fork head box protein O1 (PPAR-gamma/FoxO1) signaling pathway. Unfortunately, no additional study was conducted using FRL as an anti-aging agent, and at the end of this review, we will discuss some information related to the commercialization of FRL-based cosmetic creams.
FRL C60(OH)44 is an in-vitro inhibitor of sebum production in hamster sebocytes and of Propionibacterium acnes lipase activity. Sebum production was significantly reduced by 1.5 μM FRL in cells that had been irradiated with 10 mJ/cm2 UVB. FRL was found to inhibit Propionibacterium acnes lipase, which plays a key role in sebum formation, particularly in the facial area. 69 This study was performed more than a few decades ago; however, no additional evidence has been published since then. Small clinical trials using well-known anti-acne products, such as herbal extract tonics or clindamycin solutions, might be a good step forward in the potential applicability of FRL in sebum formation.
Xu et al 58 aimed to explore the potential use of FRL in the treatment of respiratory diseases. Many studies have focused on the antioxidative effects of FRL; however, very little information is available on its pulmonary toxicological properties. This study 58 aimed to investigate the effects of FRLs on Sprague-Dawley rats after intratracheal instillation. FRLs [C60(OH) x , x = 22, 24] were administered intratracheally at 3 different concentrations (1, 5, or 10 mg/rat). After 3 consecutive days of treatment, the lungs were assessed using bronchoalveolar lavage fluid biomarkers and a pathological evaluation of the lung tissue. Exposure to 1 mg/rat did not induce adverse pulmonary toxicity, whereas the other 2 doses induced cell injury. As expected, FRLs produced dose-dependent responses, and hence, their use as an inhaled drug was not supported. What exactly happens in the lung tissue after the administration of FRL, at the macroscopic level, is not yet well described. As stated in this paper, 58 FRL was dissolved in saline, and based on all previous papers, this type of solution could create FRL particles after administration. Authors clearly showed inflammation, at the microscopic level, in the lung tissue; however, besides dose dependency, no other acceptable reason for inflammation was found. Macroscopic evaluation of the bronchial tissue to confirm FRL behavior after inhalation would be interesting. Several studies have been published in the past few years using in-vivo models (mostly rats) to confirm the effects of FRL nanoparticles on the resumption of rat oocyte meiosis, 59 inhibition of thrombosis and blood coagulation by inhibiting the activities of thrombin and Fxa, 64 erythrocyte deformability during ischemia-reperfusion injury of the lower extremities in diabetic rats, 67 and treatment of osteoporosis via 2 different mechanisms of action.60,71 However, the formulation used for FRL administration as well as its behavior afterwards under in-vivo conditions, remain unclear. The complete scientific society is still ignorant regarding the solubility of FRL, especially its brown particle formation after contact with body fluids. While the conclusion may be too radical, all the studies till date should be repeated with the proper improved form of FRL to confirm all the hypotheses and pave the way for the next level of investigation.
The last interesting study reviewed in this section was also conducted on a Wistar rat model with the assumption of potential hepatoprotective effects of FRL nanoparticles upon alcohol-induced oxidative stress. 62 The study investigated the antioxidant effects of orally administered FRL (dissolved in ethanol) on hepatocytes. Wistar rats were divided into 9 experimental groups that were exposed to an aqueous solution of ethanol alone (0.1%, 1.0%, and 10%), FRL dissolved in ethanol (0.1%, 1.0%, and 10%; aqueous solution), or controls (PBS; PBS + FRL; vitamin C in 10% ethanol). The hepatoprotective effects of FRL against alcohol-induced oxidative stress had previously been demonstrated in rat liver primary hepatocytes. At a concentration of 400 mM, FRL presented excellent free-radical scavenging ability. It effectively eliminated alcoholic oxidative damage and accordingly inhibited the alcohol-induced over expression of TNF-α. Since alcoholism is a chronic disease, a proper study using long-term exposure to FRL would be required to prove its therapeutic effectiveness, as well as the sustainability of the outcome.
Overall impression of the presented works provided a clear conclusion that most of the positive outcomes (organ-protection) were related to the antioxidative properties of FRLs. However, in dose-dependent studies with FRL, its cytostatic effect may be much stronger than its antioxidative ability. The following 2 sections will focus on both actions (antioxidant and cytostatic), considering the therapeutic area of oncology.
FRL as Potential Organ Protector in Oncology Treatments
After more than 10 years of investigation regarding the antioxidative properties of FRLs, the first indication of their use in oncology as organ-protectors was obtained in the early 2000s. The most significant research investigations related to the potential applicability of FRL as organ-protectors in oncology treatments are listed in Table 2.11-15,19,20,22,24,75-85 Our extended research team has been publishing several papers focusing on the applicability of FRLs as antioxidants to protect organs from the side effects of chemotherapy12-27 and radiotherapy.11,77,82 Most of the tests were performed with intraperitoneal injection of FRL,11-16,18-27 whereas only 2 were administered orally.17,83 In all in-vivo investigations, mice and rats were used as animal models11-21,23-27,77-82,84; however, only one study used a swine model. 83 Most publications have focused on cardio12,14,19,82-84 and hepato-protection15,17,19,22,80,82 by FRL, as an antioxidant in chemotherapy and radiotherapy, and very few investigations have covered the protection of kidney,17,20,82 lung,24,82 erythrocytes, 13 spleen, 82 and small intestine. 82
Research Investigation Related to Potential Organ-Protector of Fullerenol (FRL) in Oncology Treatments.

In our first paper, 12 cardiotoxicity of a single DOX dose was tested (8 mg/kg) in adult Wistar rats, and potential prevention was achieved using FRL at 3 different doses ∼ 30 min before the DOX administration. Since we did not have sufficient in-vivo evidence of FRL effect after i.p. administration, we used 50, 100, and 200 mg/kg as the experimental doses of FRL. ECG was performed within 2 and 7 days of treatment. In the first 48 h, we did not observe any significant difference, whereas a week after the treatment, the effect of DOX toxicity on the heart was visible. Subsequently, additional microscopic tests were performed. In addition to ECG confirmation of myocardial changes, vacuolized tissue and tissue with edema were confirmed in the DOX group while all groups pretreated with FRL and/or with FRL (100 mg/kg) alone had results comparable to control. 12
Although the paper did not mention that visible particles of FRL were formed in the peritoneum after the i.p. administration, it was noted in the lab diary as an area requiring further improvement and focus in future studies.
Following previous experience, we designed a study to measure the activity of antioxidative enzymes in erythrocytes after a single-dose administration of DOX in rats pretreated with FRL. 13 A similar approach was used in the same animal model as that in the previous study, with the higher single-dose DOX (10 mg/kg) being the only difference. Animals were sacrificed 2 and 14 days after the treatment. The results confirm the protective effect of FRL against DOX-induced toxicity in erythrocytes. There was no significant difference between any of the FRL and control groups. Two mechanisms of action were proposed for the protection of hemoglobin and erythrocytes by FRL. One mechanism suggested the binding between FRL and hemoglobin while the other suggested a complex form with iron, which diminished free radical reactions. However, the complete mechanism of action has not yet been revealed. In addition, attention was paid to the use of different doses of FRL and their behavior in the peritoneal cavity. From a biochemical point of view, a dose of 50 mg/kg was not sufficient to protect against DOX-induced toxicity, whereas a dose of 200 mg/kg FRL was too high and did not show any superiority in its protective role compared to the middle dose. In contrast, FRL was found to be potentially toxic at the highest dose (200 mg/kg), considering the increase in GST activity. 13
From this point onwards, all future investigations were conducted using an FRL dose of 100 mg/kg or lower.
In the next set of studies, besides focusing on the protective role of FRL in DOX-induced toxicity, we focused on improving the solubility of FRL in aqueous solution with additives that would not have any side effect or influence on FRL and/or DOX actions in vivo. Obtaining as clear a solution of FRL as possible (dark brown in color) was important to start with. Different surfactants that are well-known for pharmaceutical applications were tested. SDS and a few polysorbates (Tween) showed the best improvement of solubility, whereas small volumes of different alcohols (ethanol and methanol), propylene glycol, and polyethylene glycol did not demonstrate any better solubility of FRL. Finally, polysorbate type 80, the so-called Tween 80, with the best microscopic distribution of nanoparticles, was chosen. FRL was dissolved in a sterilized apyrogenic saline solution containing Tween 80 (80:20; w/w) at a concentration of 20 mg/mL. The preparation was performed under sterile conditions in a laminar flow cabin just before it was used. 14
Once a new form of FRL was ready for use, with the possibility that particles would not be formed after administration, the next improvement of the study protocol was related to creating conditions as similar as possible to in-vivo conditions so that it could mimic real anticancer therapy. Therefore, a chemically induced cancer approach was introduced. MNU was administered via i.p. injection to female Sprague-Dawley outbred rats, aged 50 and 113 days, to stimulate the formation of breast cancer cells. One week after the second MNU dose, the first palpable mammary gland tumor was observed. On the 160th day of age, the rats were treated with a single dose of DOX (8 mg/kg) or FRL (100 mg/kg with or without DOX; 30 min before DOX treatment). The FRL dose was determined in accordance with previous investigations.9-13
Two days after the treatment with DOX and FRL, the animals were sacrificed and several biological samples were used for subsequent investigation. Blood samples and organs were used for regular hematological and serum enzyme testing, with a focus on markers related to oxidative stress. In addition, selected organs were collected, and the tissue was used for microscopic evaluation. Using the same animal models and protocols, we performed several investigations related to the potential protective effects of FRL on the heart, 14 liver, 15 kidney, 20 and lung 24 tissues after DOX treatment.
The first results related to cardio-protection were promising after the analyses of serum enzyme activity and heart tissue oxidative status. Microscopic results of the heart tissue were confirmed with the biochemical findings. DOX significantly damaged the heart tissue of rats with malignant neoplasms, whereas FRL-pretreated rats showed significantly better results and were more comparable to the control group. A single dose of FRL did not show any significant effect on the oxidative stress or blood parameters. However, our expectation related to the injection formulation with Tween 80 did not show much improvement after i.p. administration. Small brown particles were still formed in the peritoneal area, causing cell damage at the spot of particle formation. 14 This issue remains, and besides the next step focusing on acute-to-chronic toxicity testing, we still have to find a better form of FRL solution for injection.
Previous statements about particle formation and its influence in the abdominal cavity were even more pronounced in our paper that focused on FRL as a hepatoprotective agent against DOX-induced toxicity. 15 In that study, macroscopic changes in the abdominal organs, as well as micro changes in hepatocytes, were demonstrated after the i.p. administration of FRL. Brown particles, 0.1 to 2 mm in size, were found on the surface of the intestine, stomach, spleen, liver, and pancreas. Microscopic evaluation revealed significant fat necrosis and acute inflammatory reactions in the liver tissue. Both groups were administered DOX alone or DOX in combination with FRL. The tested samples treated with DOX and FRL showed slightly stronger fat necrosis, which was probably related to the accumulated particles on the liver surface. In parallel with in-vivo testing, we used an in-vitro system with HepG2 and Caco-2 cell lines. Both tests confirmed that FRL is protective in environments with high levels of oxidative stress. However, depending on the FRL concentration and other conditions (alone or with an agent that influences the creation of free radicals), FRL has dual action. It can be either antioxidant or cytotoxic in cell necrosis and apoptosis. According to serum enzyme activities related to the liver, as well as biochemical tests performed on liver tissue for oxidative stress parameters, almost all results showed better liver status in the group that received FRL 30 min before DOX injection. However, 2 parameters differed significantly; MDA and GSH levels indicated cell damage and high oxidative stress in all 3 groups (DOX, DOX + FRL, and FRL). This finding could be related to the influence of the undissolved part of FRL, which remains on the surface of the organs. 15
Two other published studies with the same animal models and protocols, focusing on the kidneys 20 and lungs, 24 showed a clear benefit of FRL as a potential protector against nephrotoxicity and pulmonary toxicity. After i.p. administration, FRL particles were not formed in the lung and kidney tissue, and there was no cell damage or any related macro or micro changes. In both cases, MDA, GSH, GSSG, SOD, CAT, GR, GSH-Px, total antioxidant status (TAS), and LDH (tissue and serum) levels were measured to compare the oxidative status across the investigated groups. In addition, for the nephrotoxicity study, α-HBDH levels in both tissue and serum were investigated. All tested parameters were significantly different in the DOX group than in the other groups. FRL with DOX or FRL alone was comparable to the control healthy and cancer groups respectively, regardless of the biomarker used or the organ investigated.20,24
Petrovic et al 80 tested the hepatoprotective effects of FRL and DOX as nanocomposites, where FRL was used as the cage/carrier and DOX was used as the active component packed in the cage. The study was conducted using healthy adult male Wistar rats. A nanocomposite was formed in saline solution with the creation of particles with an average size of 24.3 nm and a surface charge of − 9 mV. The final doses administered were DOX 2 and 4 mg/kg, FRL 0.125 mg/kg (800 times less than in a previous study), DOX 2 mg/kg plus FRL 0.125 mg/kg, and DOX 4 mg/kg plus FRL 0.125 mg/kg.
Biochemical and microscopic tests were performed to measure the oxidative status and cell damage in liver tissue after i.p. administration of the investigated compounds. Finally, the nanocomplex form was confirmed to be less harmful to hepatocytes, possibly due to avoidance of FRL-induced damage initiated by brown particles formed in body fluids and tissues after i.p. administration.14,15 From an oxidative status perspective, the results showed some unusual trends. In some cases, 2 mg/kg of DOX single dose caused greater cell damage than a high dose of 4 mg/kg. However, we could not consider the report as complete since the authors did not complete the investigation after sacrificing the animals. Only liver and blood/serum samples were preserved and tested. All microscopic tests shown in this article 80 were related to the liver tissue. Formation of a DOX/FRL complex with less use of FRL provided a better profile related to liver cell damage and oxidative stress. However, the protective effect on heart tissue, owing to the 800 times lower FRL dose, was not reported. Even in the conclusion of this article, further research for the confirmation of cardio-protection was recommended. The significance of this study can be understood from the fact that most patients with cancer who were treated with DOX, eventually developed cardiovascular problems leading to death within a few years after therapy. 21
All previous studies were based on acute DOX-induced toxicity to obtain preliminary results on the potential application of FRL as an antioxidant. Healthy animals were used first, followed by an animal model of malignant neoplasm, which provided a more realistic in-vivo model. The next objective was to take a step further. Patients with cancer receive several cycles of chemotherapy until satisfactory results are achieved. Therefore, in our next study, we aimed to focus on chronic DOX-induced cardio- and hepato-toxicity in rats with colorectal tumors after a few DOX and FRL doses, like regular oncology treatment. Since the administration was performed thrice, with a week of rest in between, lower doses were used for both DOX and FRL. DOX was administered at a dose of 1.5 mg/kg/week (4.5 mg/kg in total), whereas FRL was administered at doses of 25, 50, and 100 mg/kg/week (75, 150, and 300 mg/kg, respectively, in total). We further aimed to compare the efficacy of FRL as an antioxidant using a model in which another well-known antioxidant was administered. Vitamin C was administered at a dose of 100 mg/kg/week (300 mg/kg in total). DOX was delivered via i.p. administration alone, with FRL, and with vitamin C. DMH was administered for 15 weeks to male Wistar rats at a dose of 20 mg/kg/week to induce colorectal cancer. In addition, an extensive study on formulation improvement was performed. As shown in our previous work 19 using microscopic testing, DMSO without FRL particles provided a clear solution. Initial crystal-like FRL had 90% of particles ∼ 100 μm in size and 50% above 500 μm. This was an enormous improvement over the injectable form, for which we expected excellent behavior in the peritoneal cavity without the formation of particles. Based on the macroscopic (visual organ check), microscopic (heart and liver tissue), pharmacokinetic (serum and tissue DOX and FRL concentrations), biochemical (blood tests, serum enzyme activities, and oxidative status in blood and tissue), and pathological results (colorectal tissue/tumor tests), very interesting outcomes were recorded. Overall, FRL was very effective as a hepato- and cardio-protector. Although the starting form of FRL was a clear solution, a dose of 100 mg/kg produced visible particles in the abdominal area. That was significantly lower or almost not visible with a dose of 25 mg/kg. Repetition of the treatment (in this case, 3 times) at a dose of 25 mg/kg/week (75 mg/kg in total) was sufficient to protect the organs during free radical formation in DOX toxicity. For the first time, the kinetics of FRL elimination from the body was confirmed to be very slow. DOX concentration dropped almost 4 times between 2 and 4 h after administration, and completely disappeared within a week while FRL was eliminated by < 10% within a week. Since cell recovery and oxidative balance take time after a toxic event, the slow pharmacokinetics of FRL are indeed beneficial. Finally, an important finding was the confirmation of the superiority of FRL over vitamin C as an antioxidant in DOX-induced toxicity. 19 This study was complex yet complete, with the conclusion that ex-vivo tests should be performed on the human heart and in-vivo tests should be performed on swine models using intraperitoneal and oral administration. For ex-vivo human heart testing, getting permission from the ethics committee is difficult. Therefore, our group performed an investigation in a swine model and obtained interesting outcomes (paper under preparation for publication).
Using previous findings, the present study 84 aimed to investigate cardiac behavior using ECG results, microscopic tests, and oxidative stress parameters of the heart tissue. We focused on the combination of DOX and DOX/FRL, where FRL was used at a dose of 25 mg/kg/week (for 3 weeks). Therefore, based on a previous protocol, 3 groups were investigated from a cardiological point of view, namely the control cancer group, the DOX group (1.5 mg/kg/week for 3 weeks), and DOX/FRL (1.5 mg/kg/week + 25 mg/kg/week for 3 weeks). Several tests were conducted; microscopic evaluation of the heart tissue was followed by oxidative status tests (LDH, MDA, SOD, GSH/GSSG, and CAT) and ECG measurements (heart rate and heart rate variability parameters in time and frequency domains). 84 DOX treatment in rats with colorectal tumors induced HRV changes that were compatible with sympathetic overactivity and reduced vagal tone in the heart over time. Half-an-hour pretreatment with a 25 mg/kg dose of FRL completely prevented HRV alterations induced by DOX, decreased oxidative stress, and normalized myocardial histopathological score. 84
As mentioned previously, only 2 papers have been published till date on the orally administered FRL.17,83 The first one 17 was related to acute DOX-induced toxicity induced by a single dose of 8 mg/kg. FRL was used as an aqueous solution mixed with DMSO (80:20; w/w). The final tested FRL concentrations were 10, 14.4, and 21.2 mg/kg. Male Wistar rats were used as the animal model, and FRL was administered orally daily (for 3 days) at a dose of 10 mg/kg, followed by an additional administration of DOX 6 h after the last FRL dose. All other studies were performed 30 min before DOX administration. Oral administration and absorption require time to reach the maximum concentration in serum, which could be effective as an antioxidant and organ-protector. The investigation team performed AMF imaging to confirm the solubility of FRL. The largest particle size in the solution was < 30 nm. Considering the oral administration and solution form, it was not surprising that FRL protective effects were demonstrated in the oxidative stress tests. However, the investigators chose to use 2 organs (the liver and kidney), but not the heart tissue. Therefore, the major concern regarding DOX toxicity-related cardiotoxicity was not covered in this publication. This study was, therefore, insufficient to draw final conclusions related to the complete organ-protective profile of FRL after oral administration 6 h prior to a single dose of DOX. 17
The second study used the same approach as the previous one with the same protocol. However, this study was performed for the first time in a swine model, using pigs of the F1 generation bred from Swedish Landrace and Large Yorkshire parents. 83 DOX (10 mg/kg) was administered intraperitoneally. FRL was prepared as an aqueous solution and administered 30 min before DOX in the case of i.p. administration and 6 h before DOX in the case of oral administration. In both cases, the dose was 18 mg/kg. Two weeks after the treatment, the animals were sacrificed, and microscopic ultrastructural analysis was performed. The old hypothesis about cardio-protection after intraperitoneal administration was partially confirmed, although oral FRL administration was found to not yield any positive results. Mitotic figures were observed in the group that received FRL for 3 days, followed by DOX treatment 6 h after the last FRL dose. 83 Therefore, additional investigations using swine models and oral administration, including blood tests, animal behavior, complete key organ macroscopic evaluations, and biochemical parameters related to oxidative stress, must be performed to obtain a complete picture.
The only work published was on the potential applicability of FRL as an organ-protector in radiotherapy by comparing it with the well-known drug amifostine. 82 Jacevic et al 82 used male Wistar rats, whole-body irradiated with 8 MeV X-rays at doses of 7 and 8 Gy using a linear accelerator. Both FRL (100 mg/kg) and amifostine (300 mg/kg) were administered intraperitoneally 30 min before irradiation. The comparative radioprotective efficacy was evaluated using an absolute lethal dose of X-rays (8 Gy) and rat survival for 30 days after exposure. The team further used several organs (lungs, heart, liver, kidneys, small intestine, and spleen) for histopathological and semiquantitative analyses. Amifostine showed a dominant radioprotective profile in the heart, liver, and kidneys, whereas FRL was more prominent in the small intestine, spleen, and lungs. It would be important to reiterate that, most probably in this study, the researchers faced issues with particle formation in the abdominal cavity, which influenced the results for some of the organs. Although not so clear in this study, the particles from an aqueous solution of FRL indeed behaved the same way as in other similar rat models demonstrated previously.
The most prominent use of FRL as an organ-protector is in DOX-induced toxicity. Although it has been more than 50 years since DOX was discovered as an antibiotic with anticancer properties, even today, despite knowing all its side effects and primary cardiotoxicity, it is regularly used for chemotherapy with other cytostatics. 21 Not only is it still in use, but many reviews and original scientific papers are still being published on its usability, safety, different formulation improvements, and targeted therapies.86-108 Initially, DOX was confirmed to be effective for a few types of tumors only, although it is now used in many treatments to stop the growth of cancer cells in bones,93,94 liver,97,102 prostate,99,101 cervical tissue, 100 breasts,87,89,95,96,98,108 pancreas,86,103 lung,99,104 ovarium, 105 colon,106,107 and the brain. 92
Considering all the scientific evidence presented in this section, future research should focus on formulation improvement and the safety profile of the FRLs used as antioxidants to protect organs from toxicity in DOX-related treatments.
FRL as Potential Cytostatic
In addition to the potential medical use of FRL as well as its organ-protective function in oncology, especially in relation to DOX-induced toxicity, FRL itself might also be used as a cytostatic agent in cancer treatment. Unfortunately, very few studies have been published on its potential use in oncology9,15,19,109-120 (Table 3). Only 3 studies are currently available with in-vivo data related to its cytostatic properties.19,110,115 One investigation was performed in mice 110 and the other 2 were in the same rat model.19,115 Qin et al 110 performed tests using an in-vitro model to evaluate the effects of FRL on different cell lines. Cancer cell lines (MCF-10A, MCF-7, and MDA-MB-231) were used in the investigation. FRL at a concentration of 12.5, 25, 50, 100, or 200 μg/mL was added to the cells for 24, 48, and 72 h. Treatment with FRL (at all concentrations) did not increase the apoptosis rate of MCF-7 or MDA-MB-231 cells in contrast to control media. Following these results, the same team performed in-vivo testing to investigate the potential blockage of metastasis from breast cancer cells. 110 Five-week-old female nude mice were randomly divided into 3 groups (control, FRL, and blank groups). MDA-MB-231-luc cells were injected into the mice via the tail vein to develop a lung metastasis model of breast cancer in the control and FRL groups. One week after the injection, the mice were injected with FRL solution (35 mg/kg). After 2 weeks of treatment with FRL, the development of lung metastasis was investigated. Results indicated that FRL limited the migration of breast cancer cells and decreased metastasis to the lungs. This observation was likely attributable to the ability of FRL to disturb the dynamic actin cytoskeleton reorganization, resulting in reduced filopodia formation and altered intracellular integrin distribution. Consequently, lung metastasis of breast cancer through the vasculature was suppressed by FRL. 110
Research Investigation Related to Potential Cytostatic Effect of Fullerenol (FRL).
In 2 of our team's publications,19,115 a chemically induced colorectal cancer rat model was used to investigate the potential synergetic use of DOX and FRL in combination, or FRL alone, to block further cancer cell growth and tumor metastasis. Colorectal carcinogenesis was investigated in a dimethylhydrazine-induced rat model. Our results demonstrated that 100 mg/kg FRL effectively inhibited the formation of dysplastic aberrant crypt foci, which were observed as early histopathological lesions in the pathogenesis of colorectal cancer. 115 When the results were compared with those of the DOX-alone group or DOX plus FRL group (100 mg/kg), the best outcome was the reduction of aberrant crypt foci in the FRL group, followed by that in DOX alone for adenomas, and DOX plus FRL for carcinoma. Since this was the first study to investigate the effects of FRL on colon carcinogenesis in vivo, further studies would be required to evaluate its protective effect in other phases of carcinogenesis and to investigate the mechanism of its potential anticarcinogenic activity.
All other studies investigating the cytostatic effects or cell apoptosis involving FRLs have been conducted on blood cell samples (erythrocytes)113,116 or specific cancer cell lines.9,15,109-112,117-120 In most cases, FRL was proven to be strongly cytostatic and to induce cell apoptosis. However, programmed cell death was concentration-dependent for FRLs.
Tishevskaya et al 113 conducted a study using different concentrations of the FRL C60(OH)24 (0.1-100 μg/mL) to measure its effect on erythropoiesis in the culture of erythroblastic islets of the bone marrow. FRL at concentrations below 10 μg/mL did not show any influence on the cells, whereas in the range of 10 to 100 μg/mL, it had negative effects on the development of erythroid tissue. FRL at higher concentrations (100 μg/mL) obstructed the proliferation of erythroid cells, causing a delay in erythroblast maturation.
Following a principle like that in DOX–FRL combination treatment for the prevention of toxicity, increase of cytostatic effects, and protection of healthy tissues, Bahuguna et al 116 combined FRL with methotrexate. They reported the synthesis of FRL as a nanocarrier in conjugation with methotrexate to create a serum-stable, intracellularly hydrolyzable ester bond. The established FRL–methotrexate conjugate offered weaker protein binding and substantially higher drug loading, with pH-dependent release at both systemic pH and at cancer cell pH. Conjugate-treated cells showed a significant reduction in IC50 value compared to the cells treated with methotrexate only. FRL in combination with methotrexate demonstrated greater drug availability, both systemically and at the cellular level, with improved targeted cancer cell cytotoxicity and significant compatibility with erythrocytes.
Other studies included in-vitro testing in different cell lines, such as ovarian (L929 112 ), liver (HepG2,15,120 SMMC7721, 117 and BEL-7402 120 ), intestinal (Caco-2 114 ), lung (LLC1 9 ), breast (MDA-MB-231 9 ), melanoma (B16-F10 9 ), cervical (HeLa 109 ), kidney (LLC-PK1 118 ), and gastric (GES-1 111 ). In all in-vitro tests, FRL, alone15,110-115,117-119 or in conjugation with other medication,9,19,116,120 showed significant cytostatic effects on cancer cells and a safety profile compared to healthy cells. Considering the discussion in the previous section related to the potential use of FRL in oncology, together with DOX as an organ-protector and/or synergistic cytostatic, 2 in-vitro studies related to the DOX–FRL complex would be interesting to review in greater details.9,120
Chaudhuri et al 9 was one of the first to discuss, in 2009, the potential use of FRL as a cytostatic agent (in vitro), and reported that the challenge of particle formation of FRL depends on the media and methods used. Our first research was published with similar findings (in vitro) and observations (in vivo).14,15,19 As described in previous sections, depending on the environment and pH, FRL particles create larger forms and potentially damage healthy tissues (Figure 1). PEGylated Ful − Dox conjugate was synthesized in a study by Chaudhuri et al. 9 They used FRL as the starting material with particles smaller than 5 nm. Following PEGylation and conjugation of DOX, FRL was observed to form monodisperse aggregates with a size at least 10 times larger than the starting particle size (50-80 nm). However, these forms did not disintegrate into smaller individual FRLs. Although most of the observed particles were < 100 nm in size, clearance via the kidneys was limited. In the past, nanoparticles of FRL, < 5 nm in size, were reported (from a pharmacokinetic point of view) to be cleared by the kidney, whereas larger PEGylated nanoparticles preferentially localized in tumors through the leaky tumor neovasculature, owing to their enhanced permeability and retention effect. Unfortunately, this challenge was not investigated further in this publication, and no other research work has been published, till date, exploring this part of the challenge related to particle formation and the specific reaction, clearance, effects, and safety profile of FRLs. For the first time, in the same period (2008/2009), our work15,19 and other group's results 9 demonstrated the potential application of FRLs in cancer chemotherapy. First, the potential of increased drug loading—in this case, DOX with FRL—can facilitate the delivery of higher concentrations of active agents to the tumor. Additionally, the prolonged release of conjugates allows a longer safe exposure of tumor cells to the active agent (DOX). Furthermore, the hydroxyl groups of FRLs have a key effect on increasing hydrophilicity, which can be useful for cytotoxic agents with low solubility in water. To prove the usability of FRL with a cytostatic agent other than DOX, Chaudhuri et al 9 used cisplatin in their study in combination with FRL. The results were comparable to those for the DOX–FRL complex. One of the conclusions was that the FRL–DOX complex and FRL itself did not show equal or strong apoptosis in any of the investigated cancer cell lines. A very strong and significant effect was demonstrated in LLC1 lung and B16-F10 melanoma cancer cell lines and in the MDA-MB-231 breast cancer cell line.
In another study, 120 an upgraded version of the DOX–FRL conjugate was designed with the addition of folic acid (FA). The FA–grafted FRL–DOX conjugate was used to improve the selectivity and activity of DOX in cancer cells with reduced toxicity. FRL–DOX and FA–FRL–DOX conjugates were compared in BEL-7402 and HepG2 cell lines of human hepatocellular carcinoma. Both complexes exhibited pH sensitivity when the conjugates had a mean diameter of < 200 nm and low polydispersity. Cellular uptake studies indicated that FRL conjugation increased the intracellular accumulation of DOX in both human hepatocellular carcinoma cell lines and immortalized normal human hepatocytes (L02). The conjugation of FA with FRL and DOX protected normal cells in an FRL-dependent manner and improved their cytotoxicity against cancer cells. The authors concluded that the newly prepared FA–FRL–DOX conjugate optimized the safety and efficacy of DOX. However, this study cannot be considered complete. Knowing that FA improved FRL–DOX by targeting specific cell–drug reactions and with a better profile of normal cells, they did not include additional testing of FRL alone as a potential cytostatic agent. Based on the current findings, the mechanism of interaction using triple conjugation in vitro still remains unclear. FRL might be protective and could decrease toxicity in synergy with FA, or else, it might selectively show a combined cytostatic effect with DOX while FA acts as a protector. Without these additional scientific data, it would be difficult to reach a conclusion regarding the interesting hypothesis.
Although this review covered all findings regarding the use of FRL in oncology as a protector (antioxidant) or cytostatics (cytotoxicity), which have been published in the past 2 decades, all investigations seemed to just study the topic superficially, and there still remain many open questions yet to be answered. More in-vivo investigations regarding the potential use of FRL would be required in the future. More in-vivo models (mice, rats, and pigs) would be required for studying real-time chemically induced tumors15,19 to confirm the usability of FRL as a cytostatic agent. Both ex-vivo and in-vivo models with the proper formulation of FRL or DOX–FRL conjugates, which would not create large particles in contact with different tissues, would provide a better picture of the application of FRL in the organ-protective role of FRL in chemotherapy.
Although FRLs have been extensively researched for more than 2 decades, there are only 2 medical uses of them till date, and none is connected to oncology as such. Owing to the limited investigations, without proper clinical trials, FRL is used in cosmetics in the form of creams as an antiaging product with strong antioxidant properties.121-124 The other officially registered use of fullerenes for prevention is related to special glasses in ophthalmology. 125 The antioxidant and protective uses of FRLs should be validated in chemotherapy as well as in potential cytostatic treatments.
Conclusion and Outlook
Based on all the previous in-vitro and in-vivo tests, depending on the form, the method of administration, and the concentration of FRL, the latter can be regarded as a very strong antioxidant as well as a very strong cytotoxic agent. Therefore, extended clinical trials would be required to confirm the efficacy of cosmetic products on the skin surface, as well as the safety of FRL use in other applications, since it is partially absorbed via the skin into the systemic circulation.
The only 2 officially registered applications of fullerenes for prevention are related to cosmetology121-124 and ophthalmology. 125 C60 exhibits extraordinary quantum properties, including a unique rotational twist rate of 18 billion times/s. It efficiently interacts with the incident light, if incorporated in glass, which then, according to the seller “ultimately restores biostructures to a natural, healthy state at the quantum level.” 125 From a physical characteristics point of view, all of this is relevant; however, the real influence on the human brain, body, and overall optimal health is still difficult to confirm.
Unfortunately, for other medical uses, including oncological treatments, despite many years of research and investigation, different forms of FRLs have been far from massive global inclusion in any therapy. For patients with cancer, the most effective and useful characteristic of FRLs is the strong antioxidative property of the molecule and, in some cases, the cytostatic properties. In combination with DOX, which is still used in many chemotherapy protocols despite all side effects and toxicity to healthy tissues, FRLs can be used to prevent tumor growth and metastasis in vivo.
Overall, FRL has great potential for medical use; however, the properties of FRL and its behavior under in-vivo conditions require innovation and extended in-vitro and in-vivo trials with a focus on formulation development, regardless of whether it is used as an injection or oral dosage.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
