Abstract
Radiation therapy (RT) has been the standard of care for treating a multitude of cancer types. However, ionizing radiation has adverse short and long-term side effects which have resulted in treatment complications for decades. Thus, advances in enhancing the effects of RT have been the primary focus of research in radiation oncology. To avoid the usage of high radiation doses, treatment modalities such as high-intensity focused ultrasound can be implemented to reduce the radiation doses required to destroy cancer cells. In the past few years, the use of focused ultrasound (FUS) has demonstrated immense success in a number of applications as it capitalizes on spatial specificity. It allows ultrasound energy to be delivered to a targeted focal area without harming the surrounding tissue. FUS combined with RT has specifically demonstrated experimental evidence in its application resulting in enhanced cell death and tumor cure. Ultrasound-stimulated microbubbles have recently proved to be a novel way of enhancing RT as a radioenhancing agent on its own, or as a delivery vector for radiosensitizing agents such as oxygen. In this mini-review article, we discuss the bio-effects of FUS and RT in various preclinical models and highlight the applicability of this combined therapy in clinical settings.
Keywords
Introduction
Radiation therapy (RT) is a standard of care treatment for many different cancer types. Cells exposed to ionizing radiation experience increased levels of oxidative stress and DNA damage which led to cell death if repair mechanisms fail to ameliorate injury. 1 Currently, large doses of RT (> 60 Gy) are often delivered over the course of several weeks in smaller fractions (1.8-2 Gy). This enables the accumulated curative dose to be delivered to target tumor sites while reducing the radiotoxic effects of dose delivery to normal tissue.2,5 Current RT delivery methods are image-guided using a variety of modalities including ultrasound, computed tomography (CT), and magnetic resonance imaging (MRI) to ensure precise targeting of the tumor region.6,9 Technological advancements have allowed for the development of integrated imaging systems such as the MRI-guided linear accelerator (MR-LINAC).7,12 Despite these advancements in RT, adverse side effects and recurrences continue to plague the field of radiation oncology.13,16 For this reason, many studies look for solutions to boost the effectiveness of radiation while maintaining or reducing the treatment doses.17,18 Recently, many studies have looked at utilizing focused ultrasound (FUS) to enhance RT treatments through a multitude of potential applications.
Focused Ultrasound
Studies as early as 1942 observed the ablative effects of FUS. Lynn et al 19 observed enhanced thermal effects at the focus of concave single-element transducers in paraffin blocks and beef liver tissue, generating lesions within the test samples. Further investigations built upon by the Fry and Fry 20 lead to the early use of FUS to treat neurological disorders in humans. Since then technological development of FUS has resulted in the successful application of this modality for clinical treatments (Figure 1A and B). Most prominently, high-intensity focused ultrasound showed great efficacy in generating lesions within tumor masses and enhancing patient response.21,31 The nonionizing nature of ultrasound provides minimal risk to organs outside of the focal region, making it a safer option than conventional RT. It has successfully been used to treat a wide variety of tumor types including prostate, liver, uterine fibroids, pancreatic, and other solid tumors accessible by FUS.32,40
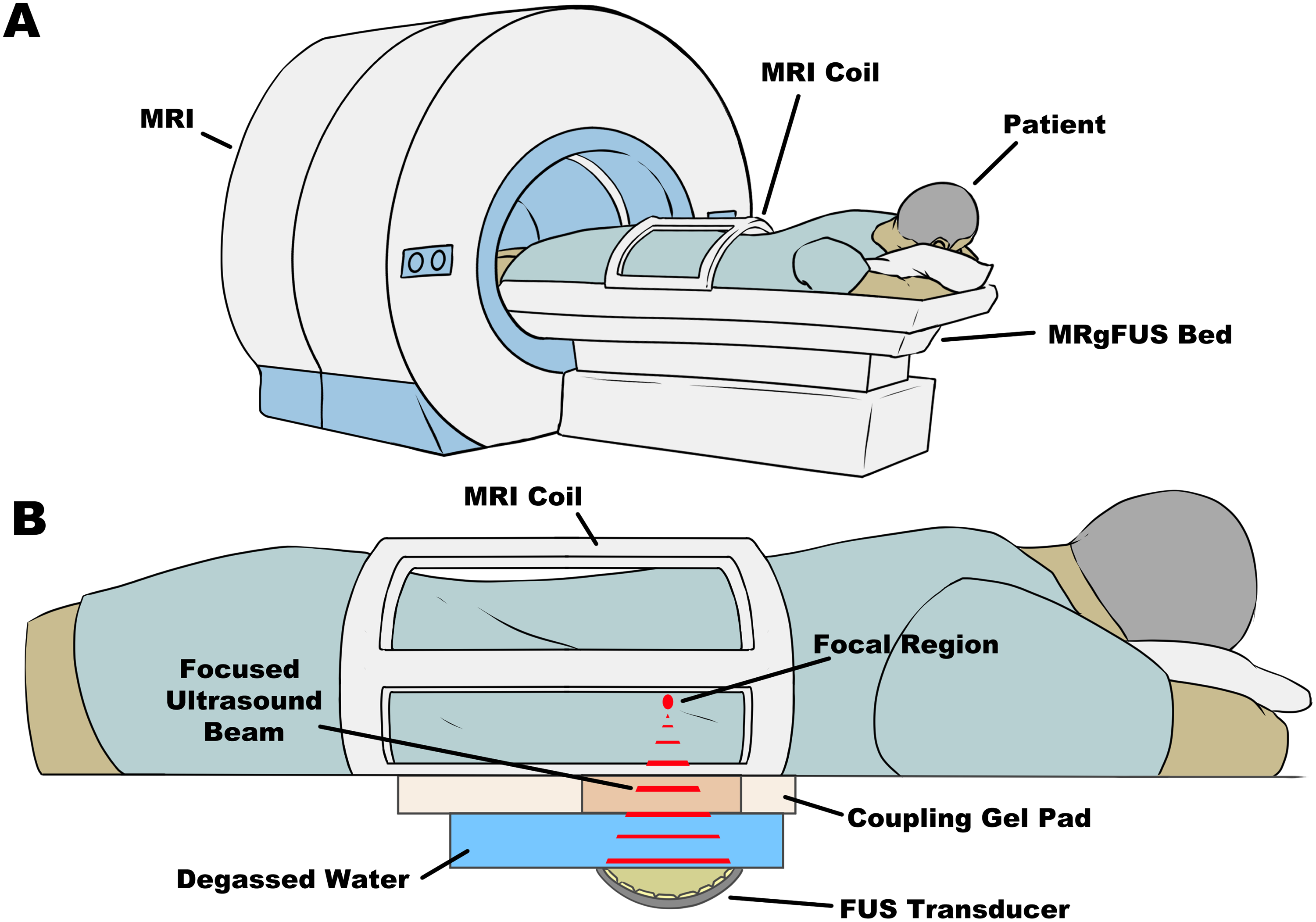
(A) Physical setup of patients on the MRgFUS system with the FUS system integrated into the bed. Patients may lay on the bed as per the required orientation with the target region of interest positioned over top of the FUS transducer. (B) The FUS system is integrated into the MRI bed. The concave transducer is submerged in degassed, deionized water with a coupling gel pad to allow for the acoustic beams to traverse into the patient with minimal energy loss. The multielement transducer is capable of focusing ultrasound waves to a targeted focal region which can generate heat and induce physical damage to the targeted tissue. All of this is done while the patient is inside the MRI after thorough planning of the treatment.
Because of the broad application of FUS, many studies have investigated its implementation in, but not limited to, the opening of the blood–brain barrier,41,44 neuromodulation,45,47 enhancing chemotherapy drug delivery,48,50 immunomodulation,51,54 and enhancing RT response.55,58
Focused Ultrasound and Radiation Therapy
The combination of FUS alongside RT has only seen preliminary studies indicating potential synergy between the 2 different modalities.59,64 An early study conducted by Jernberg et al 59 looked at the effects of FUS (1.1 MHz) and RT (2 Gy) in the Chinese hamster cell line V79-379A in vitro and observed reduced cell survival in the combination treatment compared to FUS or RT alone. It was postulated by Borasi et al61,62 that high-power FUS can complement RT in treating tumors by causing ablations and hyperthermia in the typically avascular and hypoxic core regions of the tumor where RT is potentially less effective, while RT targets the invasive margins of the tumor, a region often missed by FUS treatments. This concept was further explored by Borasi et al 63 as a mathematical model to describe the effect of RT and FUS in treating glioblastoma multiforme. The results indicate that patient survival could potentially be significantly improved with combined FUS and different fractionated radiation regimens. While these results were generated through a theoretical pipeline, they reflect the tangible effects that FUS and RT could have while suggesting safety margins similar to FUS or RT alone.
With the advancement of MRI-Guided FUS (MRgFUS) systems, Zhang et al 64 recently showed an enhanced impact of 10 Gy of RT combined with FUS in suppressing tumor growth and proliferation while increasing levels of apoptosis in PC-3 mouse xenografts. Applications of FUS and RT in clinical settings have also seen success in enhancing RT. Wu et al 65 evaluated the feasibility of replacing reduced field boost irradiation with FUS in treating patients with locally advanced and metastatic prostate cancer. Combination treatment showed a greater overall survival in patients compared to low-dose external beam radiotherapy alone. These promising results show potential for applying this combination treatment modality in a clinical setting.
FUS has only recently begun to become a standard of care treatment in the clinical setting as a monotherapy system. Interest in expanding its application is often overshadowed by optimization and integration into a clinical setting. Currently, much of FUS + RT research revolves around the use of microbubbles (MBs) to induce effects within the tumor microenvironment to enhance RT effects.
Ultrasound-Stimulated MBs
MBs have primarily been utilized as an ultrasound contrast agent to enhance the imaging of a multitude of organ structures.66,69 MBs are often 0.5 to 10 µm in diameter with a core composed of high molecular weight and low solubility gas. Often, bubbles are coated in a lipid or protein shell to enhance stability.68,70 When placed in an acoustic field of a resonant frequency, MB expands and compresses in response to the acoustic pressure applied in a process known as cavitation. MBs undergo one of 2 cavitation states; steady symmetrical oscillations at lower amplitudes (stable cavitation), or rapid, irregular oscillations leading to bubble collapse at higher amplitudes (inertial cavitation).71,72 Both forms of cavitation can generate effects on the surrounding environment such as pushing, pulling, microstreaming, generating shockwaves, and micro-jetting.71,75 These effects are capable of generating perforations in the plasma membrane, in a process known as sonoporation,74,76 which can be exploited to induce endothelial cell apoptosis or enhance the delivery of radiosensitizing agents, such as oxygen.
Ultrasound Stimulated MB (USMB) Mediated Vascular Disruption and RT
The tumor vasculature is a key component to ensuring tumor growth and survival. High doses of RT (> 8 Gy) induce increased oxidative stress to vascular endothelial cells, resulting in the activation of pro-apoptotic pathways such as the acid sphingomyelinase (ASMase)-ceramide pathway (Figure 2A).77,86 Many in vivo studies involving ASMase knockout mice (ASMase −/−) demonstrate that radiation exposure showed significant levels of endothelial cell apoptosis in wild-type (WT) mice, but not in ASMase −/− groups.77,78,84,87,92 The ASMase-Ceramide pathway was later found to also be activated through USMB mechanotransduction.93,97 This led to the idea of using USMB to enhance lower doses of RT (< 8 Gy) through ASMase activation. Combination treatments of USMB and single doses of 2 and 8 Gy in murine models demonstrated elevated levels of tumor cell death and reduced microvascular density in a dose-dependent manner.56,98,102 Furthermore, tumors in ASMase −/− mice exhibited a radioresistant phenotype against USMB + RT treatments with low single doses (2 and 8 Gy) compared to WT mice bearing fibrosarcoma tumors 56 (Figure 2B). This effect was also observed in fractionated RT regimens in mouse and rabbit xenografts where tumor growth delay and proliferation reduction were observed when combined with USMB treatments.55,100 Furthermore, in a clinical trial conducted by Eisenbrey et al, 103 the efficacy and safety of USMB alongside transarterial radioembolization (TARE) were demonstrated in treating patients with hepatocellular carcinoma. The results showed that 14 of 15 patients treated with TARE + USMB showed a partial or complete response compared to TARE alone, in which only 5 out of 10 patients showed partial or complete response, with no adverse effects. These studies offer great promise in applying USMB treatments alongside standard clinical RT regimens.

Mechanism of USMB + RT action. (A) Radiation or plasma membrane disruption induces an increase in oxidative stress and calcium influx resulting in lysosomal translocation and fusion to the plasma membrane. ASMase release into the plasma membrane initiates sphingomyelin hydrolyzation to generate ceramide, resulting in the localization of ceramide-rich lipid microdomains. Plasma membrane alteration in these microdomains result in the localization of cell death receptors and factors resulting in pro-apoptotic signaling, leading to endothelial cell apoptosis, vascular disruption, and eventually tumor cell death. (B) ASMase −/− can resist radiation and USMB leading to vascular preservation and tumor cell survival. When ASMase is knocked out, ceramide is not synthesized, thus no pro-apoptotic signaling occurs in endothelial cells.
Oxygen Loaded MBs (OMBs) and RT
In the past few years, the use of MBs as a delivery vector has been explored in great depth for chemotherapy to enhance targeting and treatment efficacy.104,105 In the context of FUS + RT, this technique can be applied in the delivery of radiosensitizers, such as oxygen, to elicit greater tumor responses to RT.
Oxygen has been of great interest as a radiosensitizing agent, as hypoxia is a known phenomenon that hinders RT efficacy.106,107 To overcome this, OMBs were developed to allow for reoxygenation in otherwise hypoxic tumor regions. Though there have been no clinical trials as of the date of this publication, many in vitro and in vivo studies have demonstrated the feasibility of utilizing OMB to elevate oxygen levels in a variety of tumors including breast, fibrosarcoma, prostate, head and neck squamous cell carcinoma, and nasopharyngeal carcinoma.108,114 Eisenbrey et al 108 demonstrated, in immunodeficient nude mice, that ultrasound-activated OMB + 5 Gy RT, resulting in breast tumor growth delay at 25 to 35 days with significantly lower growth rates and greater survival. This effect was also seen in studies conducted using higher doses of radiation (> 10 Gy), in which RT + OMB treated tumors showed lower growth rates compared to untreated tumors, demonstrating the range of doses in which OMB can enhance RT effects in single dose in external beam,109,111 and brachytherapy. 114
Overall, these studies show great promise in the efficacy of utilizing OMB to overcome hypoxia and have potential applications to be used in a clinical setting.
Current Limitations of FUS and RT
Though much of the groundwork in the preclinical studies have established the efficacy of combining FUS + RT while demonstrating its safety and application, several limitations must be considered before full implementation in the clinic. For instance, attenuation, reflections, and scattering must be accounted for when planning treatments as these effects can reduce the efficacy of the ultrasound treatment.115,116 Additionally, there are currently several commercially available USMBs with different coating and gas compositions. These properties can affect the efficacy of RT enhancement and there have yet to be studies that compare the difference between them.68,103 Furthermore, adverse effects related to FUS (e.g., thermal damage resulting in organ dysfunction) or RT (e.g., off-target exposure leading to tissue damage) must be taken into great consideration to prevent a reduction of quality of life as a result of the combination treatment.35,60,117
Motion during treatments can also provide additional challenges as there is an added risk of off-target effects in ablative FUS and RT treatments.118,121 While there have been many strategies implemented to overcome this in RT,122,124 further studies would need to be conducted to determine whether they are applicable in a FUS + RT regimen or if other strategies would be needed.
Current MRgFUS systems are used to allow for adequate planning and treatment and have been used primarily for their ablative capabilities. 125 With the state of this current technology, FUS + RT treatments would require excessive planning to ensure that targeting of the tumor is accurate as patients would be required to be moved between different systems. This can potentially be overcome by integrating FUS into MR-LINAC systems, allowing for rapid treatment of FUS and RT using the same treatment plan for precise targeting. However, as of the date of this publication, there are no such systems in place used in clinical settings.
Despite these limitations, the successful application of ultrasound combined with MB together with RT to enhance tumor response has been well documented and continues to suggest viability in safe implementation in the clinical setting.
Conclusion
FUS has progressed significantly since the initial studies began to explore its potential. While the majority of studies involving FUS have been focused on its optimization and implementation in the clinic, the value of it alongside RT must also be recognized. The use of MB cavitation has provided greater promise for FUS + RT applications as many studies have shown the effectiveness of enhancing radiation through vascular disruption or enhancing radiosensitization delivery. Thus, further studies are warranted to explore the upper boundaries of utilizing FUS by exploiting its biological effects in conjunction with RT. All of this taken together shows the feasibility of applying FUS and RT together to enhance patient quality of life while potentially reducing radiation exposure during treatments.
Footnotes
Abbreviations
ASMase, acid sphingomyelinase; ASMase −/−, acid sphingomyelinase knockout mice; FUS, focused ultrasound; LRT, low dose external beam radiation therapy; MR-LINAC, MRI-guided linear accelerator; MRgFUS, MRI-guided focused ultrasound; OMB, oxygen-loaded microbubbles; PC-3, human prostate cancer cell line; TARE, transarterial radioembolization; USMB, ultrasound stimulated microbubble; WT, wild type; RT, radiation therapy
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
Not applicable.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
