Abstract
Human cancer statistics show that an increased incidence of urologic cancers such as bladder cancer, prostate cancer, and renal cell carcinoma. Due to the lack of early markers and effective therapeutic targets, their prognosis is poor. Fascin-1 is an actin-binding protein, which functions in the formation of cell protrusions by cross-linking with actin filaments. Studies have found that fascin-1 expression is elevated in most human cancers and is related to outcomes such as neoplasm metastasis, reduced survival, and increased aggressiveness. Fascin-1 has been considered as a potential therapeutic target for urologic cancers, but there is no comprehensive review to evaluate these studies. This review aimed to provide an enhanced literature review, outline, and summarize the mechanism of fascin-1 in urologic cancers and discuss the therapeutic potential of fascin-1 and the possibility of its use as a potential marker. We also focused on the correlation between the overexpression of fascin-1 and clinicopathological parameters. Mechanistically, fascin-1 is regulated by several regulators and signaling pathways (such as long noncoding RNA, microRNA, c-Jun N-terminal kinase, and extracellular regulated protein kinases). The overexpression of fascin-1 is related to clinicopathologic parameters such as pathological stage, bone or lymph node metastasis, and reduced disease-free survival. Several fascin-1 inhibitors (G2, NP-G2-044) have been evaluated in vitro and in preclinical models. The study proved the promising potential of fascin-1 as a newly developing biomarker and a potential therapeutic target that needs further investigation. The data also highlight the inadequacy of fascin-1 to serve as a novel biomarker for prostate cancer.
Introduction
According to the latest statistics, cancer is a major public health concern, which seriously threatens human health, and is the primary cause of fatality among patients.1,2 Cancers of the urinary system include those of the bladder, kidney, renal pelvis, ureter, and other urinary organs. Males account for more than 20% of all patients with urologic cancers. 1 Bladder cancer and prostate cancer (PCa) account for the majority of the urologic cancers. The leading cause of poor prognosis and fatalities of patients with cancer is cancer metastasis. Patients with metastatic bladder cancer and PCa have a 5-year survival rate of less than 10%.3,4 Therefore, an early diagnosis would considerably improve the survival rate among these patients. Currently, there is a lack of effective treatments for metastatic bladder cancer, and the survival rate of patients is low; hence, there is an urgent need for new and enhanced treatment methods. 5 Screening and diagnosis of PCa rely mainly on analysis of the prostate-specific antigen (PSA). However, PSA cannot accurately distinguish between inert tumors and aggressive disease. 6 Therefore, new clinical prognostic PCa biomarkers are needed to better differentiate inert prostate tumors from PCa.7,8
Fascin-1 is a 55 kDa cytoskeleton protein, which binds to F-actin and participates in cell division, cell migration, and intercellular interactions.9,10 Fascin-1 is expressed only in the neurons, endothelial cells, mesenchymal cells, and dendritic cells among nondiseased adults, and it is almost absent in normal epithelial cells.11,12 However, dysregulated actin regulatory proteins are found in many common cancers. Fascin-1 is one of the most frequently upregulated regulatory protein in metastatic cancer11,13,14 and is associated with poor prognosis and increased metastasis. 15 Therefore, it is considered as a potential prognostic marker and therapeutic target.
Previous studies have identified the role of fascin-1 in gynecologic cancers, 16 gastrointestinal cancers, 17 and respiratory-related cancers. 18 This review aims to elucidate fascin-1 expression in urologic cancers (bladder, prostate, and kidney cancers and adrenocortical carcinoma [ACC]) and the signaling mechanisms of the fascin-1 gene in these urologic cancers. In addition, we evaluate the potential of fascin-1 as a prognostic marker and a therapeutic target, to support further studies.
Physiological Function and Cancer-Promoting Function of Fascin-1
Expression and Physiological Functions of Fascin-1 in Normal Cells
Current studies are focused on the intracellular physiological functions of fascin-1. The expression of fascin-1 is more pronounced during human growth stages than that in adulthood. 19 During embryonic development and fetal maturity, fascin-1 is prominently expressed in the nervous system, including the dendritic cells, and in the microvascular endothelial cells, gastrointestinal tract, and mesenchymal tissue.20–25 These cells are similar in terms of experiencing significant cell migration, indicating that fascin-1 fulfills an important role in cell migration. Moreover, fascin-1 expression is highly upregulated in cytotrophoblast cells (CTBs), promoting the proliferation of CTBs and, thus, the formation of the placenta in early gestation.26,27 During the early developmental stages of the placenta, fascin-1 regulates the epithelial–mesenchymal transition (EMT).
In healthy adult tissues, fascin-1 expression is limited to the neuronal, endothelial, mesenchymal, and dendritic cells, 12 and its expression is either inhibited or downregulated in normal epithelial cells.11,12 Fascin-1, an actin cluster protein, can cross-link with actin filaments, thus forming cell protrusions.28–30 This process is essential for intercellular adhesion, cellular interactions, motility, and migration.13,31,32 During cell development, fascin-1 can control dendrite formation, cell migration, and neuropil extension.33–35 Fascin-1 expression is highly upregulated in mature dendritic cells, thus facilitating antigen presentation to T cells and participation in the immune response.20,36 Within mature dendritic cells, fascin-1 can promote dendritic cell migration into lymph nodes by promoting podosome disassembly and increasing membrane protrusions. 36 Moreover, fascin-1 is involved in cell migration by combining with the microtubule cytoskeleton. 37 For cell migration and invasion, fascin-1 can interact with nesprin-2 to promote nuclear deformation and migration. 38 It is specifically expressed in the human retina, and mutations cause autosomal dominant retinitis. 26 Fascin-3 is also expressed in the head of the sperm at later stages and is involved in the directional movement of sperm after release; however, its exact functional role is unclear. 39
Fascin-1 plays an important role in regulating cell motility and migration, particularly during embryonic development and fetal maturation. It also participates in several cellular physiological processes, including the regulation of cell adhesion, movement, migration, and cell interaction.
The Role of Fascin-1 in Cancer
To date, Immunohistochemistry has published more than 130 studies on fascin-1 expression in various tissues and tumors, using Western blot analysis or quantitative polymerase chain reaction (qPCR). All studies have reported positive upregulation of fascin-1 expression in cancer tissues compared with that in normal epithelial or paraneoplastic tissues. However, the regulation of fascin-1 expression is tissue-specific in different tumors. In aggressive tumors, the positive expression rate of fascin-1 is higher than that in other tumors.
In vitro experiments have revealed that downregulating fascin-1 expression in various cancer cells inhibits cell growth, migration, and invasion ability.40–44 In contrast, the overexpression of fascin-1 can increase the proliferation rate of cancer cells, along with their migration and invasion ability.45–50 However, the role of fascin-1 is significantly different in melanoma, where fascin-1 and malignant melanoma are negatively correlated; fascin-1 knockdown enhances cell invasion.51,52 In addition, a report on lung cancer suggested that fascin-1 plays a role in the metastasis and colonization of cancer cells and promotes mitochondrial oxidative phosphorylation by reconstructing mitochondrial actin filaments. 53 Also, fascin-1 participates in the EMT process in several cancers. EMT is a biological process wherein epithelial cells are transformed into cells with mesenchymal phenotype through specific procedures, which enhance the migration and invasion abilities of malignant tumor cells. 54 High fascin-1 expression in cancer cells is associated with increased chemoresistance in various cancers, including breast, hepatocellular, and PCa, through different mechanisms.55–57 In conclusion, high fascin-1 expression can accelerate the growth, metastasis, and invasion of cancer cells, making them chemo-resistant.
Fascin-1 and Bladder Cancer
Results of Fascin-1 Overexpression in Bladder Cancer and its Potential as a Prognostic Marker
Fascin-1 is highly expressed in bladder tumors, with negative fascin-1 expression in the adjacent normal epithelium.41,58–62 Fascin-1 expression is also negative in inflammatory diseases of the bladder and benign urinary tumors. 59 In contrast, in invasive bladder urothelial carcinoma (BLCA), the invasive components entering the intrinsic muscular layer are all strongly positive for fascin-1, and its expression gradually increases with increasing depth of infiltration.59,63 The overexpression of fascin-1 has also been observed in most metastatic BLCA. 64 Immunohistochemical (IHC) staining analysis involving a large number of clinical samples has shown a strong positive correlation (P < .001) between fascin-1 expression and tumor pathological stage58,61,62 (Table 1) but lacks a significant correlation with tumor grade.41,58,59,62,63,65
Relationship Between High Expression of Fascin-1 and Clinical Parameters in Urologic Cancers and the Use of Fascin-1.

Abbreviations: PSA, prostate-specific antigen; DFS, disease-free survival; OS, overall survival; PCa, prostate cancer; BLCA, bladder urothelial carcinoma; IHC, immunohistochemical; RCC, renal cell carcinoma; ACC, adrenocortical carcinoma.
* [unk] = unknown; [+] = correlation (positive correlation/negative correlation); [-] = not relevant.
Effective downregulation of fascin-1 expression using different methods decreases cell migration and invasion and enhances adhesion.41,61,75 The growth of different bladder cancer cells is also significantly inhibited. 41 Thus, fascin-1 overexpression is critical in the invasion and recurrence of BLCA. A follow-up trial has found that the probability of recurrence is enhanced with an increase in the expression levels of fascin-1 among postoperative patients, and recurrence appears earlier after surgery. Log-rank univariate analysis has shown a significant independent correlation between fascin-1 expression and poor prognosis. 62 Currently, Ki-67, p53, CK20, and MDR have been extensively studied in BLCA, and the role of these 4 markers as diagnostic tools or prognostic factors is well established.76–79 A multivariate analysis has found that only fascin-1 and CK20 expression have a statistically significant independent correlation with poor prognosis. 62 Thrombomodulin, a surface glycoprotein, is the most common IHC marker of BLCA. Compared with thrombomodulin, fascin-1 has higher expression frequency, intensity, sensitivity, and specificity. 64 In addition, fascin-1 staining can aid in predicting invasiveness in biopsies. 60 Fascin-1 is associated with increased tumor invasion and recurrence.58,60,62–64 Thus, we conclude that fascin-1 overexpression can be used as a prognostic marker for BLCA.
Regulation of Fascin-1 Expression in Bladder Cancer
Chiyomaru et al found that the expression levels of miR-145 and miR-133a are downregulated in bladder cancer, and these microRNAs (miRNAs) bind to fascin-1 mRNA, thereby inhibiting fascin-1 expression. Therefore, they concluded that fascin-1 expression could be directly controlled by miR-145 and miR-133a 41 (Figure 1). Long noncoding RNAs (lncRNAs) have gained considerable attention recently. These lncRNAs represent microRNA “sponges,” which can influence biological processes by attenuating the effects of mRNAs.80–83 Xue et al found that the lncRNA uroepithelial carcinoma-associated 1 (lnc-UCA1) is highly expressed in, both, bladder cancer tissues and cells. Lnc-UCA1 can promote bladder cancer cell invasion and migration through the zinc finger E-box binding homeobox 1 and 2 (miR-145-ZEB1/2)-fascin-1 pathway (Figure 2). Moreover, they found a mutual inhibition between lnc-UCA1 and miR-145. 84 Subsequent studies found that the carcinogenic long noncoding lnc-ZEB-AS1 is highly expressed in BLCA and is negatively interrelated with miR-200b expression and positively interrelated with fascin-1 expression. Lnc-ZEB-AS1 promotes fascin-1 expression by downregulating miR-200b, which subsequently promotes BLCA migration, proliferation, and invasion and inhibits apoptosis. This result has also been validated in a xenograft model. 75 Transforming growth factor (TGF)-β is a gene associated with carcinogenesis, affecting multiple signal pathways in cancer cells.85,86 TGF-β may partially induce tumor invasion and migration by regulating the ZEB1-AS1/miR-200b/fascin-1 axis. The overexpression of lnc-UCA1 and lnc-ZEB-AS1 increases the mRNA and protein levels of N-calmodulin and waveform protein and decreases those of E-calmodulin.75,84 Moreover, lnc-UCA1 can function as an important EMT inducer in bladder cancer cells. 84 This provides a new research direction and therapeutic target for inhibiting bladder cancer invasion and metastasis.
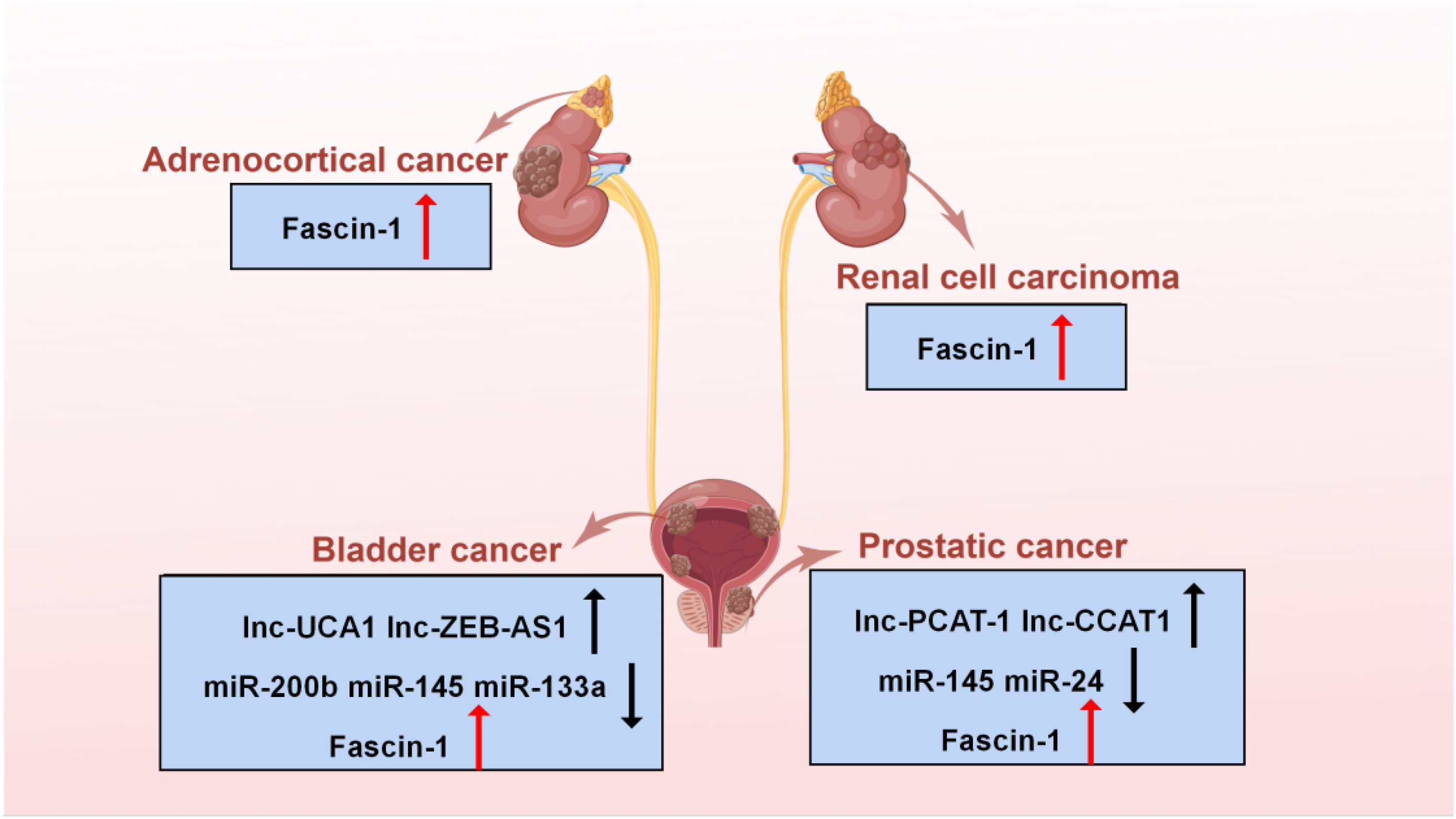
The expression of microRNA (miRNA), lncRNA, and fascin-1 in urologic cancers.

The molecular mechanism and biological effects of fascin-1 overexpression in urologic cancers. Under the regulation of various lncRNAs and growth factors, the expression of fascin-1 increases, which promotes the migration, invasion, and proliferation of tumor cells and inhibits their apoptosis.
Extracellular matrix metalloproteinase inducer (EMMPRIN) is a multifunctional cell surface glycoprotein, which can promote the production of vascular endothelial growth factor by stimulating the production of matrix metalloproteinases. 87 It is related to the poor prognosis and increased invasiveness of several cancer types (PCa, 88 tongue cancer, 89 uterine cervix cancer, 90 and esophageal 91 SCCs). EMMPRIN and fascin-1 can play a synergistic role in enhancing the invasiveness of bladder cancer cells. 58 However, the interaction mechanism is still unclear.
Fascin-1 as a Therapeutic Target in Bladder Cancer
The role of fascin-1 in bladder cancer makes it a promising therapeutic target. To date, the fascin-1 inhibitor NP-G2-044 has been used to conduct studies in this regard.5,92 First, NP-G2-044 reduces the migration of different bladder cancer cells. As reported earlier, NP-G2-044 also does not influence bladder cancer cell growth and apoptosis.5,92,93 In addition, studies in a mouse model of bladder cancer have shown that NP-G2-044, alone, or in combination with cisplatin prolongs the life span of mice,5,94,95 and combination treatment with an immune checkpoint inhibitor (PD-1) also improves overall survival. 5 Therefore, fascin-1 inhibitors could be explored as a promising new method for treating bladder cancer.
Fascin-1 and PCa
Expression of Fascin-1 in PCa and its Potential as a Therapeutic Target
The results of global cancer statistics and analysis in 2020 have shown that PCa is the second most common cancer and the fifth leading cause of death among men. 96 In China, the majority of the patients with PCa have already been diagnosed as progressed to advanced stages, thus losing out on the best operation time.97,98
Limited and hormone-refractory PCas show a significant upregulation of fascin-1 expression than that in benign prostate tissue.6,68,69,99 In addition, a follow-up of patients undergoing radical prostatectomy has shown high fascin-1 expression to be related to an increased rate of PSA recurrence. 68 High fascin-1 expression is related to Gleason score and bone metastases, 69 thus confirming its role in PCa progression. The results of cellular assays suggest that silencing the fascin-1 gene reduces tumor cell motility and invasiveness and increases cell adhesion.68,99,100 In vivo experiments have shown that reducing fascin-1 expression in mice significantly inhibits tumor development and suppresses lymph node metastasis. 70 Thus, targeting fascin-1 may be effective in preventing or treating metastatic PCa.68,99,100
Octavian and Daniel et al investigated serum fascin-1 levels.67,70 They showed that although serum fascin-1 levels differ in patients with and without PCa, the differences are not statistically significant. Moreover, although fascin-1 is overexpressed in tumors with lymph node metastasis and high Gleason score, there was no statistical difference. The expression level of fascin-1 is negatively correlated with age. 67 In contrast, studies in healthy males have found that serum fascin-1 levels are independent of patient age. 70 However, this could be attributed to the different selection of experimental participants, further suggesting that serum expression levels of fascin-1 in patients with PCa are related to age. Matthew et al examined PCa tissue and found that only 8% of the tumors exhibited >10% fascin-1-positive cancer cells and that fascin-1 status is unrelated to Gleason score, tumor stage, serum PSA levels, or postoperative biochemical recurrence. Meanwhile, fascin-1 is not superior to the ETS-related gene, a new marker of invasive PCa in the nucleus. 6 A large-scale study using protein quantitative trait loci as a tool to evaluate the correlation between gene-predicted protein levels and PCa risk identified 31 biomarkers of PCa blood proteins, but fascin-1 was not among them. 101 Thus, fascin-1 is unlikely to be used as a new biomarker in patients with PCa.6,67 Its functional role and applicability as a diagnostic marker for PCa is still controversial. There are alternate mature markers for PCa, which are superior to fascin-1. Future research should focus on its applicability in improving a patient's survival rate and prognosis.
Regulation of Fascin-1 Expression in PCa
Increasing evidence has identified miRNAs functioning in oncogenes or tumor suppressors in human cancers. MiR-145 expression is downregulated in various cancers, including PCa, and genome-wide gene expression analysis has identified fascin-1 as a candidate miR-145 target gene (Figure 1). Meanwhile, miR-145 can exert tumor suppressive effects by directly controlling fascin-1 expression in PCa cell lines, thereby inhibiting cancer cell proliferation, migration, and invasion. 100 Considering this, Xu et al found that PCa-associated lncRNA transcript 1 (lnc-PCAT-1) expression is upregulated, and miR-145-5p expression is downregulated in PCa tissues and cells. It was concluded that lnc-PCAT-1 promotes the proliferation, migration, invasion, and inhibition of apoptosis of PCa cells through the lnc-PCAT-1/miR-145-5p/fascin-1 regulatory axis (Figure 2). The RT-qPCR results showed that lnc-PCAT-1 and miR-145-5p interact to repress each other's expression. 102 LncRNA colon cancer-associated transcript 1 (lnc-CCAT1) is an oncogene for cancers such as gastric, ovarian, and nasopharyngeal cancers. It plays an important role in the prognosis and diagnosis of human cancer by regulating cell migration, cell proliferation, chemotherapy resistance, and other processes. Chen et al reported that lnc-CCAT1 contributes to the migration and invasion of PCa cells. 103 Subsequent studies have shown that lnc-CCAT1 is upregulated in PCa cells, which can promote fascin-1 expression by inhibiting the expression of miR-24-3p. Silencing CCTA1 through this regulatory pathway can improve the sensitivity of patients with PCa to paclitaxel, thus improving the treatment efficacy of PCa and the success rate of chemotherapy. 104
High expression of N-Myc proto-oncogene protein (N-Myc) leads to uncontrolled cell proliferation, affects cell metabolism, and promotes cell invasion, apoptosis, and differentiation. 105 The results of IHC staining of prostate tissue showed that N-Myc and fascin-1 levels are significantly higher than those of normal tissue and that both contribute to the malignant progression of PCa. The experimental results suggest a direct or indirect regulatory relationship between N-Myc and fascin-1, and their mutual regulation and interaction promote the clinical progression of PCa. N-Myc has many downstream regulators and a complex regulatory mechanism; hence, the role of the N-Myc/fascin-1 pathway in PCa treatment needs further elucidation. 69 Li et al discovered that decreasing fascin-1 expression in cancer cells increases the expression of E-cadherin and decreases the expression of vimentin and snail. This shows that fascin-1 can promote EMT in PCa cells. Moreover, the overexpression of fascin-1 can promote glycolysis in cancer cells. In terms of mechanism, fascin-1 enhances the invasion, migration, EMT, and glycolysis of PCa cells by activating the YAP/TAZ signaling pathway. 99
Fascin-1 and Renal Cell Carcinoma
Renal cell carcinoma (RCC) ranks ninth among all global malignancies in men and 14th in women. In the United States, it is the sixth and eighth most common malignancy in men and women, respectively. 106 However, fascin-1 has been relatively poorly studied in RCC compared to that other cancers. Fascin-1 is not expressed in normal renal tissue 107 and is overexpressed in RCC.71–73,108 IHC analysis has revealed that high fascin-1 expression is significantly linked to clinicopathological parameters such as nuclear grading, TNM stage, and disease-free survival (DFS) in RCC.71–73 Fascin-1 expression does not significantly correlate with the M stage of RCC, which may be due to the limited number of M1 stage cases.72,73 Meanwhile, fascin-1 plays an important role in the metastasis of RCC.75,76 Follow-up results show a statistically significant decrease in overall survival and 5-year metastasis-free survival in the fascin-1 expression-positive group.71,72 Thus, fascin-1 has excellent potential for applicability as a new biomarker for predicting and treating RCC in future.71–73
TGF-β1 is a multifunctional cytokine that is critical in regulating cell growth, differentiation, migration, movement, and other cellular processes. In addition, TGF-β1 can also promote the invasion and migration of tumor cells. In renal cancer cells, with the increase in fascin-1 levels, p-extracellular regulated protein kinases (ERK) and p-c-Jun N-terminal kinase (JNK) levels are also significantly increased. Nevertheless, the effect of TGF-β1 induced fascin-1 expression is inhibited by treatment with specific inhibitors of ERK and JNK signaling pathways, suggesting that TGF-β1 promotes fascin-1 expression in kidney cancer cells through these pathways 109 (Figure 2). Studies in renal fibrosis have revealed that the levels of, both, fascin-1 and CD44 (a nonkinase transmembrane glycoprotein) are apparently upregulated, while those of the miR-200b/c family declined. Overexpression through the miR-200b/c family could inhibit fascin-1 or CD44, thereby inhibiting the EMT process from improving renal fibrosis. 110 Furthermore, circular RNA ACTR2 (circACTR2) can adsorb miR-561 and upregulate NLRP3 expression levels, thereby inducing IL-1β secretion. IL-1β significantly upregulates fascin-1 expression at the protein and mRNA levels, contributing to renal fibrosis.111–113 In RCC, further research is required to confirm the role of fascin-1 metastasis and to elucidate its mechanism of action.
Fascin-1 and ACC
Compared with other cancers, ACC is a relatively rare invasive cancer with a 5-year survival rate of only 10%.114–117 Protein markers that can distinguish benign adenomas from ACC and can predict tumor progression are lacking. The first proteomic analysis of ACC identified fascin-1 as a potential malignancy marker. 118 Another study found that fascin-1 overexpression is not related to clinicopathological parameters such as age, sex, and tumor diameter but is significantly related to DFS and overall survival 74 (Table 1). Expression levels of the adrenocortical spectrum marker, SF-1, are prognostic indicators associated with tumor aggressiveness.119,120 There is a significant correlation between SF-1 and fascin-1 transcript levels, and fascin-1 participates in the invasion of SF-1 overexpressed ACC cells. The ki67 labeling index (Ki67 LI) was presumed to be the most effective predictor of disease recurrence and survival time in patients with ACC following surgery, 121 and the addition of fascin-1 may considerably improve Ki67 LI's prognostic ability. 74 Overall, fascin-1 is a reliable, independent prognostic marker for ACC and a potential therapeutic target for specific interventions in cancer spread and metastasis.
Future Perspectives
The overexpression of fascin-1 is related to some clinicopathological parameters in cancers of the urinary system. It is associated with pathological cancer staging, bone or lymph node metastasis, and reduced DFS. Studies in BLCA have found that the overall survival of mice with loaded BLCA can be improved by using molecular inhibitors of fascin-1. Thus, fascin-1 could serve as a promising diagnostic marker and a potential therapeutic target. However, the applicability of fascin-1 as a diagnostic marker in PCa is still controversial and less likely, based on current research. Future research should emphasize whether it can be used to treat patients with BLCA patients and improve prognosis.
Mechanistically, fascin-1 is regulated by several regulators and signaling pathways (such as lncRNA, miRNA, JNK, and ERK). It is overexpressed in most human cancers, including urologic cancers. There is a relative lack of research on the therapeutic application of fascin-1. Unlike other cancers, studies on the mechanism of fascin-1 expression in urologic cancers have mostly focused on long-stranded noncoding RNAs. This provides an alternative for inhibiting fascin-1 expression. Meanwhile, research on fascin-1 inhibitors (such as small molecule inhibitors, inhibitory nanosomes, and miRNAs) should be conducted to expand the number of in vivo experiments and lay the foundation for further clinical applications. Although the molecular mechanism leading to the upregulation of fascin-1 and the enhancement of tumor migration and invasion need further investigation, our study suggests that targeting fascin-1 may be a novel treatment option and prognostic marker for urologic cancers. Fascin-1 remains as a promising candidate for cancer metastasis and treatment. In future, further in vivo studies are needed to determine whether small molecule targeting fascin-1 helps treat urologic cancers.
Currently, several fascin-1 inhibitors (G2, NP-G2-044) have been discovered, and their effectiveness has been evaluated in vitro and using preclinical models.5,92,122 They function by blocking the interaction between fascin-1 and F-actin. Recently, the first human clinical trial for NP-G2-044 was conducted. The results of clinical phase 1 experiment showed that oral NP-G2-044 is well tolerated by patients, with an increased rate of progression-free survival and metastasis-free interval, particularly in patients with advanced ovarian cancer. 123 Based on this research, a phase 2A clinical experiment for ovarian cancer is in progress. In the future, the next research should increase the number of clinical trials, expand the scope of research, and further clarify the role of fascin-1 inhibitors in the treatment of other types of cancer.
Concluding Remarks
In general, fascin-1 has great potential as a novel biomarker and a potential treatment strategy in some urologic cancers (BLCA, RCC, and ACC). However, in PCa, it is less likely to become a new marker.
This research had some limitations. First, compared with the research enthusiasm for fascin-1 in other cancers with urinary cancer is relatively low, with limited available research. The association between fascin-1 and other urinary system cancers (such as those of the penis, testicules, and ureter) still lacks corresponding research. Second, the exact molecular mechanisms of fascin-1 regulation and interactions with other genes and proteins, particularly in urologic cancers, are still in the naive stages of research. For example, the mechanisms of interaction between fascin-1 and proteins such as EMMPRIN and N-Myc, the signaling pathways involved in Wnt/β-linked proteins and p38MAPK, and the mechanisms of action of fascin-1 and YAP/TAZ are still unknown. Third, there are few in vivo studies and clinical trials for evaluating the therapeutic potential of fascin-1, and further studies are needed to confirm the therapeutic efficacy and safety of fascin-1.
Footnotes
Abbreviation
Acknowledgments
The authors would like to thank Editage (www.editage.com) for English language editing. The authors made Figure 1 and ![]() by figdraw platform. Thanks for the support of figdraw platform.
by figdraw platform. Thanks for the support of figdraw platform.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Shandong Provincial Natural Science Foundation, grant numbers ZR2020MH078 and ZR2020MH070, Postdoctoral program of Affiliated Hospital of Jining Medical University, Research Support Foundation of Jining Medical University, grant number JNFC8KJ009, and the College Students’ Innovative Entrepreneurial Training Plan Program of Jining Medical University, grant number cx202109.
