Abstract
Breast cancer is the leading cause of cancer death among women worldwide. In solid tumors, the microenvironment plays a critical role in tumor development, and it has been described a communication between the different cell types that conform the stroma, including fibroblasts, pericytes, adipocytes, immune cells and cancer-associated fibroblasts. Intercellular communication is bidirectional, complex, multifactorial and is mediated by the secretion of molecules and extracellular vesicles. The extracellular vesicles are vesicles limited by two membranes that are secreted by normal and cancer cells into the extracellular space. Extracellular vesicle cargo is complex and includes proteins, miRNAs, DNA and lipids, and their composition is specific to their parent cells. Extracellular vesicles are taken up for neighboring or distant cells. Particularly, extracellular vesicles from breast cancer cells are taken up for fibroblasts and it induces the activation of fibroblasts into cancer-associated fibroblasts. Interestingly, cancer associated fibroblasts release extracellular vesicles that are taken up for breast cancer cells and promote migration, invasion, proliferation, epithelial–mesenchymal transition, changes in metabolism, chemoresistance, evasion of immune system and remodeling of extracellular matrix. In addition, the enrichment of specific cargos in extracellular vesicles of breast cancer patients has been suggested to be used as biomarkers of the disease. Here we review the current literature about the intercommunication between tumor cells and cancer associated fibroblasts through extracellular vesicles in breast cancer.
Introduction
Breast cancer is one of the most prevalent diseases in the world and the rate of new cases continues increasing. According to Global Cancer Statistics (2020), it has been estimated 19.3 million new cancer cases and 10 million cancer deaths in 2020. 1 Over the last decades, more resources and efforts have been directed at increasing our knowledge about the molecular and cellular processes involved in cancer. In solid tumors, the stroma plays an important role in every stage of the disease, from induction of early proliferation to the creation of a metastatic niche. 2 The functions carried out by stromal cells may be considered in both tumor suppression and tumor promotion, which is consistent with the complexity of this compartment. 3 The stroma consists of a variety of cell types including fibroblast, myofibroblasts, cancer-associated fibroblasts (CAFs), immune, epithelial, vascular and smooth muscle cells, which secrete a variety of molecules including cytokines and extracellular matrix (ECM) components. 4 The intercellular communication between the tumor and its microenvironment is bidirectional, complex, and multifactorial. 2 Extracellular vesicles (EVs) play an essential role in tumor progression, because they mediate a variety of cellular processes directly related with tumor progression including migration, invasion, and the creation of a tumor supporting stroma and an environment for the recruitment and transformation of CAFs.5–8 Here we review the current literature about the intercommunication between tumor cells and CAFs mediated by EVs in breast cancer.
Extracellular Vesicles
EVs are vesicles limited by two membranes that are secreted by normal and cancer cells into the extracellular space. The term “extracellular vesicle” was first used in 1971. 9 However, the foundation of the International Society for Extracellular Vesicles in 2011, proposed the replacement of the terms “exosomes” and “microvesicles” with the term “extracellular vesicles,” a generic name adopted by consensus by the international scientific community. 10 Although their designation remains controversial, based on size, biogenesis, or release pathways, EVs have been classified as microvesicles, exosomes, and apoptotic bodies.11–13
Exosomes
Exosomes were first reported in 1983 by Johnstone and colleagues as vesicles of endosomal origin secreted by reticulocytes during differentiation. 14 Exosomes are small membrane vesicles, ranging in size from 30 to 100 nm in diameter with a cup-shaped morphology and a density ranging from 1.13 g/ml (for B cell-derived exosomes) to 1.19 g/ml (for intestinal cell-derived exosomes). 15 The exosome formation process consists of four stages: initiation, endocytosis, formation of multivesicular bodies (MVBs), and exosome secretion. 16 MVBs are endocytic structures created by the budding of an endosomal membrane into the lumen of the compartment. Once these structures form, they may be sorted for cargo degradation into the lysosome or released into the extracellular space as exosomes by fusing with the plasma membrane. The mechanisms underlying the sorting of the cargo into intraluminal vesicles are not fully understood, however it is suggested that exosomal sorting is mediated by both endosomal sorting complex required for transport-dependent and -independent signals.8,17 As a consequence of their endosomal origin, nearly all exosomes, independent of the cell type from which they originate, contain marker proteins involved in membrane transport and fusion (Rab GTPases, annexins, flotillin), MVBs biogenesis (Alix and TSG101), heat shock proteins (HSP70 and HSP90), integrins, and tetraspanins (CD63, CD9, CD81, and CD82). Another feature of exosomes is their enrichment in lipids present in rafts, such as cholesterol, sphingolipids, ceramide, and glycerophospolipids with long and saturated fatty-acyl chains.17–19
Microvesicles
Microvesicles bud directly from the plasma membrane and are larger than exosomes, with a size ranging from 100 nm to 1 μm. 20 Shedding of microvesicles is considered a physiological phenomenon that accompanies cell activation, growth, and early apoptosis. A variety of extracellular stimuli are associated with an increase in the secretion of microvesicles including cytokines, endotoxins, hypoxia, oxidative stress, and exposure to shear stress.21–27 Moreover, a variety of intracellular factors influence microvesicle shedding, such as the increase in cytosolic calcium levels and the degradation of the membrane cytoskeleton.28–30 The composition of microvesicles is complex and includes scramblase, cholesterol, and phosphatidylserine in the outer membrane.25,31 Moreover, some microvesicles exhibit an enrichment of proteins associated with membrane lipid rafts.29,32,33
Exosomes and microvesicles interact with target cells through three mechanisms: (1) membrane fusion and subsequent transfer of their cargo; (2) endocytosis and release of cargo; and (3) ligand-receptor interactions.34–36 However, EVs also exert an effect on the ECM through the activity of matrix metalloproteinases (MMPs) embedded in their membrane, such as MMP-2 and MMP-937,38 (Figure 1).

Biogenesis of EVs. Microvesicles bud directly from plasma membrane, whereas exosomes arise from multivesicular bodies. EVs interact with target cells through three mechanism: (1) EVs fusion with transfer of their cargos; (2) EVs endocytosis and release of their cargos; (3) Receptor ligand interaction with activation of signal transduction pathways. Figure created with BioRender.com (2022).
Apoptotic Bodies
Apoptotic bodies are secreted exclusively by apoptotic cells during the late stages of apoptosis and their size varies between 50 nm and 2μm. 39 The content of apoptotic bodies includes nuclear material, cellular organelles, and membrane/cytosolic fractions. The distinctive characteristics of apoptotic bodies include phosphatidylserine in the outer leaflet, a high permeability membrane, irregular shape and densities greater than 1.23 g/ml.40–42
Cancer-associated Fibroblasts
Fibroblasts are the most abundant resident cells of connective tissue, and their main function is the synthesis and maintenance of ECM components including collagen, laminin, fibronectin, and proteoglycans. Moreover, fibroblasts are a heterogeneous population that are predominantly in G0 phase, however they are found in mitosis during tissue remodeling and repair.43,44 CAFs are a group of activated fibroblasts with significant heterogeneity and plasticity in the tumor microenvironment that participate by the secretion of a variety of factors in the regulation of tumor occurrence, development, metastasis, and therapeutic resistance. 44 Harold F. Dvorak stated in 1986 that “tumors are wounds that do not heal” due the similarity of some processes in the tumor microenvironment with those of wound healing, such as, proliferation, angiogenesis, survival, and migration.45,46 In agreement with Dvorak, CAFs share characteristics with myofibroblasts, a very common long-spindled cell type associated with wound healing,47,48 and constitute a significant fraction of cells in the solid tumor stroma. In breast cancer, CAFs represent around 80% of all the stromal cells, 49 and are essential in the tumor microenvironment because they secrete ECM components, cytokines, EVs, growth factors, and other soluble molecules. 50 The purpose of this review is to familiarize readers with aspects related with the communication between breast cancer cells and CAFs via EVs. The role of CAFs in cancer is a broad and very interesting topic, which is outside of the scope of this review. We refer to an excellent review that addresses this topic (Kalluri, 2016 51 ).
EVs from Breast Cancer Cells Promote CAFs Formation
The abundance of CAFs in breast tumors is well known and the role of CAFs in cancer progression has been studied in detail, being propose that CAFs may be useful biomarkers in breast cancer. 52 However, over the last decade, interest has grown with respect to the role of EVs in the development of breast cancer, because a higher number of EVs in breast cancer women compared with healthy controls has been described.53,54 CAFs from breast tumors have different origins, for instance human bone marrow mesenchymal stem cells (BM-MSCs) acquire a CAF phenotype after 30 days of exposition to tumor cell-conditioned media, 55 whereas normal fibroblasts co-cultured with MDA-MB-468 breast cancer cells result in hepatocyte growth factor secretion and the enhancement of tumor growth, a behavior typical of CAFs. 56 Moreover, EVs from breast cancer cells induce the acquisition of a CAFs-like phenotype in normal fibroblasts.57–60 Particularly, breast cancer cells release EVs containing miR-125b, which is a negative regulator of p53, and the transfer of miR-125b to fibroblasts results in the overexpression of markers associated with fibroblast activation, such as Acta2, MMP-2, and MMP-3, which promote a CAFs-like phenotype. 57 Similarly, exosomes released from MDA-MB-231 and MCF-7 breast cancer cells, transfer miR-146a to fibroblasts, which regulates the expression of thioredoxin interacting protein (TXNIP) and activates the Wnt/beta catenin pathway, and then the induction of a CAF phenotype with their recruitment to the tumor site. In contrast, EVs released from non-tumor epithelial cells MCF10A do not induce the acquisition of a CAF phenotype 58 (Figure 2).

Intercommunication between CAFs and breast cancer cells. Activation of breast cancer cells induces the release of EVs that activate normal fibroblasts into CAFs. Reprinted from “Tumor microenvironment with Callout,” by BioRender.com (2022).
It has been demonstrated that other cell types, different from fibroblasts, are able to acquire a CAF phenotype, such as adipocytes and adipose-derived mesenchymal stem cells (ADSCs). Particularly, EVs released from MDA-MB-231 breast cancer cells induce a CAF-like phenotype in ADSCs, which is mediated by activation of TGFβRI, TGFβRII, and SMAD2. ADSCs-like CAF phenotypes are functional and express proteins such as SDF1, VEGF, CCL5, and TGFβ, which act as factors that promote tumor development.
59
In addition, breast cancer cells release exosomes that activate normal fibroblasts and convert them into myofibroblasts (also known as CAFs), which is mediated by surviving, SOD1, actin alpha 2 (ACTA2) smooth muscle and vimentin, and it promotes cancer proliferation and metastasis.
60
In an
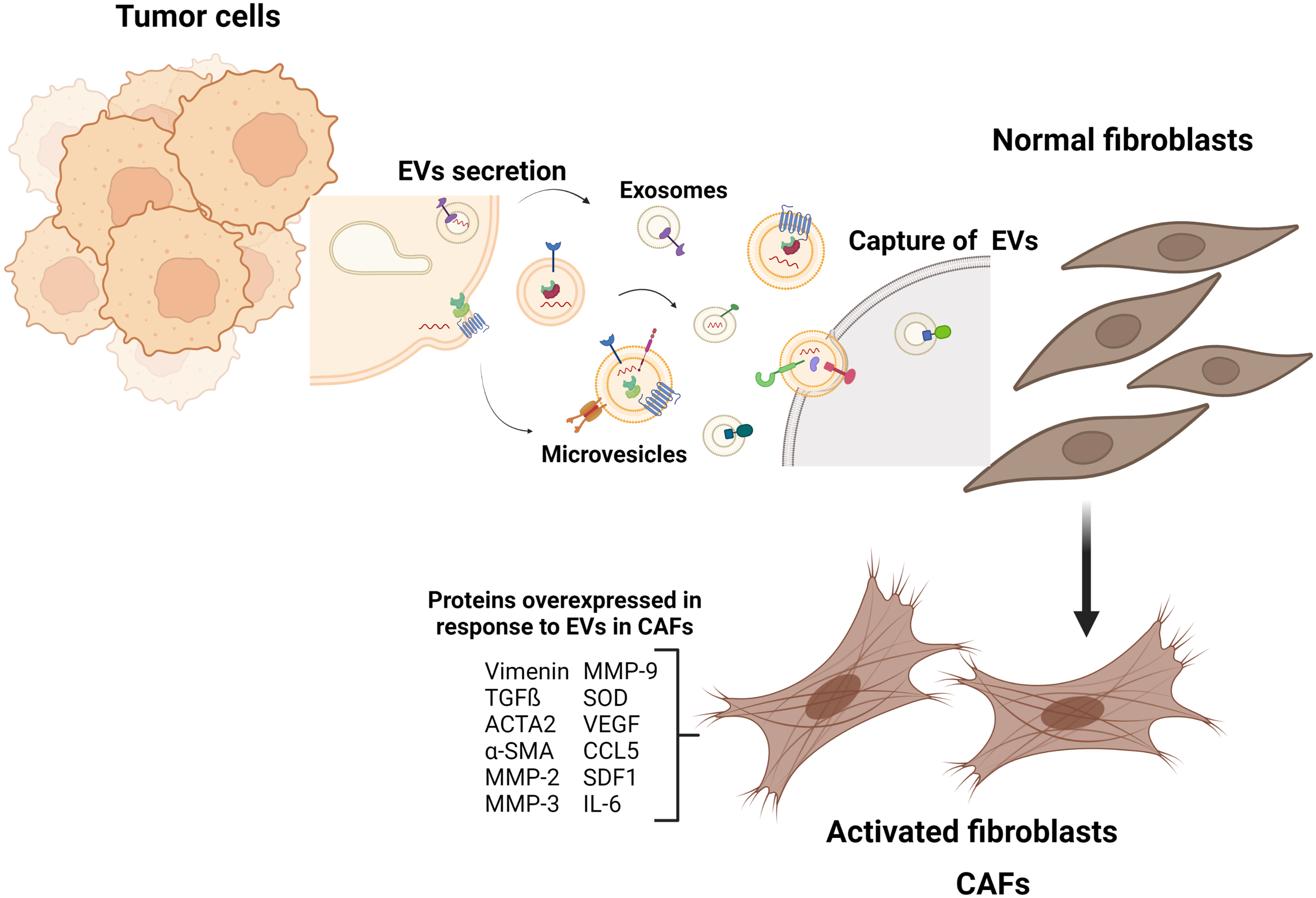
EVs from breast cancer cells induce the acquisition of CAF phenotype. Breast cancer cells release EVs carrying miRNAs, lipids, and proteins that activate normal fibroblasts into CAFs. The CAFs are functional and express proteins that act as factors to promote tumor development. Figure created with BioRender.com (2022).
CAFs-Derived EVs Promote an Aggressive Phenotype in Mammary Tumor Cells
CAFs can induce growth and tumorigenesis in breast cancer.63,64 Tumor development is regulated by the induction of a variety of cellular processes including changes in the phenotype of the tumor cells through the EMT process, which leads to an increase in the aggressiveness of the tumor cells. Proteins and miRNAs carried by EVs can promote neoplastic transformation and are widely involved in the different stages of breast cancer development.55,65,66 The focal adhesion kinase (FAK) is a non-receptor tyrosine kinase and an increased FAK expression and activity are frequently associated with metastatic and poor prognosis in breast cancer.67,68 FAK deletion in
PI3Ks are a family of enzymes involved in various cellular processes including growth, proliferation, differentiation, motility, survival, and intracellular trafficking,72,73 which are implicated in cancer.
72
The expression of the regulatory subunit p85α of PI3K is downregulated in the stroma of late-stage breast cancer tumors and in patients with lymph node metastasis. In an
During metastasis, cancer cells spread from their origin to distant parts of the body. The most common sites of breast cancer metastases are bone, brain, liver, and lung.74,75 CAFs-secreted exosomes enhance metastasis in breast cancer. Exosomes released from breast cancer cells MDA-MB-231 and MCF-7 transfer miR-146a to fibroblasts, which induces the activation into CAFs, whereas CAFs transformed by EVs promote migration and invasion in MCF-7 breast cancer cells through miR-146a, which activates the Wnt pathway. EVs released from breast cancer cells MDA-MB-231 and MCF-7 are associated with an increase in lung metastasis and proliferation in a model
The transfer of miRNAs expressed at low levels is also involved in promotion of tumor development. Treatment of MDA-MB-231 breast cancer cells with CAFs-derived EVs expressing low levels of miR-30e improves the viability, migration, and invasion.
78
Particularly, miR-30e negatively regulates the collagen triple helix repeat-containing protein 1 (CTHRC1), via the Wnt/β-catenin pathway, and the overexpression of CTHRC1 increases the migratory capacity of cells.78,79 The miR-30e downregulation and overexpression of CTHRC1 are found in breast cancer patients and in tumor-bearing mice formed with tumor cells treated with CAFs-derived EVs.
78
In an orthotopic mouse model of breast cancer, coinoculation of MDA-MB-231 breast cancer cells with fibroblasts increase tumor cell metastasis, whereas in an
Primary tumors promote metastasis by the formation of a supportive microenvironment at a secondary organ site, termed the pre-metastatic niche. Interestingly, the pre-metastatic changes in the pre-metastatic niche may be mediated by EVs.83,84 EVs secreted by breast cancer cells can travel to normal lung tissue cells, the metastatic niche, in an orthotopic nude mouse model of breast cancer.
85
Using an
Hypoxia is one of the main features of solid tumors and is associated with poor prognosis in breast cancer. 87 Exosomes released from MCF-7 cells that were subjected to hypoxic conditions express high levels of ApoE, CAIX, miR27b, and miR130B, which slightly increase the formation of mammospheres. In addition, exosomes released from hypoxic MCF-7 cells induce CAFs phenotypes with secretion of IL-6, which is mediated by PPARγ/RXR. However, treatment with the PPARγ/RXR receptor agonists, PGZ and IIF, partly inhibit mammosphere formation and Notch3 expression in MCF-7 cells. 62
A limited number of studies about the tumor suppressive properties of CAFs have been published. It is clear that CAFs can suppress tumor growth during the early stages of tumor progression, but they can support tumor progression at later stages in parallel with the dual role of TGF-β.88–90 However, the molecular mechanisms that decrease tumor growth are not fully understood. Exosomes released from mesenchymal stem cells (MSCs) are transferred to MDA-MB-231 breast cancer cells and downregulate the expression of VEGF and VEGFR-1 through the transfer of miR-100, which inhibit the expression of mTOR in conjunction with HIF-1α. Therefore, exosomes from MSCs induce changes in conditioned medium of MDA-MB-231 cells, and partly inhibit the migration and formation of angiogenesis-like tubules in endothelial cells. 91 Moreover, miR-1-3p is downregulated in breast tumor tissue and is associated with metastasis, whereas miR-1-3p is also downregulated in CAFs-derived EVs isolated from breast cancer patients. The breast cancer cell lines MDA-MB-231, SKBR3, and MCF-7 cells express low levels of miR-1-3p. In contrast, EVs released from CAFs overexpressing miR-1-3p decrease growth and metastasis in breast tumors through inhibition of EMT and proliferation. Interestingly, miR-1-3p downregulates the expression of GLIS gene, which is a promiscuous transcription factor that regulates expression of numerous genes. 92
CAFs-Derived EVs Regulate the Metabolism of Mammary Tumor Cells
Metabolic reprogramming is an important hallmark of malignant tumors, which may be regulated by the microenvironment. Changes in metabolism in the different areas of a solid tumor have been demonstrated to occur in various malignancies, including breast cancer. 93 Alterations in protein expression are associated with metabolic changes in tumor cells and the intercommunication between CAFs and tumor cells is involved with these metabolic changes93,94 Particularly, exosomes from MDA-MB-231 breast cancer cells show high levels of integrin beta 4 (ITGB4), which can be transferred to CAFs with an increase in glycolysis and lactate production. Moreover, the transfer of ITGB4 promotes the expression of mitochondrial fission proteins, such as FIS1, and the phosphorylation of dynamin-like protein 1 (DBP1) in CAFs, whereas stimulation with conditioned media of CAFs, overexpressing ITGB4, increases proliferation, migration and EMT in breast cancer cells. 94 A comparative analysis of proteins in exosomes from MSCs and exosomes from breast cancer cells MDA-MB-231 and MCF-7 demonstrate that MMP-2 and fibronectin are expressed in exosomes from MSCs, but they are not expressed in exosomes from MDA-MB-231 and MCF-7 cells. However, exosomes from MSCs transfer an active form of MMP-2 to MDA-MB-231 and MCF-7 cells and it promotes tumor invasion. 95
CAFs release EVs that transmit miRNAs and lncRNAs into adjacent tumor cells to modulate their behavior. CAFs and breast cancer cells undergo reciprocal metabolic reprogramming through horizontal gene transfer via EVs. 96 The overexpression of miR-105 in breast cancer cells enhances vascular permeability and induces metastasis, whereas the transfer of miR-105 via exosomes disrupts tight junctions and the integrity of endothelial monolayers, and it promotes breast cancer metastasis. 97 MDA-MB-231 cells release EVs expressing miR-105, a miRNA induced by MYC, which is transferred to patients-derived CAFs, and it promotes changes in glucose and glutamine metabolism with production of glutamate and lactate, compounds that nourish adjacent tumor cells. Furthermore, CAFs expressing miR-105 are able to detoxify cells for the conversion of lactic acid and ammonium into energy-rich metabolites, which nourishes tumor cells, and these CAFs promote tumor growth in NOD/SCID/IL2R-null (NSG) mice. These findings demonstrate a bidirectional communication between tumor cells and CAFs, which modify the microenvironment and contribute to tumor feeding and growth. 98
Glycolysis is one of the main energy production pathways in mammalian mammary cells. EVs released from CAFs expressing low levels of miR-7641 promote the formation of a stem cell phenotype and metabolic changes including an increase of glycolysis in MDA-MB-231 and SKBR3 tumor cells through activation of HIF-1α pathway.
99
CAFs-derived exosomes decrease the rate of oxygen consumption, mitochondrial function, lactate levels, and increase the rate of extracellular acidification in MCF-7 and MDA-MBA-453 breast cancer cells. Moreover, inhibition of SNHG3 expression, a noncoding RNA of more than 200 nucleotides, in CAFs-derived exosomes restores lactate production in target cells by reduction of miR-330 levels, whereas CAFs-derived exosomes expressing SNHG3 promote proliferation and dysregulate mitochondrial function in breast cancer cells. The miR-330 acts on pyruvate kinase (PKM) isoforms and promotes glycolysis in MDA-MBA-453 breast cancer cells.98–100 In contrast, it has been described different metabolic regulation mediated by miR-122. EVs from MDA-MB-231 cells and breast cancer patients express high levels of miR-122, and these EVs significantly inhibit the uptake of 2-NDBG, a fluorescent glucose analog, in fibroblasts compared with EVs from healthy controls.
101
Moreover, an increase of miR-122 expression in fibroblasts decreases the expression of PKM2 and the glucose transporter (GLUT1), and it decreases glucose uptake of CAFs and increase the availability of glucose for breast cancer cells. An
CAFs-Derived EVs Modulate the Immune Response in the Tumor Microenvironment
Many tumors develop mechanisms of immune evasion to survive, including secretion of tumor immunomodulatory proteins, reduced expression of antigen-presenting proteins and EVs secretion.102,103 Exosomes released from breast cancer cells can influence the immune system by interacting with T cells, natural killer (NK) cells, and macrophages.104–106 Cancer cells express the natural killer group 2 member D (NKG2D) receptor and its activation promotes the cytotoxic effect of NK cells, and provides a costimulatory signal on T cells. Exosomes are able to mediate immune evasion by downregulation of NKG2D receptor expression in the effector cells including breast cancer cells. 107 CAFs, isolated from breast tumors and adjacent tissues, secrete exosomes expressing high levels of miR-92, a miRNA implicated with the nuclear translocation of the yes-associated protein 1 (YAP1), which is a regulator of protein programmed death ligand 1 (PD-L1). 108 PD-L1 is a protein that prevents the immune cell attack to healthy cells, and its overexpression promotes T cell tolerance and escape from the host immune system during tumorigenesis. 109 Exosomes from CAFs modulate the expression of PD-L1 in MCF-7 breast cancer cells and induce upregulation of PD-L1 in NK and T cells, which promote apoptosis and a decrease in proliferation in T cells. 108 In summary, the intercommunication between CAFs and tumor cells modulate the immune response and contributes to tumor progression via EVs.
CAFs-Derived EVs Modulate Composition of ECM
Breast tumors have excessive production of connective tissue and up to 90% of mass may consist of stroma, which presents alterations in density and composition of ECM. 45 The miR-9 promotes proliferation, invasion, migration, and EMT in breast cancer cells and is associated with poor overall survival in breast cancer patients.110,111 Moreover, miR-9 is overexpressed in CAFs from TNBC tumors, but it is not overexpressed in CAFs from luminal A and luminal B breast tumors. EVs from TNBC MDA-MB-231 cells express miR-9, which is transferred to normal fibroblasts and regulates the expression of genes that are involved in the remodeling of ECM, including EGF containing fibulin extracellular matrix protein 1 (EFEMP1), collagen type I alpha 1 (COL1A1), and MMP-1. In addition, EVs from fibroblasts containing high levels of miR-9 are captured for MDA-MB-231 cells and stimulate migration and EMT process. 112 EVs depleted of FN30, a negative regulator of fibronectin expression, from Ca1a mammary mesenchymal cells promote that normal lung fibroblasts (HLF) increase fibronectin accumulation and displacement of the linear architecture of matrix, whereas these effects are not induced by EVs depleted of FN30 from Ca1h epithelial cells. 113
The alterations of stiffness and degradation of ECM contribute to tumor growth, and CAFs play a pivotal role in ECM composition. 114 Microvesicles released from MDA-MB-231 breast cancer cells promote fibroblast activation, proliferation, spreading, and stiffness on matrices, however these effects are only reported in a rigid matrix that mimics mammary tumor tissue. Likewise, tumor cell-derived microvesicles induce an increase in the traction force of fibroblasts on a rigid matrix that mimics mammary tumor tissue. Interestingly, the biological effects described above are primarily induced by microvesicles from invasive MDA-MB-231 breast cancer cells, however some effects have been demonstrated using microvesicles from non-invasive MCF-7 breast cancer cells, whereas microvesicles from mammary non-tumorigenic epithelial cells MCF10A are not able to induce these biological effects. 115
Chemoresistance in Tumor Cells is Modulated by CAFs-Derived EVs
EVs are important players in breast cancer chemoresistance. 116 Exosomes carrying HER-2 play a pivotal role in the inhibition of trastuzumab activity, 117 whereas exosomes carrying P-glycoprotein (P-gp) are proposed as another mechanism of exosomes-mediated drug resistance in breast cancer. 118 Moreover, exosomes from MCF-7 breast cancer cells resistant to tamoxifen promote proliferation of wild-type MCF-7 cells in the presence of tamoxifen, which is mediated by the transfer of miR-221 and miR-222. 119
The communication between tumor and stromal cells promotes resistance to hormone therapy in breast cancer.120,121 Estrogen receptor-positive tumors are metabolically dependent on oxidative phosphorylation rather than glycolysis,
122
whereas mitochondrial DNA (mt-DNA) is encapsulated in EVs and it can be horizontally transferred to low metastatic cells and stromal cells in the tumor microenvironment.
123
Patients with hormone therapy-resistant breast cancer show high levels of mt-DNA expressed in EVs from their plasma and the mitochondrial oxidative phosphorylation system (OXPHOS) is increased in the tumors of these patients, and when oxidative phosphorylation is inhibited in tumor cells, the cells are re-sensitized to hormonal therapy.
124
An
Endogenous RNAs under pathophysiological conditions, such as cancer, may act as damage-associated molecular patterns (DAMPs) to activate pattern recognition receptors (PRRs).125,126 A relationship between TNBC 1833 cells, a metastatic cell line obtained from MDA-MB-231 cells that respond to interferon-stimulated genes (ISGs), and cells of the stroma, which mediate communication via exosomes secretion, has been established. CAFs-derived exosomes transfer and increase the acquisition of stromal RNAs in 1833 cells compared with MCF-7 estrogen receptor-positive breast cancer cells that do not respond to ISGs. An analysis of genetic material expressed in exosomes demonstrates an enrichment of noncoding RNAs, including increased levels of 5'-PPP-RNAs.
127
Likewise, the RN75L1, a 5'-PPP-RNA, is found at high levels in CAFs-derived exosomes and its expression is regulated by NOTCH-MYC signaling pathway. Furthermore, the transfer of RN75L1 to tumor cells via exosomes induces the activation of the RIG-1 receptor, which is a PRR. The binding of RN75L1 to RIG-1 occurs in the absence of RNA-binding proteins, which generate an unshielded RN7SL1 in stroma exosomes, and then RN7SL1 acts as a DAMP. An
Cancer stem cells (CSCs) play an important role in the tumor microenvironment, 128 and enrichment of CAFs and CSCs is described in tumor xenografts and tissues of breast cancer patients resistant to hormone therapy. In a model of luminal breast cancer, the transfer of EVs released from CAFs containing increased levels of miR-222 and miR-221 promotes resistance to hormone therapy in MCF-7 cells, whereas this effect is not induced by EVs from normal fibroblasts. Furthermore, treatment with EVs from CAFs in combination with hormone therapy results in an increase in CSCs, a decrease in estrogen receptor expression and upregulation of Notch. 129 The biogenesis of miR-221 is mediated by STAT3/IL-6 pathway and inhibition of this pathway decreases the metastasis of tumor stem cells and low formation of metastatic niches. 129 In summary, the transfer of miR-222 into tumor cells through EVs from CAFs induces resistance to hormonal therapy and increase the number of CSCs, which promote the progression of the tumor (Table 1).
Summary of miRNAs in EVs Associated with CAFs in Breast Cancer.

EVs from Breast Cancer Cells and CAFs as Biomarkers
Biomarker is a biological molecule found in blood or other fluids or tissues that can be used for screening, diagnostic and/or prognosis of a disease. 130 In breast cancer, biomarkers involve a wide range of biochemical entities found in body fluids including the components of EVs from tumor cells. 131 In this regard, it has been proposed that specific miRNAs or proteins expressed in EVs could be used as biomarkers of breast cancer. 132 A study shows that EVs express a significant increase of phosphoproteins in breast cancer patients compared with healthy controls, and the expression of these phosphoproteins has been associated with membrane reorganization, intercellular communication and metastasis. 133 Particularly, the phosphoprotein FAK is expressed in EVs from breast cancer cells or CAFs, and it represents a good candidate to be evaluated as biomarker. 69 In addition, RALGAPA2, PKG1, and TJP2 are significantly elevated in EVs from breast cancer patients compared with healthy controls, and it has been suggested that these proteins are candidates for breast cancer markers. 133
We previously mentioned that miRNAs can be transferred via EVs to exert their functions. Particularly, miR-9 regulates and actively participates in tumor progression.
111
The miR-9 can be transferred via EVs to normal fibroblasts, which promotes their conversion into CAFs, with downregulation of genes involved in ECM remodeling, and then enhancing the motility of breast cancer cells.110,134 Therefore, miR-9 is a potential biomarker of tumorigenesis and metastasis in breast cancer. Moreover, an
EV cargos are specific to their parental cells, and a growing number of studies propose that EV cargos may be used as biomarkers for breast cancer, because EVs are very stable and circulate in blood and other body fluids, Therefore, EVs in the different body fluids represent the liquid biopsies. Liquid biopsies allow the analysis of proteins and miRNAs expressed in EVs and emerge as biomarkers for early detection, diagnosis and prognosis in breast cancer.131,132 However, further studies are required to establish the EV components as biomarkers of breast cancer.
Conclusion
In summary, current evidence indicates that communication between breast cancer cells and CAFs via EVs represents a novel mechanism that promotes migration, invasion, immune evasion, drug resistance, EMT, and modulates the ECM composition and promotes metastasis in breast cancer (Figure 4).

CAFs transfer their cargos to breast cancer cells through EVs supporting tumor growth and the acquisition of an aggressive phenotype. Tumor development is regulated by EVs through the induction of invasion, proliferation, migration and EMT. CAFs release EVs that mediate changes in metabolism, immune modulation and chemoresistance in breast cancer cells, and modulate the expression of ECM components. Figure created with BioRender.com (2022).
Footnotes
Abbreviations
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. Rocio Castillo-Sanchez was supported by a grant from CONACYT-Mexico.
