Abstract
Introduction
Endometrial cancer (EC) is the most common cancer of uterine cancer, and defined as the fifth leading cause of cancer death in the United States. 1 In 2020, there were an estimated 79 420 new women are diagnosed with uterine cancer and 16 880 deaths in the United States. 2 Approximately 80% of EC patients developed symptomatic presentation and the 5-year survival rate of these cases was 75% to 90%. 3 However, approximately 20% of EC patients are diagnosed with metastatic or recurrent, and these kinds of cases led to considerably poorer outcomes with a relative 5-year survival rate of 9% to 17%. 4 Therefore, it is imperative to elucidate new molecular targets and therapeutic approaches for EC.
NOD-like receptor family caspase recruitment domain family domain-containing 5 (NLRC5) is a newly identified member of the NLR family. 5 Human NLRC5 is located in the 16q13 locus and consists of 1866 amino acids (aa) while mouse NLRC5 is at chromosome 8 and contains 1915 aa. NLRC5 possesses 3 structural domains including the N-terminal atypical caspase activation and recruitment domain (CARD), which is completely distinct from the other NLRs, the central NACHT domain, which contains the nucleotide-binding domain (NBD) and 27 leucine-rich repeats (LRRs) at the C-terminal. 6 It has been demonstrated that NLRC5 is a negative regulator of inflammatory response and type I interferon (IFN) production, and an activator and synergetic component of the inflammasome. 6 Moreover, NLRC5, known as an major histocompatibility complex (MHC) class I transactivator, is a novel target for immune evasion in cancer. Downregulation of NLRC5 in cancer is crucially correlated with decreased levels of MHC class I molecules and impaired cytotoxic T cell activities. Recruitment of NLRC5 contributes to the presentation of tumor antigens to CD8+ T cells, which further increases the antitumor immunity. 7 Intriguingly, in several cancer cells, accumulation of NLRC5 contributes to cancer progression by promoting cancer cell proliferation, migration, and invasion.8,9 This interesting and even conflicting evidence might arise from the fact that the exact function of NLRC5 could be cell type- and tumor microenvironment-dependent. Our previous study in EC cells showed that NLRC5 may contribute to EC progression by promoting cell migration and invasion. 10 Nevertheless, the mechanism underlying NLRC5 in EC progression needs further determined. The programmed cell death ligand 1 (PD-L1) receptor is one of the important immune checkpoint proteins and is mainly expressed on mature cytotoxic T lymphocytes in the tumor microenvironment. PD-L1 is also expressed on cancer cells 11 and tumor intrinsic signaling pathways, 12 and restricted by the tumor suppressor genes. 13 Accumulating evidence shows that high expression of tumor cell-intrinsic PD-L1 contributes to cancer initiation, metastasis, development, and recurrence in multiple tumor types.14,15 However, the mechanism related to NLRC5 downregulation in EC patients remains unknown, and whether NLRC5 promotes EC progression by regulating PD-L1.
In our study, we detected the expressions of NLRC5 and PD-L1 in the endometrium tissue microarray. Furthermore, we analyzed the expression correlation between NLRC5 and PD-L1. We also determined the correlation of expression levels of NLRC5 and PD-L1 with different clinicopathologic features in EC patients. In addition, we explore whether NLRC5 could regulate the tumor cell-intrinsic PD-L1 in EC cell lines. Lastly, we explored the potential mechanism related to downregulated NLRC5 in EC patients. Our evidence presented that NLRC5 was downregulated and PD-L1 was upregulated in EC. Furthermore, there is a negative correlation between the expressions of NLRC5 and PD-L1. Additionally, NLRC5 promotes the expression of PD-L1 in EC cells. The mutations of ANKRD20A2, C2orf42, ADGRB3, AVPR2, GOLGA6C, and IPPK may lead to downregulation of NLRC5 in EC patients.
Materials and Methods
Patients and Tissues Microarray
The Anhui Medical University's Institutional Review Board approved the current study (Approval No.: 20180023), and all patients provided written informed consent. A tissue microarray composed of 60 EC endometrial tissue samples and 36 control endometrial tissues was analyzed. Clinical data collected for all EC patients included histological subtype, lymph node metastasis, tumor grade, tumor stage, and age. The 2009 Federation International of Gynecology and Obstetrics (FIGO) criteria were used to validate tumor staging. The World Health Organization (WHO) criteria for histological subtyping and tumor grading were followed. Age and body mass index (BMI) were collected for all 36 participants from who normal endometrial tissue samples were collected. Participants had not undergone chemotherapy, radiotherapy, immunotherapy, or other treatments before sample collection.
Immunohistochemistry
PD-L1 and NLRC5 expression levels were detected via immunohistochemistry as in prior studies. Antigen retrieval was accomplished by microwaving tissue microarray samples for 15 min in a citric saline solution after deparaffinization with xylene and dehydration with ethanol. After 15 min of treatment with 0.3% H2O2 to quench endogenous peroxidase activity, sections were blocked with 2% bovine serum albumin (BSA) and incubated overnight at 4 °C with anti-NLRC5 antibodies (ab105411, Abcam, 1:100) or PD-L1 (ab213524, Abcam, 1:100). Sections were then washed, probed for 1 h at room temperature with a biotinylated secondary antibody (G1210-2-A, Servicebio), and protein detection was then visualized with 3′-diaminobenzidine tetrahydrochloride. Following hematoxylin counterstaining, slides were dehydrated, mounted, and 5 random fields of view per slide were imaged for quantification (200x magnification) using a fluorescent microscope. Background lighting was consistent for all samples. Image-Pro Plus 6.0 (Media Cybernetics, Inc.) was used to examine staining intensity and found that dark brown staining indicated a positive staining reaction.
Cell Culture
The American Type Culture Collection (ATCC) provided the HEC-1A human EC cell line (Accession number: HTB-112), while the European Collection of Authenticated Cell Cultures supplied the Ishikawa cells (Accession number: 99040201). At 37 °C in a 5% CO2 incubator, cells were cultured in Roswell Park Memorial Institute (RPMI)-1640 (Invitrogen) containing 10% fetal bovine serum (FBS, HyClone).
Immunofluorescence Staining
HEC-1A and Ishikawa cells were permeabilized for 20 min with 0.1% Triton X-100 (Thermo Fisher Scientific) after being fixed for 30 min with 4% paraformaldehyde. Samples were then stained for 45 min at 37 °C with primary anti-NLRC5 and anti-PD-L1 antibodies, rinsed (5 min/wash at room temperature) with an immunohistochemical washing solution (Beyotime), and stained with appropriate secondary antibodies for 45 min at 37 °C. Immunofluorescence blocking solution (Sigma) was applied after 3 further washes as described above, and slices were mounted and photographed using laser scanning confocal microscopy (Nikon).
Cell Transient Transfection
The sequences of NLRC5 plasmid and siRNA-NLRC5 was performed as our previous study. 16 HEC-1A and Ishikawa cells were transfected with these constructs using Lipofectamine™ 2000 (Invitrogen) based on provided directions.
Western Blotting Analysis
To lyse cells, a protein extraction solution (Beyotime) was employed, followed by a bicinchoninic acid (BCA) assay kit (SinoBio Biotech) to determine protein quantities. Following centrifugation, lysates were transferred to polyvinylidene fluoride (PVDF) membranes (Millipore Corp). After blocking with 5% nonfat milk in tris buffered saline tween-20recipe (TBST) for 4 h at 37 °C, the blots were treated overnight with PD-L1 (ab213524, Abcam, 1:1000), anti-NLRC5 (ab105411, Abcam, 1:1000), and β-actin (GB12001, Servicebio, 1:5000) at 4 °C in primary antibody dilution buffer (Beyotime), washed 3 times using TBST, and probed at 37 °C for 1 h with an horseradish peroxidase (HRP)-conjugated secondary antibody (1:10 000). Protein bands were identified using an ECL kit (ECL-plus, Thermo Scientific) after the blots were washed 3 times with TBST. Analyses were repeated in triplicate a minimum of 3 times.
Whole-exome Sequencing (WGS)
From 3 EC patients, the genomic DNA was extracted from peripheral blood samples. All gDNA samples met the purity requirements (OD260/280 > 1.8) and concentration levels (50 ng/mL) necessary for sequencing. Upon fragmentation with NEBNext dsDNA Fragmentase into 100 to 800 bp segments with a peak size of ∼250 bp, samples were treated to end repair, dA-Tailing, and adaptor ligation with the Illumina NEBNext DNA Library Prep Reagent Set (acquired from New England Biolabs). DNA fragments were separated using a 2% agarose gel electrophoresis following adaptor ligation, and 300 to 400 bp fragments were removed for further study. Ten cycles of PCR amplification with PE primers (Illumina) and Phusion DNA polymerase (New England Biolabs) were performed on the samples. The amplified sequences were then utilized to capture exome sequences using an Illumina Truseq Exome Enrichment kit V6, which contained a 31.3 Mb CCDS (97.2% of the US NCBI CCDS Library) region covering approximately 20 794 genes within 62 Mb of coding exons (Illumina). After enrichment, 10 additional rounds of PCR amplification were performed as above, after which the sizing of amplicons was checked and a BioAnalyzer 2100 was used to quantify amplicon concentrations. The amplicons weresize-checked and quantitated using a BioAnalyzer 2100, and then subjected to 2 ×150 bp paired-end massively parallel sequencing using a Hiseq2500 SequencingSystem (Illumina).
Statistical Analysis
All data were analyzed using SPSS 23.0 (IL, USA), which is provided as means ± SEM and analyzed using one-way ANOVA tests. To find significant variations across groups, Duncan's multiple range tests were performed and to investigate correlations between samples and Pearson’s correlation analysis was utilized. P < .05 was the significance threshold.
Results
General Data
Endometrial tissues obtained from 60 patients with EC and 36 normal endometrial tissues were used for the tissue microarray. Demographic characteristics are summarized in Table 1. The 2 groups were matched for age (t = 1.169, P = .245) and BMI (t = 1.601, P = .113). Among the EC patients, 49 (81.7%) were postmenopausal and 11 (18.6) were premenopausal, and endometrioid histology was detected in 51 (85.0%) patients, whereas serous histology was detected in 9 (15.0%) patients. According to the FIGO criteria (2009), 20 (28.0%) patients had stage I EC, 28 (46.7%) had stage II EC, 10 (16.7%) had stage III EC, and 2 (3.3%) had stage IV EC. Furthermore, 54 (90.0%) patients were in the G1-G2 stage, 50 (83.3%) patients showed myometrial invasion, and 6 (10.0%) patients possessed lymph node metastasis.
Demographic Characteristics of Normal Endometrium and Endometrial Cancer (EC) Endometrium.

Data are expressed as mean ± standard deviation, unless specified otherwise.
Abbreviation: FIGO, Federation International of Gynecology and Obstetrics.
Comparisons of the Levels of NLRC5 and PD-L1 in the Normal Endometrium and the Endometrium of EC Patients and Their Correlations With the Clinicopathologic Features of EC Patients
Immunohistochemistry analysis of tissue microarrays revealed that the NLRC5 expression in the endometrium of EC patients was significantly lower than that in the normal endometrium (t = 5.053, P < .001), and PD-L1 was upregulated in the endometrium of EC patients when compared to that in normal endometrium (t = 8.704, P < .001; Figure 1A, Table 2). No correlation was found between the level of NLRC5, PD-L1 and histology, myometrial invasion, lymphatic node metastasis, FIGO stage (2009), and histological grade in EC patients (Table 3). Furthermore, there is no correlation between NLRC5, PD-L1, and cumulative survival in EC patients (Figure 1B).

Expression of NLRC5 and PD-L1 in the endometrium of control and EC patients. (A) Endometrium of control group and EC patients were subjected to immunohistochemistry analysis for NLRC5 and PD-L1 levels (scale bars, 100 μm). NLRC5 level was downregulated in the endometrium of EC patients in comparison to those in the normal endometrium of control group. PD-L1 level was upregulated in the endometrium of EC patients in comparison to those in the normal endometrium of control group. (B) Correlation of NLRC5 and PD-L1 levels with the cumulative survival of EC patients were detected by Kaplan–Meier analysis.
The Levels of NLRC5 and PD-L1 in Normal Endometrial Tissues and Endometrial Cancer (EC) Endometrial Tissues.

Data are expressed as mean ± standard deviation. There are statistically significant differences between any 2 groups (using Bonferroni’s method).
Abbreviations: NLRC5, NOD-like receptor family caspase recruitment domain family domain-containing 5; PD-L1, programmed cell death protein ligand 1.
Correlation of Mean Expression Levels of NLRC5 and PD-L1 with Different Clinicopathologic Features in EC Patients.

Correlation of mean expression levels of NLRC5 and PD-L1 with clinicopathologic features by t-test and F-test.
Abbreviations: EC, endometrial cancer; NLRC5, NOD-like receptor family caspase recruitment domain family domain-containing 5; PD-L1, programmed cell death protein ligand 1.
NLRC5 Promotes PD-L1 Expression in EC Cells
Immunofluorescence staining analysis results showed that NLRC5 and PD-L1 are co-located in the cytoplasm of HEC-1A and Ishikawa cells (Figure 2A). There is a negative expression correlation between NLRC5 and PD-L1 (r = −0.666, P < .001; Figure 2B). Moreover, western blotting analysis results showed that NLRC5 plasmid could promote NLRC5 protein expression effectively in HEC-1A cells (Figure 2C). Overexpression of NLRC5 led to PD-L1 protein being upregulated in HEC-1A and Ishikawa cells (Figure 2D). siRNA-NLRC5 could inhibit NLRC5 protein expression effectively in HEC-1A cells (Figure 2E). Downregulation of NLRC5 inhibited PD-L1 protein expression in HEC-1A and Ishikawa cells (Figure 2F). These results indicated that NLRC5 played a positive role in PD-L1 expression in EC cells.
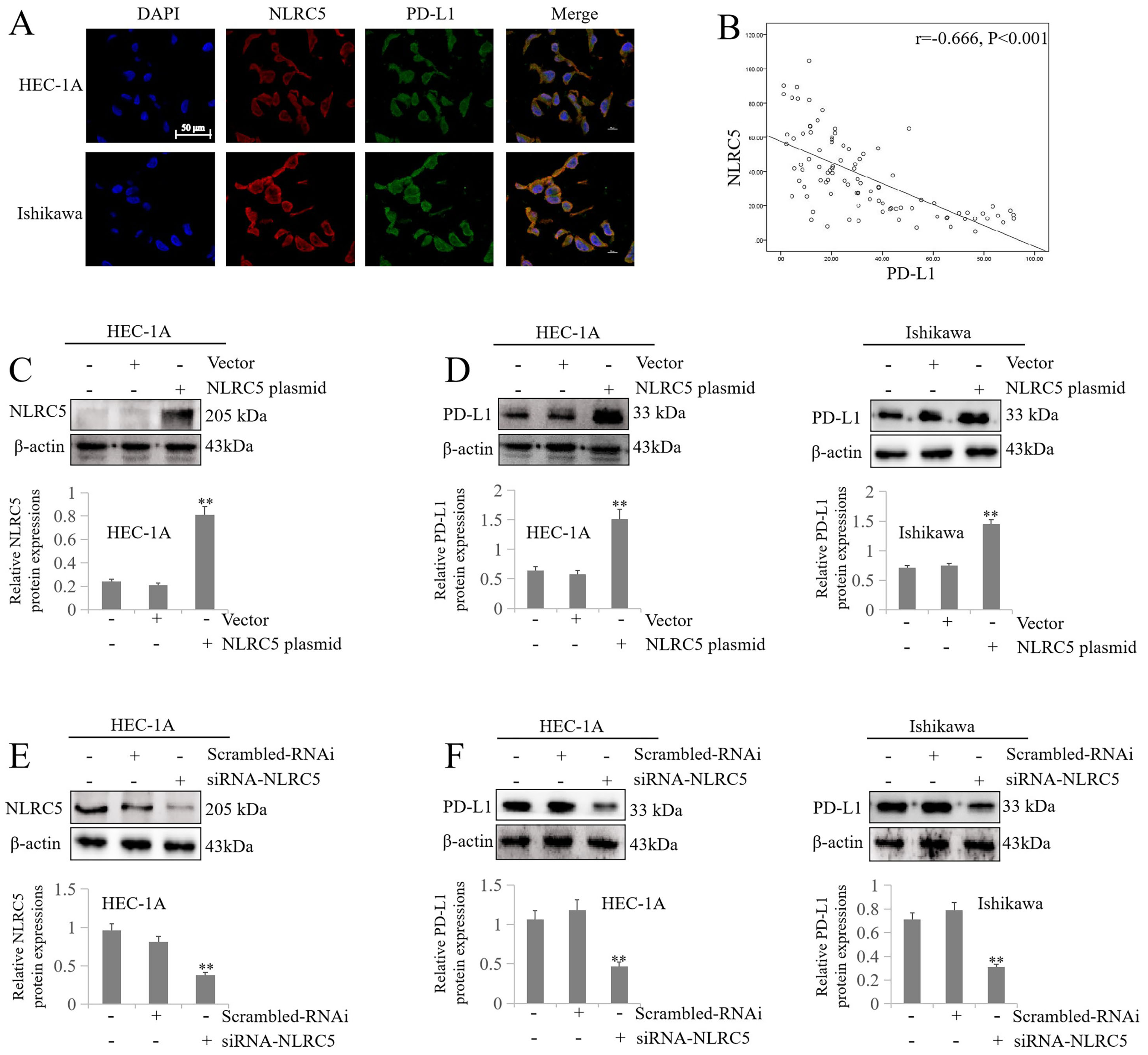
NLRC5 positively regulates the expression of PD-L1. (A) Representative image of immunofluorescence staining show co-localization of NLRC5 and PD-L1 in the cytoplasm of HEC-1A and Ishikawa cells (scale bars, 50 μm). (B) Correlation analysis between NLRC5 and PD-L1 (r = −0.666, P < .001). (C, D) Representative image of western blotting results show that the overexpression of NLRC5 promotes the levels of NLRC5 and PD-L1 HEC-1A and Ishikawa cells (**P < .01 vs vector group). (E, F) Representative image of western blotting results show that the inhibition of NLRC5 downregulates the levels of NLRC5 and PD-L1 HEC-1A and Ishikawa cells (**P < .01 vs Scrambled-RNAi group).
Mutations of ANKRD20A2, C2orf42, ADGRB3, AVPR2, GOLGA6C, and IPPK may Lead to Downregulation of NLRC5 in EC Patients
To assess the mutational statuses of the EC patients, WGS was performed for the 3 EC patients and their own peripheral blood, which was set as control group. The top 10 mutated genes were shown in Figure 3A. However, we did not find somatic mutations of NLRC5 in the samples. Furthermore, the mutation type was shown in Figure 3B. Additionally, using the muTarget database (http://www.mutarget.com/), we suggested that mutations of ANKRD20A2, C2orf42, ADGRB3, AVPR2, GOLGA6C, and IPPK may lead to the downregulation of NLRC5 in EC patients (Figure 4).

Mutational statuses of the EC patients (n = 3). (A) Summary of the top 10 mutated genes in EC patients. (B) Summary of mutation type in EC patients.

muTarget database (http://www.mutarget.com/). Mutations of genes are correlated with downregulation of NLRC5.
Discussion
EC cases are broadly classified into type I tumors, which express moderate-to-high levels of estrogen receptor (ER) and account for ∼90% of all EC cases, and type II tumors, which are estrogen-independent and generally more severe with respect to their clinicopathological characteristics. 17 Histological classification of EC tumors, however, is subject to relatively poor reproducibility with overlapping IHC and morphological features among different EC subtypes. 18 The Proactive Molecular Risk Classifier for EC instead reclassified EC tumors based on IHC findings into p53 WT, p53 abnormal, polymerase ε-mutated, and mismatch-repair-deficient, with these subtypes being better able to predict patient tumor responses to immunotherapeutic treatment. 19 Clinical immunotherapy trials have explored the use of immune checkpoint inhibitors for the treatment of EC, 20 but most patients were found to exhibit unsatisfactory responses owing to the incidence of immune escape. 21 PD-L1 is a key immune checkpoint that can mediate immune evasion in a broad range of cancers. Moreover, PD-L1 can serve as an oncogene to enhance initial tumor development, growth, and metastatic progression through the activation of intrinsic signaling pathways within these tumor cells.15,22 The expression of PD-L1 on the surface of tumor cells enables them to more effectively evade antitumor immune responses, 23 interacting with PD-1 expressed on T cells surfaces to restrict the effector functions of these cells, thus shielding tumors from immune-mediated rejection. 24 PD-L1 inhibitor treatment has been linked to improved outcomes in many cancers, including in EC, which indicated that PD-L1 represents a promising target for immunotherapy in EC.25,26 Overall, PD-L1 expression has been reported in 83% of primary EC cases and 100% of metastatic EC cases. 27 Here, significantly increased PD-L1 expression was detected in EC patients when compared to normal endometrial tissues. As a result, PD-L1 overexpression may contribute to EC formation, invasivity, metastasis, and/or immune evasion. The specific mechanisms underlying PD-L1-mediated immune evasion, however, remain to be fully clarified.
NLRC5 serves as an MHC class I transactivator that modulates MHC class I-dependent immunity. Yoshihama et al analyzed 7747 patients with 21 different types of solid cancers and determined that NLRC4 represents a potential immune evasion-related target, with the upregulation of this molecule being correlated with augmented MHC class I expression, CD8+ T cell activation, and improved survival outcomes in cancer patients. 7 From a mechanistic perspective, somatic mutations, copy number deletion, and promoter methylation can drive reductions in NLRC5 expression or activity, whereas NLRC5 recruitment can restore the immunogenicity of tumors and drive enhanced antitumor immune responses by rescuing MHC class I expression and thereby improving tumor antigen presentation to CD8+ T cells.7,28 No NLRC5 somatic mutations were detected among EC samples in the present study, however. An analysis of the muTarget database suggested that mutations in the ANKRD20A2, C2orf42, ADGRB3, AVPR2, GOLGA6C, and IPPK genes may contribute to NLRC5 downregulation in patients with EC.
Both clinical HCC samples and HCC cell lines have been found to express NLRC5 at high levels. In nude mice, NLRC5 knockdown was sufficient to dramatically reduce these cells’ proliferation, motility, and invasivity, as well as their ability to form tumors, promoting G0/G1 phase cell cycle arrest. NLRC5 overexpression, in contrast, was sufficient to enhance HCC cell proliferative, migratory, and invasive activity through the downstream activation of the Wnt/β-catenin signaling pathway. 9 NLRC5 has recently been demonstrated to enhance the proliferation of HCC tumor cells via activating the AKT/VEGF-A pathway, whereas NLRC5 knockdown was linked to decreased in vivo HCC tumor development. 29 Wang et al discovered that overexpression of the NLRC5 gene was connected to more advanced staging and poorer prognostic outcomes in patients with clear cell renal cell carcinoma (ccRCC). From a mechanistic perspective, NLRC5 was able to promote ccRCC cell proliferative, migratory, and invasive activity through Wnt/β-catenin pathway activation, while silencing NLRC5 led to the suppression of in vivo tumor growth through the inhibition of Wnt/β-catenin signaling. 8 Recent work also suggests NLRC5 to be a miR-4319 target, with NLRC5 overexpression promoting positive miR-4319-related effects on esophageal squamous cell carcinoma (ESCC) cell growth and cell cycle progression. 30 These data thus further support a role for NLRC5 as a driver of tumorigenesis, with NLRC5 inhibition thus representing a promising target for the treatment of HCC, ccRCC, and other cancer types. NLRC5, however, may exhibit tissue-specific regulatory roles. In a recent study, NLRC5 was found to be downregulated in EC tissues relative to normal endometrial tissues, promoting AN3CA cell invasion and migration through PI3K/AKT pathway activation. 10 Even so, the role that NLRC5 plays in the context of EC development and whether this role is regulated by PD-L1 has yet to be established.
Prior studies showed that PD-L1-mediated tumor immune evasion was intimately implicated in reduced activity of T cells, and PD-L1 inhibitor facilitated depletion of tumor cells by rescuing CD8+ T cells.31,32 Furthermore, it has demonstrated that there was a negative correlation between MHC class I molecules and PD-L1 expression on tumor tissues. 33 Of special note is that a recent study indicated that NLRC5 variants could interact with PD-L1 variants in colorectal cancer, which provides a novel biological information to improve colorectal cancer risk management and immunotherapy. 34 These studies provided a potential role of PD-L1 by NLRC5. In our study, we found NLRC5 and PD-L1 was co-localization in the cytoplasm of HEC-1A and Ishikawa cells. Furthermore, there is a negative correlation between NLRC5 and PD-L1. Importantly, further investigations found that NLRC5 could promote the expression of PD-L1 in EC cells. We suggest that the downregulation of NLRC5 in EC patients owning to the mutations of ANKRD20A2, C2orf42, ADGRB3, AVPR2, GOLGA6C, and IPPK. Unlike the NLRC5-mediated immune surveillance in the tumor microenvironment, NLRC5 may contribute to the cell growth by promoting PD-L1 in the single EC cells.
Conclusion
Our study presented a novel regulating role in EC cells that NLRC5 promotes PD-L1 expression, suggesting that inhibiting NLRC5 in combination with inhibiting PD-L1 may contribute to the therapy for EC. Nevertheless, the sample size in our study was relatively small. Furthermore, our study is to focus on the role of NLRC5 in single EC cells. On account of the critical role of NLRC5 in immune activation, which is essential for tumor immunotherapy, simply to increase the expression of NLRC5 may lead to tumorigenesis by promoting cell proliferation, migration, and invasion, and if simply to reduce the expression of NLRC5 may inhibit NLRC5-mediated cancer immune surveillance. Further experiments are needed to demonstrate the effect of promoting or inhibiting NLRC5 expression as adjuvant therapy in the therapy for EC, including immunotherapy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Our study was approved by The Institutional Review Board of Anhui Medical University (approval No. 20180023). All patients provided written informed consent prior to enrollment in the study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Nature Science Foundation of China, Research Fund of Anhui Institute of translational medicine and Natural Science Foundation of Colleges and Universities (grant numbers 81802586, 81871216, ZHYX2020A001, and KJ2017A197).
