Abstract
Keywords
Introduction
Lymphocytes are sensitive to ionizing radiation, and a reduction in peripheral total lymphocyte count (TLC) following radiotherapy (RT) is characterized by impaired host immune response to malignancy. 1 Numerous studies have demonstrated a link between radiation-induced lymphopenia (RIL) and poor prognosis in a wide variety of cancers, including gliomas, esophageal cancer, lung cancer, rectal cancer, pancreatic cancer, hepatocellular carcinoma, and cervical cancer.2–13 Moreover, higher TLCs are also associated with a high response rate and more durable treatment response in patients treated with immune checkpoint inhibitors.14–16 This observation further emphasizes the importance of preserving and maintaining circulating lymphocytes in cancer patients who receive the new therapeutic strategy that combines RT and immunotherapy.
Many risk factors for RIL have been described and include higher radiation dose, higher target volume, larger field size, more fraction number, and the use of photon RT (compared with proton RT).17–21 RIL most likely results from direct toxicity of radiation to circulating lymphocytes as these cells pass through the field of radiation.18,22 Thus, longer radiation time likely exposes more blood to the radiation, yielding more lymphocyte depletion; however, little is known about the impact of radiation time on RIL and the subsequent lymphocyte count recovery (LR). As a result, we sought to evaluate the potential impact of radiation time on RIL and subsequent LR after stereotactic body radiation therapy (SBRT) and to examine the association of these parameters with patient outcomes in early-stage non-small cell lung cancer (NSCLC).
Materials and Methods
Patient Population
The reporting of this study conforms to STROBE guidelines. 23 This study was approved by the X Hospital Ethics Committee (B2022-294). Written informed consent was obtained from each patient for the use of his or her clinical data in clinical studies. We retrospective analyzed our clinical database of early-stage NSCLC who received definitive SBRT between December 2014 and May 2018 at Zhongshan Hospital, Fudan University. The 18F-fluorodeoxyglucose positron emission tomography/computed tomography (18F-FDG PET/CT) scans before SBRT in patients without pathological confirmation of disease were necessary. Patients who received SBRT with curative intent for early-stage NSCLC and had no history of another concomitant malignancy were selected for inclusion if there were available blood count values from within 10 days prior to commencement of SBRT, within 10 days immediately following completion of SBRT, and from at least 4 more dates within 6 months after SBRT. Those with a history of prior RT, other anti-cancer treatment, pre-SBRT TLCs < 1000 cells/µL, and other diseases that may affect TLCs (eg, severe infection or immunosuppression) were excluded.
Data Collection
The SBRT treatment plan and delivery method at our institution have been previously described.24,25 All patients were treated with SBRT using the Helical TomoTherapy (HT) Hi-Art treatment system (Accuray). Patient demographics, tumor details, treatment-related characteristics, and follow-up data were extracted from medical records. Patients with asthma, chronic obstructive pulmonary disease, or other lung diseases that cause the forced expiratory volume in 1 s (FEV1) to be <1.6 were considered to have underlying respiratory system disease. Dosimetric parameters and radiation time were extracted from the treatment plan system. Radiation time for the individual patient was defined as the length of time of circulating lymphocyte exposure to radiation, and this time was calculated by beam-on time per fraction multiplied by fraction number. To visualize the TLCs before and after SBRT, complete blood cell (CBC) count data, which included a pre-SBRT, a post-SBRT, and then a value at monthly intervals for 6 months, were assessed. For patients with missing CBC data at a time point of interest, the closest value to the desired date was used. We further examined the alteration of TLCs (ATLCs, post-SBRT-pre-SBRT TLCs) to evaluate the extent of reduction in TLC after SBRT. According to the National Cancer Institute Common Terminology Criteria for Adverse Events version 4.0, post-SBRT TLCs < 1000 cells/µL were considered to indicate lymphopenia, and post-SBRT TLCs ≥ 1000 cells/µL were non-lymphopenia (G0). Lymphopenia was further categorized into grade 1 (G1, <1000 cells/µL), grade 2 (G2, <800 cells/µL), grade 3 (G3, <500 cells/µL), and grade 4 (G4, <200 cells/µL). Considering the variations between different blood tests, only patients with post-SBRT TLC levels that were restored back to pre-SBRT levels at least twice during the first 6 months follow-up were considered to have recovered from RIL and were classified as the LR group.
Statistical Analyses
Data are presented as median (interquartile range [IQR]) for continuous data or number (proportion) for categorical variables. The TLCs change over time was assessed using Student's
Results
Patient Characteristics
A total of 115 patients with early-stage NSCLC met the selection criteria and were included in this study. The median number of blood tests per patient was 8 (IQR, 6-11). The median number of days for pre-SBRT counts was 2 days (IQR, 0-4) prior to commencement of SBRT, while that for post-SBRT counts was 3 days after SBRT completion (IQR, 2-5). The median age of all patients was 71 years (IQR, 64-78 years), and the median diameter of the primary tumor was 20.2 mm (IQR, 15.45-30.00 mm). Additional information about patient demographic, tumor, and treatment characteristics are listed in Table 1.
Baseline Patient, Tumor and Treatment Characteristics for All Patients (n = 115).

Abbreviations: IQR, interquartile range; ECOG, Eastern Cooperative Oncology Group; GTV, gross tumor volume; PTV, planning target volume; SBRT, stereotactic body radiation therapy.
Lymphocyte Counts After SBRT
To understand the pattern of changes in CBC response to SBRT, CBC values were assessed with respect to time (Figure 1 and Supplemental Figure S1). Most patients (100/115, 86.96%) experienced significantly decreased median TLCs following SBRT (1700 vs 1100 cells/µL;
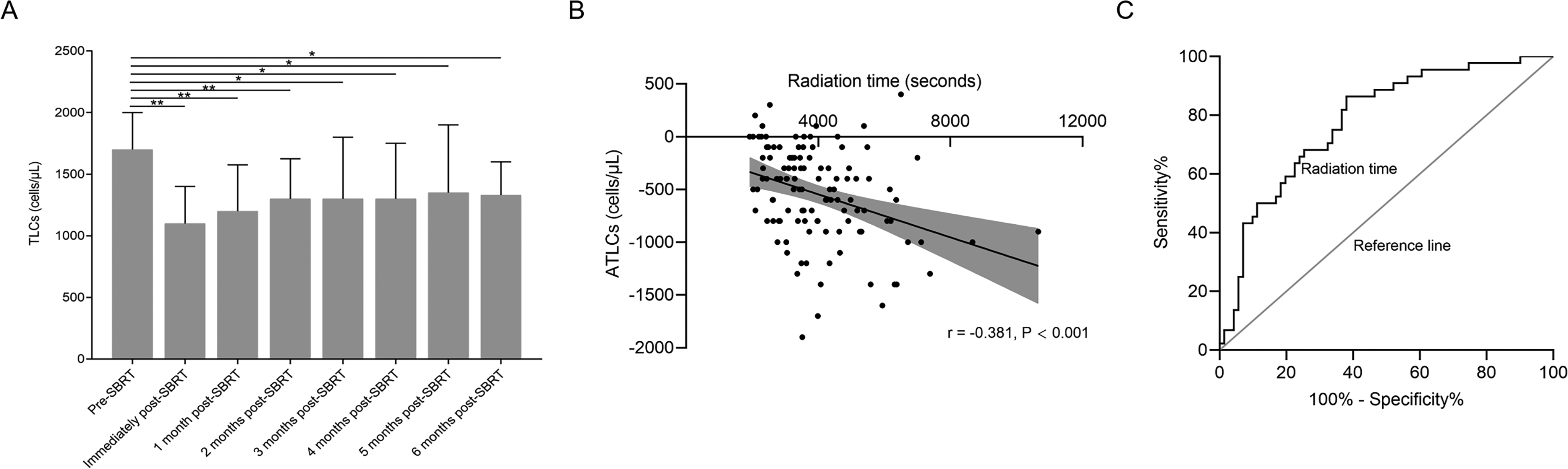
(A) Distribution of TLCs from baseline levels before SBRT to 6 months after SBRT in patients with early stage NSCLC. Note: Not all patients had TLCs data available at each time point. (B) Correlation between ATLCs and radiation time. (C) ROC curve analysis of radiation time for LR.
In addition, 6 months after SBRT, 44 patients (38.26%) exhibited recovery, including 17 patients in the lymphopenia group and 27 patients in the non-lymphopenia group (
Association of Radiation Time and ATLCs After SBRT
Analysis revealed a negative correlation between radiation time and ATLCs (
Univariate and Multivariate Liner Regression Associating Baseline Factors With ATLCs.

Abbreviations: ATLC, alteration of total lymphocyte count; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; SBRT, stereotactic body radiation therapy; GTV, gross tumor volume; PTV, planning target volume; Lung V5, the percentage of total lung volume receiving at least 5 Gy; Heart V15, the percentage of total heart volume receiving at least 15 Gy.
Impact of Radiation Time on LR After SBRT
ROC curve analysis was performed to determine the predictive value of radiation time on LR. The optimal cut-off value of radiation time was determined as 3950 s (
We further identified patient and treatment characteristics associated with LR within 6 months after SBRT (Table 3). According to univariate analyses, radiation time, tumor diameter, pre-SBRT TLCs, ATLCs, GTV, PTV, lung V50, and dose fractionation were all significantly associated with LR (
Univariate and Multivariate Logistic Regression of Factors Associated With LR After SBRT.

Abbreviations: RL, lymphocyte recovery; SBRT, stereotactic body radiation therapy; OR, Odds ratio; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; TLCs, total lymphocyte counts; ATLC, alteration of TLCs; GTV, gross tumor volume; PTV, planning target volume; Lung V50, the percentage of total lung volume receiving at least 50 Gy.
Outcome According to RIL and LR
The median follow-up period was 28 months (range, 5-68 months) for the entire cohort and 31 months (range, 5-68 months) for survivors. At the time of analysis, 37 patients (32.17%) had locoregional or distant progression, and 23 patients (20.00%) had died. The 2-year PFS and 2-year OS rates for the whole cohort were 72.3% and 81.3%, respectively.
To assess the relationship between the lymphocyte kinetic and patient outcomes after SBRT, we stratified patients according to PFS and OS at 2 years after SBRT. As shown in Figure 2, patients with superior survival had higher median TLC values at most time points compared to those with shorter PFS and OS. Patients with G1-3 lymphopenia had inferior PFS and OS compared with those with G0 (
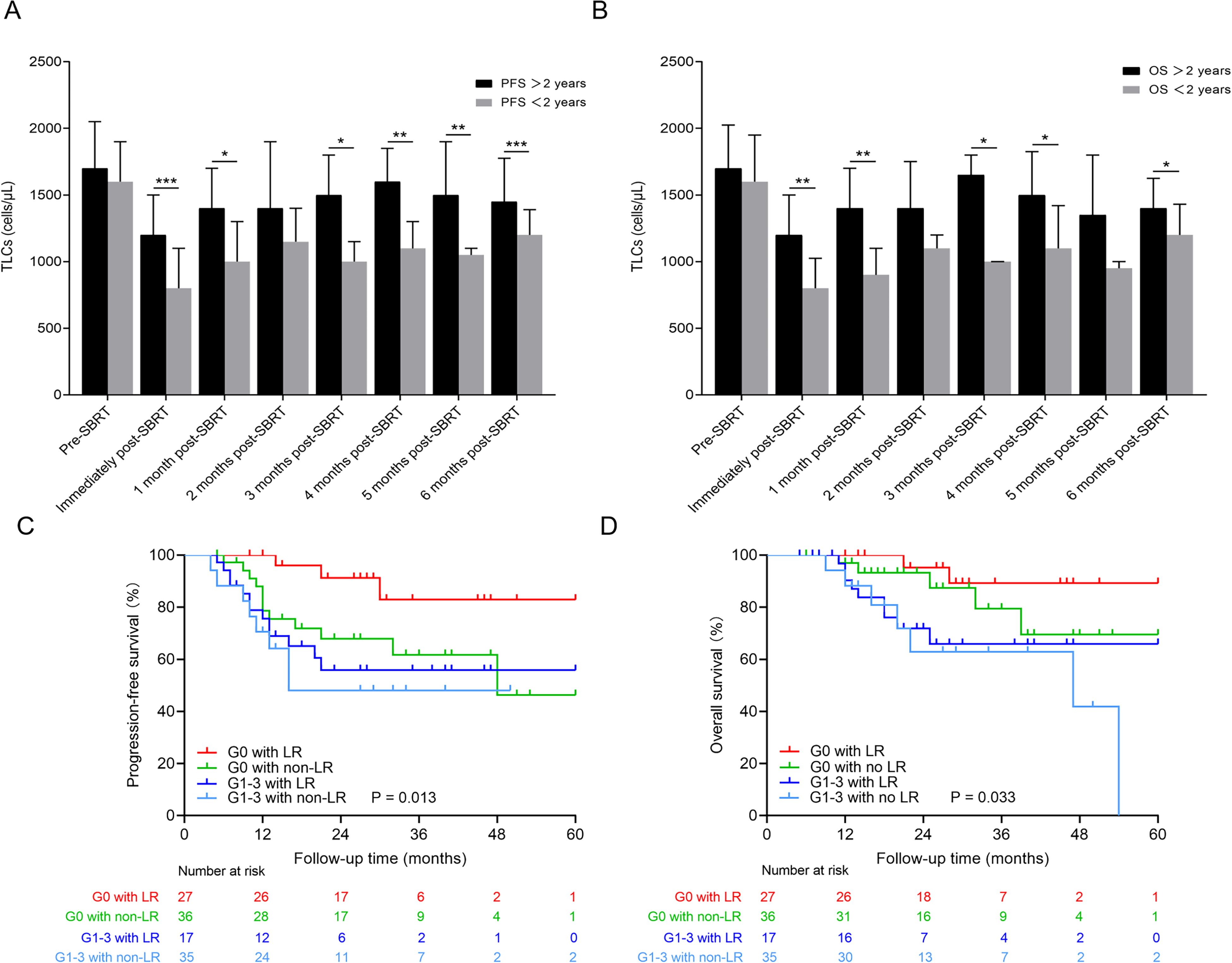
(A and B) Distribution of TLCs at each time stratified by survival (PFS > 2 years vs PFS < 2 years and OS > 2 years vs OS < 2 years). ***
Given the relatively short follow-up time and limited number of deaths, univariable and multivariable Cox proportional hazard regression analyses were performed only with PFS data (Table 4). By univariate analysis, tumor diameter (<22.5 mm), GTV (<8.2 cm3), PTV (<32 cm3), fractionation (50 Gy/5 fractions), post-SBRT TLCs (≥1000 cells/µL), and G0 status in patients with LR were all associated with improved PFS (
Univariable and Multivariable Cox Proportional Hazard Regression Analyses of Factors Associated With PFS.

Abbreviations: PFS, progression-free survival; OR, odds ratio; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; GTV, gross tumor volume; PTV, planning target volume; SBRT, stereotactic body radiation therapy; TLCs, total lymphocyte counts; LR, lymphocyte counts recovery.
Discussion
In this study, we observed significant and durable clinical lymphocyte depletion following SBRT in patients with early-stage NSCLC, and most patients failed to restore their TLCs to pre-SBRT levels within 6 months after SBRT. Furthermore, we found that decreased values of TLCs varied with respect to different pre-SBRT TLC levels, RT fraction number, and PTV, whereas LR with 6 months after SBRT was associated with underlying respiratory system disease, ATLCs, lung V50, and radiation time. Finally, we found that LR within 6 months after SBRT was associated with improved survival in patients without lymphopenia but did not mitigate poor survival with lymphopenia. These findings suggest that shortening radiation time may promote recovery from lymphocyte depletion following SBRT and ultimately improve patient prognosis.
Currently, reports have indicated that the risk factors of RIL include a higher radiation dose, higher target volume, larger field size, and a greater number of fractions.3,16,19–21 Similarly, we also found that PTV and fraction number were inversely correlated with ATLCs. To date, however, few studies have focused on the impact of radiation time on TLC reduction and subsequently LR. Based on clinical observations, Yovino et al postulated a mathematical model for the estimation of the radiation dose on circulating lymphocytes during a course of external beam conventional fractionated RT (60 Gy in 30 fractions) for glioblastoma. These investigators found that shorter radiation time was associated with a lower dose of RT to the circulating lymphocytes at the first 10th fraction during conventional fractionated RT with 30 fractions, thereby reducing the risk and severity of RIL.
26
Here, we also found that ATLCs were inversely proportional to radiation time in actual patient data; however, in multivariate analysis for ATLCs, only pre-SBRT TLCs, fractionation, and PTV remained significantly associated, while radiation time did not (
The absolute number of peripheral lymphocytes is essential for an efficient anti-tumor immune response. Several studies have demonstrated a strong link between severe and persistent RIL and poor clinical outcomes, including in early-stage NSCLC patients treated with SBRT.4,12 In our study, although no lymphopenia and G0 patients with LR exhibited superior PFS in univariate analysis, only G0 patients with LR along with age and dose fractionation emerged as significant prognostic variables in multivariate analysis. ATLCs likely reflect a transient response to SBRT, while LR is likely associated with a sustained response. Therefore, our data suggest that a combination of the degree of the TLC decline and the subsequent recovery of the TLCs within 6 months after SBRT is a better predictor of persistent anti-tumor immune function than the presence of lymphopenia alone. The fact that lymphopenia no longer predicts prognosis when the dynamic pattern of lymphocytes is factored in further lends support to the conclusion that lymphocytes kinetics following SBRT serve as a better predictor for prognosis in these patients. In addition, Deng et al reported that recovery from lymphopenia to (near) normal levels did not mitigate poor survival in esophageal cancer patients with severe RIL (G4) after chemoradiation therapy. 27 This result coincides with our finding that LR did not mitigate poor survival in NSCLC patients with lymphopenia. Although the extent of lymphocyte depletion after SBRT appears to have been compensated for in some patients, one aspect of effective immunity against the tumor remains impaired and is not recovered. This ineffective tumor immunity occurs primarily because the newly generated lymphocytes do not guarantee anti-tumor response. 28 This finding also suggests that recovered immune function exerts a crucial role in repressing the remaining tumor and delaying disease recurrence.
It is reasonably assumed that the degree and rapidity of decreased lymphocyte count would mostly depend on the volume of circulating blood exposed to a certain level of radiation. The estimated blood volume is the complexity of target volume size, the proportion of circulating blood volume covered by isodose line, radiation exposure time, velocity of blood flow, and so on. As shown in Supplemental Figure S3 of the isodose distribution of 4 representative early-stage lung cancer patients treated with SBRT, different isodose lines covered the heart (Supplemental Figure S3A) or great vessels (left pulmonary artery in Supplemental Figure S3B, aortic arch in Supplemental Figure S3C, and right pulmonary artery in Supplemental Figure S3D) in mediastinum. However, the velocity of blood flow in the heart, artery, and vein are different and changes from day to day. In addition, lymphocytes concentration in the blood changes from day to day and it also affects the degree and rapidity of decreased lymphocyte count after RT. It is difficult to estimate these data and blood volume even in a prospective analysis. Figure 2 showed the correlation between ATLCs and radiation time. In addition, multiple logistic regression analysis revealed that the contribution of radiation time to LR was still statistically significant after adjusting other confounding factors in our revised manuscript. These results suggested that the importance of radiation time on ATLCs after RT has long been neglected when we evaluated RT planning.
Our findings must be interpreted in light of the following limitations. First, the retrospective nature of this analysis is subject to all the limitations of post-hoc analyses. Second, we didn't perform the sample size/power analysis and included a relatively small sample size. Third, all patients in our study received SBRT using HT, which creates highly conformal plans at the target but usually results in a larger area of tissue receiving low doses of radiation and longer radiation times.29–31 Whether the long radiation time of HT increased lymphopenia risk and compromised the patient outcomes have prompted additional future investigations. Finally, our work still requires external validation to support our findings.
Conclusion
In conclusion, our study supports a strong link between radiation time and TLC kinetics following SBRT and their associated LR and survival outcomes in early-stage NSCLC. Our findings add to the evidence that supports the clinical importance of radiation time and fraction number for maintenance of lymphocytes after SBRT. These data require prospective validation and support further testing.
Supplemental Material
sj-tif-1-tct-10.1177_15330338221112287 - Supplemental material for Shortened Radiation Time Promotes Recovery From Radiation-induced Lymphopenia in Early-Stage Non-small Cell Lung Cancer Patients Treated With Stereotactic Body Radiation Therapy
Supplemental material, sj-tif-1-tct-10.1177_15330338221112287 for Shortened Radiation Time Promotes Recovery From Radiation-induced Lymphopenia in Early-Stage Non-small Cell Lung Cancer Patients Treated With Stereotactic Body Radiation Therapy by Qianqian Zhao, Tingting Li, Shisuo Du, Jian He and Zhaochong Zeng in Technology in Cancer Research & Treatment
Supplemental Material
sj-tif-2-tct-10.1177_15330338221112287 - Supplemental material for Shortened Radiation Time Promotes Recovery From Radiation-induced Lymphopenia in Early-Stage Non-small Cell Lung Cancer Patients Treated With Stereotactic Body Radiation Therapy
Supplemental material, sj-tif-2-tct-10.1177_15330338221112287 for Shortened Radiation Time Promotes Recovery From Radiation-induced Lymphopenia in Early-Stage Non-small Cell Lung Cancer Patients Treated With Stereotactic Body Radiation Therapy by Qianqian Zhao, Tingting Li, Shisuo Du, Jian He and Zhaochong Zeng in Technology in Cancer Research & Treatment
Supplemental Material
sj-tif-3-tct-10.1177_15330338221112287 - Supplemental material for Shortened Radiation Time Promotes Recovery From Radiation-induced Lymphopenia in Early-Stage Non-small Cell Lung Cancer Patients Treated With Stereotactic Body Radiation Therapy
Supplemental material, sj-tif-3-tct-10.1177_15330338221112287 for Shortened Radiation Time Promotes Recovery From Radiation-induced Lymphopenia in Early-Stage Non-small Cell Lung Cancer Patients Treated With Stereotactic Body Radiation Therapy by Qianqian Zhao, Tingting Li, Shisuo Du, Jian He and Zhaochong Zeng in Technology in Cancer Research & Treatment
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
The study was approved by the ethics board of Zhongshan Hospital, Fudan University (B2022-294).
Supplemental Material
Supplemental material for this article is available online.
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
