Abstract
Keywords
Introduction
During thoracic-abdominal tumor radiotherapy, tumor motion will induce interplay and fuzzy effects along with spatial deformation of the dose distribution,1,2 leading to a lack of dose coverage in the target and excessive exposure of normal tissues. 3 This in turn will worsen the curative effect of radiotherapy and causes the surrounding normal tissue to receive an additional dosage of radiotherapy. 4 It is therefore very important to manage a patient's respiratory motion during radiotherapy. Conventional respiratory management approaches include motion-encompassing,5,6 breath-holding,7–10 respiratory gating,11,12 abdominal compression, 13 4-dimensional computed tomography (4DCT), 14 and direct real-time tracking techniques of implanted marker;15–18 unlike all the above-mentioned techniques, indirect real-time tracking predicts the position of tumor based on the external/internal respiratory motion correlation model by external surrogate respiratory signals acquired using optical or infrared devices while the patient breathes freely.19,20
Indirect real-time tumor tracking technique based on the use of optical surface imaging is considered to be a type of surface-guided radiation therapy (SGRT). Owing to its non-radiological and non-invasive nature,21,22 SGRT can be continuously used throughout the radiotherapy process and is well suited to tumor tracking.23,24 Applications of SGRT in the radiotherapy of lung 25 and breast tumors 26 as well as laryngeal cancer 27 have been reported. Its principle is to input a surface real-time signal obtained from a patient by optical equipment into an external/internal correlation model to predict a real-time tumor motion signal. This signal can then be used to adjust the beam or the treatment couch to allow the beam to continuously follow the moving tumor and to eliminate the influence of respiratory motion. 28 The algorithms used to implement the external/internal correlation model include linear,29,30 piecewise linear, 31 multinomial, 32 and combined multinomial 33 models as well as adaptive filters, machine learning, 34 and deep neural network algorithms.35,36
A patient's respiratory motion will cause their respiratory baseline to undergo systematic changes during breathing; breathing patterns can vary in magnitude, period, and regularity.37–40 The motion of thoracic-abdominal tumors is complicated, and many variations in motion associated with tumor location and pathology lead to distinct individual patterns in terms of displacement, direction, and phase of tumor motion. 41 In liver cancer patients, Zhang et al found that the liver motion trajectory model included perfect linearity, sample linearity, hysteresis, and area. 42 Park et al analyzed the liver motion characteristics of 20 patients and observed large inter- and intra-fractional variations in motion. 43 In lung cancer patients, the anatomy and air volume in the lung continually change during breathing, 41 and some lung tumors behave as rigid objects during respiration whereas others show systematic shape changes. 44 Seppenwoolde et al analyzed the motion trajectories of 21 lung tumors and found that the direction and amplitude of motion varies with each tumor and that every tumor follows different paths during inhalation and exhalation. 37
In total, both the respiratory and tumor motion vary depending on the patient, resulting in patient-specific correlations between the external respiratory and the internal tumor motion (external/internal correlation). If the external/internal correlation in a particular patient is poor, it will be difficult to establish a relationship between the external respiratory and tumor motion through an external/internal correlation model; for such patients, indirect real-time tracking techniques based on an external/internal correlation model might not be suitable, implying that patient-specific radiotherapy is preferable. The goal of this study on liver and lung cancer patients is to explore the relationship between the external/internal correlation and several patient clinical characteristics to provide a basis to qualitatively judge whether a particular patient is suitable for indirect real-time tracking based on an external/internal correlation model prior to radiotherapy. The external/internal correlation of patients can be assessed using methods such as the phase shift 45 or normalizing real-time position management (RPM) trace data to compare them directly with the fiducial location. 46 However, there may be a non-linear correlation between the external respiratory and internal tumor motion. To perform more accurate correlation fitting, we introduce machine learning methods and calculate 2 correlation coefficients to quantitatively reveal the strength of specific patients’ external/internal correlation in 3 dimensions and attempt to identify patient characteristics that affect the strength of the patients’ external/internal correlation.
This study aimed to find a general rule of internal and external correlation that applies to patients with thoracic and abdominal tumors, or to find the main factors that reflect the internal and external correlation of thoracic and abdominal tumor patients, for achieving rapid correlation judgment through patient characteristics and for judging specific patients before radiotherapy. Whether it is suitable to accept SGRT will undoubtedly provide significant convenience for the development and promotion of SGRT.
Materials and Methods
Dataset
In this study, data from 90 liver and lung cancer patients treated with respiratory-gated radiotherapy using the Varian RPM system (Varian Medical Systems) in the radiotherapy department were consecutively collected (Table 1) to perform a retrospective analysis, and all patient data were anonymized. The internal tumor motion data, corresponding external motion data, and other clinical data, all of which may be related to the external/internal correlation in a patient, were collected. The reporting of this study conforms to STROBE guidelines. 47
Basic Data for the 90 Cancer Patients Assessed in This Study.

Acquisition of Internal Tumor Motion Data
Internal tumor motion data is acquisited from 4DCT. After the 4DCT scanning was completed on the Revolution CT scanner (GE Healthcare), the images were then loaded into an Advantage Workstation V4.7 (General Electric Co.) and were split into 10 respiratory phase CT (0%-90%). Using the Raystation treatment planning system (TPS) V4.7.6 (RaySearch Laboratories AB), a doctor outlined tumor on 10 respiratory phase CT images for each patient. Therefore, the coordinates of the tumor centroid (Px, Py, and Pz) of 10 respiratory phases can be obtained. In this manner, internal tumor motion data corresponding to 10 respiratory phases in the right-left (R-L), anterior-posterior (A-P), and superior-inferior (S-I) directions were obtained from 90 cancer patients. For patients with multiple tumors, the internal motion data for each tumor were obtained.
Acquisition of External Motion Data
The Varian RPM system records the one-dimensional real-time external motion signals corresponding to respiratory phase during 4DCT scanning via markers placed on a patient's anterior abdominal surface (midway between the xiphoid process and the umbilicus). The multiple sets of external real-time motion coordinate data were grouped according to the corresponding respiratory phase, and the average value was used as the external motion data of the phase. Using the scanning and recording processes, we obtained the external motion data and the corresponding internal tumor motion data over 10 respiratory phases.
Acquisition of Other Clinical Data
The factors that may affect the external/internal correlation in a patient include age, gender, tumor volume Vt, internal tumor motion magnitude Ai, external marker motion magnitude Ae, etc.41,48 For lung tumors, the type of location of the tumor can also be obtained. 49 All these data were collected from the Raystation TPS and were analyzed in this study.
Evaluation of External/Internal Correlation
The Spearman's rank correlation coefficient (SCC) was used to evaluate the external/internal correlations in the patients. The Spearman rank correlation coefficient is a nonparametric technique for evaluating the degree of linear association or correlation between 2 independent variables.
50
Because the technique operates on the ranks of the data and is relatively insensitive to outliers, it can be used with very small sample sizes, and it is suitable for non-normally distributed datasets.
51
Considering the small amount of data and non-normal distribution characteristics of the dataset collected in this study, the Spearman rank correlation coefficient is an indicator suitable for measuring the strength of the external/internal correlation, which is in line with the characteristics of the data in this study. For the calculation of the Spearman rank correlation coefficient, each variable is ranked separately from lowest to highest and the difference between ranks for each data pair is recorded. If the data are correlated, then the sum of the square of the difference between ranks will be small. The magnitude of the sum is related to the significance of the correlation.
51
The Spearman rank correlation coefficient can be calculated as follows: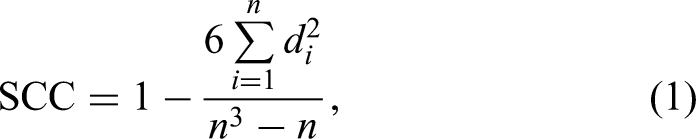
The SCC was further combined with the support vector regression (SVR) network fitting method to calculate the SCC after the SVR fitting (SCCsvr). To do this, an SVR network was first used to fit the external/internal motion data for each patient to obtain a fitter. The internal tumor motion data were then input into the fitter to obtain fitted external motion data. Finally, the fitted external motion data and the real external motion data were calculated to obtain the SCCsvr. An SVR network is a type of network that can transform the nonlinear problem in an actual space into a linear problem in a higher-dimensional feature space using a nonlinear transformation to enable solving of the original nonlinear problem through the construction of a linear decision function. 52 Owing to this characteristic of the SVR network, it is suitable for solving nonlinear problems such as respiratory motion signal fitting. Furthermore, it should be noted that the strength of the patient's external/internal correlation is mainly related to the absolute value of the SCC and the SCCsvr, and the absolute value of them, were used in the study. The cut-off values, weak (r<0.3), moderate (r = 0.30-0.69), and strong (r = 0.70-1.0) were used to interpret the external/internal correlation.
Statistical Analyses
All statistical comparisons were performed using the nonparametric Wilcoxon signed rank test based on Statistical Package for the Social Sciences software (IBM SPSS, version 25.0), with P<.05 indicating statistical significance.
Results
Correlation Between External/Internal Correlation and Patient Characteristics
For the liver cancer patients, there was a statistically significant correlation between the external/internal correlation and their age in the S-I direction and the value of Ae in the A-P direction. For the lung cancer patients, the correlation coefficients between the external/internal correlation and the value of Ai and Ae were statistically significant. In both the liver and lung cancer patients, the correlation coefficients between the external/internal correlation and Vt were not statistically significant. Totally, the correlation coefficients between the external/internal correlation and the patient's characteristics (Ai, Ae, Vt, and age) were smaller than 0.5, and 77.1% of them are <0.3 (Table 2), that is, the effects of Ai, Ae, Vt, and age on the external/internal correlations in liver and lung cancer patients were all weakly to moderately correlated.
Spearman Correlation Coefficients and Significance Scores Between External/Internal Correlation and Patient Characteristics.

* P<.05.
** P<.01.
External/Internal Correlations in Liver and Lung Cancer Patients
The statistical results for all patients (see Figure 1) revealed that the external/internal correlations in liver cancer patients were stronger than those in lung cancer patients, with an average SCCsvr value of 0.913 (range: 0.552-1.000) and 0.813 (range: 0.006-0.988), respectively, for liver and lung cancer patients in the S-I direction. The proportions of liver and lung cancer patients at different levels of correlation are listed in Table 3. The external/internal motions of most of the liver (96.6%) and lung (81.5%) cancer patients were strongly correlated in the S-I direction. For both liver and lung cancer patients, the SCC values in all 3 directions increased following the SVR fitting. The mean SCC values of the liver cancer patients increased by 0.019, 0.060, and 0.050, in the S-I, A-P, and R-L directions, respectively. Moreover, in these directions, the corresponding increase in the mean SCC values for lung cancer patients were 0.045, 0.306, and 0.274, respectively. In addition, the proportions of liver and lung cancer patients at the “strong” level increased by 3.4%, 10.2%, and 6.8% and by 5.8%, 51.4%, and 44.3%, respectively, in the S-I, A-P, and R-L directions. This statistical data suggests 2 conclusions: first, following the SVR fitting the SCC values of the lung cancer patients increased more than they did in the liver cancer patients; second, following the SVR fitting, the SCC values increased more in the directions with smaller SCC (ie, A-P and R-L directions).
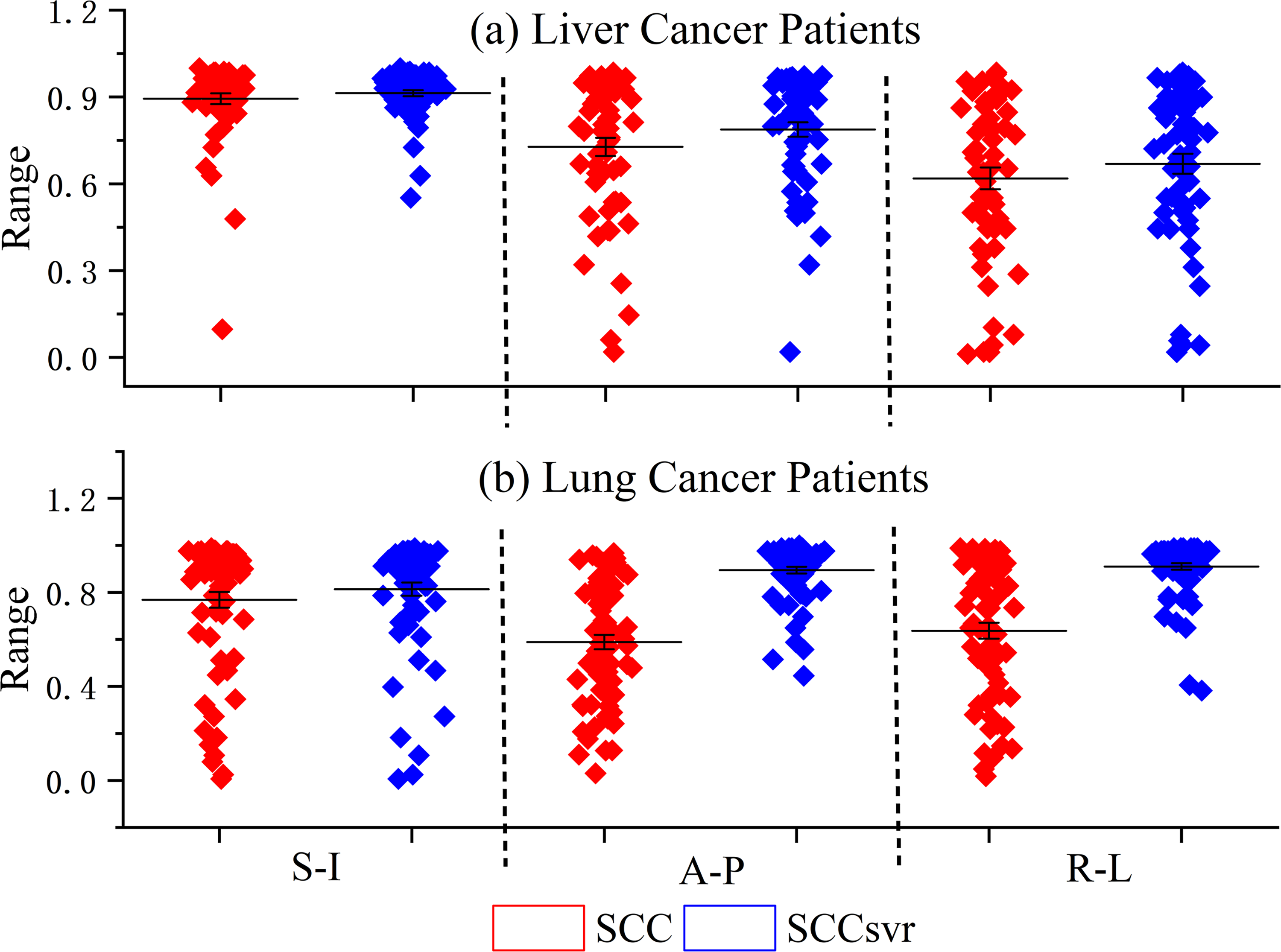
External/internal correlations in (a) liver and (b) lung cancer patients. Red and blue dots indicate SCC and SCCsvr values, respectively.
Proportions of Liver and Lung Cancer Patients With Different SCC and SCCsvr Scores.

External/Internal Correlations for Individual Cancer Patients
In Figure 2(I), the SCC and SCCsvr scores of each liver and lung cancer patient are presented in an ascending order of the SCC. Figure 2(II) illustrates the deviation between the SCC and the SCCsvr for each (a) liver and (b) lung cancer patient. In the region in which the SCC is large, the SCCsvr scores are nearly the same as the SCC scores, with a negligible deviation between the two (see Figure 2). In addition, the deviations between the SCC and SCCsvr scores decreased as the SCC increased (see Figure 2(II)). In other words, the increase in the SCC following the SVR fitting primarily occurred in patients with small SCC scores, where small SCC scores correspond to greater deviations, particularly in the R-L and A-P directions in which the mean values of “SCCsvr − SCC” are greater than those in the S-I direction (see the line in Figure 2(II)). Furthermore, the lung cancer patients were more likely than the liver cancer patients to have deviations that are greater than zero between the SCC and SCCsvr, that is, the situation in which the SCC increased after SVR fitting occurred more frequently in the lung cancer patients. These 2 phenomena are consistent with the results presented in Figure 1 and Table 3.
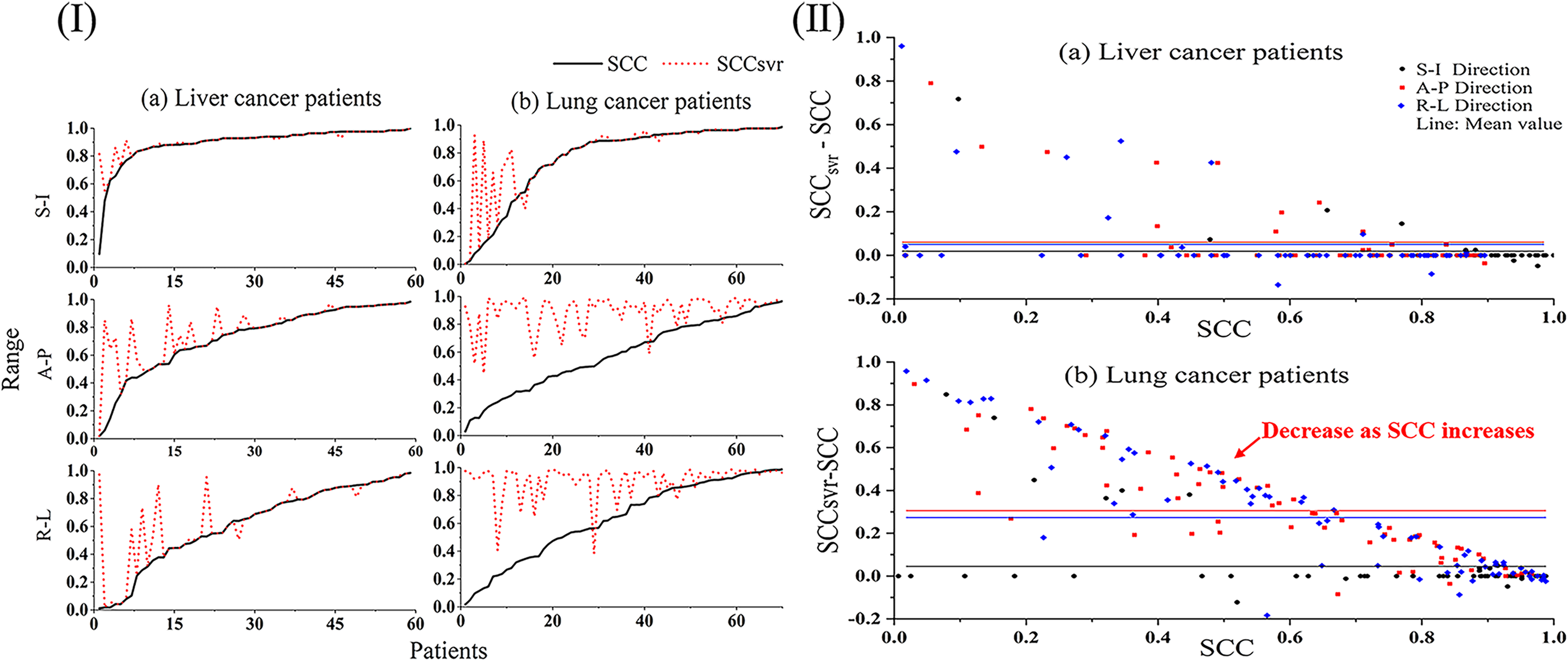
(I) SCC and SCCsvr scores for each patient. Solid and dotted lines indicate SCC and SCCsvr scores, respectively. (II) Deviations between SCC and SCCsvr in (a) liver and (b) lung cancer patients in the SI, AP, and LR directions, which are plotted in different colors. The solid line of the same color corresponds to the mean deviation value.
An inspection of the SCC and SCCsvr scores for each patient revealed that the external/internal correlation varied significantly among patients (see Figure 2) not only between patients with different types of cancers but also between patients with the same type of cancer, indicating that the external/internal correlation exhibits significant individualized differences for each patient.
Relationship Between Patient Characteristics and Level of External/Internal Correlation
The differences in terms of all 4 characteristics among the 3 groups (weak, moderate, and strong) are compared in Figure 3. For the liver cancer patients, none of the 4 characteristics (Ai, Ae, Vt, and age) are related at a statistically significant level to the strength of external/internal correlation. For lung cancer patients, only the differences in the Ai values between the weak and moderate groups in the S-I direction and the differences in the Ae values between the moderate and strong groups in the A-P direction are statistically significant. Despite this, the figure presents the following trends: (1) Ai and Ae are positively correlated with the strength of the external/internal correlation, (2) the patient's age and the strength of the external/internal correlation are negatively correlated, and (3) Vt has a positive correlation with the strength of the external/internal correlation in liver tumors but a negative correlation in lung tumors.

(a) Liver and (b) lung cancer patient characteristics with different levels of SCCsvr scores. A P value of .05 was defined as statistically significant.
External/Internal Correlations in Liver and Lung Cancer Patients of Different Genders
There were no consistent trends for either the male or female patients in terms of the SCC, SCCsvr, or Ai values along the 3 directions; furthermore, the intergender differences in terms of these 3 characteristics were small. For the female patients, the Ae and Vt values for both liver and lung cancer patients were smaller than they were for the male patients; however, in terms of age, the situation was reversed; the female liver as well as lung cancer patients tended to be older than the male patients, with an average difference of 3 years.
External/Internal Correlations in Patients With Central/Peripheral Lung Cancers
For patients with central/peripheral lung cancers, Ai was 0.729/0.996, 0.272/0.324, and 0.187/0.242 cm in the S-I, A-P, and R-L directions, respectively. The values of Ae and Vt and the ages of the central/peripheral lung cancer patients were 0.981 ± 0.214/0.955 ± 0.289 cm, 43.314 ± 63.626/5.384 ± 10.074 cm3, and 55.458 ± 11.209/58.609 ± 15.122 years, respectively. The SCC, SCCsvr, Ai, and patient age were all larger in patients with peripheral lung cancers than those with central lung cancers. The Ae values of patients with central and peripheral lung cancers were roughly equal, whereas the Vt of patients with peripheral lung cancers were much smaller than those of patients with central lung cancers. This result is consistent with the conclusion in the section “Relationship Between Patient Characteristics and Level of External/Internal Correlation” that the Vt of lung cancers are negatively correlated with the strength of the external/internal correlation.
Discussion
Because the external/internal correlation for a patient is a complex relationship, the simultaneous motion of different anatomical structures in the process of free breathing is not a coupled equilibrium state involving simple harmonic motion but rather a complex temporally and spatially correlated process that can be modeled in terms of phase difference and time change.53,54 That is, different parts of the anatomy can move along different temporal and spatial pathways, with the external/internal correlation in a patient following a relationship involving complex phase differences and spatial transformations.
Thus, we use 2 methods, SCC and SCCsvr, to evaluate the correlation. Figure 2 indicates that in regions in which the SCC values are large, the SCCsvr and SCC scores are nearly the same, indicating that the SVR fitting primarily increases the SCC values occurring in patients with small SCC values. Understandably, the respiratory motion varies markedly between patients, 41 and therefore for some patients, SCC alone can accurately reflect the real external/internal correlation but might underestimate the correlation in other patients. For the latter, SCCsvr can better reflect the real external/internal correlation because it more faithfully reflects the complexity of this correlation through the use of an SVR network to solve it as a nonlinear problem. 52 In fact, comparing SCC and SCCsvr for both the liver and lung tumor, the proportion of patients with SCCsvr belonging to the “weak” and “moderate” grades is much lower than those with SCC, and the proportion of patients belonging to the “strong” grade with SCCsvr is more than twice those with SCC (Table 3). Therefore, for patients whose SCCsvr scores are much larger than their SCC scores (Figure 2), the external/internal correlation will be strong, and the SCC value will not fully reflect the real external/internal correlation. However, for patients with small SCC and SCCsvr values (see Figure 2) which indicate that their external/internal correlation is weak, the SCC value alone accurately reflects the real external/internal correlation and the SVR fitting does not increase this value. The results in Figure 2 also indicate that the external/internal correlation varies to a high degree depending on the patient, indicating that the external/internal correlation is highly individualized. This can be explained by the fact that respiratory and tumor motion are complicated and vary for each patient.37–41
In this work, we explored the relationship between the external/internal correlation and several patient clinical characteristics. It is seen from Figure 3 that both Ai and Ae are positively correlated with the strength of the external/internal correlation, whereas patient age is negatively correlated with the strength of the external/internal correlation. This indicates that, patients with large Ai or Ae values as well as those who are younger often have better respiratory functioning, leading to strong external/internal correlation. Vt is positively correlated with the strength of the external/internal correlation in patients with liver tumors but negatively correlated in patients with lung tumors. For liver cancer patients, larger values of Vt correspond to a higher degree of consistency between the tumor and liver motion. Because there is also a strong correlation between liver motion and patient respiratory motion, the external/internal correlations in liver cancer patients with larger tumor volumes, Vt, will be stronger. For lung cancer patients with smaller Vt values, the tumors will generally have a reduced degree of involvement with the surrounding lung tissues and bronchi and the patient’s tumor motion will be driven more by their respiratory motion, leading to stronger external/internal correlations in lung cancer patients with smaller Vt.
The association of gender with breathing type (with females tending to perform chest breathing and males performing abdominal breathing) might affect the external/internal correlations in cancer patients of different genders. 41 In the section “External/Internal Correlations in Liver and Lung Cancer Patients of Different Genders”, we explored the relationship between the patient’s external/internal correlation and their gender, with the results in Figure 4 depicting that there was no consistent intergender trend in terms of the SCC or SCCsvr scores and that there was only a small intergender difference in the external/internal correlation along the S-I direction. These results indicate that gender is not an important factor in determining a patient’s external/internal correlation. In the section “External/Internal Correlations in Patients With Central/Peripheral Lung Cancers”, we explored the relationship between the patient’s external/internal correlation and the type of location of the lung tumor, with the results indicating that the Ai values and the external/internal correlations in patients with peripheral lung tumors were larger than those of patients with central lung tumors. Central lung tumors are involved with and adhere to the bronchi and other tissues because they are closer to them; as a result, their Ai values will be smaller. According to the results presented in Figure 3, Ai is positively correlated with the strength of the external/internal correlation, indicating that the external/internal correlations in patients with central lung cancers are smaller than those in patients with peripheral lung cancers (Figure 5).
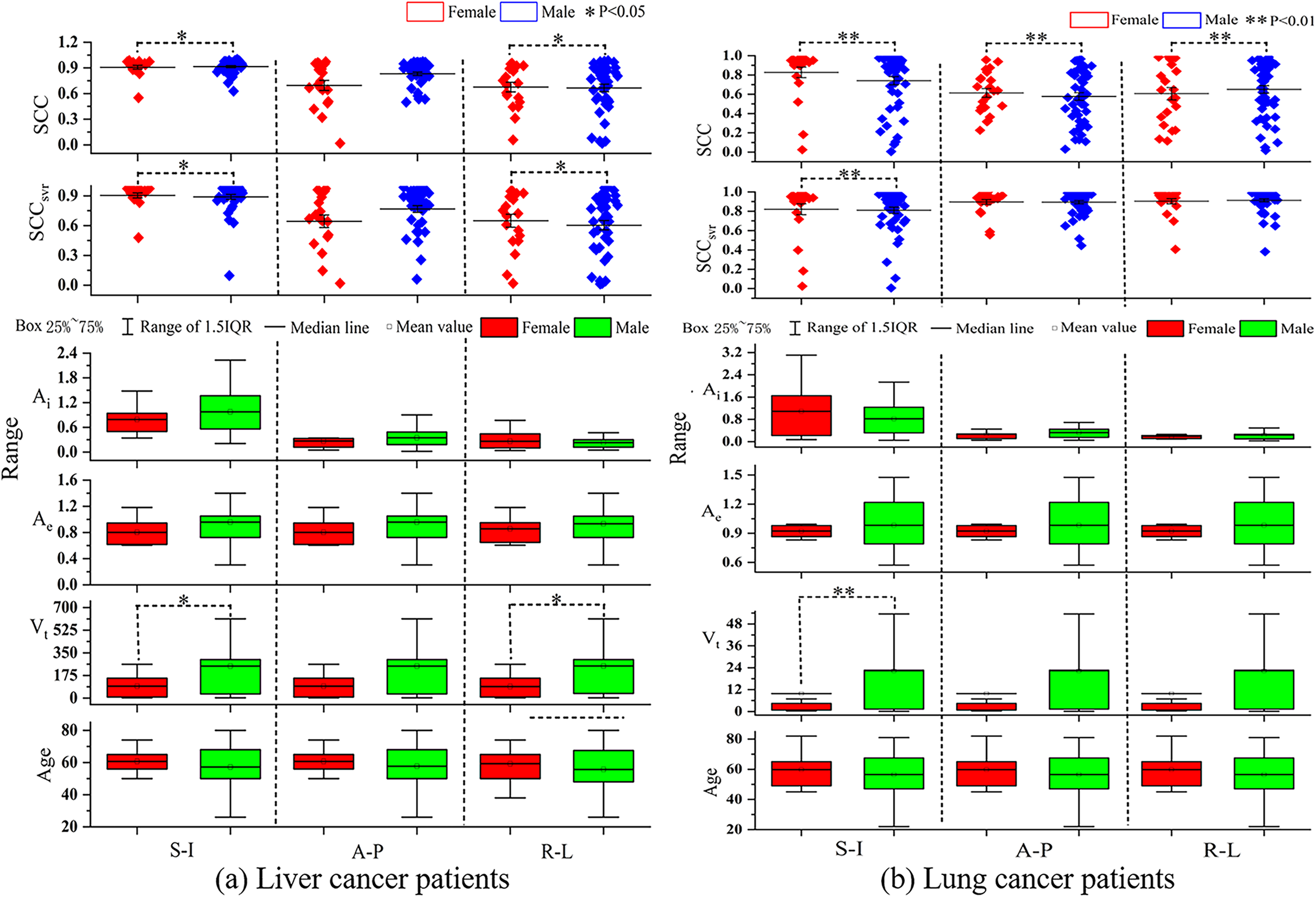
External/internal correlations and other characteristics of (a) liver and (b) lung cancer patients of different genders in the SI, AP, and LR directions. The box represents that the values lie between 25% and 75% and the middle line the mean value of each characteristic.
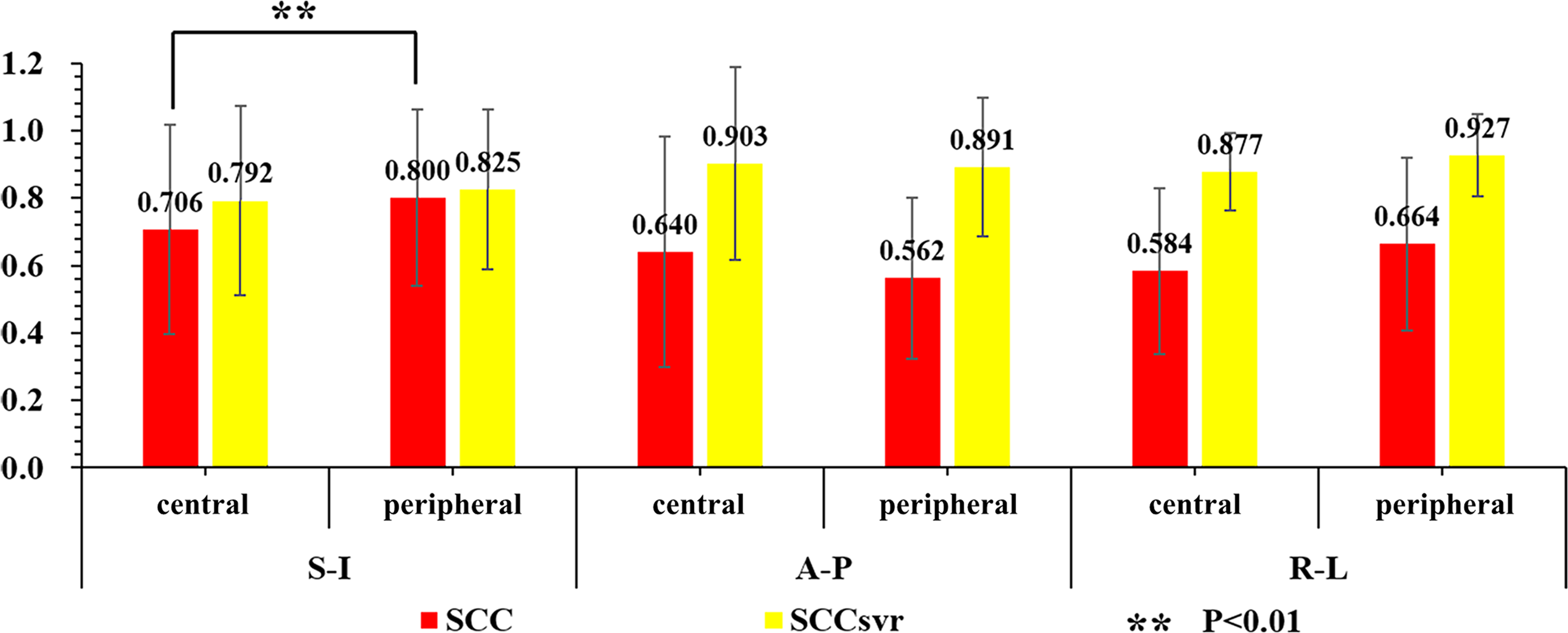
External/internal correlations of patients with central/peripheral lung cancers. A P value of .05 was defined as statistically significant.
Totally, as the SCC and SCCsvr values between the external/internal correlation and patient characteristics are all smaller than 0.5, and 77.1% of them are less than 0.3 (Table 2), the effects of Ai, Ae, Vt, and age on the external/internal correlation in both liver and lung cancer patients were weakly to moderately correlated. It was also found that the external/internal correlations in both types of cancer patients differed to a high degree depending on each patient. Together, these findings suggest that, in cases in which the characteristics of a liver or lung cancer patient may not provide strongly correlated guidance in conjunction with their external/internal correlation, the SGRT technique should be individualized to assess the patient’s external/internal correlation prior to using it for breath motion monitoring.
Our approach was limited in terms of the obtaining of internal tumor motion data. Data based on 4DCT scans lacks real-time tumor motion information and only a small amount of tumor motion data could be obtained for each patient. In the future, we will seek to actively obtain synchronous external/internal real-time motion signals from tumor patients to enable a quantitative study to determine the threshold of external/internal correlation with the goal of deciding whether specific patients are suitable for indirect real-time tracking techniques based on the use of an external/internal correlation model. In addition, as breathing training may be beneficial in many cases, 41 we will further verify and quantitatively evaluate whether such training can help improve patients’ external/internal correlation.
Conclusion
In this study, we determined that the external/internal motions in liver and lung cancer patients are strongly correlated in the S-I direction; however, the external/internal correlation may differ by a high degree for various patient. In addition, the effects of the factors Ai, Ae, Vt, and patient age on the external/internal correlations were all weakly to moderately correlated for both liver and lung cancer patients. Our results suggest that, before the SGRT technique is used for lung or liver cancer patient breath motion monitoring, it should be individualized to assess the patient’s external/internal correlation.
Footnotes
Ethical Approval
This study was approved by the ethics committee of the West China Hospital with no need for patient consent and registered by the Chinese Clinical trial Registration Center with registration number ChiCTR2100042714.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant numbers 81972848, 81472807, and 81803056), and the Sichuan Science and Technology Program (grant number 2021YFS0143).
