Abstract
Osteosarcoma is the most common primary bone malignancy. We aim to investigate that role of M-phase phosphoprotein 8 (MPP8) on proliferation and apoptosis in osteosarcoma. Briefly, the current research reported an in vitro study investigating the role MPP8 in OS tumorigenesis. Consequently, we found that the MPP8 expression was upregulated in osteosarcoma tissues and in osteosarcoma cell lines. Interestingly, MPP8 knockdown via shRNA restrained the cell viability and proliferation of U2OS and Saos-2 cells. In addition, MPP8 knockdown promoted the apoptosis of U2OS and Saos-2 cells, while MPP8 overexpression promotes proliferation and inhibited the cell apoptosis of osteosarcoma cells. These results suggested that MPP8 may serve as a contributor for osteosarcoma growth and inhibition of MPP8 may help restrain the development of osteosarcoma. Importantly, we found that MPP8 overexpression suppressed the protein levels of HOXA5, p38αMAPK, increased cell proliferation and inhibited cell apoptosis, while co-transfection with HOXA5 overexpression suppressed the cell proliferation and increased cell apoptosis. These results indicated that MPP8 contributed to cell proliferation and the underlying mechanism might be involved with HOXA5/ p38αMAPK pathway.
Introduction
Osteosarcoma is one of the most common primary malignant bone tumors. 1,2 Through surgical resection and multidrug chemotherapy, up to 70% of osteosarcoma patients can survive for a long time. 3 Despite surgical excision and multidrug chemotherapy, the prognosis of osteosarcoma is still poor for patients with unresectable, primary metastasis, or recurrence. 4 Therefore, it is still meaningful to find more therapy biological target for osteosarcomas.
M-phase phosphoprotein 8 (MPP8) is considered to be a highly phosphorylated protein at mitosis. 5 It consists of 2 functional domains, a carboxy-terminus junction domain and an amino terminus staining domain, related to the residues of trimethylated histone H3 lysine 9 (H3K9me3). 5 During the course of the cell cycle, MPP8 is mainly localized on chromatin and isolated in the interphase and early mitotic stages. 6 In particular, MPP8 expression has been demonstrated to be increased in breast cancer cells, MPP8 has been indicated to promote tumor cell movement and invasion via mediating E-cadherin. 7 In addition, MPP8 has been identified to promote the gastric cancer cell growth and metastasis via regulating Bcl-2 and EMT related signaling pathways, 8 suggesting that MPP8 may serve as an oncogene for tumor progression. Several articles demonstrated the oncogenic role by conducting loss-function experiments of MPP8. For instance, MMP8 downregulation has been proved to inhibit proliferation and metastasis of colon cancer cells. 9 MPP8 knockdown has been demonstrated to inhibit melanoma cell growth both in vitro and in vivo via inducing S-phase cell cycle arrest. 10 However, whether MPP8 has an effect on osteosarcoma has not been reported.
Studies have shown that HOXA5 was upregulation in osteosarcoma 11 -13 and down-regulation of MPP8 can lead to increased expression of HOXA5 in NSCLC cells at mRNA and protein levels. 14 Upregulation of HOXA5 in osteosarcoma can inhibit cell proliferation, promote apoptosis, and activate the P38 MAPK pathway by increasing the level of p53. 13 Therefore, we hypothesized that MPP8 may regulate the P38 MAPK pathway through HOXA5 in osteosarcoma, thereby regulating osteosarcoma cell survival. Our present study revealed that MPP8 contributed to cell proliferation in osteosarcoma cells and MPP8 knockdown showed suppressed role in osteosarcoma cells, down-regulation of MPP8 can lead to increased expression of HOXA5 in osteosarcoma, which might be involved with the activation of p38αMAPK pathway.
Materials and Methods
Human Tissues and Cell Culture
18 pair of osteosarcoma tissues and normal tissues from patients were obtained from Qinghai Provincial People’s Hospital, the written consent will be obtained from all participates, which confirms the participants’ voluntarism. This study has been approved by the ethics committee of Qinghai Provincial People’s Hospital. The use of human tissues has been conducted in accordance with the tenets of the 1975 Declaration of Helsinki, as revised in 2000. Human osteoblastic cell hFOB1.19 and human osteosarcoma cell lines 143B, HOS, U2OS, SaOS-2 were obtained from ATCC (Manassas, VA, USA). The hFOB1.19 cells were maintained in Ham’s F12 and Dulbecco’s Modified Eagle’s medium supplemented with 10% fetal bovine serum (FBS). 143B and U2OS cells were cultured in RPMI 1640 medium (Invitrogen, Carlsbad, CA, USA) containing 10% FBS, 100 mg/mL streptomycin, and 100 U/mL penicillin. Saos-2 cells were kept in McCoy’s 5A culture medium (Gibco, Grand Island, NY, USA). All cells were cultured in a humidified chamber at 37°C supplied with 5% CO2.
Cell Transfection
The MMP8 shRNAs used in this experiment were designed and synthesized by Ruibo (Guangzhou, China), China. PcDNA ™3.1 was obtained from Invitrogen, USA. The U2OS and Saos-2 cells were seeded into a 6-well plate with a density of 1×10 4 cells / mL. After 24 h of culture, MMP8 shRNAs, sh-NC, MPP8 overexpression vector, or HOXA5 overexpression vector were transfected into the U2OS and Saos-2 cells with liposome 3000 (Invitrogen) alone or together. The expression of gene was detected by quantitative PCR(Q-PCR) after 24 h culture.
Q-PCR
Total mRNAs of osteosarcoma/normal tissues and cells were extracted with TRIzol Reagent (Invitrogen, Carlsbad, CA, USA) as the manufacturer’s instruction. Complementary DNA (cDNA) was synthesized by a Transcriptor cDNA Synthesis Kit 2 (Roche, Mannheim, Germany). Q-PCR reactions were performed using SYBR Green PCR Master Mix (Applied Biosystems, USA). Q-PCR conditions were listed as follows: 10 min, 94°C, 1 min, 94°C, 1 min, 56°C, and 1 min,72°C for 35 cycles, then 10 min, 72°C. Primers were listed as: left primer: ATCACAAAAGGCGCGAAAGT; right primer: TACAGGCCTTCATCAGTGCA; β-actin: left primer: ACCTGACCGACTACCTCATG; right primer: CTCGTAGCTCTTCTCCAGGG. β-actin was used as the internal control, and the target gene relative expression was normalized to β-actin.
Western Blot
Total proteins were extracted from osteosarcoma/normal tissues and cells, and the protein concentration was determined with a BCA Assay kit (Abcam, Cambridge, MA, USA). Then, 40 µg proteins were loaded onto a 10% SDS-PAGE and transferred onto a polyvinylidene fluoride membrane. After blocked with 5% skim milk, the membrane was incubated with primary antibodies separately, HOXA5 (1 µg/ml; Abcam), Bcl-2(1:2000; Abcam), Bax (1:10000; Abcam), Cleaved caspase-3(1:1000; Abcam) and P38 (1:1000; Abcam) at 4°C overnight. After washing with TBST, the membrane was incubated with horseradish peroxidase-conjugated anti-rabbit immunoglobulin G at 25 °C for 1 h. The relative expression of target proteins was normalized to the GAPDH expression.
MTT Assay
U2OS and SaOS-2 cells were cultured at a density of 4000 cells/well in 96-well plates. Cell proliferation was detected by MTT (3-[4,5-dimethylthiazolyl-2]-2,5-diphenyltetrazolium bromide) assay. U2OS and SaOS-2 cells were cultured for 48 h and then incubated with 0.5% (w/v) MTT regent for 4 h at 37°C. After that,150 µl of dimethyl sulfoxide (DMSO) was added to each well to dissolve the intracellular Formazan. OD value at 490 nm was determined by a spectrophotometer.
Clone Formation Assays
U2OS and SaOS-2 cells were inoculated in 6-well plates at a density of 200 cells/well and then kept at 37°C for 2 weeks. When the incubation was finished, the cells were fixed with absolute methanol and stained with 0.1% of Crystal Violet. Megascopic cell colonies were counted with Image-Pro Plus 5.0 software (Media Cybernetics, Bethesda, MD, USA). Each experiment was conducted at least 3 times.
TUNEL Assay
Cell apoptosis was assayed by TUNEL assay. Briefly, an In-Situ Cell Death Detection Kit-POD (Roche) was used as the manufacturer’s protocols. U2OS and SaOS-2 cells were fixed by 4% paraformaldehyde, penetrated by 0.2% Triton X-100, and incubated at 37°C for 1 h with TUNEL reagent containing TMR red labeled nucleotides. The cells were washed in 1X PBS and placed in a medium containing DAPI. Images were obtained with a fluorescence microscope (OLYMPUS, micropublisher 3.3RTV, 20X). The number of TUNEL positive cells and number of DAPI positive cells were counted. Each experiment was repeated 3 times.
Statistical Analysis
All data in this study are presented as mean ± SD and were analyzed using SPSS software (version 17.0). One-way ANOVA followed by Host-poc, Turkey-test was used to analyze the significant differences between groups. P values less than 0.05 were considered statistically significant. Each experiment was conducted at least 3 times (n = 3).
Results
MPP8 Expression Is Increased in Osteosarcoma Carcinoma Tissues and Cells
MPP8 has been demonstrated to be an oncogene in several cancers, however the role of MPP8 in osteosarcoma remains unknown. Therefore, we detected the mRNA and protein expression of MPP8 in osteosarcoma tissues and normal tissues. As a result, both of the mRNA and protein expression of MPP8 in osteosarcoma tissues were increased compared with the normal tissues (Figure 1A-B). In addition, the protein expression of MPP8 in human osteosarcoma cells (143B, HOS, U2OS, SaOS-2) was also increased compared with human osteoblastic cell hFOB1.19 (Figure 1C). The results indicated that MPP8 is highly expressed in osteosarcoma, and MPP8 may be an oncogene in osteosarcoma.

MPP8 expression is increased in osteosarcoma carcinoma tissues and cells. (A) Q-PCR was used to detect the mRNA level of MPP8 in osteosarcoma and normal tissues. (B) Western blot was used to detect the protein expression of MPP8 in osteosarcoma and normal tissues. (C) Western blot assay was performed to detect the protein expression of MPP8 in Human osteoblastic cell hFOB1.19 and human osteosarcoma cell lines 143B, HOS, U2OS, Saos-2. The experiments were repeated 3 times. **p < 0.01, compared with the normal/HFOB1 group.
MPP8 Knockdown Inhibits Osteosarcoma Cell Proliferation
To confirm the role of MPP8 in osteosarcoma, MPP8 functional complementarity experiment was carried out in U2OS and SaOS-2 cell. The MPP8 overexpression vector and MPP8 knockdown were successfully established as shown in Figure 2A. Significantly, MPP8 overexpression increased the cell viability compared with control and MPP8 knockdown restrained the cell viability, especially ShRNA1 showed satisfactory inhibitory effect, thus we chose ShRNA1 for subsequent experiments (Figure 2B). Moreover, the clone formation assay demonstrated that MPP8 overexpression increased the cell proliferation potential and MPP8 knockdown inhibited the cell proliferation (Figure 2C). The results indicated that MPP8 overexpression contributes to osteosarcoma cell proliferation, and inhibition of MPP8 lead to inhibition of osteosarcoma cell proliferation.

MPP8 knockdown inhibits osteosarcoma cell proliferation. U2OS and Saos-2 cells were transfected with (I) control, (II) MPP8 overexpression, (III) shNC, (IV) shRNA1, (V) shRNA2. (A) Western blot assay was performed to detect the protein expression of MPP8 in U2OS and Saos-2. (B) Cell viability was detected by MTT assay. (C) Cell proliferation was detected by clone formation assay. The experiments were repeated 3times. **p < 0.01, compared with control group; ## p < 0.01compared with the shNC group.
MPP8 Knockdown Promotes Apoptosis of Osteosarcoma Cells
To further confirm the role of MPP8 in osteosarcoma, the apoptosis was detected in the above cells by TUNEL assay. As a result, MPP8 overexpression decreased the cell apoptosis and MPP8 knockdown promoted the cell apoptosis (Figure 3A). The apoptosis related protein showed consistent results (Figure 3B). The results furtherly demonstrated that MPP8 contributed to osteosarcoma cell growth, while MPP8 knockdown help inhibit osteosarcoma cell growth.

MPP8 knockdown promotes apoptosis of osteosarcoma cells. (A) Cell apoptosis was performed by TUNEL assay. (B) Western blot assay was performed to detect the protein expression of related proteins. The experiments were repeated 3 times. **p < 0.01, compared with control group; ## p < 0.01compared with the shNC group.
MPP8 Regulates P38 MAPK Pathway Activity Through HOXA5
As the role of HOXA5/ p38αMAPK pathway in osteosarcoma has been studied well, 11 -13 we speculated that the MPP8 may work on osteosarcoma via HOXA5/ p38αMAPK pathway. As shown in Figure 4A, MPP8 overexpression inhibited the protein expression of HOXA5 and p38αMAPK, while MPP8 knockdown upregulated the expression of HOXA5 and p38αMAPK. To figure out the mechanism of MPP8 on osteosarcoma, the HOXA5 overexpression alone or MPP co-transfection with HOXA5 overexpression were constructed. As shown in Figure 4B, MPP8 inhibited the expression of HOXA5 and p38αMAPK, while HOXA5 overexpression restored the expression of HOXA5 and p38αMAPK. The results indicated that MPP8 regulated p38αMAPK pathway via HOXA5.
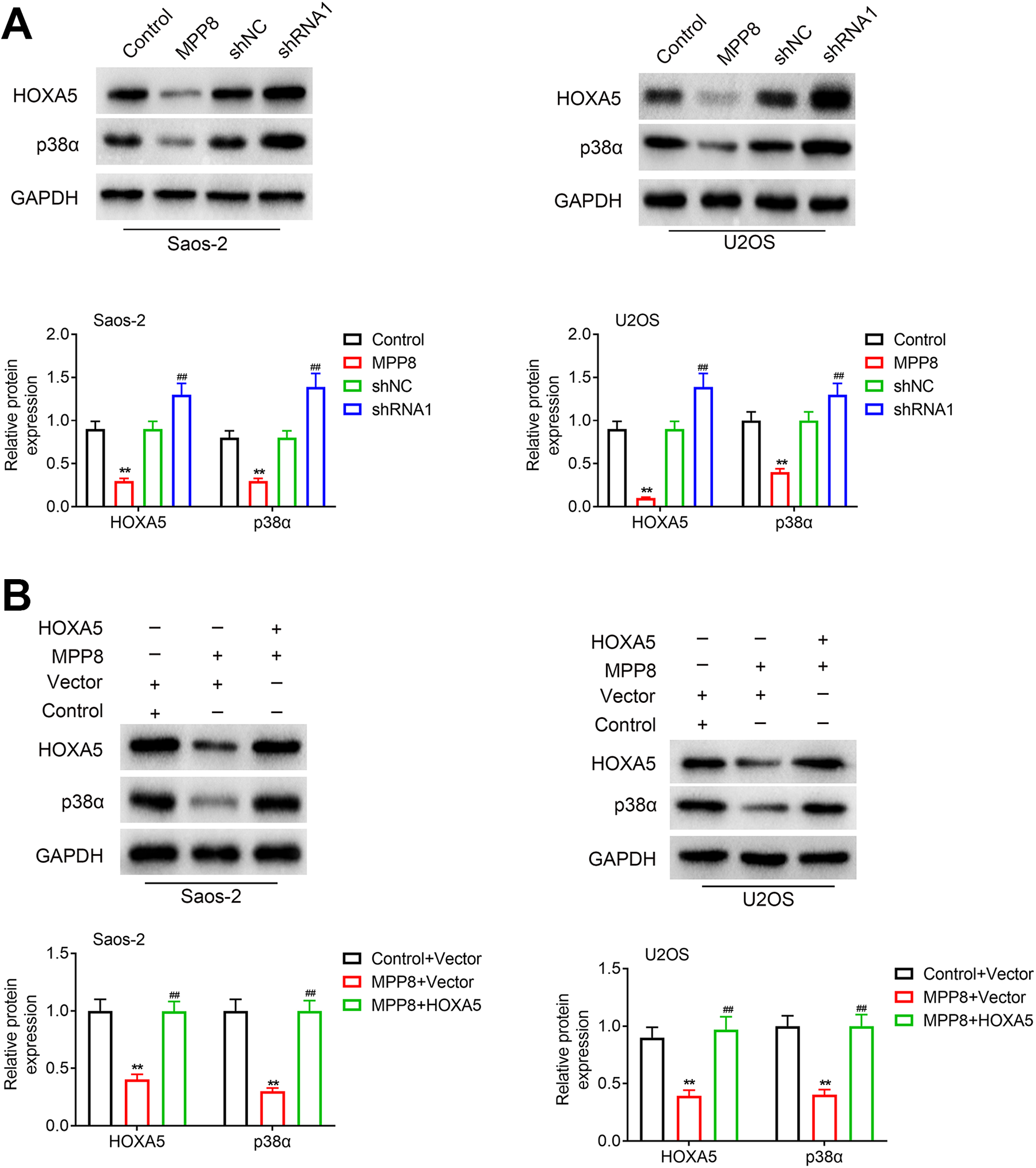
MPP8 regulates P38 MAPK pathway activity through HOXA5. (A) U2OS and Saos-2 cells were grouped into (I) control, (II) MPP8 overexpression, (III) shNC, (IV) shRNA1. Western-blot assay was performed to detect the protein expression. **p < 0.01, compared with control group; ## p < 0.01compared with the shNC group. (B) U2OS and Saos-2 cells were grouped into (I) control + vector, (II) MPP8 overexpression + vector, (III) MPP8 overexpression + HOXA5 overexpression. Western-blot assay was performed to detect the protein expression. The experiments were repeated 3 times. **p < 0.01, compared with control + vector group; ## p < 0.01compared with the MPP8 overexpression + vector.
To confirm whether MPP8 regulated cell proliferation and apoptosis via HOXA5, the biological experiments were conducted in above cell. Consequently, MPP8 overexpression increased cell proliferation (Figure 5A-B) and inhibited cell apoptosis (Figure 5C), while co-transfection with HOXA5 overexpression suppressed the cell proliferation and increased cell apoptosis (Figure 5A-C). These results indicated that MPP8 contributed to cell proliferation and the underlying mechanism might be involved with HOXA5/p38αMAPK pathway.

MPP8 regulates osteosarcoma cell proliferation and apoptosis via HOXA5. U2OS and Saos-2 cells were grouped into (I) control + vector, (II) MPP8 overexpression + vector, (III) MPP8 overexpression + HOXA5 overexpression. (A) Cell viability was detected by MTT assay. (B) Cell proliferation was detected by clone formation assay. (C) Cell apoptosis was performed by TUNEL assay. The experiments were repeated 3 times. **p < 0.01, compared with control + vector group; ## p < 0.01compared with the MPP8 overexpression + vector.
Discussion
Osteosarcoma is still a disease with a high mortality rate among children and adolescents, but early detection and timely treatment have greatly improved the survival rate of this disease. 15 After pathologic diagnosis of osteosarcoma, prior chemical or radiation therapy is initiated. Resection of tumor tissue is an important step in the treatment of osteosarcoma. 16 With the improvement of surgical techniques and the development of implants, limb preservation therapy shows a better therapeutic prospect. Therefore, prevention and early biological target screening are particularly important. More effort should be made in finding new target for the diagnosis and treatment of osteosarcoma.
The previous studies have not revealed the relationship between MPP8 and osteosarcoma, despite several studies have demonstrated the role of MPP8 in several cancers. For example, MPP8 has been shown to promotes gastric cancer growth and metastasis via Bcl-2 and EMT-related signaling pathways. 8 Interestingly, repression of MPP8 has been demonstrated to inhibited malignant behaviors like proliferation in colon cancer, 9 melanoma 10 and non-small cell lung cancer. 14 These studies suggest the oncogenic role of MPP8 in cancers. Moreover, MPP8 inhibition also contributes to apoptosis induction. 8,9 Our present study demonstrated that MPP8 is abnormally upregulated in osteosarcoma tissues and MPP8 knockdown by shRNA can suppressed the osteosarcoma cell proliferation and promote cell apoptosis, which is consistent with the previous studies.
The activation of p38αMAPK pathway is related to the suppression of cancers. 13,17,18 In a previous study, lower expression of HOXA5 was observed in human osteosarcoma cells. HOXA5 overexpression in osteosarcoma cells markedly reduced cell survival and proliferation and elevated cell apoptosis and caspase-3 activity. HOXA5 also activated the p38α MAPK pathway by increasing p53. 13 In this article, MPP8 overexpression inhibited the expression of HOXA5 and p38αMAPK, while MPP8 knockdown upregulated the expression of HOXA5 and p38αMAPK. The results indicated that MPP8 knockdown activated HOXA5/p38αMAPK pathway.
We aim to clarify the underlying relationship between MPP8 and HOXA5. Another interesting paper has revealed that MPP8 knockdown suppresses cell proliferation via regulation of HOXA5 in non-small cell lung cancer cells, 14 which give a new sight for our study. In our study, MPP8 overexpression inhibited the expression of HOXA5 and MPP8 knockdown released the suppression of HOXA5 expression from MPP8. Therefore, we can assume that MPP8 knockdown suppresses cell proliferation via regulation of HOXA5 in osteosarcoma cells. Indeed, our study revealed that MPP8 regulated cell proliferation and apoptosis via HOXA5.
In summary, MPP8 expression was abnormally upregulated in osteosarcoma tissues and cells, and MPP8 knockdown could suppress the proliferation of osteosarcoma cells, promote apoptosis, and increase the activity of P38 MAPK pathway. HOXA5 overexpression decreased MPP8 regulation of osteosarcoma cell growth and P38 MAPK pathway. Our present study revealed that MPP8 contributed to cell proliferation in osteosarcoma cells and MPP8 knockdown showed suppressed role in in osteosarcoma cells, which might be involved with the activation of p38αMAPK pathway.
Footnotes
Abbreviation
Authors’ Contributions
Tao Li and Na Li designed the study, supervised the data collection, Lei Wang analyzed the data, interpreted the data, Jia Li and Xin Zhang prepare the manuscript for publication and reviewed the draft of the manuscript. All authors have read and approved the manuscript.
Availability of Data and Materials
All data generated or analyzed during this study are included in this published article.
Ethics Approval
Ethical approval was obtained from the Ethics Committee of Qinghai Provincial People’s Hospital (Approval No. 2016-12).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
