Abstract
Objectives:
To construct a simplified prognostic risk model to predict overall survival after adjuvant radiotherapy for parotid gland carcinoma patients with stage T1-4aN1-3M0.
Materials and Methods:
We evaluated 879 patients who were pathological diagnosed as stage T1-4aN1-3M0 parotid gland cancer. Those eligible patients treated with parotidectomy and neck lymph node dissection between 2004 and 2015 in the Surveillance Epidemiology and End Results database. All cases received adjuvant radiotherapy. Independent prognostic factors included in the original model were identified by Cox regression analysis. The primary endpoint was overall survival. The model’s prediction power was evaluated by the concordance index. The entire cohort was categorized into new low- and high-risk groups using X-tile software according to the results of prognostic model. Kaplan-Meier method was used to depict the survival curves. And the statistical significance was determined by log-rank test. Besides, a heat map was visually described the association between the survival time and 2 most significant prognostic factors.
Results:
In the univariable and multivariate analyses, 4 independent factors for overall survival were age, tumor size, pTNM stage, and the number of positive lymph nodes, which were all selected in the parsimonious prognostic model. The concordance indices of the prognostic model and pTNM stage were 0.652 and 0.565, respectively. Patients in the low-risk group had better overall survival over patients in the high-risk group [unadjusted hazard ratio = 2.578, 95% confidence interval 2.095-3.172, P < 0.001]. The results of the heat map revealed that patients with smaller tumor size and fewer positive lymph nodes had much longer survival time.
Conclusions:
This parsimonious prognostic model could estimate the long-term survival after adjuvant radiotherapy for parotid gland carcinoma with stage T1-4aN1-3N0M0. The tools may be valuable to guide multidisciplinary team in making treatment decisions.
Introduction
Salivary gland cancer accounts for 1%-5% of all head and neck cancers, 1 and among which parotid gland carcinoma (PGC) accounts for 70%, with 24 pathological subtypes. 2 -4 Adenocarcinoma, adenoid cystic carcinoma, and mucoepidermoid carcinoma are the most common histological types in the salivary gland carcinoma. 5 Surgery is the main treatment for PGC, and adjuvant radiotherapy has been retrospectively reported to improve the local control effect and survival rate. 6 -8 The prognosis of PGC is complicated because of the pathological diversity and the rarity of salivary adenocarcinoma that complicates the selection of appropriate treatment strategies. According to previous reports, 5-year disease-free survival rates range from 69% to 93.6% 9,10 while the 5-year overall survival (OS) rates range from 76% to 94.6% for PGC. 10,11 Some prognostic factors of PGC have been reported, including tumor-node-metastasis (TNM) stage, age at diagnosis, molecular biomarkers, and extracapsular spread. 9,12 -15 The TNM system provides a framework for evaluating treatment outcomes and allowing prognostic evaluation of OS. 16 Previous studies had confirmed lymph node metastasis was a poor prognostic factor. 9,14 Therefore, National Comprehensive Cancer Network guidelines recommend patients with lymph node metastasis after parotidectomy should receive adjuvant radiotherapy. 16 However, patients could have heterogeneous survival outcomes in the same pathological stage diseases according to above studies. Besides, the prognoses of patients with adjuvant radiotherapy remain unclear. In practice, clinicians give follow-up recommendations and adjuvant therapy based on prediction for patients’ prognoses. Therefore, it’s important to predict the prognoses of those patients with adjuvant radiotherapy. Thus, we used patients’ data obtained from Surveillance Epidemiology and End Results (SEER) database. This study aimed to obtain an appropriate tool to improve predictive ability of long-term survival in PGC patients with adjuvant radiotherapy.
Patients and Methods
Patients
The Clinical Research Ethics Committee of our institutional center considered this study exempt because we used existing data without patient identifiers. A total number of 879 patients who underwent parotidectomy between 2004 and 2015 were enrolled retrospectively in this study. Eligible cases for this cohort study had pathologically confirmed stage T1-4aN1-3M0 based on the 8th edition of the American Joint Committee on Cancer Staging Manual. These patients were included in this study who met the following conditions: (1) pathologically diagnosed as parotid gland carcinoma; (2) 1 primary only; (3) surgical code was not “0”; (4) age was over 18 years old; (5) complete follow-up. The exclusion criteria of patients were shown in Figure 1.

The flow chart of the patient screening process in the surveillance epidemiology and end results database.
Follow-Up
In the SEER database, the median follow-up time from surgery to the last contact with patients was 34 months (range 2-154 months). The primary observed endpoint was overall survival. Overall survival was the last follow-up time or death time minus the parotidectomy time. The selection of overall survival as a primary clinical endpoint was considered most clinically relevant.
Statistical Analyses
Statistical analysis was performed using R 4.0.3 software (https://www.r-project.org/), X-tile version 3.6.1 (http://www.tissuearray.org/rimmlab), and SPSS Statistics 25.0 software (IBM SPSS, Inc., Chicago, IL, USA). X-tile software was used to determine the optimal cut-off value of positive lymph nodes, examined lymph nodes, and model’ risk score. 17 Previous studies have shown that X-tile software was similar to the time-varying receiver operating characteristic curve analysis and could provide the best cut-off value for continuous data. 18,19 Hazard ratios (HR) with 95% confidence interval (CI) were calculated by univariable and multivariable Cox proportional hazard regression analyses. Univariable and multivariable analyses were performed to identify independent prognostic factors for overall survival. Variables with P < 0.05 in univariable analysis or affecting prognosis (such as race/ ethnicity, and pathological subtypes) were selected to enter the multivariable analysis to further confirm the independent prognostic factors. All cases were categorized into 2 new risk subgroups (low-risk group and high-risk group) using X-tile software based on model’s scores. Moreover, survival curves between different groups were compared by Kaplan-Meier analysis and the log-rank tests. It was considered statistically significant when the results of all statistical tests met a 2-sided P < 0.05.
The predictive ability of the simplified prognostic model was evaluated by the concordance index. Previous studies revealed that the value of concordance index was associated with the predictive accuracy. 18 The heat map was performed to visualize the relations of 2 most significant independent factors with survival time based on regression coefficient. We used the gglot2 and rms packages of R 4.0.3 software to calculate the concordance index and formulate the heat map.
Results
Patient Characteristics
The clinical characteristics of the patients from SEER database are listed in Table 1. Among the 879 cases, 604 (68.7%) of them were men and 275 (31.3%) were women. The patients’ age ranged between 18 and 104 years old (median, 64 years). In this cohort, the 3-, 5- and 10-year overall survival rates were 55%, 46%, and 37%, respectively, with the median survival time of 34 months. The cutoff value of examined lymph nodes and positive lymph nodes were 17 and 7, respectively. The majority pathological subtypes were adenocarcinoma, squamous cell carcinoma, mucoepidermoid carcinoma and ductal carcinoma. The number of N1 diseases was the most (58.0%), and most patients were white patients (85.0%).
Clinical Characteristic of Parotid Gland Cancer Patients With Stage T1-3N1-3M0 From Surveillance Epidemiology and End Results Database.

Univariable and Multivariable Analyses
As shown in the Table 2, univariable and multivariable analyses identified the 4 variables as independent prognostic factors for parotid gland carcinoma patients with stage T1-4aN1-3M0: age >65 (adjusted HR = 1.529, P < 0.001), positive lymph nodes >7 (adjusted HR = 1.867, P < 0.001), pTNM stage (adjusted HR = 1.287, P = 0.049), and tumor size (≥4.1 cm: adjusted HR = 2.128, 2.1-4.0 cm: adjusted HR = 1.615, all P < 0.001).
Univariable and Multivariable Cox Proportional Hazard Regression Analyses in Parotid Gland Cancer Patients.a,b
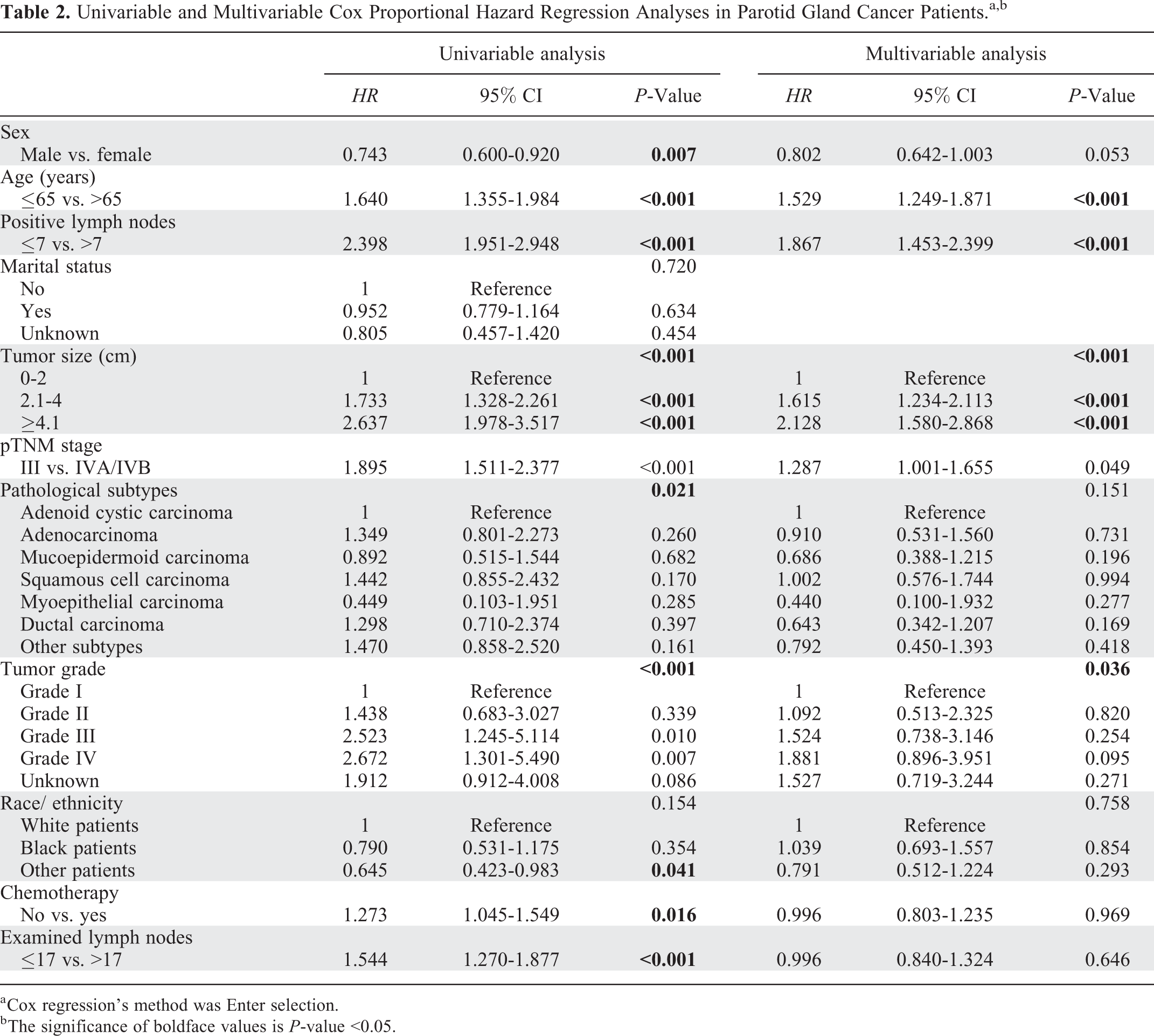
a Cox regression’s method was Enter selection.
b The significance of boldface values is P-value <0.05.
The Construction of Heat Map and Prognostic Model
A parsimonious prognostic model was generated by Cox regression after categorization of the numeric variables (age, tumor size, and positive lymph nodes). Four prognostic factors were included the original model (Table 2). According to the regression coefficient, we assigned values to the above variables. A score of 1 point was assigned to the factor with the smallest hazard ratio at Cox regression (pTNM stage), and proportionally weighting the other 3 variables: tumor size, positive lymph nodes, and age. Every patient got a total prognostic score (range 0-8) by summing the points of above 4 factors. A heat map was provided to visually observe the relation between prognostic factors of the largest regression coefficient (tumor size and positive lymph nodes) and survival time (Figure 2). The concordance index of the model for predicting overall survival was 0.652 (95% CI 0.651-0.653). To confirm the advantage of this model, we also calculated the concordance index (0.565, 95% CI 0.564-0.566) of pTNM stage. Besides, the results revealed that prognostic model had a better stratified effect than pTNM stage alone (Figure 3). We used X-tile to determine the optimal cutoff value of prognostic score based on the model as 3.5 points. The entire cohort were classified into 2 new risk groups, low-risk group (prognostic score < 3.5, n = 388) and high-risk group (prognostic score ≥3.5, n = 491).
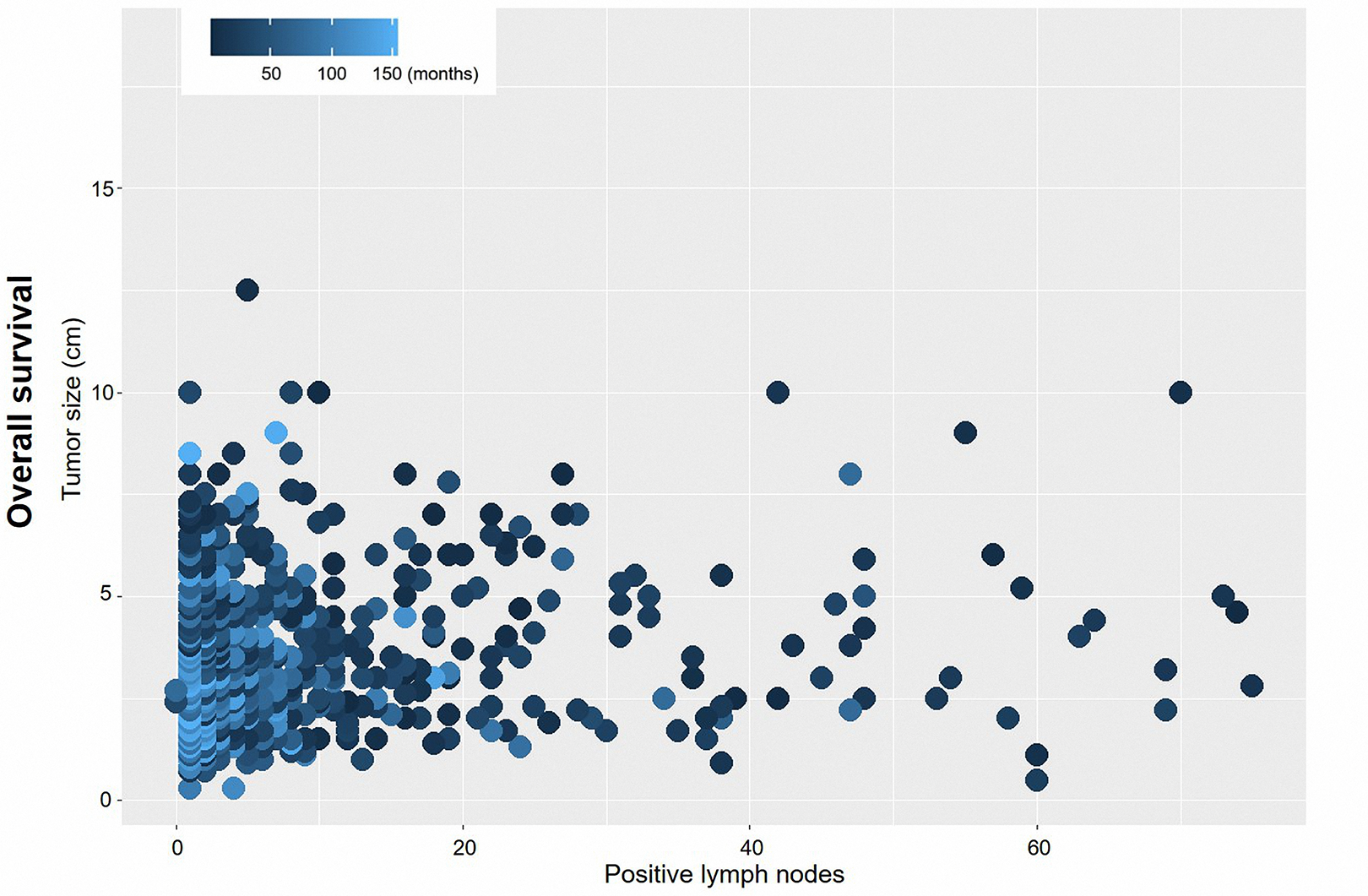
A heat map for positive lymph nodes (x-axis) and tumor size (y-axis) corresponding to survival time. Deep blue regions show relatively poorer survival outcomes than light blue regions.

Kaplan-Meier survival curves for overall survival according to the status of pTNM stage (A) and the prognostic model (B).
Patients in the high-risk subgroup had a significantly lower overall survival over the low-risk subgroup (unadjusted HR = 2.578, 95% CI, 2.095-3.172, Log-rank test: P < 0.001, Figure 3B). Our results revealed that the 3-year, 5-year and 10-year overall survival in the low-risk subgroup vs. that in the high-risk subgroup was 71% vs. 41%, 62% vs. 32% and 58% vs. 20%, respectively (Figure 3B, Log rank P < 0.001).
Sub-Group Analysis for OS
To further explore the effect of prognostic model on cohort with different variables, univariable analysis was used to estimate the correlations between prognostic model and overall survival. Our results revealed that unadjusted HR exceeded 1 or, in other words, model’s score could be a risk indictor among population of different variables except cohort with grade I, adenoid cystic carcinoma, and other races (Figure 4).

Univariable analyses of prognostic factors for overall survival in the cohort.
Discussion
In the present study, the data of SEER was analyzed by univariable and multivariable Cox regression, and then the prognostic model was constructed. Age, positive lymph nodes, pTNM stage and tumor size were considered as independent prognostic factors for those patients with PGC. Furthermore, new risk subgroups based on model’s score were also identified to have a good distinction efficiency. We also found that prognostic model had a better stratified effect than pTNM stage alone. Therefore, this model was considered to have certain clinical utility. The relative data for the prognostic model could be easily obtained from patients’ medical record, which makes it rather practical to validate our results by multi-institution study and to be applied in clinical work.
Previous studies had constructed prognostic score based on proportional Cox regression analysis. 20,21 In this study, we used same method to construct a prognostic model. Poorten et al retrospectively analyzed the medical data of 168 patients with PGC by multivariable Cox regression, and suggested that prognostic score was a useful tool to estimate recurrence-free survival for PGC patients with stage T1-4N0-3M0. 20 Thanks to their team’s effort, they validated the utility of prognostic indices in another 231 patients. 21 In the present study, our observed endpoint was overall survival, and the selection of stage was different from above studies. Because eligible cases in this study all belonged to metastasis disease of lymph nodes, those patients needed to receive adjuvant radiotherapy according to the guidelines. 16 Besides, present study aimed to evaluate and predict the prognoses in the PGC patients who received standard treatment. Therefore, there were some differences between the present study and Poorten’s study. In our study, the sample size was much larger than the above studies. Moreover, the simplified prognostic model provided an acceptable stratified effect on overall survival for PGC patients.
Histological subtypes might significantly affect the prognoses based on findings of other study. 22 Thus, we selected histological subtypes to enter into the multivariable analysis, though the P value was not smaller than 0.05. In the sub-group analysis, the prognostic model couldn’t have good performance in the adenoid cystic carcinoma and myoepithelial carcinoma, as the sample size was small. In the present study, the results revealed that chemotherapy couldn’t provide survival advantages for PGS patients with stage T1-4aN1-3M0. In fact, Kordzinska-Cisek et al had similar findings. 22 Regretably, due to the lack of detailed information about chemotherapy in the SEER database, we couldn’t make further analysis. We hope that we can use the detailed data of our cancer center to further analyze the effect of adjuvant chemotherapy on prognoses in the next study.
There are some limitations in present study. First, the sample size of PGC patients was relatively small, and the proportion of each TNM stage disease was not consistent. To address this issue, the sample size would need to be expanded in next studies. Second, the data information limited in the SEER database only, which might impact the accuracy of this prognostic model, so a multicenter study was necessary to confirm our results. Third, these findings could only provide certain reference information to the clinicians. Clinicians only used this prognostic model to evaluate the survival outcomes for PGC patients. Doctors would need to make decisions on the patients’ treatment according to the relevant guidelines and clinical experience. Fourth, data of molecular diagnoses and some pathology (such as programed death 1, programed cell death-ligand 1, vascular lymphatic invasion, and status of surgical margin) was absent, which might be effective prognostic factors contributing to a more accurate predictive effect. Thus, substantial research about the molecular level and pathological status is still needed.
Conclusions
In conclusion, this parsimonious prognostic model could estimate the long-term survival after adjuvant radiotherapy for parotid gland carcinoma with stage T1-4aN1-3N0M0. The tools may be valuable to guide multidisciplinary team in making treatment decisions.
Footnotes
Abbreviations
Authors’ Note
Conception and design: Quan Zhang, Wen-Mei Jiang, Lei-Lei Wu. Administrative support: Quan Zhang. Provision of study materials or patients: Quan Zhang, Qi-Long Ma. Collection and assembly of data: Quan Zhang, Wen-Mei Jiang, Lei-Lei Wu, Huan-Ye Wei. Data analysis and interpretation: Quan Zhang, Wen-Mei Jiang, Qi-Long Ma. Manuscript writing: Quan Zhang, Wen-Mei Jiang. Final approval of manuscript: Quan Zhang, Wen-Mei Jiang, Lei-Lei Wu, Huan-Ye Wei, Qi-Long Ma. All authors agree on the order in which their names will be listed in the manuscript. Cases were identified using the PGC participant data file from the Surveillance, Epidemiology, and End Results database, which contain deidentified patients level data that do not identify hospitals, health care providers, or patients. Therefore, institutional review board approval was waived by the Ethics Committee of Sun Yat-sen University Cancer Center. Any researchers interested in this study could contact us for requiring the data.
Acknowledgments
Our sincere thanks to our colleagues in the Department of Head and Neck Surgery, and staff in the Surveillance Epidemiology and End Results database. Last but not least, we thank our patients who are willing to provide personal information for medical research and they are the best teachers for doctors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
