Abstract
Introduction:
Stereotactic body radiotherapy (SBRT) currently adopts non-discriminative prescription regimen. This study attempts to investigate an individualized fraction regimen (IFR) method for SBRT patients with non-small cell lung cancer (NSCLC) based on Uncomplicated and Cancer-free Control Probability (UCFCP).
Methods:
Twenty patients with NSCLC were retrospectively prescribed with 40 regimens, ranging from 8Gy×5f to 12Gy×5f in step of 0.1 Gy. Taking into consideration of the age and the BMI index of each patient as well, the tumor control probability (TCP), the normal tissue complication probability (NTCP) of the total lung, chest wall and rib, and the secondary cancer probability (SCP) of the total lung were calculated for each plan of the patients. For the 40 regimens, the UCFCP was calculated and the maximum value of UCFCP was the IFR of the specified patient. Besides, IFR of UCP approach which only took account of the TCP and NTCP was also derived and to be compared with the IFR based on the UCFCP method.
Results:
For all the patients, the UCFCP value showed a bell-shaped trend with the change of prescription dose. Among the 20 patients, the IFRs of 16 patients were different from the original fixed regimen. Of the 16 patients, the IFR of 5 patients exhibited slight changes between UCP and UCFCP methods.
Conclusion:
The method based on the maximum value of UCFCP function may be helpful to provide IFR for specific SBRT patients with NSCLC, differentiating the patient specific characteristics such as anatomical structures and locations.
Keywords
Introduction
Stereotactic body radiotherapy (SBRT) is an alternative treatment modality for inoperable non-small cell lung cancer (NSCLC) patients. Since the total survival rate of patients treated with SBRT is similar to that of surgery, 1 SBRT has attracted wide attention and has become more and more accepted as a management option. In our center, for patients who receive SBRT with stage I-IIa peripheral NSCLC, the treatment prescriptions are normally non-discriminative without considering patient specific characteristics such as details of anatomic structures, BMI and ages.
Previous study
2
showed that average 3-year local control (LC) rate was 86.7% and 3-year overall survival (OS) rate was 59.6%. Though SBRT is a viable and effective treatment option for the NSCLC patients who meet the treatment criteria, many of them still suffer from chest wall pain (CWP), rib fracture (RIF), radiation pneumonia (RP) and other side effects. For SBRT patients with stage I-IIa NSCLC, the overall survival time is longer but the above mentioned side effects may seriously debase their quality of life. Mutter
In addition to side effects, secondary malignant neoplasms (SMNs) are among the most serious consequences of cancer treatment. Statistics, Epidemiology, and End Results (SEER) 6 demonstrated that the cancerous patients who received radiation treatment are 14% more likely to have SMNs than the general population. The main inducements of SMNs are the scattered and leakage photon radiation. IMRT provides better conformability and dose uniformity in the target than 3D-CRT, yet the higher monitor unit (MU) will lead to a large number of low-dose areas of OARs. It is reported that IMRT could double the risk of potentially fatal SMN compared with 3D-CRT. 7 -11 It was reported that the risk of secondary cancer in 6-MV 3D-CRT and IMRT prostate treatment could be 0.6-1.5% and 1-3.0%, respectively. 9,10,12
Currently, the fractionation scheme of SBRT is usually the same for the patients, although there are differences in patients’ specific anatomical characteristics, BMI, age, tumor size and distance between tumor and OARs. Without considering patient specific character, the use of a single prescription may cause suboptimal dosing to several patients. Thus, there is a need to further improve the prescription regiment based on patient specific characteristics, and the research on individualized fraction regimen (IFR) is warranted. Sukhikh
In this study, we propose an IFR method based on Uncomplicated and Cancer-free Control Probability (UCFCP). By investigating a range of fractional doses an individualized SBRT prescription regiment can be derived based on normal organ toxicity, tumor control probability, and secondary cancer recurrence rate, which are computed by using patient specific characteristics.
Materials and Methods
Patient Selection and Contouring
Twenty patients with NSCLC who underwent SBRT at our center from 2018 to 2019 were retrospectively investigated. All patients were diagnosed with clinical stage I-IIa peripheral NSCLC and underwent CT simulation scans on a SOMATOM Definition AS (Siemens Healthcare GmbH). The slice thickness of CT images was 3 mm.
Four-dimensional CT images were acquired to allow delineation of internal target volume (ITV) for lung SBRT. Planning tumor volume (PTV) included the entire delineated ITV plus a 5 mm margin. For OAR contouring, total lung was limited to the air-inflated lung parenchyma, and the ITV and trachea/ipsilateral bronchus were excluded. Chest wall (CW) was a 2 cm 2-dimensional expansion of the ipsilateral lung excluding the lung volume, the mediastinal soft tissue, and anterior vertebral body. The CW contour was extended 3 cm above and below the PTV. The rib contour that was within or closest to the target was delineated under a window level of 750 and a window width of 1400. Contouring of target and OARs were taken from the original clinical contours, which were delineated by experienced radiation oncologist.
Treatment Planning and SBRT Techniques
The original patient treatments were all prescribed with the standard regiment of a total dose of 50 Gy in 5 fractions. SBRT plans were generated for each of the 20 patients, strictly following the clinical and dosimetric objectives of RTOG 0915. The plans were generated using Pinnacle3TM treatment planning system (TPS, v9.10, Philips Medical Systems, Cleveland, USA) for an Edge linear accelerator (Varian, Palo Alto, CA) equipped with 120 MLC leaves (Millennium MLC) and 6MV photon beam. All the patients received SBRT on a linear accelerator equipped with CBCT using online IGRT (Varian, Palo Alto, CA) for every fraction.
Introduction of UCFCP Function
UCFCP is an integral radiobiological approach proposed by Beatriz Sánchez-Nieto

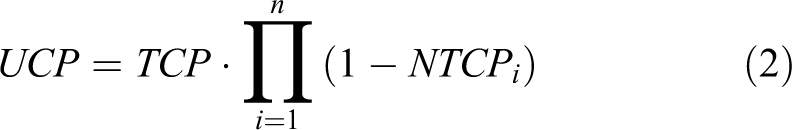
where


TCP, NTCP and SCP were calculated by using an in-house code on MATLAB 7.0 (MathWorks, USA). Those latest radiobiological models were chosen based on a large literature review. 17 -22
UCP Model
The chosen TCP model was the modified regrowth model proposed by Tai
The multivariate model of Shaun
Stam
For Radiation pneumonitis grade 2 or more (RP2 +) prediction, the universal survival curve (USC) model of Wennberg
Secondary Cancer Probability
Schneider
Individual Fractional Dose Prescription Regimen
The IFR of patient was determined based on the maximum value of UCFCP function (equation 3). The maximum value of UCFCP represents the highest treatment gain.
Each of the 20 patients were respectively re-prescribed with 40 prescription regimens, ranging from 8Gy×5f to 12Gy×5f in step of 0.1 Gy for fractional dose. Forty sets of DVH data of each patient were then obtained. Dose volume histogram (DVH) of PTV (DVHPTV), total lung (DVHLung), CW (DVHCW), and rib (DVHRib) were extracted from the TPS at a resolution of 1 cGy. According to patient’s specific age and BMI index, the TCP and the NTCP of total lung (NTCPLung), CW (NTCPCW) and ribs (NTCPRib), and the SCP of total lung (SCPLung) were calculated. Finally, the UCFCP of the 40 regimens were calculated and the maximum value of UCFCP was determined to be corresponding to the IFR of the specific patient. A flow chart of the fractional regimen individualization is presented in Figure 1.

A schematic diagram of the IFR method.
Results
Patient Characteristics
The median age of the enrolled patients was 72.9 years old. The investigative prescription dose ranged from 40Gy/5f to 60Gy/5f (BED10 = 72-132 Gy, biological effective dose, α/β = 10). The median GTV and PTV were 11.50 cm3 and 35.17 cm3, respectively.
Dependence of UCFCP on the Prescription Dose
The dependence of the UCFCP value on prescription dose for a typical patient (patient 14) is shown in Figure 2. It can be seen that as the prescription dose increased, the UCFCP value presented a bell-shaped trend. UCFCP reached a maximum value of 0.74 at a total dose of 47 Gy with the corresponding fractional dose at 9.4 Gy. The dependence of UCFCP as a function of prescription dose is similar to the one shown in Figure 2 for all patients, although the maximum value and its corresponding prescription regimen varied from patient to patient.

An example of UCFCP values dependence on prescription dose. Star (★) stands for the maximum value of UCFCP.
Individualized Fraction Regimen
Figure 3 lists the maximum values of UCFCP for all of the 20 patients, as well as the corresponding TCPs, total doses, fractional doses, NTCPs, and the SCPs. As can be seen in Figure 3A, the maximum value of UCFCP, which represents the optimal radiotherapy gains, was quite different from patient to patient, with the largest value of 0.79 (patient 4) and the smallest value of 0.65 (patient 15). It is observed in Figure 3B that the TCP value was greater than 0.92 (range: 0.92-0.97) for all patients.

(A) UCFCP, (B) TCP, (C) prescription dose, (D) fraction dose, (E) NTCPLung, (F) NTCPCW, (G) NTCPRib, and (H) SCP of all patients.
Figure 3C and Figure 3D show the optimal total prescription dose and fractional dose. They varied from patient to patient, with the optimal total doses ranging from 45.5 Gy to 51.5 Gy, and fractional doses ranging from 9.1 Gy/f to 10.3 Gy/f.
Figure 3E, 3F and 3G are the NTCP values of total lung, chest wall, and ribs of 20 patients, respectively. Figure 3H shows the SCP of patient. Among them, the SCP value is relatively small.
Comparison of IFR Based on UCFCP and UCP Method
Table 1 lists the IFR comparison of the 20 patients derived based on UCFCP and UCP methods, respectively. The IFRs of 16 of the patients were different from the non-discriminative standard prescription of 10.0 Gy × 5 f. They ranged from 9.1 Gy × 5f to 10.3 Gy × 5 f. Among the 16 patients, 5 slight differences of IFR between UCP and UCFCP approaches were observed for 5 patients. For example, for patient 4, the IFR based on the UCP model was 10.4Gy/f, and it slightly decreased to 10.3 Gy if the UCFCP method was applied. For patient 10, the optimal fractional dose obtained by the UCP function was 9.7 Gy, while the utilization of UCFCP model increased the IFR to 9.8 Gy/f. The above examples suggest that when the SCP was included in the consideration of treatment gain, the IFR could change, although slightly.
Characteristics of Patients.

RUL: Right upper lobe; RLL: Right lower lobe; LUL: Left upper lobe; LLL: Left lower lobe.
The PTV-CW distance equals to zero represents that PTV partly invades neighboring CW.
Analysis for IFR of Typical Patients
CT images of 4 patients with representative targets are presented in Figure 4, and their corresponding UCFCP curves are shown in Figure 5. Figure 4A shows the CT image of a relatively small target (18.03 cc), far away from CW (distance is 5.50 cm), and the derived IFR was 10.3 Gy×5 f. Figure 4B presents a case with small target (9.67 cc) which was close to the CW (0.4 cm); The derived IFR for this patient was 10.1Gy×5 f. Figure 4C shows the image of a patient whose treatment target was relatively large (61.23 cc) and was far away from CW (1.28 cm); and the IFR was 9.4 Gy×5 f. In Figure 4D, the CT image of a patient with large target (61.71 cc) close to CW (0 cm) was presented; and the IFR was 9.1Gy×5 f. It is interesting to observe that the smaller the target was and the farther away from the chest wall the derived IFR was with a larger fractional dose.

CT images of 4 representative patients: (A) Small target which is far from CW, (B) small target close to CW, (C) large target which is far from CW, and (D) large target close to CW.

The UCFCP of 4 typical patients varies with the prescribed dose, and star represents the IFR corresponding to the largest value of UCFCP.
Discussion
Individualized prescription regimen may be one of future directions for radiotherapy. Due to its nature of high fractional dose, the efficacy of SBRT is directly related to the dose fractionation regimen. Therefore, the demand for customizing prescription based on the patient’s specific characteristics is more warranted than the conventional fractionation regiment. The use of IFR may improve the therapeutic effect. Sukhikh
Many clinical studies have been conducted to search for the optimal dose of SBRT for early stage NSCLC. Onishi
Our research on IFR was based on UCFCP function, which can be used to explore alternative regimen of SBRT based on the expected value of tumor control rate, normal tissue damage and secondary cancer probability. According to the value of UCFCP, the treatment gains generated by different regimens were sorted, and the regime corresponding to the maximum value of UCFCP was deemed as the optimal prescription. Our study showed (Figure 2) that as the total dose of prescription increased (the number of fractions remained the same), the UCFCP value presented a bell shape trend. In addition, due to the direct or indirect impacts of age, BMI index, target size and location, the IFR among the patients varied, ranging from 9.1Gy×5f to 10.3Gy×5f, and 16 of all 20 patients’ IFRs were different from the standard prescription, indicating that it is very likely that the optimal prescription regiment is indeed patient specific. In all the investigated cases in this study, the TCP of the derived IFR was all higher than 92%.
Table 2 lists the comparisons of IFR based on the UCP and UCFCP methods. The UCP method comprises TCP and NTCP indicators. UCFCP function adds SCP index on the basis of UCP function. The comparison shows that as the SCP was included in the study of IFR, 5 of 16 patients had slight changes in IFR, which indicates that inclusion of SCP had impacts to IFR. In our research, the range of SCP was 0.4%-1.2%, which is consistent with the data from SEER, 6 confirming the appropriateness of the model we chose.
IFR Comparisons of UCFCP and UCP Methods.

Bold text represents patients who have different IFRs between UCFCP and UCP methods.
The size of tumor, the distance between PTV and the chest wall, and the volume of total lung may affect TCP, NTCP, and SCP, which also affect IFR. Figure 5 shows that patients with small target or target far from the chest wall could be prescribed with relatively large fractional dose, while fractional dose of patients with larger target or target close to the chest wall needed to be appropriately decreased. The derived results are consistent with clinical observations. Clinically, a patient with a larger target has a higher irradiation dose of normal lung tissue and a greater probability of radiation pneumonia. It is necessary to properly reduce the risk of pneumonia while ensuring a certain local control rate. Similarly, if target areas are close to the chest wall, high radiation dose to the target areas may inevitably spread doses to the chest wall leading to a much higher probability of chest wall pain and rib fractures. Therefore, radiation oncologists will usually consider reducing the target dose to protect the chest wall and ribs. For the target that is small or far away from the chest wall, the radiation doses to chest wall are likely to be relatively small, and radiation oncologist will likely think about increasing the target dose appropriately to improve the local control rate.
In this study, the TCP, NTCP, and SCP are all predicted with a power-law relationship with the radiation dose of target or normal tissue, which relies on the use of an oversimplified population average model, and some of the inherent functions or physiological heterogeneity of OARs are ignored by default. In addition, the parameters in the radiobiological models are questionable to a certain extent, which is the reason why the clinical validation is required to confirm the results.
Besides, UCFCP adopts equal weight for local control and each type of toxicity, which is a simplified assumption. In actual situations, weights may need to be shifted to the TCP. However, considering the complex elements involved in clinical treatment, we finally chose the simplified model. One limitation of this article is that the retrospective patients we selected are generally aged, since the elderly patients are more likely to choose SBRT because of their physical limitations. Moreover, with the development of medical technology, the life span of cancer patients has also increased to a certain extent. Therefore, radiation oncologists need to consider the possibility of secondary cancer for the elderly patients. Meanwhile, the IFR method based on UCFCP proposed in this paper is also suitable for younger patients. In addition, we have considered the individual anatomical parameters such as BMI when calculating the NTCP of CW. However, the patients are generally thin and the BMI is relatively low, our result shows that there was no correlation between BMI and IFR. Therefore, we didn’t discuss this aspect in this paper. In addition, we only studied the IFR of fractional dose, and did not investigate the effect of number of fractions. Our considerations are as follows, there is a strict fraction scheme in clinical practice for SBRT, and we choose the number of fractions prescribed by radiation oncologists before the careful and long-term clinical trial verification are conducted.
In the future research, deep learning based automatic feature extraction of tissue characteristics can be applied to predict the radiotherapy tumor control and radiotoxicity and may help to further improve the IFR studies.
Conclusion
We establish an UCFCP-based method to investigate IFR for SBRT patients with NSCLC. This method comprises TCP, NTCP, and SCP indexes to obtain the IFR based on patient specific characteristics. The developed method has the potential to design SBRT IFR for a variety of cancers beyond NSCLC.
Footnotes
Abbreviations
Acknowledgments
The author would like to thank Nurture projects for basic research of Shanghai Chest Hospital (No.2019YNJCM05) for its financial support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Nurture projects for basic research of Shanghai Chest Hospital (No.2019YNJCM05).
