Abstract
Background and Purpose:
We aimed to validate the usefulness of prescriptions based on gross tumor volume for stereotactic body radiotherapy for small peripheral lung tumors.
Materials and Methods:
Radiotherapy treatment planning data of 50 patients with small peripheral lung tumors (adenocarcinoma: 24, squamous cell carcinoma: 10, other: 1, unknown: 15) receiving breath-hold computed tomography-guided stereotactic body radiotherapy at our institution during 2013–2016 were analyzed. For each case, 3 dose prescription methods were applied: one based on 95% (PTVD95%) of the planning target volume, one based on 50% of the gross tumor volume (GTVD50%), and one based on 98% (GTVD98%) of the gross tumor volume. The maximum (GTVDmax), minimum (GTVDmin), and mean gross tumor volume dose (GTVDmean) and the dose covering 98% of the gross tumor volume were calculated to evaluate variations in the gross tumor volume dose.
Results:
Upon switching to GTVD50%, the variations in GTVDmax and GTVDmean decreased significantly, compared with variations observed for PTVD95% (p < 0.01), but the variation in GTVDmin increased significantly (p < 0.01). Upon switching to the GTVD98%, the variation in GTVDmean decreased significantly compared with that observed for PTVD95% (p < 0.01).
Conclusion:
Switching from prescriptions based on 95% of the planning target volume to those based on 98% of the gross tumor volume decreased variations among cases in the overall gross tumor volume dose. Overall, prescriptions based on 98% of the gross tumor volume appear to be more suitable than those based on 95% of the planning target volume in cases of small peripheral lung tumors treated with stereotactic body radiotherapy.
Introduction
Stereotactic body radiotherapy (SBRT) has been used for small (size ≤5 cm) tumors in peripheral lung cancer radiotherapy. In this study, we only focused on small peripheral lung tumors to discuss the concept of dose prescription in SBRT. SBRT has been reported to offer good local control for patients with small peripheral tumors. 1 -4 Relatively consistent outcomes have been reported at several institutions, with local control rates of 80%–90%. However, many aspects of SBRT, such as equipment choice, irradiation method, administered dose, and fractionation have not been standardized across institutions. 5 Currently, one of the widely used methods in Japan is prescribing a dose covering 95% of the planning target volume (PTVD95% prescription) using a superposition algorithm. However, when using the superposition algorithm, Monte Carlo, or other dose calculation algorithms in the lung region, the use of PTVD95%-based prescription results in variable gross tumor volume (GTV) doses among cases, even when the prescribed doses are equal. An example of such a variation is presented in Figure 1. Clinically, this variation cannot be overlooked as GTV dose variation among cases leads to treatment heterogeneity, thus making it difficult to examine the radiation dose-response relationship of small peripheral lung tumors.

Example of GTV dose variation between cases with PTVD95% prescription. Dose distributions and dose volume histograms are shown for the PTVD95% prescription. The GTV dose was much higher in case B than in case A. GTV: gross tumor volume; GTVD98%: dose covering 98% of the gross tumor volume; PTV: planning target volume; PTVD95%: dose covering 95% of the planning target volume.
GTV-based prescriptions have been proposed for minimizing GTV dose variations among cases with identical prescribed doses. However, only a few studies have investigated and compared the GTV dose variations that are associated with PTVD95%- and GTV-based prescriptions. These prior analyses consist of simulations based on a few clinical cases 6 and phantom studies. 7,8 To date, no report has analyzed a large number of clinical cases. Furthermore, prior reports have mainly used Monte Carlo methods for dose calculation algorithms, 6 -9 and almost none have used superposition methods.
Therefore, in the present study, we aimed to validate GTV dose variations by using the dose prescription method based on the GTV. The primary endpoints were the GTV dose variations among cases. The secondary endpoints were the changes in the GTV dose variation observed among cases after switching the dose prescription method. To this end, we performed and analyzed simulations based on radiotherapy treatment planning data of 50 cases who received SBRT for small peripheral lung tumors at our institution.
Materials and Methods
We used radiotherapy treatment planning data of 50 patients who received breath-hold computed tomography (CT)-guided SBRT for small (≤5 cm) peripheral lung tumors at our institution between April 2013 and December 2016. Eligibility criteria for inclusion in this study was pathologically proven or clinically diagnosed Stage I (Union for International Cancer Control TNM classification of malignant tumor Seventh Edition 10 ) lung cancer with the tumor located at least 2 cm from a proximal bronchial tree. The cases were enrolled consecutively. Table 1 shows the background information of the 50 cases.
Characteristics of the 50 Cases.

ECOG: Eastern Cooperative Oncology Group; PS: performance status; Adeno: adenocarcinoma; Sqcc: squamous cell carcinoma; GTV: gross tumor volume; PTV: planning target volume.
We used an Aquilion LBCT scanner (Toshiba Medical Systems, Tokyo, Japan) for treatment planning with the following CT imaging parameters: field of view, 55 cm; tube current, 300 mA; voltage,120 kVp; beam pitch, 0.8; slice spacing, 2 mm; and slice thickness, 2 mm. To measure the breath-holding error (in millimeters), we used CT images taken 3 times during breath-holding in 3 directions (craniocaudal, lateral, and dorsoventral) on a CT console or radiotherapy planning support software (MIM Maestro®; MIM Software, Cleveland, OH, USA). A 3-dimensional treatment planning system (Pinnacle version 9.2–9.8; Philips, Eindhoven, The Netherlands) was used, and the treatment device was Synergy (Elekta, Crawley, UK; Agility gantry head; multi-leaf collimator width, 5 mm). Treatment planning was conducted according to the Japan Clinical Oncology Group (JCOG) 0702 11,12 protocol, but we placed no limit on the homogeneity index (HI), which was set to <1.60 in the JCOG0702 protocol. For calculation, we used the collapsed cone convolution algorithm with a heterogeneity correction. The GTV consisted of the tumor that was visible on CT (the attending physician set the GTV for each patient based on the CT image). The clinical target volume (CTV) was identical to the GTV. The internal target volume (ITV) was obtained by expanding the CTV in all directions to compensate for breath-holding error. First, 3 breath-hold CT images that at least included the tumor were acquired at the planning simulation. Then, the coordinates of a landmark in the tumor volume for each CT image were evaluated using a CT console or radiotherapy planning support software (MIM Maestro®). The displacement of coordinates between the 3 CT images was calculated in 3 directions (craniocaudal, lateral, and dorsoventral), and the obtained value was added as the margin for CTV to ITV expansion. The planning target volume (PTV) was obtained by expanding the ITV by 3–5 mm in all directions. The beam shape (irradiation field) was set by adding a 0–5 mm margin to the PTV periphery. The port margins in every direction were defined according to tumor location, tumor size, and mass density in each case; the margins were set using the auto-surround function. The multi-leaf collimator (MLC) positions were set outside the PTV. SBRT was delivered by non-coplanar fixed multi-port (7–11 ports) irradiation with 4–10MV x-rays. During the radiation treatment, one fraction lasted about 30 minutes. Planning of the original treatment was randomly assigned to one of 5 radiation oncologists to minimize individual biases in plan details (such as GTV settings, beam placement, or beam weight). We explicitly decided not to perform re-planning in this study and instead chose to use the target (GTV and PTV) and treatment beam settings (number of beams, energy, placement, shape, and weight) of the original plans to make the results of this investigation more generalizable.
For GTV-based prescriptions, we selected doses covering 50% and 98% of the GTV (GTVD50% and GTVD98%, respectively). We evaluated the degree of GTV dose variations among cases with PTVD95%, GTVD50%, and GTVD98% prescriptions, and the changes in GTV dose variations among cases when the dose prescription method was switched from PTVD95% to GTVD50% and GTVD98%.
This study was approved by the Research Ethics Committee of Faculty of Medicine, University of Yamanashi (approval no. 1702). The procedures for this study were performed in accordance with the ethical standards of the Helsinki Declaration (1964, amended most recently in 2013 by the World Medical Association). Written informed consent from patients was not required because this study had a retrospective observational design.
GTV Dose Variations Among Cases
We selected the maximum GTV dose (GTVDmax), minimum GTV dose (GTVDmin), mean GTV dose (GTVDmean), and GTVD98% as the GTV dose elements.
To examine the effects of the 3 dose prescription methods on GTV dose variations, we conducted 3 simulations for each case (1 per dose prescription method). First, we performed calculations for the administration of 5000 cGy (D1) for each case using the PTVD95% prescription method. For these calculations, we used the collapsed cone convolution superposition algorithm with a heterogeneity correction. The grid resolution was 2 mm. Subsequently, we used the same dose calculation algorithm for each patient to administer 6260 cGy for the GTVD50% prescription method [D2: the mean value of the GTVDmean for the 50 cases under the PTVD95% prescription method (5000 cGy)] and 5632 cGy for the GTVD98% prescription method [D3: the mean of GTVD98% for all cases under the PTVD95% prescription method (5000 cGy)]. For each of the 50 cases and 3 dose prescription methods, we obtained the GTVDmax, GTVDmin, GTVDmean, and GTVD98% values (all in cGy) with a dose-volume histogram (DVH). We calculated the maximum, minimum, mean, median, and standard deviation for each GTV dose element among cases for the 3 dose prescription methods. Variation was evaluated in terms of standard deviation. Furthermore, we displayed the DVHs of the GTV and PTV for the 50cases with the 3 prescription methods using graphs to obtain a visual representation of the variations in DVH for comparison and evaluation.
Changes in GTV Dose Variations Among Cases Caused by Switching the Dose Prescription Method
We calculated the GTVDmax, GTVDmin, GTVDmean and GTVD98% values (all in cGy) for the PTVD95%, GTVD50% and GTVD98% prescription methods and evaluated the changes in GTV dose variations among cases following switching of the prescription method from PTVD95% to GTVD50% and GTVD98%. The variation was evaluated in terms of variances, and we used the F-test to test for significant differences. P-values < 0.05 were considered statistically significant. We used Microsoft Excel 2008 (Redmond, WA, USA) for these calculations.
Results
GTV Dose Variations Among Cases
Tables 2 -4 displays the maximum, minimum, mean, median, and standard deviation values for GTVDmax, GTVDmin, GTVDmean, and GTVD98% with the 3 prescription methods. Figure 2 displays the DVHs of the GTV and PTV for the 50 cases under each dose prescription method. Out of the GTV dose elements, GTVDmax exhibited the highest variation (standard deviation) for all 3 dose prescription methods. For both PTVD95% and GTVD98% prescriptions, there was more variation in GTVDmean than in GTVDmin. Regarding the DVHs of the GTVs and PTVs for the 50 cases with the 3 prescription methods, the DVH of the GTV was relatively dispersed; particularly, the variation in the DVH of the GTV was large in the high-dose area with the PTVD95% prescription method. Although the DVH of the GTV was relatively non-dispersed in the GTVD50% prescription method, this prescription method showed increased variation in the DVH of the GTV in the low-dose area, as compared with the variation observed for the other 2 prescription methods. The GTVD98% prescription method was characterized by relatively low variability in the DVH of the GTV in the low-dose area, but the variability increased toward the high-dose area. Comparison between the PTVD95%- and GTV-based prescription methods showed that the variations in the DVHs of the GTV were non-significant for the GTVD98% and PTVD95% prescription methods but were significant for the GTVD50% prescription method.
Variation in GTV Dose According to Dose Prescription Method (OR) PTVD95%-Prescription.

GTVDmax: maximum gross tumor volume dose; GTVDmean: mean gross tumor volume dose; GTDmin: minimum gross tumor volume dose; GTVD98%: dose covering 98% of the gross tumor volume; GTVD50%: dose covering 50% of the gross tumor volume; PTVD95%: dose covering 95% of the planning target volume.
GTVD50%-Prescription.

GTVDmax: maximum gross tumor volume dose; GTVDmean: mean gross tumor volume dose; GTDmin: minimum gross tumor volume dose; GTVD98%: dose covering 98% of the gross tumor volume; GTVD50%: dose covering 50% of the gross tumor volume; PTVD95%: dose covering 95% of the planning target volume.
GTVD98%-Prescription.
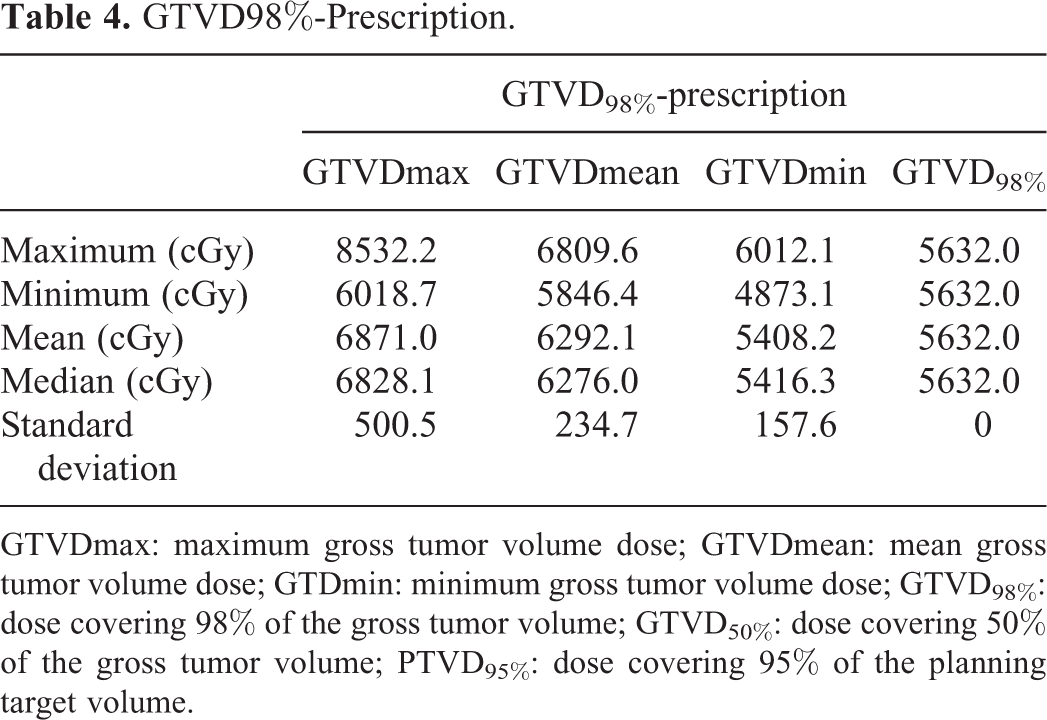
GTVDmax: maximum gross tumor volume dose; GTVDmean: mean gross tumor volume dose; GTDmin: minimum gross tumor volume dose; GTVD98%: dose covering 98% of the gross tumor volume; GTVD50%: dose covering 50% of the gross tumor volume; PTVD95%: dose covering 95% of the planning target volume.

Dose volume histograms for the 50 cases, as shown for the PTV and GTV. The prescriptions are PTVD95% (a: PTV; b: GTV), GTVD50% (c: PTV; d: GTV), and GTVD98% (e: PTV; f: GTV). PTV: planning target volume; GTV: gross tumor volume; GTVD50%: dose covering 50% of the gross tumor volume; GTVD98%: dose covering 98% of the gross tumor volume; PTVD95%: dose covering 95% of the planning target volume.
Changes in GTV Dose Variations Caused by Switching the Dose Prescription Method Among Cases
Tables 5 and 6 display the variances in GTVDmax, GTVDmin, GTVDmean, and GTVD98% with the PTVD95%, GTVD50%, and GTVD98% prescription methods as well as results of the F-test. The values of GTVD98% were always identical with the GTVD98% method and, thus, have been omitted. Switching from PTVD95% to GTV-based prescription methods reduced the overall GTV dose variations among cases. Upon switching to the GTVD50% prescription method, the variations in GTVDmax and GTVDmean decreased significantly compared with the variations observed for the PTVD95% prescription (p < 0.01), whereas the variation in GTVDmin increased significantly (p < 0.01). Although there were trends of increased variation with GTVD98%, the difference was not significant (p = 0.08). Upon switching to the GTVD98% prescription method, the variation in GTVDmean decreased significantly compared with that observed for the PTVD95% prescription method (p < 0.01). The variations in GTVDmax and GTVDmin showed decreasing trends, but the difference was not significant (GTVDmax p = 0.05, GTVDmin p = 0.15).
Changes in the GTV Dose Variation Observed Among Cases After Switching the Dose Prescription Method From PTVD95% to GTVD50%.
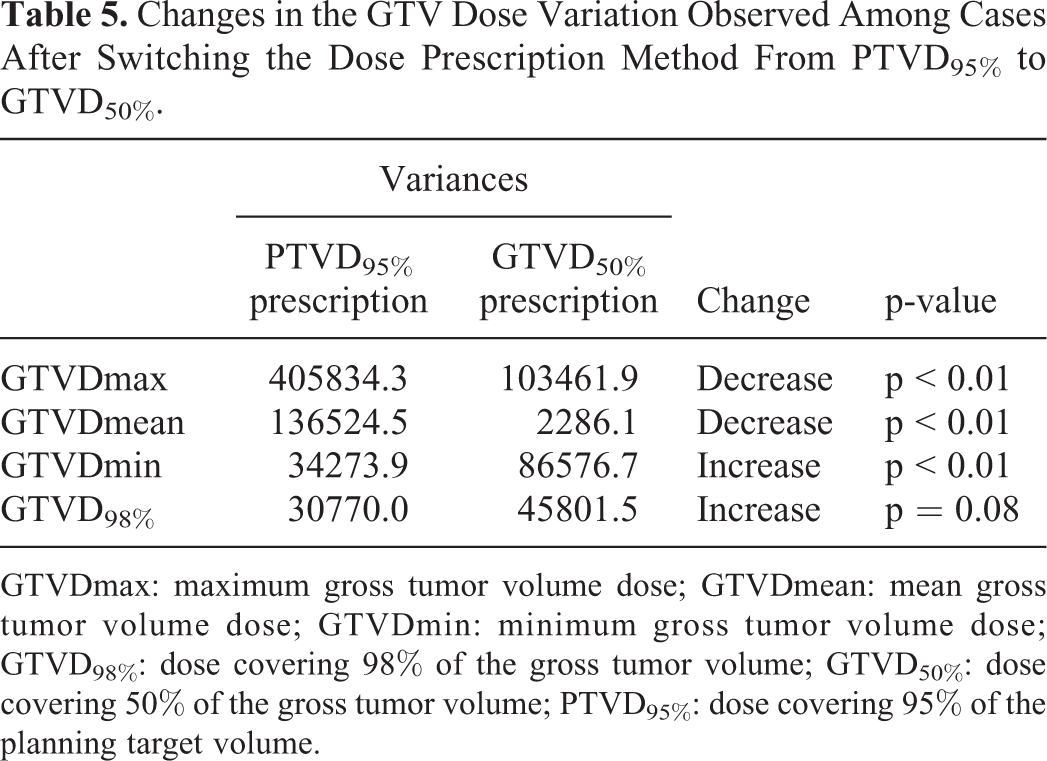
GTVDmax: maximum gross tumor volume dose; GTVDmean: mean gross tumor volume dose; GTVDmin: minimum gross tumor volume dose; GTVD98%: dose covering 98% of the gross tumor volume; GTVD50%: dose covering 50% of the gross tumor volume; PTVD95%: dose covering 95% of the planning target volume.
Changes in GTV Dose Variation Observed Among Cases After Switching the Dose Prescription Method From PTVD95% to GTVD98%.

GTVDmax: maximum gross tumor volume dose; GTVDmean: mean gross tumor volume dose; GTVDmin: minimum gross tumor volume dose; GTVD98%: dose covering 98% of the gross tumor volume; PTVD95%: dose covering 95% of the planning target volume.
Discussion
In some recent reports on SBRT for small lung tumors, it has been suggested that dose prescriptions should be based on the GTV rather than the PTV, because of the inadequacy of the latter parameter. 6,7 In comparison with the GTV, which is a purely anatomical and clinical target, the PTV is a virtual target based on geometric concepts to secure a prescribed dose for the GTV. The process of determining the PTV includes a setup margin that compensates for positioning error before and during each irradiation, as well as an internal margin that compensates for internal motion of the target. Therefore, we considered the PTV as the volume for securing the GTV dose and not for dose prescription. The variations in all GTV dose elements decreased among cases with the GTVD98% prescription method compared to the PTVD95% prescription method. On the contrary, the variation in GTVDmin increased among cases with the GTVD50% prescription method. This lack of robustness of the GTV dose may affect the therapeutic effect. The GTV dose variations were lower in low-dose areas and higher in high-dose areas among cases where the PTVD95% and GTVD98% prescription methods were used. This may indicate that there is a greater potential risk of excessive irradiation of the GTV, although it is relatively easy to ensure GTVDmin. Regarding individual curability, intratumoral dose heterogeneity is permitted to a certain degree to ensure the minimum dose. A high intratumoral dose is favorable from the perspective of antitumor effects, but large intra-patient dose variations due to excessive irradiation of the GTV are problematic in clinical trials, as they make experimental treatment evaluation difficult. For the GTVD50% prescription method, GTV dose variations among cases were almost zero near the GTVDmean, while the variation increased in both low-dose and high-dose areas. Hence, cases are unlikely to receive excessive irradiation of the GTV with the above 2 prescription methods, but such a guarantee cannot be made for GTVmin.
The prescribed doses for the PTVD95%, GTVD50%, and GTVD98% prescription methods were 5000 cGy (D1), 6260 cGy (D2), and 5632 cGy (D3), respectively. However, since the prescribed doses were not the same, it was difficult to make a direct comparison between the GTV doses associated with the different dose prescription methods. Therefore, we evaluated changes in GTV dose variations among cases in terms of the observed variances. The variations in GTVDmean and GTVDmax notably decreased upon switching to the GTVD50% or GTVD98% prescription methods; thus, we considered this decreasing variation among cases as a characteristic of the changes in the GTV dose variations associated with the switch from PTVD95%- to GTV-based prescriptions. Lacornerie et al. 6 compared 3 different platforms and PTVD95% and GTVD50% prescriptions with 2 different dose calculation algorithms for small lung lesions. Using the Monte Carlo method, they reported that the inter-platform and inter-patient variations in GTVD98% were lower for the GTVD50% prescription method than for the PTVD95% prescription method. This is contrary to our results; however, the reasons remain unclear. Switching from the PTVD95%-based method to the GTVD98%-based method decreased the overall GTV dose variations among cases. Less treatment variability is expected to make SBRT substantially more homogenous in clinical practice and to enable the detailed examination of the radiation dose-response relationship of small lung tumors. Overall, we believe that the GTVD98% prescription method is more suitable than the PTVD95% prescription method for dose prescription and evaluation in patients with small peripheral lung tumors being treated with SBRT.
There are substantial inter-patient differences in factors that affect the GTV dose and increase the GTV dose variations among cases; these inter-patient differences may be involved in the strong correlation between the GTV dose elements and influencing factors. Physical density of the lungs, irradiation field size, and target size are some of the influencing factors that are correlated with GTV dose elements. We infer that the components of air in PTV are the most influential factors for GTV dose variation among cases. In SBRT for a larger lung tumor or central lung tumor, there are few components of air in PTV compared with SBRT for small peripheral lung tumors. Therefore, the GTV dose variation in central or larger cases is smaller than SBRT for small peripheral lung tumors.
This study was conducted using SBRT data for treatments performed with respiratory management based on “breath-holding.” With this method, the positional shifts and changes in mass density caused by respiratory movements of the irradiated tumor, lungs, and peripheral organs are minimal. This renders the GTV, the setting, and evaluation of the mass densities closer to actual values rather than to hypothetical values. Accordingly, it generates highly accurate and reliable results, even with a complex radiation dose calculation algorithm, such as the superposition method with a heterogeneity correction. Nevertheless, this point is also a limitation of the study. If one wished to perform a similar study in facilities that practice different methods of preventing breathing-related shifts of the lungs during radiotherapy (such as free-breathing or breathing synchronization), then some additional hypothesized calculations would have to be made to set and evaluate the GTV and mass density 13 ; this prevents the procedures and results of the present study from being directly applied or reproduced. In this study, we insisted on using the GTV prescription policy for 3-dimensional conformal radiation therapy (3DCRT) planning under the restriction of MLC positions outside of PTV. When using intensity-modulated radiation therapy, it would be desirable to prescribe doses for not only PTV but also for GTV to maintain the robustness of GTV doses.
Conclusions
In patients receiving SBRT for small (<5 cm) peripheral lung tumors, switching from the PTVD95%-based to GTVD98%-based prescription method decreased the overall GTV dose variations. Less treatment variability is expected to make SBRT substantially more homogenous in clinical practice and to enable the detailed examination of the radiation dose-response relationship in small peripheral lung tumors. In summary, the GTVD98%-based prescription method appears to be more suitable than the PTVD95%-based prescription for dose prescription and evaluation of patients receiving SBRT for small peripheral lung tumors. Therefore, we suggest that GTVD98%-based prescription should be the standard method for prescribing and evaluating the SBRT dose for small peripheral lung tumors. Though this study is more theoretical than clinical, this stage is important before the clinical implementation. Therefore, our findings will be further evaluated in our future clinical practice.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
