Abstract
Cancer cells undergo metabolic changes that support their malignant growth. These changes are often associated with increased expression of the rate-limiting glycolytic enzyme hexokinase 2. Hexokinase 2 is an enzyme that catalyzes the conversion of glucose to glucose-6-phosphate. In this study, we utilized Gene Expression Profiling Interactive Analysis (GEPIA) database analysis and clinical sample analysis to find that hexokinase 2 was highly expressed in cervical cancer. Furthermore, we found that high hexokinase 2 expression in cervical cancer demonstrated a positive correlation with tumor size (P = .009696), pathological grade (P = .028551), and prognosis (P = .00069) but not with age (P = .956201) or lymph node metastasis (P = .131379). At the cellular level, we knocked down the expression of hexokinase 2 in the human cervical cancer cell line SiHa. The results demonstrated that knockdown of hexokinase 2 inhibited the proliferation and migration of SiHa cells and promoted cell apoptosis. During this process, knockdown of hexokinase 2 inhibited phosphorylation of AKT and mammalian target of rapamycin and promoted p53 expression. At the same time, overexpression of human papillomavirus 18 oncogenes E6 and E7 significantly promoted the expression of hexokinase 2. Most importantly, we discovered a novel upstream regulatory microRNA for hexokinase 2: miR-9-5p. Luciferase reporter assays and Western blot assays demonstrated that hexokinase 2 expression was inhibited by miR-9-5p by directly binding its 3′-untranslated region in SiHa cells. Next, we determined that miR-9-5p could suppress the proliferation and migration of SiHa cells and induce apoptosis. In conclusion, we found that hexokinase 2 serves a carcinogenic role in cervical cancer through the miR-9-5p/hexokinase 2/AKT pathway, which serves as the basis for potential therapeutic targets and prognostic indicators.
Introduction
Cervical cancer is the second most common malignant tumor in females worldwide, and it is the second most common cause of fatality in gynecological oncology in developing countries. 1 -4 Targeted therapy has become a popular research topic in recent years, and estimated glomerular filtration rate and cyclooxygenase 2 are targets of current therapies utilized in the treatment of cervical carcinoma. 5 -8 Unfortunately, the survival rate of cervical cancer has not significantly increased, and there has been an increase in adverse events. Therefore, further research regarding prognostic biomarkers and therapeutic targets is necessary.
During glucose metabolism, glucose is phosphorylated by hexokinase (HK) to be converted into glucose-6-phosphate. 9 The process of glycolysis in tumor cells not only provides energy to support the rapid proliferation, it also satisfies the huge metabolic need for the synthesis of macromolecules. 10 So far, 4 different isoforms of HKS, HK1-4, have been characterized. 11 Hexokinase-2 (HK-2) is considered the principal regulated isoform in numerous cell types. 12 Analysis of clinical specimens demonstrated that HK2 overexpression occurs in a variety of cancers, including breast cancer, 13 gastric cancer, 14 esophageal carcinoma, 15 and cervical cancer. 16 In hepatocellular carcinoma (HCC), HK-2 was found to be overexpressed in 55.67% of clinical specimens and was closely correlated with poor patient survival. 17 In addition, hyperactive aerobic glycolysis in HCC cells demonstrated a close association with high HK-2 expression. 18 Thus, interfering with tumor glycolysis would be an effective method for the clinical treatment of HCC. 19,20 3-Bromopyruvate, which suppresses tumor glycolysis via inhibition of HK-2 activity, has been designated by the Food and Drug Administration as an orphan drug. However, it has been reported to show promising efficacy and to substantially prolong the lives of patients with HCC. 21
MicroRNAs (miRNAs) are an important class of endogenous noncoding RNAs consisting of about 18 to 25 nucleotides in length. The miRNAs are responsible for the posttranscriptional regulation of the expression of about one-third of all protein-coding genes. The miRNAs suppress expression of target genes through base pairing to the 3′-untranslated region (3′-UTR) of their mRNAs, and either destabilizing or inhibiting protein translation. 22 The miRNAs have drawn much attention because of their diagnostic, prognostic, and predictive potential. They are implicated in a variety of cellular processes including differentiation, self-renewal, proliferation, metabolism, and apoptosis 23,24 and several diseases including cancers 25 and autoimmune disorders. 26
In the present study, the roles of HK2 against tumorigenesis and its underlying mechanisms in cervical cancer were investigated. Our study provides a novel strategy for treatment of cervical cancer.
Materials and Methods
Patients and Tumor Tissues
A total of 36 pairs of human cervical tumors and matched normal adjacent cervical epithelia were collected from Qilu Hospital of Shandong University from January 2015 to June 2017. Informed consent was obtained from all patients, and the experiment was approved by Qilu Hospital of Shandong University. All patients who had not received chemotherapy, radiotherapy, or other treatment were clinically diagnosed at this hospital, and their clinical data were recorded completely. None of the enrolled patients had associated tumors or serious diseases. All clinical and pathological information obtained from the case are shown in Table 1.
HK2 Expression Associated With the Clinicopathological Parameters in Cervical Tumors.

Abbreviation: HK2, hexokinase 2.
aP<0.05
Immunohistochemistry
EliVisionTMplus kit (KIT-9902) (MXB.Biotechnologies, Fuzhou, China)was used for immunohistochemical analysis. 27 Using the 40× objective lens, 3 to 5 fields of view were randomly chosen for analysis. The staining was scored as follows: Staining intensity (A) was scored 0 for no staining; light yellow as 1; light brown as 2; and dark brown as 3. The ratio of stained cells (B) was scored 1 for 1% to 10%; 2 for 11% to 50%; 3 for 51%; and 4 for over 80%. The final score was calculated by multiplying A and B. A final score between 1 and 4 was considered low expression, and anything more than 4 was considered high expression.
Cell Culture
Human cervical cancer cell line SiHa was ordered from the cell bank of the Chinese Academy of Sciences (Shanghai, China). Cells were cultured in Dulbecco modified Eagle medium containing 10% fetal bovine serum (FBS), 100 U/mL Penicillin, and 0.1 mg/mL streptomycin at 37°C with 5% CO2.
Plasmid Construction and Transfection
Lipofectamine2000 was utilized for siRNA-HK2 transfection following the manual instructions. p-Super vector was used as negative control (NC). SiRNA-HK2 was obtained from Ruibo (Guangzhou, China). Expression vectors for human papillomavirus (HPV) 18 E6/E7 have been previously described. 28 Wild-type and mutated HK2 3′-UTRs, including the predicted target sites of miR-9-5p, were cloned into a pmirGLO dual-luciferase reporter vector (Promega, Madison, Wisconsin). MiR-9-5p mimics and their NCs (miR-NC) were synthesized by GenePharma (Shanghai, China) and were also transfected into cells using Lipofectamine2000.
Fluorescence Quantitative Polymerase Chain Reaction
Total RNA was extracted using Ultrapure RNA Extraction Kit, and complementary DNA (Cdna) was synthesized using HiFiScript cDNA Synthesis Kit. The mRNA expression was detected using fluorescent quantitative polymerase chain reaction, and the expression volume was calculated with the 2− △Ct method. The primers are listed as below: HK2 sense: 5′-TGGCTGCCCACAAGATAATG-3′, anti-sense: 5′-TCAATGTCGGCGCCTATTTC-3′; E6 sense: 5′-GAACAGCAATACAACAAACCG-3′, anti-sense: 5′- TCTGCAACAAGACATACATCG-3′; E7 sense: 5′-ATGCATGGAGATACACCTAC-3′, anti-sense: 5′-CTCCTCTGAGCTGTCATTTA-3′; GAPDH sense: 5′-TGACTTCAACAGCGACACCCA-3′, anti-sense: 5′-CACCCTGTTGCTGTAGCCAAA-3′.
Western Blot
After 48 hours of transfection, cells were collected and lysed with ice-cold RIPA buffer, and the protein concentration was detected using the BCA method. Twenty micrograms of protein samples were heated at 95°C for 5 minutes for denaturing and then loaded onto each lane on 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis. After transferring, the polyvinyl difluoride membrane was blocked with 5% nonfat milk for 1 hour at room temperature and incubated with primary antibodies in blocking solution at 4°C overnight. Then, the membrane was incubated with secondary antibody in blocking buffer at room temperature for 1 hour. After washing, electrochemiluminescence substrate was added to the membrane for signal development. Images were taken using darkroom development techniques for chemiluminescence.
The primary antibodies used in this study were as follows: anti-HK2 (1:1000, Rabbit polyclonal antibody, GTX48542; GeneTex, Irvine, California); anti-BCL2 (1:2000, Mouse polyclonal antibody, 12789-1-AP; Proteintech, Manchester, United Kingdom); anti-Bax (1:1000, Rabbit polyclonal antibody, 23931-1-AP; Proteintech); anti-Caspase3 (1:1000, Rabbit polyclonal antibody, 25546-1-AP; Proteintech); anti-p-AKT (1:1000, Mouse monoclonal antibody, Ser473, 66444-1-Ig; Proteintech); anti-AKT (1:500, Mouse monoclonal antibody, #9272; Cell Signaling Technology, Beverly, Massachusetts); anti-p-mammalian target of rapamycin (mTOR; 1:1000, Rabbit monoclonal antibody, Ser2448, ab109268; Abcam, Cambridge, Massachusetts); anti-mTOR (1:1000, Mouse monoclonal antibody, ab109268; Abcam); and anti-P53 (1:1000, Rabbit monoclonal antibody, 10442-1-AP; Proteintech).
Cell Counting Kit 8 Assay
Approximately 1000 cells were seeded into each well of the 96-well plate. Cell vitality was detected every 24 hours by adding 10 μL of Cell Counting Kit 8 (CCK8) reagent. After incubation at 37°C for 90 minutes, optical density value of excitation light was detected by using enzyme standard instrument at a wavelength of 450 nm.
Transwell Assays
Cells transfected for 24 hours were trypsinized and resuspended in serum-free culture medium; 100 μL of cell suspension containing 1 × 104 cells was added into each Boyden chamber (8-µm pore size), and 600 μL of medium containing 10% FBS was added outside the chamber. The medium was incubated at 37°C for 24 hours, and nonmigrating cells were removed. Migrated cells were fixed with 4% paraformaldehyde (PFA) at room temperature for 15 minutes and stained with 0.1% Crystal violet for 5 minutes. Microscopy was utilized to observe, count, and image the invaded cells.
Wound Healing Assays
Cells of 5×105 were plated into each well of a 6-well plate and incubated at 37°C overnight to reach 100% confluent cell monolayer. After transfection for 24 hours, the cell layer was scratched with a tip to destroy a small area. Cells were then incubated in medium without serum. The open area was then detected by microscopy at a time of 0 and 48 hours, and the size was measured with Image J (http://rsbweb.nih.gov/ij/) at 6 to 8 different points.
Cell Apoptosis Analysis With Flow Cytometry
After an additional 24 hours of incubation in serum-free medium, cells were trypsinized with EDTA-free trypsin. Cells were resuspended in 1× binding buffer with 3 to 5 × 105 cells/mL. AnnexinV-FITC of 5 μL was added to the 100 μL cell suspension, which was then incubated at 25°C in the dark before the addition of 10 μL of 20 μg/mL Propidium Iodide (PI) for 2 more minutes of double staining. Results were analyzed with a flow cytometer (BD FACSC anto II; BD Biosciences, San Jose, California).
Dual-Luciferase Reporter Assay
After incubation for 24 hours, luciferase reporter plasmids, pmirGLO-HK2-3′ UTR (wild type, WT) or pmirGLO-HK2-3′ UTR mutant (Mut), were co-transfected with miR-9-5p mimic (miR-9-5p) or miR-NC into SiHa cells with by Lipofectamine 2000. After transfection for 48 hours, the luciferase activity was measured.
Statistical Analysis
SPSS 18.0 software was used for statistical analysis. The results were denoted as plus or minus standard deviation, indicated in the legend of the figure. The data represented 3 separate experiments. The Student t test is used to determine the meaning of all pairs of interest comparisons. P < .05 was determined to be statistically significant.
Results
Hexokinase 2 Shows Abnormally High Expression in Cervical Cancer and Has a Significant Association With Prognosis
Messenger RNA expression data of 306 cervical squamous cell carcinoma and endocervical adenocarcinoma samples and 13 normal cervical epithelia samples were included in the GEPIA database as the basis for the analysis of HK2 expression in cervical cancer. After online data analysis and box plot drawing, we found that the expression of HK2 in cervical cancer was significantly higher than that in normal tissues (Figure 1A, P < 0.05), suggesting that HK2 played a role in the progression of cervical cancer.

Hexokinase 2 (HK2) shows abnormal high expression level in cervical cancer and is significantly associated with prognosis. (A) The boxplot of HK2 expression level. Red and gray boxes represent cervical cancer tissue and normal cervical tissue, respectively. The data came from the Gene Expression Profiling Interactive Analysis (GEPIA) database, including 306 cervical cancer tissues and 13 normal cervical tissues. (B) The prognostic value of HK2 in patients with cervical cancer. Red and blue represent high and low expression of HK2, respectively. The data came from the Cancer Genome Atlas (TCGA) database, including 292 cervical cancer tissue. (C) Immunohistochemical staining of HK2 in cervical cancer tissues and normal cervical tissue. (D) Fluorescence quantitative polymerase chain reaction (PCR) demonstrated the expression of HK2 was reduced by small-interfering RNA1 (siRNA1). (E and F) The protein levels of HK2 were detected by Western blot. (G) The proliferation was detected by CCK8 assay. *P < .05, compared to negative control (NC). All experiments were performed independently 3 times.
The TCGA database provides the survival information of 292 patients with cervical cancer. On this basis, we analyzed the prognostic value of HK2 in tumors. Through online data analysis, we found that the patients with low HK2 expression in cervical cancer had an overall better prognosis compared to patients with high HK2 expression, hazard ratio = 2.4. The difference was significant (Figure 1B, P = .00069). The abovementioned analysis indicates that HK2 has potential clinical significance in cervical cancer and provides a database for further experiments.
To investigate the clinical significance of HK2 in cervical cancer, we detected the expression of HK2 by immunohistochemistry in 36 pairs of cervical tumors and normal cervical epithelia. As shown in Figure 1C, HK2-positive signals (the yellow staining) were mainly located in the cytoplasm. Furthermore, HK2 was highly expressed in 86.1% of cervical tumors (31/36) but was highly expressed in only 11.1% of cervical epithelia (4/36, P < .001; Table 2). This suggests that HK2 expression is upregulated in cervical cancer compared to normal cervical epithelia. Table 1 shows the correlation analysis between HK2 expression and clinical characteristics of cervical cancer. As expected, high HK2 expression correlated with larger tumors (P = .009696) and pathological grade (P = .028551). In addition, the expression of HK2 in cervical cancer was not related to age (P = .956201) or lymph node metastasis (P = .131379). Taken together, these findings indicate that HK2 is highly expressed in cervical cancer and is associated with tumor growth and prognosis.
HK2 Expression in Cervical Tumors and Normal Cervical Epithelial Tissues.

Abbreviation: HK2, hexokinase 2.
aP<0.05
Knockdown of HK2 Inhibits Proliferation and Migration of Cervical Cancer Cells
To investigate the function of HK2 in cervical cancer, SiHa cell line was selected as the study subject. The HK2 knockdown cell line (siHK2) was constructed using RNA interference technology. As shown in Figure 1D, transfection of siRNA1 effectively inhibited the mRNA expression of HK2, which was used in subsequent experiments. Protein level of HK2 was also decreased by transfecting siRNA1 (Figure 1E and F). We next detected the effect of HK2 knockdown on the proliferation of SiHa cells. As shown by CCK8 assay, the relative proliferation curve of siHK2 was significantly decreased at 72 hours (Figure 1G). Additionally, knockdown of HK2 also inhibited the migration of SiHa cells. As shown by Transwell assay, there was a significant decrease in the number of migrated cells in siHK2 (Figure 2A and B). Through wound healing assay, we found that the migration ability of siHK2 was significantly reduced compared to NC (Figure 2C and D). Taken together, knockdown of HK2 inhibited the proliferation and migration of cervical cancer cells.
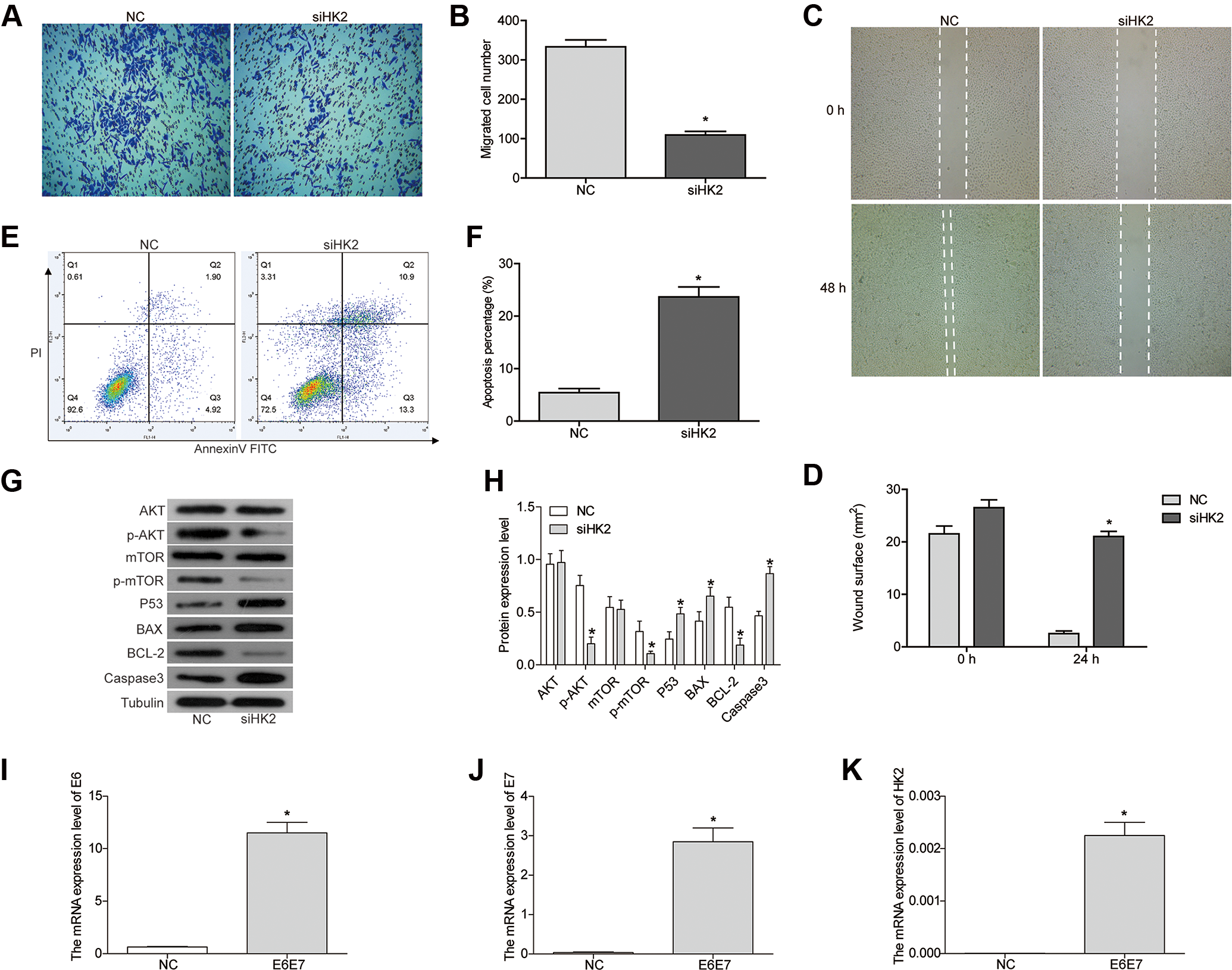
Knockdown of hexokinase 2 (HK2) inhibits the migration of SiHa cells and induces apoptosis. (A and B) Transwell assay and (C and D) wound healing assay showed that knockdown of HK2 inhibited cell migration activity. (E and F) Apoptosis was detected by flow cytometry. (G and H) The protein expression was detected by Western blot. (I-J) The messenger RNA (mRNA) expression of E6, E7, and HK2 was detected by Fluorescence quantitative polymerase chain reaction (PCR). *P < .05, compared to negative control (NC). All experiments were performed independently 3 times.
The Effect of HK2 on Apoptosis of Cervical Cancer Cells
Next, we investigated the role of HK2 in apoptosis using flow cytometry analysis. As shown in Figure 2E and F, the rate of apoptosis in siHK2 increased significantly compared to NC (P < .05). We also detected the expression of apoptotic associated proteins. As shown in Figure 2G and H, the expression of proapoptotic proteins, BAX and caspase 3, was upregulated after HK2-siRNA transfection, and the expression of antiapoptotic protein BCL-2 was downregulated. Taken together, we concluded that HK2 knockdown induced the apoptosis of SiHa cells.
The Mechanism of HK2 in Tumorigenesis of Cervical Cancer
To elucidate the molecular mechanism of HK2 knockdown in the inhibition of cervical cancer, we detected the expression of members of the PI3K pathway. The key proteins in the PIK3 signaling pathway, such as AKT and mTOR, are involved in the proliferation and metastasis of tumor cells. As shown in our results, the expression of AKT and mTOR was not changed, but the phosphorylated forms p-AKT and p-mTOR were significantly decreased (Figure 2G and H). This suggested that knockdown of HK2 decreases the phosphorylation of AKT and mTOR. p53 is a tumor suppressor gene that plays an important role in cell cycle control and apoptosis. 29 The expression of p53 was significantly increased in siHK2 cells (Figure 2G and H).
The Overexpression of E6E7 Enhances the Expression of HK2
Human papillomavirus, which is a small DNA virus, is an important risk factor for the progression of cervical cancer. 30,31 Most anogenital cancers contain HPV DNA expressing the viral oncogenes E6 and E7, the primary HPV oncoproteins. 31,32 HPV E6 proteins can inactivate the tumor suppressor p53, which leads to genetic instability and uncontrolled cell growth. 33 E7 targets the retinoblastoma tumor suppressor protein and abrogates its function through ubiquitination. 33,34 Both E6 and E7 expression can become dysregulated due to either genetic or epigenetic changes leading to E6 and E7 overexpression in a full-thickness epithelial lesion. 35 In our experiment, we overexpressed E6 and E7 in SiHa cells to observe how this affected HK2 expression. Our results showed that the expression of E6 and E7 was significantly increased after E6 and E7 plasmid transfection (Figure 2I and J). The HK2 expression was robustly increased in the E6 and E7 transfection group compared to that in NC (Figure 2K).
Hexokinase 2 Is a Target Protein of miR-9-5p
To investigate the mechanisms responsible for the functions of HK2 in cervical cancer, we searched for upstream miRNAs using publicly available databases (TargetScan and miRanda). MiR-9-5p was predicted as the potential upstream miRNAs of HK2. To confirm whether HK2 is a direct and specific target of miR-9-5p, the HK2 3′-UTR (WT) or mutant 3′-UTR (Mut) luciferase reporters were co-transfected with miR-9-5p mimic (miR-9-5p) or miR-NC (Figure 3A). Luciferase reporter assays showed that miR-9-5p significantly decreased HK2 3′-UTR reporter activity but not mutant 3′-UTR activity (Figure 3B). In agreement with the results, Western blot analysis showed that miR-9-5p mimic inhibited HK2 expression in SiHa cells (Figure 3C and D). Taken together, these results indicate that HK2 expression was inhibited by miR-9-5p by directly binding its 3′-UTR in SiHa cells. Next, we found that miR-9-5p could suppress the proliferation and migration of SiHa cells and induce cell apoptosis (Figure 3E-I).

Hexokinase 2 (HK2) is a target protein of miR-9-5p. (A) The sequences of miR-9-5p, HK2 3’-UTR wild-type (WT) and mutant 3’-UTR (Mut). (B) Luciferase reporter assays in SiHa cells transfected with WT or Mut HK2 reporters plus miR-9-5p mimic or miR-NC. (C/D) The protein levels of HK2 were detected by Western blot. (E) The proliferation was detected by CCK8 assay. (F and G) Cell migration was detected by Transwell assay. (H and I) Apoptosis was detected by flow cytometry. *P < .05, compared with MiR-9-5p negative control (miR-NC). All experiments were performed independently three times.
Discussion
Cervical cancer is one of the most common gynecological tumors. It has been the fourth leading cause of cancer-associated death among women worldwide and is becoming the second most commonly diagnosed cancer in developing countries. 36 Additionally, the 5-year survival rate is about 15% among advanced stage patients; therefore, there is a poor prognosis of late stage patients. 37,38 It is essential to study the potential molecular mechanisms of different biological expression levels in order to understand the occurrence and progression of cervical cancer. In this article, we explored the roles of HK2 in the tumorigenesis of cervical cancer with a goal of obtaining new information that could improve the prevention and treatment of cervical cancer.
First, we determined that HK2 was highly expressed in cervical cancer. Furthermore, the high HK2 expression showed a higher incidence of larger tumors sizes (P = .009696) as well as high pathological grade (P = .028551) and poor prognosis (P = .00069). It has been confirmed by various research that HK2 expression was significantly higher in a variety of malignant tumors, including glioblastoma, hepatoma, colon, breast, laryngeal, and renal cell carcinoma. 39 -43
Most malignant tumor cells prefer to metabolize glucose by glycolysis even in the presence of sufficient oxygen. Increased glycolysis provides tumor cells with 3 benefits: rapid energy production, macromolecular biosynthesis for growth and proliferation, and an antiapoptotic phenotype. 44 Hexokinase is the rate-limiting enzyme in the first step of the glycolytic pathway. Consistent with this theory, our results indicate that knockdown of HK2 significantly inhibits cell proliferation and migration and promotes apoptosis in SiHa cells.
Western blot results also showed that knockdown of HK2 resulted in a decrease in the phosphorylated form of AKT and mTOR, while there was no significant change in AKT and mTOR. Furthermore, the expression of p53 was significantly increased in HK2 knockdown SiHa cells, which means that the loss of HK2 provides the cells with more tumor suppressor activity. We also found that ectopic overexpression of E6 / E7 can directly upregulate the expression of HK2.
Most importantly, we discovered a new HK2 upstream regulatory miRNA—miR-9-5p. Hexokinase 2 expression was inhibited by miR-9-5p by directly binding its 3′-UTR in SiHa cells. Furthermore, miR-9-5p could suppress the proliferation and migration of SiHa cells and induce cell apoptosis. Li et al found that miR-9-5p could promote cell growth and metastasis in non-small cell lung cancer through the repression of TGFBR2. 45 This conclusion seems to contradict our findings. However, it is also possible that miR-9-5p has multiple target genes and plays different roles in different tumor types.
Conclusion
In conclusion, we found that HK2 exerts a cancer-promoting effect in cervical cancer through the miR-9-5p/HK2/AKT pathway and could serve as a potential therapeutic target and prognostic indicator of cervical cancer.
Footnotes
Authors’ Note
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request. Chunyan Liu and Xiuli Wang contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from National Key R&D Program of China (2016YFC1302900 and 2016YFC0902901), the National Natural Science Foundation of China (NSFC, 81572559) and the Key Research Project of Shandong Province (2017CXGC1210).
