Abstract
Background:
Brucellosis is a zoonotic bacterial disease that can affect humans and animals. It is often transmitted to humans through the consumption of contaminated animal products or by direct contact with infected animals. In Kenya, data on human and animal brucellosis are limited.
Methods:
To investigate the seroprevalence and molecular prevalence of human brucellosis in Kenya, we randomly selected from the healthy participants with possible exposure to animals and animal products and randomly selected 348 out of 2,779 human blood samples, that were obtained from a larger longitudinal cohort study that investigated of dengue, chikungunya and Rift Valley Disease exposure in western (Kisumu) and coastal (Ukunda) Kenya.
Results:
Our study included 126 males (36%), 222 females (64%) in different age categories, and 61 children aged 16 years and younger (18%), with an overall median age of 29.5 years (2- to 75-year age range). Samples were tested by Abnova Brucella IgG enzyme-linked immunosorbent assay (ELISA) Kit (KA0954). Of the tested individuals, anti-Brucella IgG antibodies were detected in 96 (28%) of 348 randomly selected participants. All samples yielded negative results in quantitative polymerase chain reaction (qPCR) analysis. There was no statistically significant correlation between Brucella exposure and study site, gender, age, socioeconomic status, ownership of particular livestock (sheep, goats, and cattle), or consumption of raw animal products. However, highly educated individuals were more likely to have Brucella exposure (odds ratio = 2.02, 1.20–3.41, p = 0.01).
Conclusion:
In comparison to previous seroprevalence-based studies conducted in nonpastoral Kenyan communities, our study revealed relatively higher seropositivity. This article emphasizes the importance of conducting surveillance for brucellosis in urban areas. Urban brucellosis surveillance within the framework of One Health could serve as a baseline to guide future research on brucellosis in humans.
Introduction
Brucellosis is a zoonotic disease caused by the Brucella species that is transmitted to humans by direct contact through infected animal excretions such as milk, abortion materials, and consumption of unpasteurized milk products and contaminated animal products (Corbel et al., 2006; Di Bonaventura et al., 2021; Franco et al., 2007; Laine et al., 2023; Memish et al., 2000; Pappas et al., 2005). Individuals who work with livestock, carcasses, aborted fetuses, or placentas that are contaminated are at risk for occupational risks, including veterinarians, slaughterhouse workers, and farmers (Corbel et al., 2006; Di Bonaventura et al., 2021). Brucella spp. can cause an acute febrile disease (undulant fever) that can progress to chronic forms and cause serious complications in the musculoskeletal, cardiovascular, and central nervous systems, followed by involvement, pain of the bones and joints, undulant fever, sweating, and nonspecific constitutional symptoms and that can mimic several other infectious diseases like typhoid, malaria, dengue (DENV), and chikungunya (CHIKV) (Franco et al., 2007; Memish et al., 2000; Pappas et al., 2005).
Common diagnostic procedures for brucellosis in both humans and animals include laboratory cultures, polymerase chain reaction (PCR), and serology tests such as the rose Bengal test (RBT), Serum Agglutination Test (SAT), and enzyme-linked immunosorbent assay (ELISA) (Corbel et al., 2006; Di Bonaventura et al., 2021; Franco et al., 2007; Yagupsky et al., 2019). Although blood cultures are the gold standard for detection of brucellosis in humans (Corbel et al., 2006; Franco et al., 2007; Pappas et al., 2005), low-to-middle-income countries mainly use serologic tests due to their cost-effectiveness, ease of use and strong negative predictive value (Laine et al., 2023; Pappas et al., 2006; Potter, 2013) In addition, PCR tests (nested, RT-PCR, conventional) can also be used to diagnosis of human and animal brucellosis (Al-Nakkas et al., 2005; Becker and Tuon, 2021; Di Bonaventura et al., 2021; Probert et al., 2004; Yagupsky et al., 2019). In order to control brucellosis in animals, vaccination strategies for healthy livestock, slaughter of suspected brucellosis cases, and sero-surveying with screening tests have all been implemented (WOAH, n.d.a; Nielsen, 2002; Nielsen et al., 1995).
The ongoing risk of brucellosis to public health is attributed to the expansion of the animal industry, urbanization, and inadequate hygienic measures in food processing and animal husbandry (WOAH, n.d.b; Laine et al., 2023; Potter, 2013). The disease is prevalent in the Mediterranean Basin, the Middle East, Latin America, Africa, and Asia (Corbel et al., 2006; Pappas et al., 2006; Potter, 2013). Brucellosis is highly prevalent in Africa, affecting people with a prevalence rate of up to 58% (Djangwani et al., 2021; Laine et al., 2023; McDermott and Arimi, 2002; Muema et al., 2022; Munyua et al., 2021), the disease continues to be inadequately diagnosed and underreported (Dean et al., 2012; Franco et al., 2007; Godfroid et al., 2005). Kenya exhibits a high incidence of human brucellosis compared to other African countries, but reports zero incidences of the disease in their animal populations each year. The relationship between human disease and animal disease is directly correlated. This reveals a notable inconsistency in reporting systems (Laine et al., 2023; Laine et al., 2022; Munyua et al., 2021). In Africa, the lack of systematic surveillance systems has resulted in little information about the prevalence of brucellosis and hints that up to 50% of cases are misdiagnosed as malaria (Laine et al., 2022; McDermott and Arimi, 2002; Njeru et al., 2016), with 4–11% of the total malaria cases later identified to be brucellosis (Laine et al., 2023; Mehari et al., 2021; Njeru et al., 2016). The majority of the data on brucellosis in humans in Sub-Saharan Africa is obtained from sero-epidemiological investigations conducted among pastoralist communities that rely on livestock for their livelihood, putting them at high risk for brucellosis infection (Kahariri et al., 2021; Mehari et al., 2021; Muema et al., 2022; Njeru et al., 2016). Domestic animals and their waste provide an abundance of resources for synanthropic species in Africa’s fast-growing cities, where small-scale animal husbandry is common. These habitats’ relatively low biosecurity implies that there is a significant possibility of contact between wildlife that uses these resources for food, livestock, and livestock products (such as feces), which can lead to the spread of infections. Occupational exposure to livestock and their products is a recognized risk factor for the spillover of zoonotic pathogens (Hassell et al., 2023; Plowright et al., 2017). However, current research primarily addresses the risks associated with sourced food value chains, leaving the potential threats that livestock present to their caretakers and the wider urban population inadequately explored (Hassell et al., 2023).
In this study, we utilized samples obtained from a larger longitudinal cohort study to investigate the seroprevalence of brucellosis among urban populations in two sites in western and coastal Kenya (Gerken et al., 2023; Grossi-Soyster et al., 2017; Khan et al., 2023).
Materials and Methods
This study is a substudy of a 2-year-long (December 2019–February 2022) longitudinal cohort study, which aimed to identify risk factors for exposure to DENV and CHIKV among two urban populations in Kenya: Kisumu in the west and Ukunda on the coast (NIH, R01AI102918; PI: A.D.L.). Ukunda is a coastal town 25.8 south of Mombasa and is located in Kwale County, Coast Province, adjacent to and to the immediate west of Diani Beach at the border with Tanzania (Ukunda in Kwale (Coastal Kenya, n.d.); Wikipedia.org, n.d.b) (Fig. 1). Kisumu is the third-largest city in Kenya, located in the Lake Victoria area. The city serves as the capital of Kisumu County and was the immediate former capital of now-defunct Nyanza Province. It is the second largest city after Kampala in the Lake Victoria Basin. The city has a population of slightly over 600,000 (The Kenya National Bureau of Statistics, n.d.; Wikipedia.org, n.d.a). For the parent study, healthy participants consented to blood sampling for baseline testing of IgG antibodies against DENV, CHIKV, Malaria, and Rift Valley fever virus (RVFV), and other arboviruses and consented to further sample use. Additionally, participants underwent a comprehensive clinical examination, completed a questionnaire, and captured demographic and household information was followed every 6 months for the duration of the study. The enrollment-structured survey for the current study covered livestock ownership, consuming raw animal products, and working in high-risk occupations (Gerken et al., 2022). The cross-sectional study design was used to analyze the risk variables and identify those which are the selection criteria in the cohort that had exposure to animals and animal products: livestock ownership, consumption of raw blood, unpasteurized milk, milk products, or raw meat, working at butcher shops, slaughtering animals, and exposure to abortion products from animals.
Map of urban study sites, Kisumu and Ukunda, Kenya. Base Map: https://africaopendata.org/dataset/kenya-counties-shapefile. (Gerken et al., 2022). Link text: https://doi.org/10.1371/journal.pgph.0000505.g001.
Declaration of ethics
To This Statement;'The institutional review boards of Stanford (31488) and the Kenya Medical Research Institute, KEMRI (SSC95 2611), approved this study. The participant received explicit consent by being given the time to review the consent form, having any questions addressed, and confirming their comprehension of the consent process through a checklist prior to signing the written consents. Parental or legal guardian consent was acquired for each participant who was under the age of 18. Verbal assents were collected from children above the age of 7 years. Only participants who had selected “second use” authorization were used in this substudy (Gerken et al., 2023; Grossi-Soyster et al., 2017; Khan et al., 2023).
Questionnaire administration
The data from the parent study were gathered by study personnel who had received extensive training and were conducted in the participants’ language of choice, which included English and Kiswahili, as well as indigenous languages such as Digo (coast) and Luo (west). The data were electronically gathered and securely stored using a REDCap database.
Sample selection
The selection of participants was based on the questions presented in the Supplementary Data(Questionnaire 1). The inclusion criteria were responding positively to any one of the following risk factors: owning any livestock/ruminants, raw animal blood use, drinking raw milk (fresh/fermented), participation in animal slaughtering, and providing care to animals. A random sampling was conducted from individuals who possess at least one risk factor, as outlined in our inclusion criteria which consisted of those who demonstrated positive responses to any of the following risk factors: ownership of livestock or ruminants, usage of raw animal blood, consumption of raw milk (either fresh or fermented), involvement in animal butchering, and providing of animal care. The samples were selected using the Open EPI Random Number generator. In the process of randomization, we systematically arranged the sample IDs in descending order to maintain a structured approach. Subsequently, random numbers were generated using a reliable random number generator accessible through the openEPI website.
The sample corresponding to each generated random number was selected for analysis and subsequently removed from the Masterfile to ensure avoidance of duplication and to maintain the integrity of the random selection process. This procedure was repeated up to three times to accommodate instances where duplicate random numbers were generated, thereby ensuring the validity and randomness of the selected samples for analysis. The sample size for frequency in a population was obtained using a 95% confidence level. We included an additional 10 samples to account for any absence or inadequacy of serum samples.
Serological testing
To investigate the seroprevalence of brucellosis in Kenya, we selected 2,779 samples from participants who had exposure to at least one brucellosis risk factor assessed for in the parent study (Questionnaire 1). From these 2,779 samples, we then selected a random sample of 348 using random numbers generated through the OpenEPI website for testing by Abnova Brucella IgG ELISA Kit (KA0954) was used to test the samples.
Briefly, the reagents and the serum samples were allowed to stand at room temperature (20–25°C) and gently mixed before use. A 1:21 dilution of test samples was prepared by adding 10 μL from the sample to 200 μL of sample diluent. About 100 μL of diluted sera, calibrator, and controls were dispensed into the appropriate wells, and the kit protocol was followed by each step. Negative control, positive control, and calibrator were used as recommended in the kit protocol. The optical densities were measured at 450 nm using an ELISA reader within a 15-min window, employing the recommended dual-wavelength reference filter of 600–650 nm. Interpretation of the results was based on the Antibody Index, where readings below 0.9 indicated the absence of detectable antibodies to Brucella IgG by ELISA. Values falling between 0.9 and 1.1 were considered borderline positive, while readings surpassing 1.1 indicated the presence of detectable antibodies to Brucella IgG by ELISA.
Molecular testing
Molecular testing was performed to detect potential acute Brucella infections. DNA extraction was performed utilizing the Mag Mind Viral DNA/RNA 96 Kit, followed by a quantitative PCR (qPCR) protocol modified from the method outlined by Probert et al. (2004). Positive controls comprised genus-specific Brucella reference strains (Brucella melitensis 16M, Brucella abortus 544, and Brucella abortus biovar 3 Tulya), obtained from the United States Department of Agriculture Animal and Plant Health Inspection Service. Primers and probes were utilized from Integrated DNA Technologies at a concentration of 100 nmol under ambient conditions in accordance with the established protocol. Amplification and real-time fluorescence detection were performed using the Bio-Rad C1000 Touch Thermal Cycler (Bio-Rad Laboratories, Hercules, CA) with the following cycling parameters: an initial denaturation and polymerase activation step at 95°C for 10 min, followed by 45 cycles of denaturation at 95°C for 15 s and annealing/extension at 57°C for 1 min. Samples demonstrating a fluorescence signal at least 30 times greater than the mean standard deviation of all wells assessed between cycles 2 and 10 were classified as positive, whereas those falling below this threshold were deemed negative.
Statistical methods
Our primary outcome of interest was the binary result (positive or negative) of the anti-Brucella IgG ELISA test, indicating the presence or absence of antibodies against brucellosis. Descriptive statistics were calculated to evaluate demographic factors, and comparisons were made between subjects who tested positive and negative for antibodies. Fisher’s exact tests were utilized to analyze the relationship between demographics and animal exposures, specifically for categorical and binary variables. The composite variable “exposure to raw animal products” includes responses from participants who confirmed consuming any of the following: unpasteurized fresh milk, unpasteurized fermented milk, raw meat, or raw blood, as well as coming into contact with animal abortion material, touching animal blood and tissues without personal protective equipment, working in butcher shops, being a veterinarian, and other occupational exposures. Livestock ownership, site, socioeconomic status (SES), education, and age were also evaluated. A univariate logistic regression analysis was performed to identify the risk factors associated (p < 0.05) with brucellosis exposure in our study population. All data were analyzed using SAS (R) Proprietary Software 9.4 (Copyright 2016 by SAS Institute Inc., Cary, NC, USA).
Results
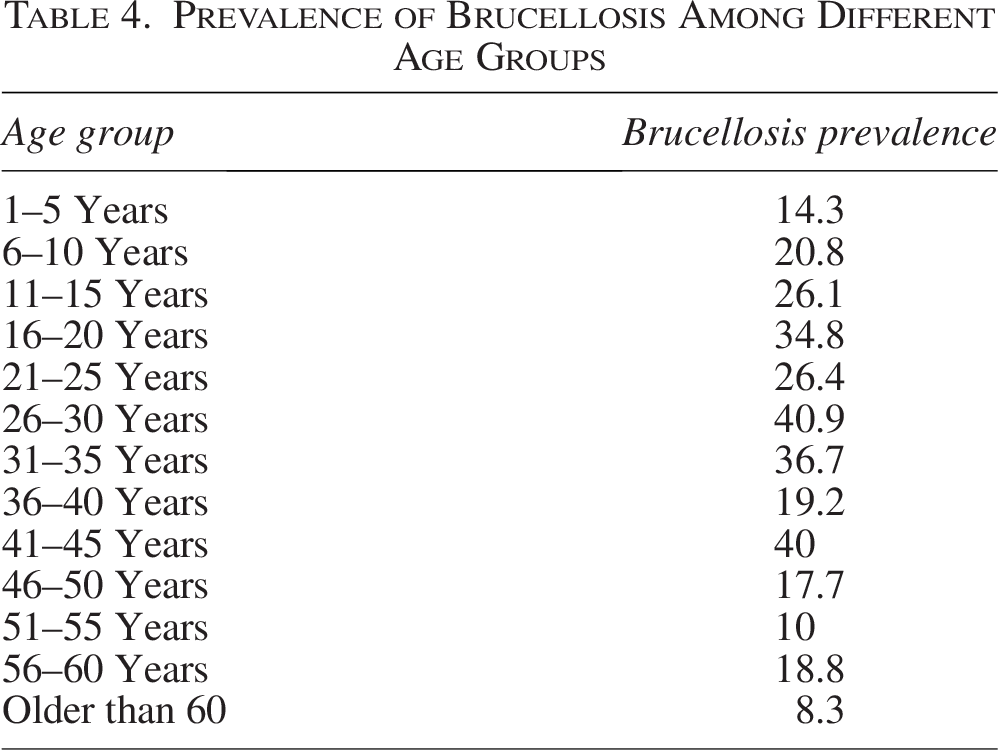
Our study included 126 males (36%), 222 females (64%), and 61 (18%) children aged 16 and younger, with a median age of 29.5 years between the 2 and 75 age range. Characteristics of the study population were included in detail in Table 1, and brucellosis seroprevalence by month 2019–2022 were included in (Fig. 2, Table 2). The findings demonstrate that Brucella spp. exposure exhibited no significant correlation with study site, gender, age, SES, livestock ownership (cattle, goats, and sheep), employment in butcher shops, or consumption of animal products, as well as seropositivity for IgG antibodies against RVFV, DENV, CHIKV, malaria, and brucellosis (Table 3). Of the tested individuals, anti-Brucella IgG antibodies were detected in 96 (28%) individuals. The children between the ages of 0 and 16 showed 20.04% positivity, since the adults between the ages of 16 and 50 showed 30.81 positivity (Fig. 3, Table 4). Among those with brucellosis exposure, 19%, 27%, and 34% also tested positive for DENV, CHIKV, and malaria exposure, respectively, in the parent study testing. The univariate analysis revealed secondary level or higher-educated individuals to be more likely to be Brucella-exposed than lower-educated individuals (OR = 2.02, 1.20–3.41, p = 0.01) (Table 3). Participants were selected based on prior exposure to animals, thereby restricting the evaluation of additional risk factors. All samples in our study yielded negative results for quantitative PCR (qPCR). No significant relationships were found in univariate analysis between brucellosis seropositivity and study site, gender, age, socioeconomic position, livestock ownership (cattle, goats, and sheep), work in butcher shops, or consumption of animal products (Table 3).
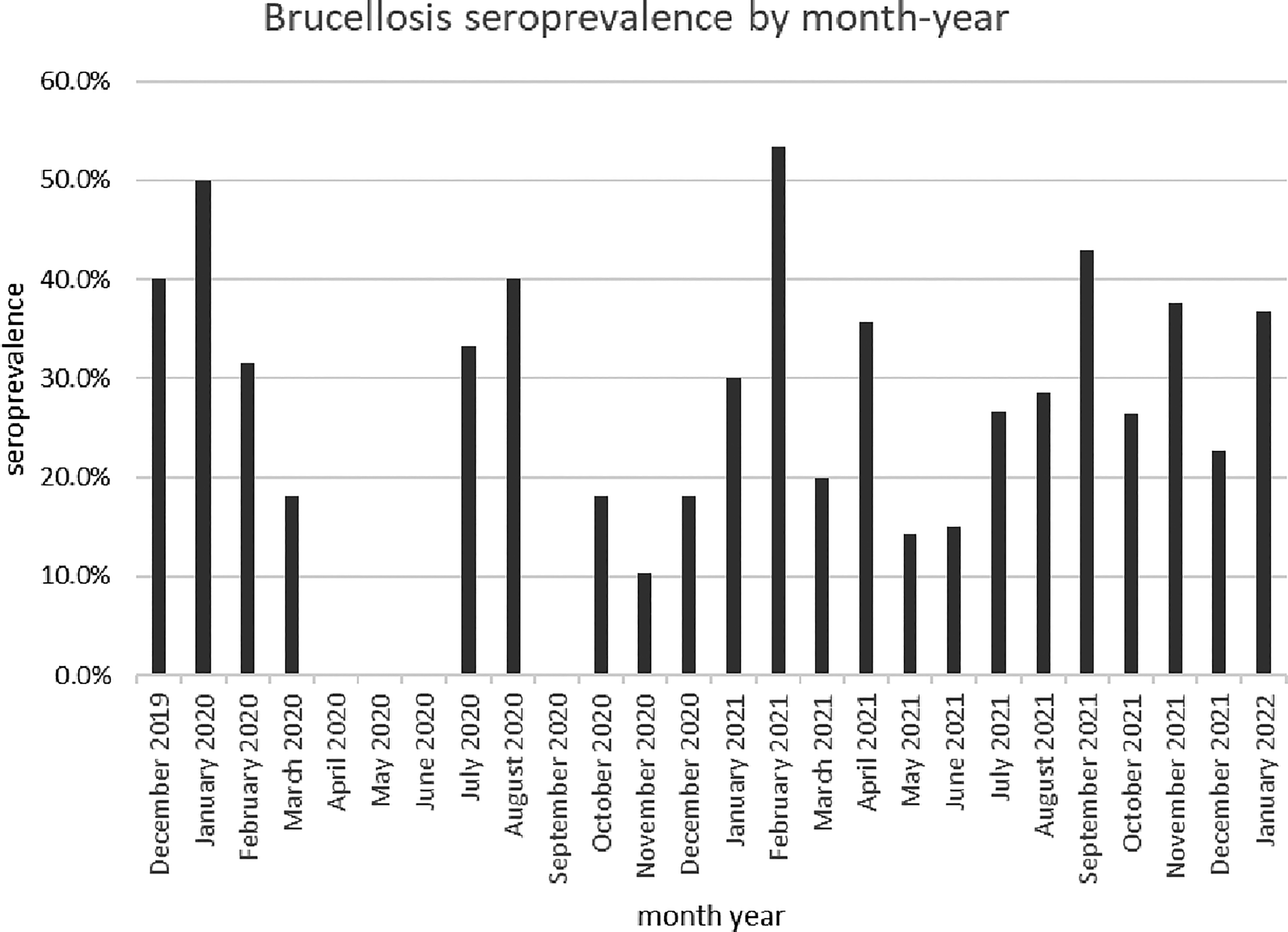
Brucellosis seroprevalence by month 2019-2022.
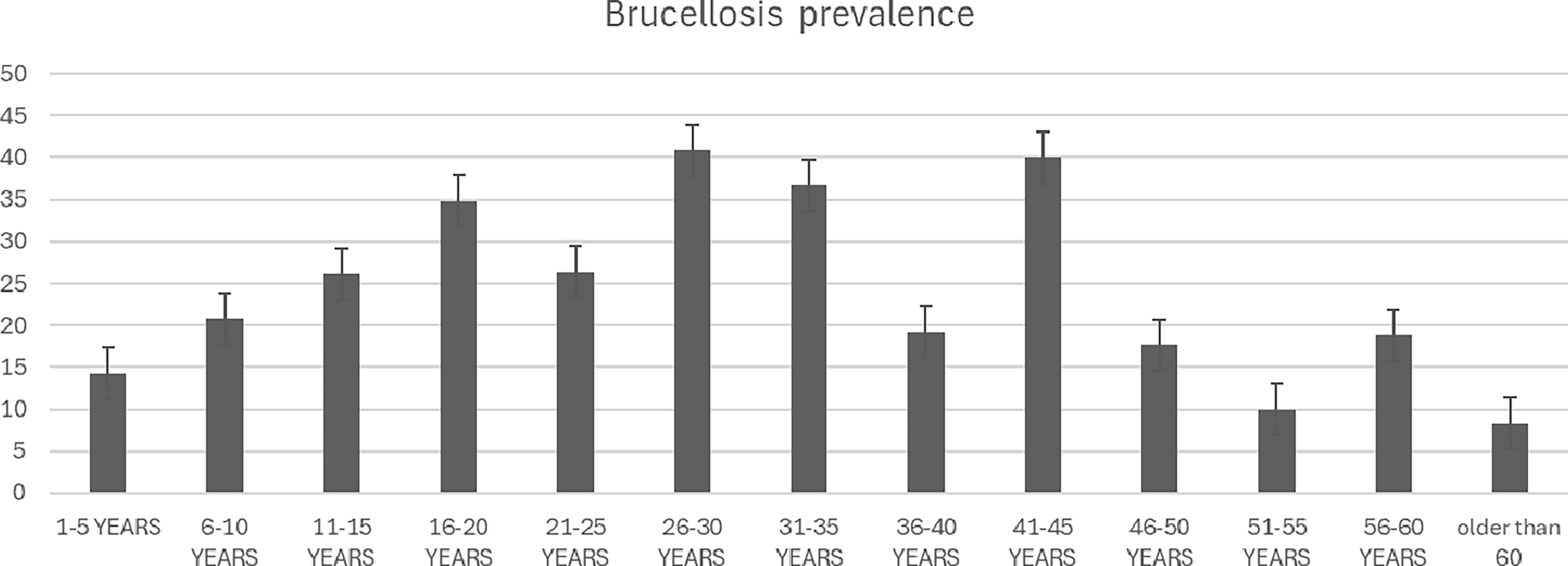
Brucellosis prevalence across age.
Characteristics of the Study Population
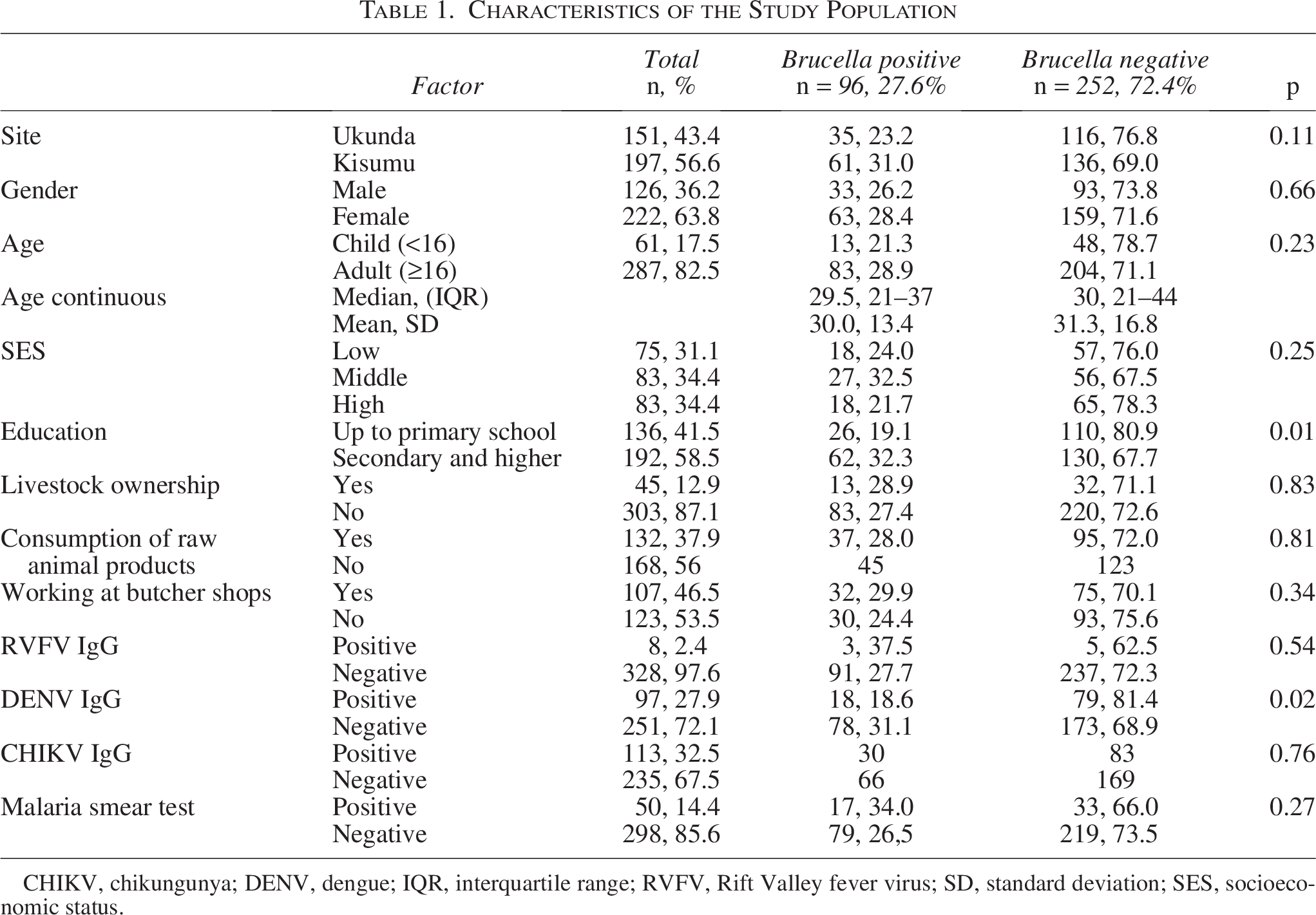
CHIKV, chikungunya; DENV, dengue; IQR, interquartile range; RVFV, Rift Valley fever virus; SD, standard deviation; SES, socioeconomic status.
The Prevalence of Brucellosis Seropositivity Between December 2019 and January 2022

Univariate Analysis of Risk Factors for Brucellosis Exposure
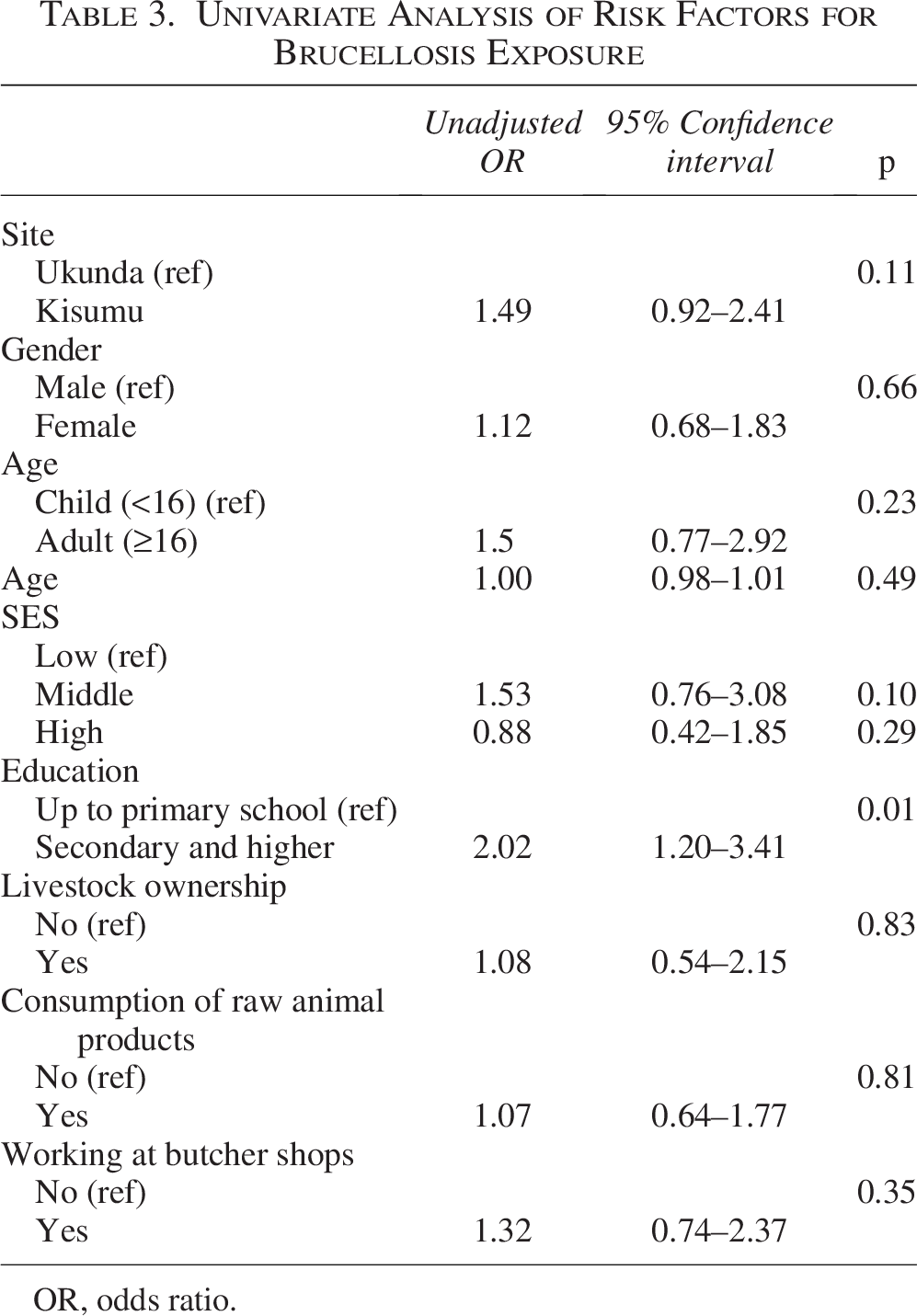
OR, odds ratio.
Prevalence of Brucellosis Among Different Age Groups
Discussion
Twenty-eight percent of our urban participants had Brucella exposure, as evaluated by the presence of Brucella IgG positivity. The frequency recorded in pastoral communities in Africa is higher than the prevalence observed in nonpastoral human communities in Africa, which is higher than what has been observed previously in diverse human communities across Africa(Akoko et al., 2023; Djangwani et al., 2021; Muema et al., 2022; Munyua et al., 2021; Mwatondo et al., 2023). Seroprevalence was slightly higher in western Kisumu compared to coastal Ukunda, which may be due to the increased animal population in the western area. According to previous studies, the prevalence of brucellosis in Kenya ranges from 0% to 47% (Djangwani et al., 2021; Kairu-Wanyoike et al., 2019; Muema et al., 2022; Osoro et al., 2015) in humans and from 1% to 38% (Muema et al., 2022; Nthiwa et al., 2019; Osoro et al., 2015) in animals; the highest estimates were found in areas home to pastoralists who live around their livestock. In a research done by Osoro et al. (2015), it was found that the county of Marsabit, which is a pastoralist area, had a higher prevalence rate of 46.5% for brucellosis among people compared to the other two counties included in the study. Our study region is located on the coast and in the western part of Kenya. Insufficient research has been conducted to categorize and understand Kwale County as a possible region with the potential for epizootic Brucella spp. transmission from animals to humans (Akoko et al., 2023, 2021; Munyua et al., 2021; Mwatondo et al., 2023; Osoro et al., 2015).
In our study, higher-educated individuals were more likely to contract brucellosis than lower-educated individuals (odds ratio [OR] = 2.02, 1.20–3.41, p = 0.01); however, Osoro et al. (2015) suggested that attaining at least a high school education and above was a protective factor for human seropositivity (adjusted OR [aOR] = 0.3, 95% CI = 0.3–0.4). Typically, higher education is associated with greater health awareness and lower risk of zoonotic infections due to improved hygiene practices and occupational choices (Dean et al., 2012; McDermott and Arimi, 2002). However, this relationship may be context-specific. One plausible explanation is confounding by age or rural–urban origin. In many low-resource settings, older individuals, who are more likely to have prolonged cumulative exposure to Brucella spp. through occupational or household contact with livestock, often have lower levels of formal education (Lindahl et al., 2015). Conversely, younger participants with higher education levels may have recently entered environments or professions that place them at elevated risk, such as veterinary medicine, laboratory work, or public health field operations (Mangen et al., 2002) These roles have been associated with increased Brucella exposure due to direct contact with infected animal materials or clinical specimens (Corbel et al., 2006). Furthermore, sampling bias may have influenced this association if participants with higher education were overrepresented in a study site with higher transmission levels. We also hypothesize that one of the reasons for these increased odds is that individuals with higher education are more likely to are more to own animals and thus more likely to have direct interaction. Individuals in higher education and medium socioeconomic status are more connected to social life and are likely to be more influenced by external factors such as work and social environments compared to those in low or high SES and low education. Similar findings have been reported in other studies where education level did not confer a protective effect, possibly due to occupational exposures among educated individuals (Kunda et al., 2007). Given that no significant associations were observed with other conventional risk factors—such as gender, SES, livestock ownership, butcher shop employment, or consumption of animal products—it is essential to further investigate whether the observed association with education reflects a true epidemiological trend or is a result of residual confounding or selection bias. Further research is recommended to better understand the nature of this relationship.
Due to the large volume of human-livestock contacts, previous studies have shown that pastoralist communities have a greater human brucellosis prevalence than metropolitan areas (Chota et al., 2016; Djangwani et al., 2021; Muema et al., 2022; Osoro et al., 2015). Human samples were gathered from two metropolitan areas, Ksimu and Ukunda, for this research. The selection bias arises from the criteria that we utilized to determine our inclusion. Based on our selection criteria, certain individuals may have been omitted from the research. The fact that this study was not conducted in an agricultural, semiarid, or pastoralist environment, as well as the method of sampling, might influence our findings.
We identified a significantly higher brucellosis seropositivity among participants aged 26–30 compared to other age groups in our research. Seroprevalence displayed a concave profile with the maximum acceleration in the 26–30 age range and a steady decline in acceleration until the 46–50 age range as the age ranges increased for individuals aged 16 and older. This can be explained by the fact that young individuals begin to physically participate in family affairs (managing cattle, preparing food, etc.) at a higher rate starting at age 16, and that engagement starts to decline around age 46–50.
None of the samples in our study tested positive for qPCR. This contrasts with the ELISA results, indicating that acute brucellosis has not been detected in the healthy individuals without fever in our collection.
The present study aimed to determine that owning livestock, consuming raw animal products, and working in a butcher shop were associated with a higher risk of animal interaction. The association between these parameters and Brucella spp. seropositivity (p value) was determined to be insignificant. We hypothesize that the rationale behind these odds is the potential presence of bias in the outcomes. The study did not address the awareness or understanding of participants who were likely to have intimate contact with animals regarding brucellosis. A large-scale arbovirus research cohort study provided the samples for our study; issues pertaining to zoonosis were excluded. Consequently, the potential risks of contact between humans and animals have not been addressed.
Fever is a frequent symptom of human brucellosis, and we found overlap with malaria, DENV, and CHIKV exposure. This poses an important challenge to proper laboratory diagnosis in regions where appropriate diagnosis and therapy are not easily accessible. Dutta et al. (2016) found in a study that among 37 cases of DENV confirmed through serological testing, eight cases (21.6%) exhibited coinfection with both brucellosis and dengue fever. This emphasizes the need to investigate febrile diseases and their results in patients with febrile illness to provide future opportunities for minimizing case deaths. In Africa, CHIKV, DENV, and malaria are the most common febrile diseases (Hotez et al., 2019; Khan et al., 2023; Mordecai et al., 2020), and Brucella cases could remain ignored in the region compared to these dominating infections.
The samples were analyzed in a biosecurity level 2 laboratory, as recommended for Brucella (Corbel et al., 2006; Di Bonaventura et al., 2021; WOAH, n.d.b; Yagupsky et al., 2019) while following the set biosafety and biosecurity measures for the biosafety level 2 laboratory. The brucellosis cohort study was conducted in Kenya, Africa, where animals are vaccinated against Brucella using live attenuated S19 and Rev 1 strains. However, the only animal vaccination used in the United States to combat brucellosis in cattle herds is the Brucella abortus RB51 vaccine (WOAH, n.d.b; Zhang et al., 2019). Testing for potential brucellosis disease in humans is severely restricted in the United States because of the administration of RB51 vaccinations to animals. Ordering tests for serology for research purposes requires approval from the Centers for Disease Control and Prevention (CDC) and the United States Department of Agriculture (USDA). Due to the circumstance that our laboratory, where we conduct the experiments, is classified as a biosafety level 2 (BSL 2) facility, we were unable to acquire the import documentation required by the CDC and the USDA. Laboratories working specifically with RB51 isolates should perform all manipulations in a class II biological safety cabinet, utilizing biosafety level 3 (BSL-3) practices as described in Biosafety in Microbiological and Biomedical Laboratories, 6th edition (U.S. Department of Health and Human Services, 2020).
Most African countries and many of the significant Asian countries have little awareness and knowledge about brucellosis. This is predictable due to inefficiencies in Public Health and Veterinary Services, as well as notification mechanisms. In Kenya, the practice of vaccinating animals against brucellosis is limited and frequently occurs randomly rather than as part of a well-organized nationwide initiative (WOAH, n.d.a). There were limited publications (Akoko et al., 2023) available at the time of the study that described national programs for the control of brucellosis. The overall reported studies were limited to research on the prevalence of antibodies in a population. Like many other African nations, control programs have significant challenges in terms of insufficient public and veterinary health services. The lack of public and veterinary health services, due to limited governmental funding and insufficient private sector engagement, constitutes a significant barrier to control programs in this and other African countries. To establish an effective management strategy in Kenya, it is essential to account for the unrestricted movement of cattle across borders between neighboring countries via trade routes, as well as the migratory actions of individuals seeking pastures and water sources (Njeru et al., 2016). These activities have the potential to facilitate the introduction and transmission of sick herds, posing a challenge to internal control measures and demanding regional coordination (Njeru et al., 2016; Van Seventer and Hochberg, 2017).
We had some limitations in the research. The research includes data from December 2019 to February 2022. Data were unavailable during the period of April to June 2020 owing to the entire shutdown caused by the COVID-19 pandemic. Animal serum samples were not analyzed in the present study. The study utilized data from an NIH-funded parent study to identify DENV and CHIKV, and therefore, the survey questions were not a priori selected for a Brucella study, which may introduce bias and insufficient information. This study exhibits selection bias since it exclusively examined samples that have at least one risk factor for Brucella. Due to restrictions enforced by the USDA, the serological tests mentioned, such as RBPT (Rose Bengal Plate Test), SAT (Serum Agglutination Test), Coombs test, complement fixation test, and microagglutination tests, could not be imported into the United States without proper authorization. Consequently, we were unable to verify and validate the results using these alternative serological tests. The sensitivity and specificity of the test were assessed in accordance with the manufacturer’s declarations regarding the commercial kit. False-positive results underwent retesting to confirm the results and to eliminate human errors. The ELISA test utilized in this study detected IgG antibodies were detected but not IgM. We were unable to get sufficient data on the livestock owners’ animals being exposed to potential infections with other diseases, and other health record information from those who participated in our study. Due to these limitations, it is impossible to determine the herds’ history of brucellosis or whether livestock owners who may have come into contact with the herds are affected. Random selection of participants makes it impossible to monitor home dynamics and whether cases were clustered within individual households.
In Africa, as well as in other parts of the world, brucellosis continues to be a neglected zoonotic infection. To distinguish this very significant zoonosis from other febrile diseases, the only way to do so is to determine the high prevalence of the disease in the region and to raise awareness among the overall population. It is necessary to administer immunizations to ruminants on a constant basis in Kenya and in other regions across the world where brucellosis is prevalent. It is also essential to conduct serological tests on the animals at periodic intervals of 6 months to monitor the state of brucellosis antibodies. As a result of the increased likelihood of brucellosis transmission to individuals in pastoral settings that involve a substantial amount of interaction between humans and animals, it is of the utmost importance to put into practice effective preventative measures. Furthermore, it is of the highest priority to spread information to the general population regarding the importance of avoiding the consumption of unpasteurized milk and dairy products like unpasteurized milk, fresh cheese, as well as other raw animal products, such as fresh blood and raw meat. The public should be made aware of the fact that there is a risk of transmitting brucellosis disease by consuming animal products that have not been pasteurized or cooked, and municipalities and other health organizations should work to raise public awareness. Additionally, brucellosis seroprevalence should be conducted on humans in areas where the disease is endemic. Collaboration between the human and animal health sectors can improve public health by recognizing brucellosis cases early, before the transmission of the disease. The interdisciplinary One Health approach emphasizes the value of and requirement for global, multidisciplinary, and cross-sectoral communication and collaboration at local, national, and international levels since it acknowledges the interconnectivity of global health concerns (Majiwa et al., 2023; Travis et al., 2014). Human cases can serve as a valuable indicator of disease occurrence in animal populations and may be the only reliable source of information for surveillance (Mj & Sm, 2016; Munyua et al., 2021; Osoro et al., 2015). Determining the disease’s origin and whether items were made locally or imported are crucial (Corbel et al., 2006). Public health awareness and continuous screening for brucellosis in humans and animals are required.
Conclusion
This study demonstrates the neglected significance of brucellosis exposure among urban human populations in Kenya. Our analysis demonstrated considerably greater seropositivity compared to other seroprevalence-based investigations in Kenyan villages among nonpastoralists. To determine the mode and rate of human exposure to brucellosis and the underlying causes of the disease, further investigation is required.
Impacts
Brucellosis is a zoonotic bacterial disease that can affect humans and animals. This study demonstrates the neglected significance of brucellosis exposure among urban human populations in Kenya.
In Kenya, data on human and animal brucellosis are limited. Our analysis demonstrated considerably greater seropositivity compared to other seroprevalence-based investigations in Kenyan villages among nonpastoralists.
Authors’ Contributions
Conceptualization: E.B., B.A.B., A.P., C.I., F.M.M., B.A.N., and A.D.L. Data curation: E.B., C.I., and B.A.B. Formal analysis: E.B., B.A.B., and A.P. Funding acquisition: A.D.L. Investigation: E.B., B.A.B., A.P., C.I., F.M.M., B.A.N., and A.D.L. Methodology: E.B., B.A.B., C.I., and A.D.L. Project administration: E.B., B.A.B., C.I., and A.D.L. Resources: A.D.L. Supervision: A.D.L. Validation: E.B., C.I., and B.A.B. Visualization: E.B. Writing—original draft: E.B. Writing—review and editing: E.B., B.A.B., A.P., C.I., F.M.M., B.A.N., and A.D.L.
Footnotes
Author Disclosure Statement
The authors declare no conflicts of interest.
Funding Information
The institutional review boards of Stanford (31488) and the Kenya Medical Research Institute, KEMRI (SSC95 2611), approved this study.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
