Abstract
Background:
In the Netherlands, Usutu virus (USUV) is endemic in birds, and recently West Nile virus (WNV) was also detected in birds, mosquitoes and humans. Here we investigated the possible role of rodents in the viruses’ transmission ecology.
Materials and Methods:
We sampled rodents at six locations including sites where WNV had been previously detected. Brains (n = 668), oral swabs (n = 282), and ticks (n = 91) collected from rodents were tested for arboviruses via Reverse Transcriptase Polymerase Chain Reaction (RT-PCR). Also, sera from 118 rodents were tested for WNV- and USUV-antibodies.
Results and Conclusion:
Brain samples, swabs, and ticks tested negative for viral RNA. However, 2.5% (3/118; two wood mice, one field vole) of rodents had WNV-neutralizing antibodies (WNV-NAbs). USUV-NAbs were detected in a wood mouse. Two bank voles had NAbs against both viruses. The WNV and USUV antibody-positive rodents were found at locations with previous WNV and USUV circulations, suggesting that rodents may be involved in the ecology of WNV and USUV.
Introduction
Over the past decade, two vector-borne viruses of medical and veterinary importance were detected in the Netherlands. West Nile virus (WNV) was initially identified in 2020 in Haarzuilens, Utrecht Province (Sikkema et al., 2020), and again in 2022 in Hippolytushoef, North Holland (RIVM, 2022). Usutu virus (USUV) has been circulating in the Netherlands since 2016 (Rijks et al., 2016). While birds are recognized as the primary reservoir and amplifying hosts for both WNV and USUV, previous studies have detected RNA of WNV (Root and Bosco-Lauth, 2019) and USUV (Diagne et al., 2019) in rodent species. The ability of both viruses to grow in rodent cell lines (Benzarti and Garigliany, 2020) has further raised questions regarding the susceptibility and involvement of rodents in the transmission cycles of USUV and WNV. Here, we investigated WNV and USUV in rodents (rats, mice, and voles) at various locations across the Netherlands, including sites where WNV and USUV had previously been detected in wild birds.
Materials and Methods
Rodent sampling
Rodents were live-trapped (Heslinga Traps) and sampled from three rural locations in the Netherlands: Haarzuilens (Utrecht), Hippolytushoef (Noord-Holland), and Kraaijenbergse Plassen (Noord-Brabant), including locations where WNV (Utrecht and Noord-Holland) and/or USUV (all locations) had previously been detected. Utrecht sampling was conducted three times (April, June, and December 2021) and once each at Noord-Holland and Noord-Brabant: March and October 2023 (Fig. 1). Trapped rodents were identified to species level and weighed. For all animals weighing >20 g, blood (serum) was collected under anesthesia (isoflurane) from the retro-orbital plexus. Oral swabs and ticks were also collected. Rodents were sampled in the field and released at the individual trapping locations immediately after sampling.
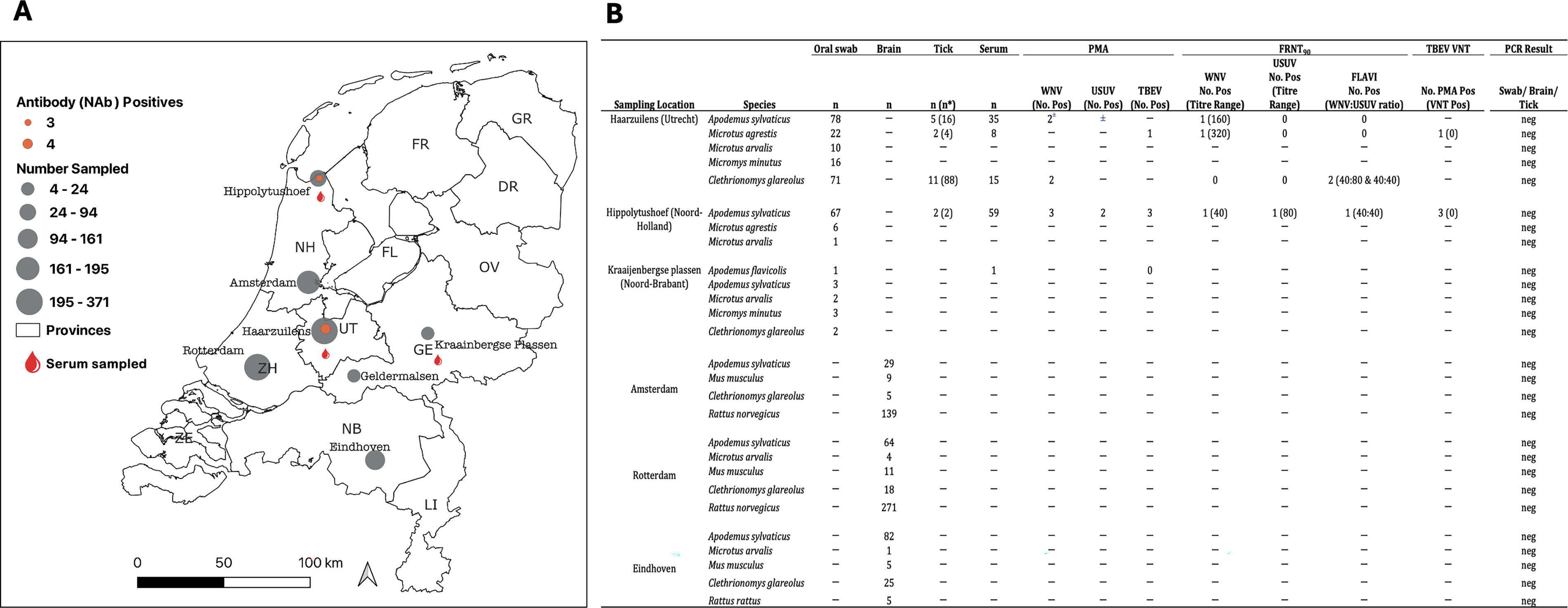
Sampling locations in the Netherlands and rodent species tested for binding and neutralizing antibodies (IgG) to WNV, USUV, and TBEV.
Brains were collected from rodents sampled at urban locations in Amsterdam (Noord-Holland), Rotterdam (Zuid-Holland), and Eindhoven (Noord-Brabant) as part of a study on the impacts of urban greening on the epidemiology and ecology of rat-borne zoonotic diseases (de Cock et al., 2024). In short, rodents were collected using snap traps between June and October 2020 and March and December 2021. Mice were collected incidentally (bycatch) while targeting mainly rats; hence, only a few mice were sampled. Following carcass storage (−20°C) and necropsy, brain samples were stored in −80°C until testing.
Sample processing and testing
Polymerase chain reaction
Total nucleic acids were extracted from oral swabs and brain tissues using the Roche MagNA Pure system (Atama et al., 2022). Ticks collected from each rodent were morphologically identified under the microscope and identified as nymphs or adults, then pooled per rodent (a maximum of five ticks per pool). RNA extraction was performed as previously described (Bakker et al., 2024). All samples were tested using real-time RT-PCR for USUV and WNV RNA (Atama et al., 2022). Ticks were identified molecularly as previously described (Bakker et al., 2024).
Serology
Sera were screened on an in-house multiplex protein microarray (PMA) for binding of IgG antibodies to USUV (The Native Antigen company), WNV (Sino Biological), and TBEV (Immune Technology) NS1 antigens (Atama et al., 2022)—validated using USUV postinfection mice sera. Samples with positive signals (median fluorescence ≥2500 at 1:80 dilution) on the PMA were further tested for neutralizing antibodies (NAbs) via a micro-Focus Reduction Neutralization Assay (FRNT90) (de Bellegarde de Saint Lary et al., 2023). Sera with WNV neutralizing antibody (NAb) titers (cutoff ≥40) fourfold higher than USUV were considered WNV positive on FRNT90 and USUV positive if the reverse. Positive signals without a fourfold difference were considered Orthoflavivirus-positive (FLAVI). Samples with positive signals for TBEV-NS1 were further confirmed on an in-house TBEV virus microneutralization test (VNT) with a predetermined cutoff (titer ≥32) (Reusken et al., 2019).
Results
Sera were collected from 118 rodents of four species, and oral swabs from 282 individuals of five species (Fig. 1). In addition, 110 Ixodes ricinus ticks were collected from 20 rodents of three species, with individual rodents carrying a median of 1.5 ticks (interquartile range [IQR]: 1.0–6.8).
Thirteen of 118 sera showed antibodies to either WNV-NS1 (n = 5), USUV-NS1 (n = 2), both WNV and USUV NS1 (n = 2), or TBEV-NS1 antigens (n = 4) in the PMA (Fig. 1). Three rodents (two wood mice: Apodemus sylvaticus and a field vole: Microtus agrestis) were confirmed positive for WNV-neutralizing antibodies (WNV-NAb prevalence 2.5%) and one (wood mouse) was confirmed positive for USUV (USUV-NAb prevalence 0.8%). Two of the WNV-positive rodents (field vole and wood mouse) were sampled from Utrecht in June and December 2021. The third WNV-positive (wood mouse) and the USUV-positive rodent (a wood mouse) were both sampled from Noord-Holland in March 2023. Three other PMA-positive rodents exhibited NAbs against both WNV and USUV with titer differing ≤twofold and were considered Orthoflavivirus-positive (Fig. 1). None of the samples exhibited NAbs to TBEV. All brain samples, swabs, and ticks tested negative for WNV, USUV, and TBEV in RT-PCR.
Discussion
Samples positive for WNV-NAb were obtained from two wood mice (A. sylvaticus) and one field vole (M. agrestis)—species in which natural infection and susceptibility have not been reported before. These novel detections, coupled with previous detections of viral RNA and antibodies to WNV in other closely related species such as the yellow-necked wood mouse (Apodemus flavicollis) and bank vole (Clethrionomys glareolus) (Root and Bosco-Lauth, 2019), further emphasize the exposure and possible involvement of rodents in WNV circulation dynamics.
Most sera, including the WNV- and USUV-antibody positives, were collected from locations where WNV and/or USUV were previously detected. Across other locations with no history of WNV detection, serum was collected from only one rodent (Kraaijenbergse Plassen, Noord-Brabant), which was negative. Due to the limited sample size, exposure levels at these locations could not be inferred. Also, only a few rodents expressed binding antibodies to TBEV-NS1 antigen on the PMA, and none showed TBEV-specific NAbs, despite the known circulation of TBEV in the Netherlands. This disparity between the assays may be a result of cross-reactivity on the PMA or due to low-level exposure in the rodents that may be insufficient to generate robust NAb responses (Shevtsova et al., 2016) or due to antibody decay to undetectable levels for the neutralization assay.
The finding of WNV and USUV NAbs in rodents at two locations, where WNV and USUV were previously detected, proves exposure and indicates possible involvement of rodents in the ecology of WNV and USUV.
Authors’ Contributions
N.C.A.: Conceptualization (equal), methodology (equal), investigation, field data collection, writing—original draft (lead), and laboratory investigations (equal). B.B.M., M.G.v.H., E.L.P., M.P.d.C., M.M., H.J.E., and C.J.M.K.: Investigation and field data collection. F.C. and C.H.: Laboratory investigations (equal). N.C.A., B.B.M., M.G.v.H., F.C., E.L.P., M.P.d.C., M.M., C.H., H.J.E., C.J.M.K., M.K., M.S., and R.S.S.: Writing—review and editing Writing—review and editing (equal). M.K.: Conceptualization (equal) and methodology (equal). M.S. and R.S.S.: Conceptualization (equal), methodology (equal), investigation, and field data collection. All authors read and approved the final article.
Footnotes
Acknowledgments
The authors thank the Dutch Mammal Society (NJN), Tijmen Hartung, and Wilma Blauw for their invaluable expertise, support, and coordination with fieldwork. They also thank Suchada Khemthong and Anne van der Linden for their support in the lab and Jeroen Nagtegaal and Leon Kelder for their assistance in planning and their knowledge of ecology and field site setup.
Author Disclosure Statement
The authors declare no competing interests.
Funding Information
This work is part of a One Health PACT research consortium, which is (partly) funded by the Dutch Research Council (NWO) with project number 109986. R.S.S. and M.K. received funding from the European Union’s Horizon 2020 research and innovation programme under the grant agreement No. 874735 (VEO). Fieldwork was partly funded by Kappa Flu budget (project number: 1414961).
Ethics Approval and Consent to Participate
Protocols for handling and sampling of rodents were approved by both the Netherlands Ministry of Economic Affairs (approval no. FF/75A/2015/014) and the Animal Experiments Committee of Wageningen University under the permit 2017.W-0049.009.
Consent for Publication
All authors consent to the publication of the article.
Data Availability
All data generated during the study will be made available on the EMBL-Pathogen Portal.
