Abstract
Nano-arrays deposition has emerged as a powerful strategy for enhancing the surface and mechanical properties of plant fibers. However, there is a lack of in-depth understanding of the underlying relationship between nano-array formation and deposition conditions. In this study, the complex interactions between the surface characteristics of plant fibers and the sol-gel process parameters are elaborated, providing insights into the mechanisms driving in-situ nucleation and growth dynamics of nano-arrays. For the fiber surface, the introduced carboxymethyl groups and the concave morphology facilitate the heterogeneous deposition of nano-arrays by strengthening the interaction between the fibers and the deposited structures, creating ideal deposition sites for the layer-by-layer self-assembly of nano-arrays. In the sol-gel process, the precursor concentration acts as the driving force for heterogeneous nucleation and growth of nano-arrays. The catalyst concentration influences the morphologies of nano-structures, dictating whether thin nano-gels, well-aligned nano-arrays, or aggregated nano-particles. The reaction time determines the nucleation and growth cycles of nano-arrays. In this case, the well-aligned SiO2 nano-arrays are deposited on the carboxymethyl pre-treated jute fibers in the ethanol/water solution containing 0.60 mol/L ammonia (catalyst) and 0.08 mol/L TEOS (precursor) for 6 h. Through the grey relational analysis, the influence of these three factors on particle size follows this order: reaction time < precursor concentration < catalyst concentration. The relationship between the surface features of plant fibers and sol-gel parameters enables the formation of well-aligned nano-arrays that effectively repair surface defects, ensuring precise control over the efficient fabrication of plant fibers with significantly enhanced properties.
Introduction
Nature-friendly and cost-effective bio-composites derived from sustainable resources have recently gained attention as promising alternatives in engineering applications, particularly in the architectural and transportation fields.1–3 Plant fibers, including jute and hemp fibers, are emerging as promising reinforcements for advanced functional and structural bio-composites due to their exceptional mechanical properties.4,5 However, these bio-composites are susceptible to the interface debonding, resulting in the mechanical properties that are significantly lower than their ideal values. 6 It has been established that the hydrophilic nature and surface defects (some narrow parallel grooves in the longitudinal direction of the fibers and some flaws on the fiber surface) of plant fibers are the primary causes of the interface debonding of these bio-composites.7,8 Therefore, enhancing the interface bonding of bio-composites relies on two key strategies: (1) Optimizing the chemical structure of the fiber surface and (2) Repairing the surface defects of the fibers.
Various strategies, including physical and chemical modifications such as heat treatment and alkaline treatment, have been employed to optimize the surface structure of plant fibers.9–11 In contrast, nano-deposition offers an effective and feasible method for enhancing the performances of the fibers, as shown in Supplemental Table S1 and S2. This approach can modify the fiber surface at the molecular scale to achieve the desired chemical structure while simultaneously repairing surface defects at the nanoscale.12–14 Yang et al. 15 demonstrated that jute fiber/polypropylene composites exhibited outstanding mechanical properties due to the deposition of zinc oxide (ZnO) nano-rods. The ZnO nano-rods on the jute fibers acted as the anchor structures, increasing the contact area and the physical interlocking with polypropylene. This enhanced the interface interaction, thereby improving the mechanical properties of the composites. Prasad et al. 16 evaluated the mechanical and water absorption properties of TiO2 nano-coated flax fiber reinforced composites. Their findings showed that the TiO2 nano-particles acted as a bridge between the fibers and the matrix, improving the tensile, flexural, and interlaminar shear strength by 22%, 24%, and 16%, respectively. Inspired by the hierarchical structures and specialized functionalities found in nature, Ekbatani et al. 17 introduced a simple and versatile spray coating technique to deposit cellulose nano-crystals (CNCs) on flax fibers, eliminating the need for any chemical treatments. The mechanical properties of the hierarchical nano-engineered plant fiber reinforced composites were significantly improved due to the presence of CNCs on the flax fibers.
In previous studies, researches reported a nano-array interphase to achieve excellent interface bonding in plant fiber reinforced composites under both dry and wet conditions.18–21 Compared to thin nano-gel and dispersed nano-particles, nano-arrays with a well-aligned structure are more likely to achieve the desired results. However, the formation of nano-arrays requires strictly controlled conditions, including the surface features of plant fibers and the process parameters. The relationship between nano-array formation and in-situ deposition conditions remains unclear. Moreover, there is a notable gap in the literature regarding the nucleation mechanism and growth behavior of nano-arrays on plant fibers. In this study, the underlying mechanism behind the in-situ deposition of nano-arrays on plant fibers is explored. Jute fibers and SiO2 nano-arrays were selected as the models for plant fibers and nano-arrays, respectively. By integrating the surface characteristics of plant fibers with the sol-gel process parameters, a systematic investigation into the deposition effects of nano-arrays under different conditions was conducted. This included examining the morphologies, deposition amounts, and particle sizes, enabling us to gain a deeper understanding of the underlying mechanisms driving in-situ nucleation and growth behavior of nano-arrays on plant fibers.
Experimental
Materials and reagents
Jute fibers (sourced from Hunan, China) were chosen as the representative of plant fibers. Tetraethoxysilane (TEOS), 4-dimethylaminopyridine, ClCH2COONa, NH3·H2O, H2SO4 (>98%), NaOH (>96%) and ethanol were provided by Chemical Reagent Co. Ltd, China.
Fabrication of fiber samples
First, jute fibers were functionalized by the carboxymethyl pre-treatment. As shown in Supplemental Figure S1, the fiber samples (1 g) were kept in the NaOH (2 wt%)/isopropanol (20 wt%) solution at 40°C for 1.5 h, followed by adding the ClCH2COONa (2.5 mol/L)/isopropanol (8.0 wt%)/4-dimethylaminopyridine (0.04 mol/L) solution and stirring at 60°C for 4 h. Then the samples were rinsed with distilled water and then immersed in the sulfuric acid (0.05 mol/L) solution at 25°C for 2 h. After that, the functionalized jute fibers were rinsed with distilled water, and dried in a baking oven.
Second, SiO2 nano-arrays were introduced on the functionalized jute fibers using the sol-gel method as follows. The fiber samples (0.1 g) were placed in the ethanol (30 mL)/TEOS solution, followed by adding the ethanol (10 mL)/distilled water solution and the ethanol (5 mL)/ammonia solution, separately. Then the solution was stirred ultrasonically for 1 h at 25°C and stirred mechanically for some time at 40°C. The functionalized jute fibers coated with SiO2 nano-arrays were washed with deionized water to remove residual chemicals and dried at 70°C for 2 h.18,19
Measurements and characterization
Wettability of the test liquids.

Grey relational analysis
To gain a comprehensive understanding of the quantitative relationship between the process parameters and the particle size of nano-arrays, grey relational analysis was performed to determine the correlation degree (
First, the normalizing process for the series was performed using the higher-is-better method according to the following equation:

This grey relational coefficient matrix (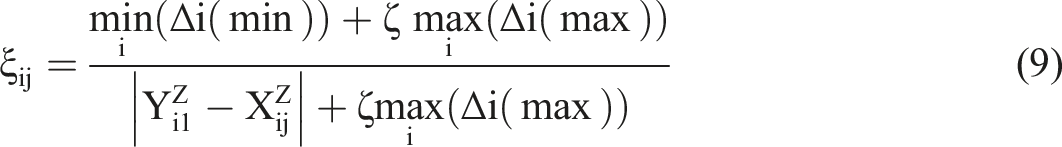

Results and discussion
Surface composition and microstructural analysis of fiber samples
The surface composition of the deposition substrate was confirmed by characterizing the FT-IR spectra of both the jute fibers and functionalized jute fibers. As illustrated in Figure 1(a), the peak at 1737 cm−1, associated with the carboxymethyl groups (-OCH2COOH) of the waxy substance, disappears following the carboxymethyl pre-treatment, indicating the removal of the waxy substance. In contrast, new bands appear at 1611 cm−1 and 1423 cm−1, corresponding to the vibrations of carboxymethyl groups after the pre-treatment.28,29 These observations confirm the successful grafting of functionalized groups onto the jute fibers. To examine the microstructure, the surfaces of fiber samples were imaged using SEM. The jute fiber exhibits a surface covered with a waxy substance, whereas the surface of functionalized jute fiber reveals narrow and parallel defects aligned along the longitudinal direction (Figure 1(b)). AFM scans also demonstrate that the functionalized pre-treatment effectively increases the surface roughness, as shown in Supplemental Figure S2. Additionally, XRD spectra show a significant decrease in the crystallinity of fiber samples (Figure 1(c)), providing strong evidence for the increased amorphous region. The crystallinity of functionalized jute fibers (71.3%) was decreased by 8% than that of control samples (63.2%). The presence of functionalized groups and the increased amorphous region impart a hydrophilicity to jute fibers (surface energy: 45.27 mN·m−1), which is approximately 60% greater than that of control samples (surface energy: 27.89 mN·m−1), as shown in Figure 1(d). Figure 1(e) shows the representative models of the jute fibers and functionalized jute fibers after the energy minimization and structural relaxation. Comparison of the surface compositions and microstructures of the jute fibers and functionalized jute fibers, including (a) FT-IR spectra, (b) surface morphologies, (c) XRD spectra, (d) wettabilities (contact angles and surface energies) and (e) the schematic diagram for equivalent models.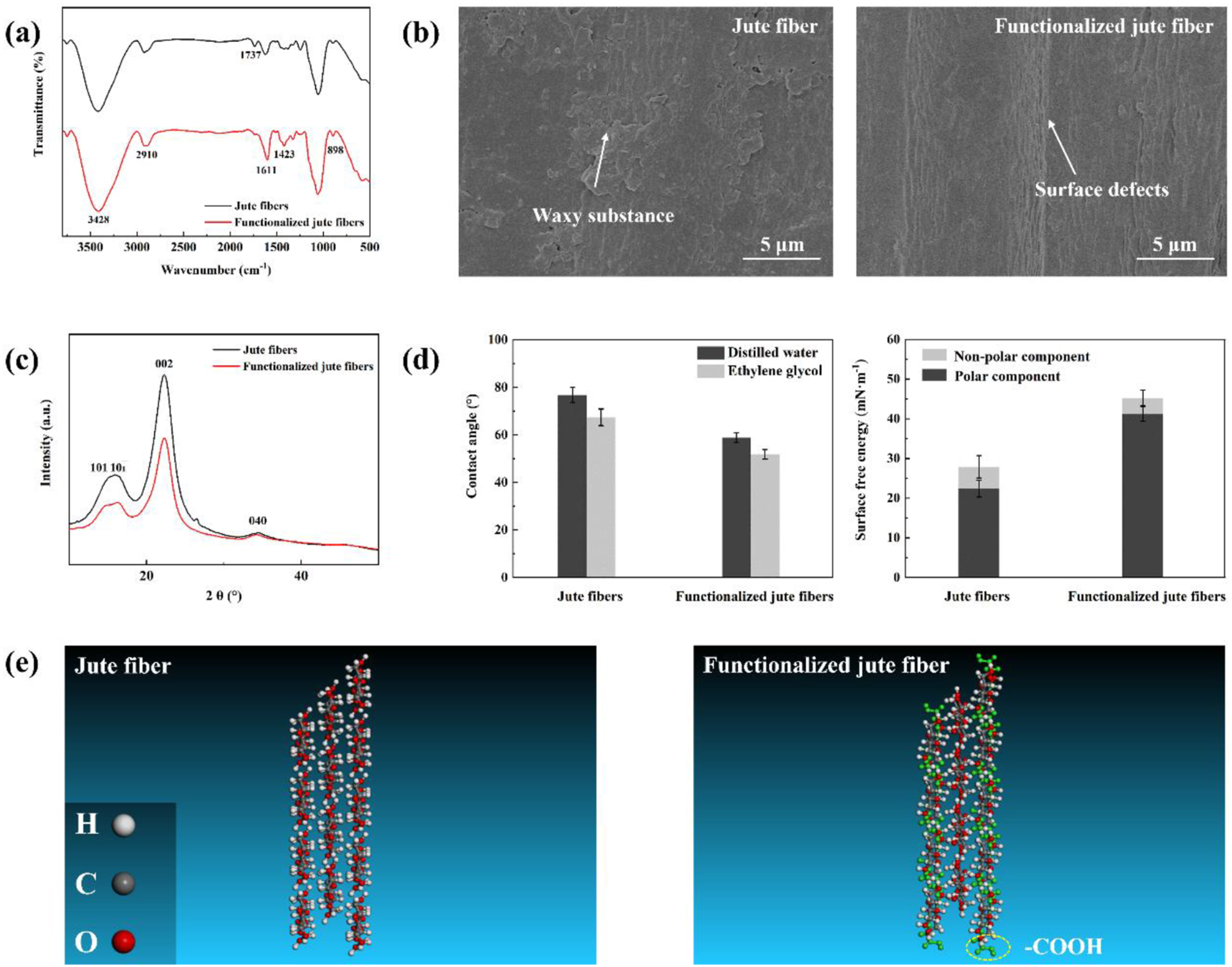
Effect of varying sol-gel parameters on deposition characteristics
Catalyst concentration
Figure 2 illustrates the morphologies of nano-deposition on the functionalized jute fibers, along with the particle sizes and deposition amounts. The deposition of nano-arrays on the functionalized plant fibers is highly influenced by the catalyst concentration. At a low catalyst concentration, a thin and uniform nano-gel layer, with an average thickness of 85 nm, completely covers the surface of the functionalized jute fibers. This results in a surface appearance similar to that of jute fibers with the waxy substance. At this stage, the hydrolysis and condensation rates of precursor are too slow to generate nano-particles on the fibers or in the solution. As shown in Supplemental Figure S3, the reaction solutions are colorless and transparent at the catalyst concentrations of 0.15 mol/L and 0.30 mol/L. The reaction solution gradually saturates as the catalyst concentration increases, which can be seen in Supplemental Figure S3. At the catalyst concentration of 0.60 mol/L, nano-arrays align in an orderly and uniform manner through self-assembly, imparting a neat, nanoscale structure to the functionalized surface of the jute fibers. By analyzing the morphology alongside the trends in particle size and deposition amount, the optimized deposition results achieve the desired control objective, where the surface defects of jute fibers are filled with nano-arrays. However, further increasing the catalyst concentration leads to a slight increase in deposition amount but causes significant aggregation of nano-particles. At this moment, the reaction solution turns non-transparent milky white, which is in a supersaturated state (Supplemental Figure S3). Additionally, the difference in modulus between the fibers substrate and the thick nano-coating results in cracking and flaking, as shown in Figure 2(a). This result aligns with previous research.
30
(a) shows SEM images of nano-deposition on the functionalized jute fibers under the different catalyst concentrations (precursor concentration: 0.08 mol/L; reaction time: 6 h). (b) Comparison of the particle size and deposition amount of nano-deposition under the different catalyst concentrations.
Precursor concentration
As depicted in Figure 3, the particle size of nano-arrays on the functionalized jute fibers increases proportionally with the precursor concentration (Figure 3(b)). A similar trend is observed in the deposition amounts. These findings demonstrate that the deposition characteristics of nano-arrays can be regulated by modulating the precursor concentration. Even slight variations in the precursor concentration result in significant changes in both particle diameter and deposition amount. It can also be proved by the saturated state of the reaction solution in Supplemental Figure S4. The reaction solution gradually saturates with the increasing of the precursor concentration. During the initial stages of deposition, nano-nuclei preferentially arrange in an orderly manner within the surface defects of the fiber samples. As expected, both the particle size and deposition amount of nano-particles increase markedly with rising the precursor concentration. Notably, nucleation requires the solution to reach supersaturation, leading to the formation of nano-particles that are present not only on the samples but also dispersed in the solution (Supplemental Figure S4). At a higher precursor concentration, both the particle size and the deposition amount of the nano-arrays exhibit further increases. When the precursor concentration exceeds the optimal level, the growth in particle size and deposition amount becomes less pronounced due to the limited availability of deposition sites on the functionalized jute fibers. Consequently, a significant proportion of particles remains suspended in the solution rather than deposited on the fiber surface, as illustrated in Supplemental Figure S4. Under these conditions, cracks are also observed in the thicker nano-arrays. (a) shows SEM images of nano-arrays on the functionalized jute fibers under the different precursor concentrations (catalyst concentration: 0.60 mol/L; reaction time: 6 h). (b) Comparison of the particle size and deposition amount of nano-arrays under the different precursor concentrations.
Reaction time
Figure 4 illustrates that extending the reaction time significantly influences both particle size and deposition quantity. A line-shape fitting model was applied to evaluate the particle sizes within the initial 4-h period for detailed analysis. The model reveals a growth rate of approximately 15.24 nm/h and a nucleation time of about 1.26 min, indicating that the nucleation of nuclei occurs within a relatively short timeframe. After 2 h of reaction, a thin nano-gel layer containing some nuclei forms on the functionalized jute fibers. However, this layer is insufficiently thick to repair the surface defects. As the reaction progresses, the nuclei grow and organize into arrays. Additionally, more nano-particles form in the solution, as seen in Supplemental Figure S5. Interestingly, the saturation state of the solution barely changes after 4 h. It means that the nucleation may stop, whereas the growth continues. By 6 h, the nano-arrays nearly fill up the surface defects of the fibers. With further extension of the reaction time, the nano-arrays, influenced by short-range interactions between adjacent particles, may form an additional gel layer over the fiber surface. (a) shows SEM images of nano-arrays on the functionalized jute fibers under the different reaction times (catalyst concentration: 0.60 mol/L; precursor concentration: 0.08 mol/L). (b) Comparison of the particle size and deposition amount of nano-arrays under the different reaction times.
Grey relational analysis
According to equation (6), the matrix (R) formed by the comparative series (X) and the reference series (Y) is structured as follows: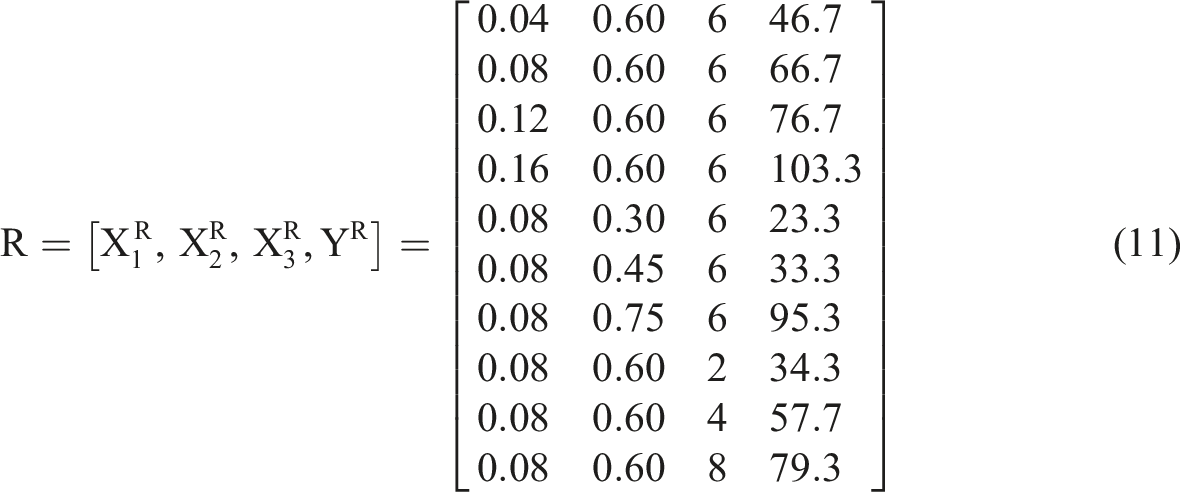
Based on equations (7) and (8), the normalized matrix (Z) is given as follows:
According to equation (9), the grey relational coefficient matrix (
By using equation (10), the grey relation grade (
Nucleation and growth mechanisms of nano-arrays on plant fibers
Effects of the surface composition and microstructure of plant fibers
The surface of plant fibers, serving as the substrate for nano-deposition, plays a crucial role in determining the nucleation and growth behaviors of nano-arrays. Well-aligned nano-arrays can be formed on the functionalized plant fibers for the following reasons. First, the active groups introduced on the fiber surface facilitate interactions between the fiber and the nano-sol, thereby promoting the heterogeneous deposition of nano-sol. This finding has been quantitatively confirmed through molecular dynamics simulations in the previous researches.18,31 Second, the functionalized pre-treatment enhances the amorphous regions on the plant fibers, where the introduced active groups are loosely bound to the molecular chains. The functionalized pre-treatment provides easier access for the reaction solution to interact with the plant fibers. Third, the removal of the waxy substance exposes surface defects, enhancing the surface roughness of the plant fibers. At the initial deposition stage, the nucleus assumes a truncated spherical shape (Figure 5). Based on the Wenzel model, the relationship between the contact angle of the rough surface ( Micro modellings of the heterogeneous nucleation on the functionalized plant fibers with the different morphologies.

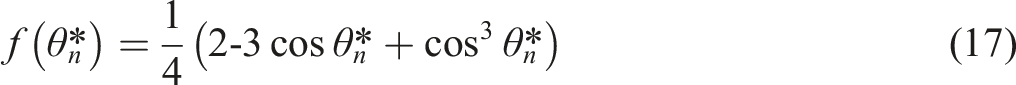

From equation (15), an increase in roughness factor (
Effect of process parameters in sol-gel synthesis
Manipulating the process parameters is an effective approach to achieving the desired nano-array structure. Under basic catalyst conditions, the single precursor undergoes hydrolysis with water, which controls the supersaturation of the reaction solution and provides the driving force for the heterogeneous nucleation and growth of the nano-arrays.37–39 As the precursor concentration increases, both the particle size and deposition rate of the nano-arrays improve simultaneously until they reach their maximum values. However, once the maximum deposition amount is exceeded, a significant proportion of the nano-particles remains in the solution rather than depositing on the fibers. The nucleation and condensation rates of the precursor are highly dependent on the concentration of the basic catalyst. At low catalyst concentrations, the hydrolysis rate of the precursor is slow, leading to the formation of hydrolysis products primarily as M(OC2H5)3OH, M(OC2H5)2(OH)2. 39 After condensation, a thin layer of nano-gel with a three-dimensional cross-linking network structure forms on the fiber. When sufficient basic catalyst is present, the hydrolysis rate of the precursor increases, leading to the formation of hydrolysis products, primarily MOC2H5(OH)3 and M(OH)4. As a result, the complete condensation of the nano-sol promotes the formation of spherical particles on the fiber.37–39 Notably, achieving monodispersed particles becomes challenging at excessively high concentrations of the basic catalyst. There are two primary pathways for nano-particle growth: atom/molecular addition and aggregation.40,41 Once the concentration of the basic catalyst exceeds the critical value, the hydrolysis and condensation of the precursor occur too rapidly to be controlled. This results in aggregation becoming the dominant pathway for nano-particle growth rather than the atom/molecular addition process. Furthermore, the excess -OH groups in the reaction solution can enhance the surface adsorption of nano-particles, thereby increasing the likelihood of aggregation. 37 The reaction time plays a critical role in the nucleation and growth cycle of nano-arrays, particularly during the growth period. Sufficient reaction time is essential to fully deplete the supersaturation and smaller nuclei, thereby promoting the formation of stable nano-arrays.
Evolution of nano-array structures on plant fibers
Based on the above results, the evolution process of nano-arrays on plant fibers with functionalized pre-treatment is outlined. As shown in Figure 6, the hydrolysis of precursor takes place under the base-catalyzed condition, leading to the formation of nano-sol. In the presence of a basic catalyst, the hydrolysis rate exceeds the condensation rate, leading to the primary hydrolysis product being M(OH)4 rather than (C2H5O)M(OH)3 or (C2H5O)2M(OH)2.42,43 Subsequently, M(OH)4 undergoes condensation to form two-dimensional chains and three-dimensional cross-linked networks, which result in the production of a thin nano-gel layer that covers the fiber. This process is facilitated by the active groups, as illustrated in Figure 7. When the critical nucleus size is reached, the nano-gel forms nuclei with the uniform size on the fiber, serving as the seed layer. Notably, the initial nuclei align in an orderly manner within the surface defects of the fiber, which is attributed to short-range interactions. The distribution of seed size is controlled during the nucleation stage.
37
Nucleation ceases once the reaction concentration falls below the nucleation concentration, while the growth persists until the first layer of nano-array is formed.
44
The concave nanostructures, which exhibit the uniform curvature between the nano-particles, create nucleation sites for the next layer of nano-arrays.32,45 Similarly, due to the presence of these concave structures, well-spaced nano-arrays form in the surface defects of the fibers through a layer-by-layer self-assembly process.
46
The hydrolysis and condensation of precursor on the functionalized plant fibers. The evolution process of nano-arrays on the functionalized plant fibers.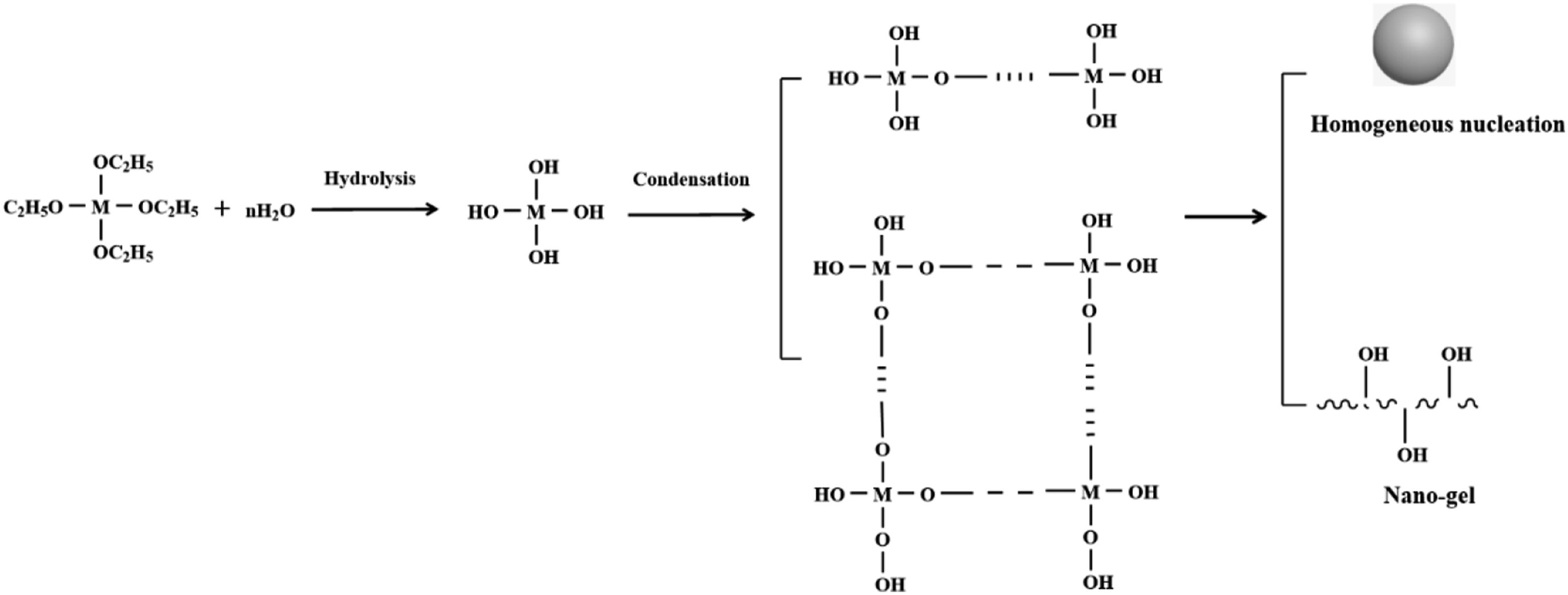

Conclusion
To summarize, the synergistic effect of the surface features of plant fibers and the sol-gel process parameters facilitate the layer-by-layer self-assembly of nano-arrays. The key conclusions drawn from this study are as follows: (1) Regarding the surface features of the plant fibers, the introduced active groups and the concave structures strengthen the interaction between the fibers and the deposited structures, creating the suitable sites for heterogeneous deposition and thus enabling the formation of nano-arrays through layer-by-layer self-assembly. (2) Regarding the sol-gel parameters, the precursor concentration provides the driving force for the heterogeneous nucleation and growth of nano-arrays. The catalyst concentration controls the morphologies of the nano-deposition, including the thin nano-gel, well-aligned nano-arrays, and aggregated nano-particles. The reaction time is closely linked to the nucleation and growth cycle of nano-deposition. By the grey relational analysis, the influence of these three factors on particle size follows this order: reaction time < precursor concentration < catalyst concentration.
The mechanisms of in-situ nucleation and growth behavior of nano-array on plant fibers discussed in this study provide a theoretic basis for preparing advanced plant fibers with the controllable surface structure and properties. This, in turn, supports the development of eco-friendly and cost-effective bio-composites with competitive mechanical performances.
Supplemental Material
Supplemental Material - Sol-gel derived nano-array structures on plant fibers: Nucleation and growth mechanisms
Supplemental Material for Sol-gel derived nano-array structures on plant fibers: Nucleation and growth mechanisms by Xuan Liu in Journal of Industrial Textiles
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.





