Abstract
Pancreatic cancer remains one of the most challenging malignancies to treat due to its dense stromal microenvironment and resistance to conventional therapies. This study introduces a novel localized drug delivery system design to target residual cancer cells following surgery. Electrospun fibrous carriers were fabricated using needle-less electrospinning from polycaprolactone (PCL), silk fibroin (SF), and their blend. Among these, PCL carriers (average fiber diameter: 141 ± 28 nm) exhibited the highest and most sustained paclitaxel (PTX) release in vitro. Coating the PCL carrier with hyaluronic acid (HA) increased the fiber diameter to 535 ± 116 nm and modulated PTX release, shifting from an initial rapid release phase in uncoated carrier to a more gradual and sustained release over 120 hours. PTX-loaded HA-coated electrospun PCL carriers significantly reduced MiaPaCa cell viability, with only 13% viability at 96 hours compared to 22% for the non-coated carrier. This HA-coated electrospun PCL carrier offers a scalable and efficient solution for localized PTX delivery, providing sustained drug release, prolonged cytotoxic efficacy, and reduced off-target effects. Its industrial scaleability, combined with its potential for post-surgical pancreatic cancer management, presents an innovative approach to minimizing reliance on systemic chemotherapy and its associated toxicities. The use of needle-less NanospiderTM electrospinning technology further emphasizes its clinical potential, with future in vivo studies needed to confirm carrier’s safety, pharmacokinetics, and therapeutic benefits.
Keywords
Introduction
Pancreatic cancer remains one of the most lethal malignancies, characterized by poor prognosis and limited treatment options. The 5-year survival rate for patients diagnosed with distant metastases is alarmingly low, ranging from 3% to 10%, depending on the stage at diagnosis. 1 Current treatment modalities include surgery, chemotherapy, radiation therapy, and palliative care. Surgical resection remains the gold standard for localized disease. 2 For patients with locally advanced or metastatic disease, chemotherapy is the cornerstone of treatment. Common regimens, such as gemcitabine with paclitaxel (PTX) and FOLFIRINOX, have shown some success in extending survival. 3 However, chemotherapy presents significant drawbacks, including systemic side effects, technical limitations, and cancer recurrence due to incomplete tumor removal during treatment. 4
Drug delivery systems have the potential to lower these limitations during pancreatic cancer treatment. They offer a significant advantage by releasing drugs efficiently at the target site, reducing systemic side effects and lowering the likelihood of tumor recurrence. 5 Drug delivery systems can be classified as either systemic (e.g., nanoparticles) or localized (e.g., nanofibers, films, hydrogels) based on their application. 6 Systemic drug delivery, such as nanoparticles, are beneficial due to their small size, specificity, and non-invasive nature. These features allow nanoparticles to travel through smaller vessels and target specific receptors without invasive procedures.7,8 However, nanoparticles may lose specificity or face functionalization challenges due to the complexities of the delivery mechanism. 9 For example, hyaluronic acid (HA) nanoparticles have gained attention for targeting cancer cells, particularly those expressing the CD44 receptor, a common marker in pancreatic cancer. This receptor-targeting improves the delivery of chemotherapeutic agents like PTX, increasing therapeutic efficacy while reducing toxicity. 10 HA’s biocompatibility reduces immune responses, enhancing its potential as a drug delivery system. 11 Additionally, HA can modify the tumor microenvironment, potentially improving drug penetration in dense tumors. 12 However, challenges such as rapid degradation in biological environments may limit sustained drug release. 13 Developing stable and effective HA nanoparticles remains complex and requires further optimization. 14
Localized drug delivery systems offer an alternative by releasing drugs directly at the target site through diffusion or degradation. 15 Examples include foams, hydrogels, and nanofibers.16–18 While hydrogels and foams rely primarily on rapid diffusion, which limits sustained therapy, electrospun fibrous materials have shown superior potential for controlled, long-term drug release. 19 Additionally, localized systems can complement surgical tumor removal, reducing recurrence risk. 20 Electrospun fibrous materials have gained attention due to their high surface area, porosity, and ability to deliver drugs in a controlled and sustained manner. These materials are biocompatible and biodegradable, minimizing immune responses while offering customization in composition and drug release properties. 21 Recent advances have explored electrospun fibrous materials for targeting pancreatic cancer cells. These materials, engineered to deliver antibodies, have demonstrated significant potential in minimizing off-target effects and enhancing treatment efficacy. 22 Nanofibrous carriers loaded with chemotherapeutics, such as irinotecan, have also been investigated for post-surgical adjuvant therapy. 23 Moreover, co-delivery systems that combine microRNA-21 antisense oligonucleotides with gemcitabine have demonstrated synergistic effects in inhibiting cancer metastasis. 24 These studies highlight electrospun fibrous drug delivery systems as a promising strategy for enhancing therapeutic efficacy in pancreatic cancer.
Electrospinning is a widely used technique for producing electrospun fibrous carriers. It involves applying an electric force to draw charged polymer solutions into nanofibers. While needle-based electrospinning allows for precise control of fibers, it has limitations like needle clogging and scalability issues. 25 Needle-less electrospinning technique, such as free-liquid electrospinning, addresses these limitations by offering higher production rates and more uniform fiber diameters. 26 NanospiderTM technology, a needle-less electrospinning method, enables high-quality nanofiber production with minimal solvent use. This makes it suitable for industrial-scale applications.27,28 The use of polycaprolactone (PCL) and silk fibroin (SF) electrospun fibrous materials in pancreatic cancer treatment is a promising area of research due to their unique properties. These polymers enhance drug delivery even though direct studies targeting pancreatic cancer with PCL or SF nanofibers are limited. Several papers suggest their potential in related biomedical applications, which could be extrapolated to pancreatic cancer treatment. 29 PCL is a biodegradable polymer known for its mechanical properties and biocompatibility, making it suitable for drug delivery systems. Its slow degradation rate enables sustained drug release, which is crucial for aggressive cancers like pancreatic cancer, where prolonged therapeutic exposure enhances efficacy. 30 For instance, study has shown that PCL-based nanofibers could effectively deliver quercetin, an anticancer agent, with a controlled release profile suitable for cancer treatment. 31 In another study, nanofibers incorporating tamoxifen citrate for use in the treatment of breast tumors, highlighted their potential as an alternative for local chronic tamoxifen citrate use for breast cancer treatment. 32 SF, derived from silkworms, has favorable mechanical properties, biocompatibility, and the ability to form nanofibers that mimic the extracellular matrix, facilitating cell adhesion, proliferation, and differentiation. Study has reported that SF electrospun materials could be engineered with tunable mechanical properties, critical for supporting tumor cells or healthy tissue during treatment. Additionally, the high β-sheet content in SF electrospun materials contributes to their stability and controlled drug release capabilities. 33 The combination of PCL and SF offers a synergistic effect that could enhances their application in pancreatic cancer treatment. For example, PCL/SF hybrid nanofibers have been shown to improve cell viability compared to PCL alone, suggesting enhanced biological performance. 34 Furthermore, PCL/SF core-sheath nanofibers have demonstrated the ability to encapsulate drugs effectively, offering a controlled release profile suitable for cancer therapies. 35
Building on the recognized potential of electrospun PCL and SF fibrous materials for localized drug delivery systems, this study investigated the PTX release profiles from these carriers, targeting applications in pancreatic cancer treatment. To facilitate potential industrial-scale production, electrospun fibrous carriers were fabricated using needle-less NanospiderTM technology and surface-loaded with PTX, a chemotherapeutic agent renowned for its efficacy against pancreatic cancer. The fibrous carriers—consisting of PCL, HA-coated PCL, SF, and SF/PCL blends—were systematically evaluated to determine their PTX release kinetics and suitability for localized drug delivery. Additionally, in vitro cytotoxicity assays were conducted to evaluate the therapeutic potential of HA-coated electrospun PCL fibrous carrier loaded with PTX against pancreatic cancer cell line MiaPaCa. This novel approach integrates electrospun PCL fibrous material with HA to create a localized drug delivery system aimed at targeting residual cancer cells post-surgery. Unlike systemic chemotherapy, which often results in off-target effects, localized delivery through fibrous materials offers controlled and sustained drug release, enhancing therapeutic efficacy. The proposed system serves as a complementary approach to systemic therapies, particularly for localized treatment, with the potential to minimize systemic side effects, optimize drug delivery at the target site, and ultimately improve patient outcomes when used alongside conventional treatments. This research stands out by introducing a distinctive combination of materials—PCL, HA, and PTX—processed via NanospiderTM technology, a method that has not been previously explored for localized pancreatic cancer treatment.
Materials and methods
Materials
PCL (Mw 80,000 g/mol) and HA (sodium salt, Mw ∼ 1 MDa) were obtained from Sigma-Aldrich, USA. Acetone, sodium hydroxide (NaOH), and ethanol were sourced from Penta, Czech Republic. Thai silk cocoons of Bombyx mori Linn. silkworms (Nang-Noi Srisakate 1) were collected from Amphoe Mueang Chan, Si Sa Ket Province, Thailand. ECE phosphate reference detergent (FBA free, Union TSL Co., Ltd, Thailand) was used as a soaping agent during the degumming process. The chemicals used for the preparation of SF spinning solutions included calcium chloride (Fluka AG, Switzerland) and 98% formic acid (Penta, Czech Republic). PTX injection solution (Hospira, 6 mg/mL) was purchased from Pfizer, USA. MiaPaCa pancreatic cancer cells, along with cell culture medium (Dulbecco’s Modified Eagle’s Medium, DMEM), phosphate-buffered saline (PBS), trypsin, and the tetrasodium salt WST-1 assay reagent, were all supplied by Sigma-Aldrich, USA. All materials and reagents used in this study were of analytical grade.
Preparation of electrospun fibrous carriers
All fibrous layers were electrospun from polymer solutions using the NanospiderTM NS-Lab 1WS500U electrospinning system (Elmarco, Czech Republic). This system combines industrial production capabilities with the precision of a high-end laboratory instrument. The NanospiderTM technology utilizes a spinning electrode, shaped as a thin wire, with a head that applies the polymer solution along the entire length of the wire. When exposed to a strong electric field, fibrous materials are generated from the thin polymer layer deposited on the wire electrode, see Figure 1. Needle-less electrospinning system and resulting fibrous material. (a) Schematics of the needle-less electrospinning process. The photography of NS-Lab 1WS500U: (b) front view of the electrospinning system. (c) Side view of the electrospinning system, (d) detail of the resulting fibrous material (arrow), deposited onto a spun-bond substrate (asterisk).
The polymer solutions (listed below) were sequentially transferred into a 500 mL head with a polymer solution connected to the electrospinning apparatus. To achieve the production of defect-free fibrous layers with consistent morphology and high productivity using NanospiderTM technology, it was imperative to optimize both the polymer solution preparation and the electrospinning parameters. The electrospinning parameters were systematically optimized through a series of trials to achieve defect-free electrospunPCL, SF, and SF/PCL fibrous materials with high throughput efficiency. The optimization process involved adjusting key parameters such as voltage, collector distance, environmental conditions, as discussed in our previous study. 36 These refinements ensured consistent and reproducible results, which are crucial for the needle-less electrospinning of fibrous materials. A voltage of 50 kV was applied between the wire and the collector, with the two positioned 100 mm apart. Environmental conditions were maintained at 21°C and 60 ± 2% relative humidity using an AC system to optimize the electrospinning process.
The fibrous materials were deposited onto a moving baking paper advancing at 30 mm/min along a negatively charged collector electrode, resulting in a 500 × 600 cm fibrous sheet. After electrospinning, the fibrous material was stabilized, and removed from the baking paper before PTX loading onto the carriers, as detailed in the following sections.
Polycaprolactone fibrous carrier
The solvent mixture composition and PCL solution concentration were thoroughly optimized through numerous trials to achieve defect-free fibrous material. This process involved adjusting the solvent ratios to ensure complete polymer dissolution and fine-tuning the polymer concentration to balance viscosity and needle-less spinnability. These parameters were selected based on our findings to ensure consistent fiber morphology and high production efficiency. Further details on this optimization process can be found in our previous work. 36 The solvent mixture, consisting of acetone and ethanol in a volume ratio of 7:3 (v/v), was used to dissolve solid PCL polymer. The polymer solution was stirred at a rotational speed of 200 RPM at room temperature for 24 hours using a magnetic stirrer, resulting in an optimal polymer concentration of 16 wt%. Electrospinning of the PCL solution was performed under the conditions described above. For comparison, half of the electrospun PCL fibrous sheets were pretreated with 3M NaOH for 5 minutes to enhance the wettability of the fiber surfaces.
Silk fibroin fibrous carrier
The optimization of the degumming process and SF solution parameters was achieved through systematic trials, ensuring the production of high-quality, defect-free fibers. These parameters were carefully selected to control both the degumming and electrospinning conditions, which are essential for enhancing the properties of the final fibrous structure. This approach is consistent with our practices in SF needle-less processing, as outlined in our previous work. 36 The degumming process of raw silk cocoons was conducted twice, utilizing a 0.1 M sodium carbonate solution and a 0.5% standard reference detergent at 100°C for 30 minutes each. This process effectively removed sericin from the fiber surface, after which the fibers were rinsed with warm water and allowed to dry at room temperature. Subsequently, SF solution was prepared by dissolving degummed silk fibers in 98% formic acid, supplemented with 3 wt% calcium chloride, resulting in a polymer solution with a concentration of 12 wt%. This solution was subjected to magnetic stirring at room temperature for 24 hours to ensure homogeneity before electrospinning under the specified conditions described above. The electrospun SF fibrous carrier underwent crystallization by immersion in ethanol for 30 minutes, which contributed to a reduction in water solubility. After drying at room temperature, the treated fibrous carrier was immersed in distilled water overnight to facilitate the removal of residual salts, followed by rinsing with distilled water and air drying.
Silk fibroin/polycaprolactone fibrous carrier
The preparation of SF solution was following the protocol outlined in previous section. The PCL solution was prepared by dissolving PCL in formic acid to achieve a concentration of 20 wt%, followed by magnetic stirring to ensure complete dissolution. For the production of the SF/PCL blend fibrous carrier, electrospinning was conducted using a mixture of the SF and PCL solutions in an 8:2 (v/v) volume ratio. This ratio was selected based on findings from our previous research, which indicated that the proportion of PCL in the blend significantly influences the properties of the resulting electrospun fibrous material. Specifically, increasing the PCL content in the spinning solution has been shown to reduce fiber diameter and enhance electrospinning performance while simultaneously decreasing the brittleness of the fibrous carriers. Furthermore, the incorporation of PCL contributes to improved hydrophobicity of the fibers, making this blend particularly suitable for applications where such properties are desired. To examine the effects of the hydrophobic and hydrophilic characteristics of the fibers on the loading of PTX, we opted to investigate this specific 8:2 SF to PCL volume ratio. The electrospinning process for the SF/PCL blended solution was carried out under the conditions described above.
Loading of PTX onto the electrospun fibrous carriers
The electrospun fibrous carriers were cut into 1 cm2 pieces and immersed in a 0.6 wt% PTX solution for 48 hours to ensure uniform drug adhesion and absorption throughout the entire volume of the fibrous carriers. Following this loading process, the fibrous carriers were dried at 37°C for 2 hours. Importantly, this method did not alter the physical or morphological properties of the fibrous layers. For comparative analysis, an additional set of electrospun fibrous carriers was prepared by first soaking them in the PTX solution, followed by immersion in the HA (1 wt% in 0.1 M NaOH) for 1 minute. These HA coated carriers were then dried in oven at 30°C for 24 hours (refer to Figure 2 for a schematic representation and photograph of electrospun carrier loaded with PTX and coated by HA). Three batches of these carriers were prepared. Schematic representation and photograph of electrospun fibrous carrier. (a) Cross-section of electrospun fiber loaded with PTX, (b) cross-section of electrospun fiber loaded with PTX and coated by HA, and (c) photograph of the actual electrospun fibrous carrier.
Methods
Morphological characterization
The surface morphology of the electrospun fibrous carriers was characterized using a Vega TS 5130 scanning electron microscope (SEM, Tescan, Czech Republic). SEM images were obtained with an accelerating voltage of 30 kV. Prior to imaging, each fibrous carrier was sputter-coated with gold (Q150R ES, Quorum Technologies Ltd, England) to a thickness of 7 nm. The SEM images were analyzed using NIS-Elements AR software. The average fiber diameter and its distribution were determined by measuring 100 randomly selected fibers from the PCL, SF, SF/PCL, and HA-coated PCL fibrous carriers. This approach ensured a comprehensive assessment of fiber morphology and consistency across all tested materials.
The gram per square meter (GSM) of the electrospun fibrous carriers was measured according to the ISO 3801:1977 standard.
Contact angle measurement
Optical tensiometry, commonly referred to as contact angle goniometry, is a widely employed technique for determining the contact angle of a liquid droplet on a solid surface, thereby characterizing its surface properties. 37 To evaluate the hydrophilicity of the electrospun fibrous carriers, contact angle measurements were performed at three distinct positions on the carrier per material using a Kruss Drop Shape Analyzer DS4 at room temperature. A 2 μL droplet of deionized water was carefully placed on the surface of the dry carrier, and the average contact angle values were calculated from these measurements.
In vitro PTX release test
The in vitro release test for PTX was performed by immersing 1 cm2 electrospun fibrous carriers into 1 mL of PBS buffer (pH 7.4, 0.15 M ionic strength) in 4 mL glass vials. The PBS buffer was chosen because it closely mimics the physiological conditions of the human body, maintaining a pH of 7.4, which is optimal for both the stability of PTX and the biological relevance of the system. Additionally, PBS has an ionic strength similar to that of bodily fluids, making it suitable for simulating the extracellular environment where the drug release would occur. The vials were incubated at 37°C and agitated at 100 rpm using an Innova 4000 incubator shaker (New Brunswick Scientific, NJ, USA). At predetermined time intervals (0, 1, 2, 4, 6, 24, 48, 96, and 120 hours), 300 μL of the release medium was collected for UV-vis analysis, and an equal volume of fresh PBS buffer was added to the incubation solution to maintain a constant volume and ensure that the drug release process was not disrupted by changes in the volume or composition of the medium. The UV-vis absorbance of the collected release medium was measured at 290 nm using a SpectraMax Plus 384 UV-vis spectrophotometer (Molecular Devices, CA, USA). A linear correlation (R2 = 0.9982) was established between absorption intensity and PTX concentration for standard samples ranging from 0 to 50 μg/mL. All measurements were conducted in triplicate to ensure accuracy and reproducibility of the results.
Drug release modelling
The release profile of PTX from the electrospun carriers was evaluated using two established mathematical models to characterize the underlying drug release mechanisms. These models provide insight into the relative contribution of different processes, such as diffusion, erosion, and degradation, in the release behavior. The experimental data on cumulative drug release at different time points was used to fit the following models. Higuchi model is based on Fickian diffusion and assumes that the drug is released from the matrix via diffusion through the medium. The model relates the cumulative drug release to the square root of time. The mathematical expression is given by:
Where Mt is the cumulative drug release at time t, M
∞
is the total drug amount, and k is the diffusion constant.
38
Hixson-Crowell model is particularly useful for systems where the drug release is governed by the erosion or disintegration of the matrix. This model assumes that the decrease in surface area available for release controls the process. The equation is:
Where Mt is the cumulative drug release at time t, M ∞ is the total drug amount, and kHC represents the rate constant for matrix erosion. 39 For each of these models, the parameters were fitted using a linear regression approach, and the goodness of fit was assessed using the R 2 value. A higher R 2 value suggests a better fit of the data to the model, indicating that the model can accurately describe the release kinetics. The drug release data were plotted, and the models were fitted to assess the release rate constants and the dominant release mechanism (diffusion, erosion, or a combination of both). The analysis of the release kinetics allows for a deeper understanding of the processes controlling the release of PTX from the HA coated carriers, which is important for optimizing sustained release formulations.
In vitro cytotoxicity evaluation
In vitro cytotoxicity of the electrospun fibrous carriers was evaluated in accordance with ISO 10993-5. 40 Prior to testing, the fibrous carriers were sterilised using short-wavelength UV light for 10 minutes to ensure sterility without compromising the structural or chemical properties of the carriers. For the cytotoxicity assay, MiaPaCa pancreatic cancer cells were cultured in DMEM growth medium supplemented with 10% fetal bovine serum (Sigma-Aldrich, Cat# F7524), 1% L-glutamine (Sigma-Aldrich, Cat# G7513) and 1% Penicillin-Streptomycin (Sigma -Aldrich, Cat# P0781). Cells were kept at 37°C in an incubator with an atmosphere containing 5% CO2. When the cells reached 70% confluence, they were trypsinized with 1x trypsin (Sigma-Aldrich, Cat# T3924). Subsequently, MiaPaCa pancreatic cancer cells were seeded in 24-well plates at a confluence of 1 × 105 cells per well and cultured for 24 hours at 37°C to form monolayers of tumor cells covering the 1.9 cm2 growth area of each well. After confirming cell layer consistency, the supplemented DMEM was removed, and the fibrous carriers loaded with PTX (0.5 cm2) were placed in each well. The electrospun carriers along with MiaPaCa pancreatic cells were incubated in 1 mL of fresh-added DMEM medium at 37°C for 24, 48, and 96 hours. At the end of each time period, the fibrous carriers were removed, and cell viability was determined using the WST-1 assay. This assay is based on the reduction of tetrazolium salt WST-1 to soluble formazan by electron transport across the plasma membrane of dividing cells. The level of WST-1 reduction into formazan reflects cell metabolism. The sample aliquots (10 per each time point) of 100 μL were transferred to a 96-well plate, and the absorbances were measured using a SpectraMax Plus 384 UV-vis spectrophotometer at a wavelength of 450 nm. For reference, MiaPaCa cells were seeded in DMEM fresh culture medium under the same seeding conditions.
Cytotoxicity of electrospun fibrous carriers was calculated based on the absorbance values obtained from wells containing cells incubated with the carrier/eluate (A
Statistical analysis
All experiments were performed in triplicate (at a minimum) to ensure accuracy and reproducibility. Results are expressed as the mean ± standard deviation (SD). Statistical analysis was conducted using one-way or two-way ANOVA test to compare data between carriers. Significance levels are indicated: p < 0.001 (***) and p < 0.0001 (****).
Results and discussions
Morphological analysis of electrospun carriers
The primary goal of any drug delivery system, particularly in cancer treatment, is to ensure the targeted release of drugs at the tumor site, maximizing therapeutic efficacy while minimizing side effects. Electrospun fibers are particularly advantageous for delivering high local drug concentrations required in cancer therapies due to their ability to absorb significant amounts of therapeutic agents. The drug release profile is influenced by various factors, including fiber diameter, polymer composition, and the nature of the drug. Moreover, fiber uniformity plays a critical role in ensuring consistent release profiles. Smaller fibers, with their higher surface area, typically release drugs more rapidly, while larger fibers provide a more sustained release, helping to maintain therapeutic drug levels over extended periods.41,42 Achieving a balance between electrospun fiber size and uniformity is essential for optimizing drug delivery performance. One challenge in needle-less electrospinning is ensuring uniformity and consistent fiber morphology at scale. Variations in the electrospinning environment, such as humidity and temperature, can affect fiber deposition. To address this, we optimized solution parameters and controlled process conditions (voltage, spinneret distance, temperature and humidity) to maintain carriers uniformity. In this study, NanospiderTM technology was utilized to enable the continuous and stable production of electrospun fibrous materials with high uniformity. Additionally, the AC chamber in the NanospiderTM system, which we utilized in this study, helps stabilize the electrostatic field, minimizing variations and improving fiber uniformity for large-scale production.
In this study, PCL, SF, and SF/PCL fibrous carriers were successfully needle-less electrospun, exhibiting no beads or structural defects. The fibers demonstrated a narrow diameter distribution and a smooth, round morphology, as illustrated in Figure 3. The average fiber diameters for the PCL, SF, and SF/PCL carriers were determined using SEM. For each material, a total of 100 fibers were measured from the carrier to ensure reliable data representation. The average diameters of the fibers for the PCL, SF, and SF/PCL carriers were found to be 141 ± 28 nm, 903 ± 331 nm, and 772 ± 187 nm, respectively. Notably, NaOH-treated PCL fibers maintained their original diameter of 150 ± 31 nm while exhibiting significant alterations in surface morphology. Specifically, the treated fibers presented a disturbed surface characterized by protruding fluff, indicating that NaOH treatment effectively modifies surface characteristics without impacting the fiber diameter. SEM images of electrospun fibrous carriers. (a) PCL carrier, (b) PCL carrier treated with 3M NaOH, (c) SF carrier, and (d) SF/PCL carrier. The images illustrate the fiber diameter and surface texture for each carrier type. Corresponding histograms of fiber diameter distributions are provided, with measurements taken from the carrier for each material (n = 100 fibers).
The results demonstrated that the electrospun SF fibrous carrier exhibited the largest fiber diameter, while the PCL fibers displayed the smallest diameter. The SF/PCL blend fibers had a diameter slightly smaller than that of pure SF, likely due to the incorporation of PCL, which tends to facilitate the formation of finer fibers. Generally, smaller fiber diameters correlate with a higher surface area-to-volume ratio, thereby enhancing drug absorption. As a result, PCL fibers, characterized by their smaller diameters, provide a larger surface area, thereby should improve drug loading capacity. To compare PTX release from the electrospun fibrous materials, we produced carriers with the same GSM value, specifically 6.5 ± 0.2 g/m2. By maintaining this consistent GSM, we ensured that the amount of material and fiber density remained the same across samples, allowing for a more accurate comparison of the release profiles of PTX between the different fiber compositions. This approach helps isolate the influence of the material properties (such as PCL and SF) on the drug release rate, ensuring that any observed differences are not due to variations in carrier mass or thickness.
Wettability of electrospun carriers
The surface hydrophobicity or hydrophilicity of electrospun fibrous carriers, as indicated by the contact angle, plays a critical role in determining the interaction between loaded drugs and the fibrous surface, significantly influencing drug release behavior. Contact angle measurements serve as essential parameter for assessing the wettability of fibrous materials, directly correlating with their hydrophilic or hydrophobic nature. Specifically, fibrous materials with a contact angle greater than 90° are classified as hydrophobic, while those with a contact angle below 90° are deemed hydrophilic. 43 This distinction is crucial for drug delivery systems, particularly for chemotherapeutics like PTX, where the interaction between the drug and the electrospun fibrous carrier can significantly affect the efficiency of drug delivery to tumor sites. 44
Wettability characteristics of electrospun PCL, PCL (NaOH treated), SF, and SF/PCL carriers. The table include images of water droplets on each fibrous carrier.

The observed variations in contact angle and corresponding wettability can substantially affect both the loading capacity and release kinetics of PTX, a hydrophobic drug. The release dynamics of PTX are closely linked to the surface characteristics of the drug carrier, such as its hydrophobicity or hydrophilicity. Additionally, factors such as carrier porosity, surface charge, and environmental conditions (e.g., pH or biological fluids) also play a significant role in modulating the drug-carrier interactions. Understanding of these variables is essential for the development of advanced drug delivery systems aimed at maximizing the therapeutic efficacy of PTX.
In vitro PTX release from electrospun carriers
As mentioned above, the hydrophobicity or hydrophilicity of fibrous carriers significantly influences the release profile of PTX, a hydrophobic chemotherapeutic widely used in pancreatic cancer treatment. Hydrophobic fibers, such as those made of PCL, strongly interact with PTX due to their compatibility with hydrophobic environments, resulting in a slower diffusion rate and sustained release. This controlled release is crucial for maintaining therapeutic drug levels over prolonged periods, enhancing cancer cell eradication while minimizing systemic exposure. 45 In contrast, increasing the hydrophilicity of fibers—through modifications like surfactant application or sodium hydroxide treatment— enhances water penetration and accelerates drug dissolution and diffusion, often leading to an initial rapid release phase. While beneficial for rapid drug action, this may reduce long-term therapeutic effects. 46 Optimizing these properties is key to designing drug delivery systems that balance immediate and prolonged release based on the specific demands of cancer treatment. Understanding this interplay helps create fibrous materials tailored for effective cancer therapies while minimizing adverse effects. 47
In this study, 0.6 wt% PTX injectable solution was used which is prepared using PTX, polyoxyl 35 castor oil, and dehydrated alcohol. These components facilitate the dissolution of the castor oil and stabilize the formulation, resulting in a solution where PTX exhibits a markedly hydrophobic character. As discussed in Section 3.2, the electrospun fibrous carriers utilized in this study demonstrated varying degrees of hydrophobicity and hydrophilicity, which significantly may influence the interaction between the fibrous materials and PTX, ultimately affecting the drug release profile. The release behavior of PTX from four types of electrospun fibrous carriers—PCL, NaOH-treated PCL, SF, and SF/PCL blend—was evaluated in triplicates, and the data were analyzed using a one-way ANOVA test to assess statistical differences between the groups. The results, illustrated in the accompanying Graph 1, revealed distinct differences in PTX release behavior among the electrospun fibrous carriers types. PTX release from electrospun fibrous carriers. The release behavior of PTX from four types of electrospun fibrous carriers—PCL, NaOH treated PCL, SF, and SF/PCL blend—was evaluated. Statistical analysis using a one-way ANOVA test revealed significant differences in the release profiles among the carriers (p < 0.0001, n = 3).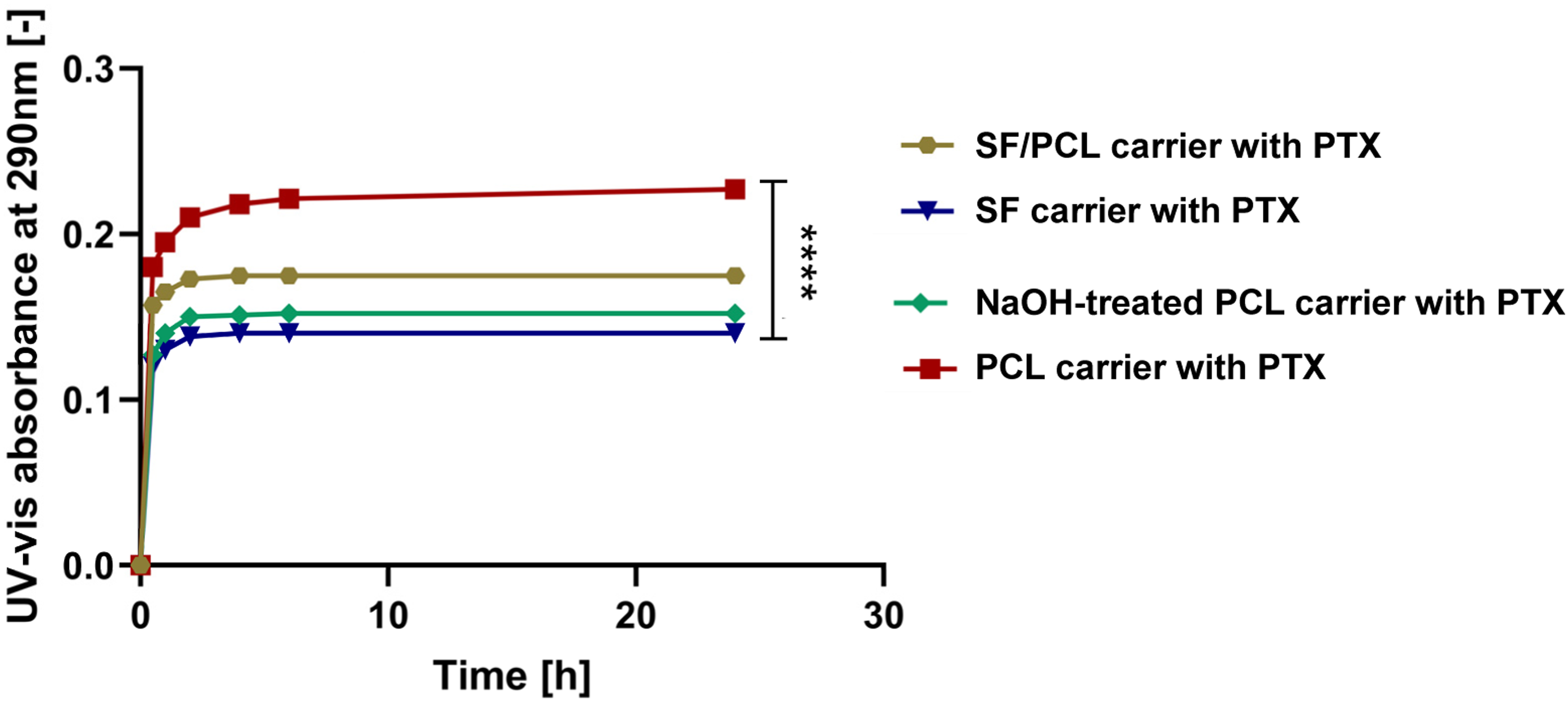
The electrospun PCL fibrous carrier, characterized by its hydrophobic nature and fine fiber structure, exhibited the highest and most sustained PTX release. The sustained release curve indicates a controlled and gradual release over time, with a delayed onset. This sustained release is attributed to strong hydrophobic-hydrophobic interactions, driven by van der Waals forces, which enable PTX molecules to adhere tightly to the electrospun PCL fibrous carrier, thus facilitating prolonged drug delivery. In contrast, SF fibrous carrier, being more hydrophilic, exhibit a weaker affinity for PTX due to hydrophobic-hydrophilic interactions, resulting in an initial rapid release phase curve that shows a rapid increase in drug release early on. While this rapid release can be beneficial for achieving quick therapeutic action, it is less suited for applications requiring sustained drug release. The SF/PCL blend demonstrated intermediate release characteristics. Notably, the highest UV-vis absorbance 0.23 was recorded for PCL fibrous carrier, indicating a greater absorption of PTX compared to SF carrier, which exhibited the lowest absorbance 0.14. This observation aligns well with the contact angle measurements, highlighting the relationship between the hydrophobicity of the PCL fibrous carrier and its ability to absorb and release PTX. Statistical analysis using a one-way ANOVA test revealed significant differences in the release profiles among the carriers. In summary, the contact angle measurements and corresponding PTX release profiles clearly demonstrate the pivotal role that surface hydrophobicity or hydrophilicity plays in controlling drug release kinetics. Hydrophobic electrospun PCL fibrous carrier offers a more sustained release of PTX, ideal for applications requiring prolonged therapeutic effects. This is particularly relevant for conditions like pancreatic cancer, where long-term drug exposure is crucial for effective treatment. In contrast, hydrophilic fibrous carrier, such as SF, facilitate an initial rapid release phase of PTX, making them suitable for scenarios requiring immediate therapeutic action. The findings highlight that selecting fibrous materials tailored to the desired release kinetics is essential for optimizing drug delivery systems for cancer treatment. Given the sustained release profile of PTX, PCL fibrous carrier has been identified as the most suitable candidate for further investigation.
Morphology and in vitro release of PTX from electrospun carrier coated by HA
Biopolymer coatings such as alginate and polyethylene glycol (PEG) have been widely explored for drug delivery applications due to their biocompatibility and ability to control drug release. Alginate, a naturally derived polysaccharide from brown seaweed, is frequently used in drug delivery due to its gel-forming ability and structural similarity to the extracellular matrix. Alginate-based hydrogels can encapsulate drugs, providing a protective barrier and enabling sustained release. However, alginate lacks intrinsic bioactivity, limiting its interaction with specific cellular receptors, which may be a drawback for targeted therapies. 48 Similarly, PEG, a synthetic polymer, is known for its hydrophilicity and non-immunogenic properties, making it an excellent material for reducing protein adsorption and prolonging circulation time in drug delivery systems. PEG-based hydrogels offer tunable degradation rates and mechanical properties, allowing for controlled drug release. However, PEG lacks specific cellular interactions, which can limit its bioactive potential. 49 In contrast, HA presents unique advantages in drug delivery due to its intrinsic bioactivity. The incorporation of HA as a surface coating on fibrous carriers may significantly enhanced the release profile and therapeutic efficacy of PTX. HA, a naturally occurring polysaccharide, is renowned for its biocompatibility and capacity to create a hydrophilic environment, which is particularly beneficial for maintaining therapeutic drug levels over extended periods. 10 This characteristic helps reduce dosing frequency and minimize associated side effects. 50 The mechanism through which HA improves PTX’s release profile is multifaceted. Firstly, HA enhances the solubility and stability of PTX, addressing the drug’s inherent hydrophobicity. Additionally, the molecular weight of HA is critical in modulating the release kinetics of PTX; higher molecular weight HA can create a more viscous barrier, potentially slowing drug diffusion, while lower molecular weight HA may facilitate a faster release profile. 51 This balance is essential for optimizing therapeutic outcomes, allowing for tailored drug release rates according to specific treatment requirements.
Coating the PTX-loaded PCL fibrous carrier with HA resulted in significant changes in both morphology and release profiles, as shown in Figure 4 and Graph 2. The HA coating formed a continuous film on the surface of individual fibers. Fiber diameters were measured across three batches of HA-coated electrospun PCL carriers, with a total of 100 fibers analyzed to ensure batch-to-batch consistency, as illustrated in the SEM images in Figure 4(a)–(c). Variability in fiber diameter was observed, with Batch 1 averaging 549 ± 120 nm, Batch 2 averaging 544 ± 124 nm, and Batch 3 averaging 512 ± 121 nm. Despite these differences, the overall average fiber diameter across all batches was 535 ± 115 nm, indicating relatively consistent fiber morphology. The observed variation in fiber diameters is attributed to the HA coating process rather than the electrospinning. Before HA coating, the standard deviation of fiber diameters was 28 nm, indicating tightly distributed morphology, whereas after HA coating, it increased to 115 nm, reflecting typical variability from surface modification. Despite this, the consistency across batches confirms the reproducibility of the HA coating process, demonstrating that both electrospinning and coating techniques are reliable for scalable production. The porous fibrous structure indicates that the HA primarily coated the fiber surfaces, increasing their diameter without compromising the carrier’s integrity. This ensures maintained functional properties, enhanced drug absorption, and improved controlled release capabilities. SEM image of electrospun PCL fibrous carrier loaded with PTX and coated with HA. (a–c) SEM images of three different batches, confirming reproducibility. (d) Detailed SEM image of the fiber structure. The accompanying histogram presents the fiber diameter distribution, with measurements taken from three batches of HA-coated carriers (n = 100 fibers in total). Release kinetics of PTX from electrospun PCL fibrous carrier coated with HA. The graph demonstrates a gradual release of PTX over time, highlighting the effect of HA coating on PTX delivery dynamics. Statistical analysis using a one-way ANOVA test revealed significant differences in the release profiles among the carriers (p < 0.0001, n = 3).

To comprehensively evaluate the carrier’s performance, the PTX loading efficiency (DLE) and loading capacity were determined. The DLE, calculated as the ratio of the total amount of PTX released to the initial amount applied, was 97.5% for the non-coated electrospun PCL carrier and 90.9% for the HA-coated PCL carrier. Additionally, the PTX loading capacity was found to be 533 mg PTX per 1 g of carrier, demonstrating the carrier’s high drug absorption efficiency. Since the electrospun carriers were immersed in the PTX solution, drug loading predominantly occurred on the fiber surface rather than within the fiber matrix. In an ideal scenario without degradation or irreversible binding, the theoretical DLE would approach 100%. However, slight deviations from this value were observed, likely due to PTX adsorption onto fiber surfaces or partial entrapment within the fibrous structure, as reflected in the measured DLE values.
Moreover, statistical analysis using a one-way ANOVA test revealed significant differences in the release profiles between PCL carriers with and without HA coating, as illustrated in Graph 2. For the electrospun PCL carrier without HA, UV-Vis absorbance measurements indicated a steady release during the first 2 days, followed by a rapid increase after 48 hours. This suggests that the release from the uncoated fibers was primarily diffusion-controlled, with a faster initial release and a slight variation after 48 hours. In contrast, the HA-coated PTX-loaded electrospun PCL carrier exhibited a more controlled and gradual increase in release over time. The release rate remained stable for the first 3 days, followed by a significant increase in PTX release after 72 hours, continuing until 120 hours. The HA-coated carrier achieved a substantially prolonged drug release compared to the uncoated PCL carrier, supporting the hypothesis that HA enhances drug retention and modulates release characteristics. As a control, PTX alone (without the electrospun carrier) was tested and exhibited a nearly constant release profile throughout the test period. This underscores the superior efficacy of the HA-coated electrospun PCL carrier in providing a more favorable drug release environment, promoting regulated PTX release over time.
Additionally, the release profile of PTX from HA-coated electrospun PCL carrier was analyzed using the Hixson-Crowell and Higuchi models to gain insight into the mechanisms governing drug release. The Hixson-Crowell model, which describes drug release through matrix erosion, demonstrated a good fit with an R2 value of 0.85. This indicates that HA disintegration is one of the key mechanisms for PTX release, with the HA coating degrading over time and gradually increasing the exposed surface area of the drug-loaded carrier. The derived rate constant (k = 0.001) reflects the rate of HA coating degradation, facilitating sustained drug release as the coating progressively breaks down. The Higuchi model, commonly used to describe diffusion-driven release, showed an R2 value of 0.74. This suggests that diffusion also plays a role, particularly in the later stages of the release process as the HA coating continues to degrade. The rate constant (k = 2.9) indicates that as the HA coating degrades, diffusion becomes more dominant, contributing to the sustained release of PTX. By applying these two models, it is evident that the release of PTX from HA-coated electrospun PCL carrier occurs in multiple stages, initially driven by HA degradation and later governed by diffusion. Early in the release process, a rapid PTX release is observed as the HA coating degrades. Over time, as the HA coating continues to break down, diffusion becomes the primary release mechanism, resulting in a prolonged release phase that sustains the therapeutic effect of PTX. These findings are consistent with existing literature on HA-based electrospun fibrous drug delivery systems, which have consistently demonstrated comparable sustained release profiles. For example, HA/gelatin coaxial nanofibers loaded with arginine effectively controlled the release of cationic biomolecules, highlighting the potential of HA-based electrospun fibers in drug delivery applications. 52 Similarly, studies have reported an initial rapid release phase followed by sustained release over extended periods, further confirming HA’s efficacy in modulating drug release kinetics. 53 Building on these observations, biocompatible electrospun polyvinyl alcohol/chitosan/HA fibrous materials were evaluated for the sustained release of human growth hormone, showing an initial rapid release phase of 11% within 2 hours, followed by sustained release reaching 64% over 48 hours. 54 Furthermore, electrospun HA/polyvinyl alcohol/2-hydroxypropyl-β-cyclodextrin fibrous materials demonstrated sustained naproxen release over 48 hours, attributed to the structural stability of the carriers. 55 These examples collectively emphasize the ability of electrospun HA-based fibrous carriers to provide controlled release of therapeutic agents through structural and material composition.
The degradation behavior of HA-coated PCL carrier, while tested in vitro in PBS, is expected to differ significantly in vivo, where enzymatic process mediates HA breakdown. Under physiological conditions, HA is primarily degraded by hyaluronidases, which cleave glycosidic bonds between disaccharide units. The degradation rate of HA is influenced by the presence and concentration of these enzymes, often upregulated in tumor environments. 56 Additionally, non-enzymatic processes, such as hydrolysis, also contribute to HA degradation. Physiological conditions, including pH, temperature, and the ionic environment, may further modulate the rate of degradation, either accelerating or slowing the process. 57 In contrast, PCL degrades much more slowly in physiological conditions, with in vivo studies showing only partial degradation over 90 days, ensuring sustained drug release. 58 The combination of HA and PCL creates a dual-release system—an initial rapid release from HA, driven by rapid enzymatic degradation, followed by a sustained release from the slowly degrading PCL matrix. This approach optimizes drug retention at the target site while maintaining prolonged therapeutic effects, making it particularly valuable for controlled drug delivery in pancreatic cancer therapy. This intricate balance between enzymatic and non-enzymatic degradation is crucial for ensuring a controlled and sustained release of the therapeutic agent, a key factor in the localized treatment of pancreatic cancer. Understanding these processes is important to fine-tuning the release profile for optimal therapeutic efficacy. Future studies focusing on the precise degradation kinetics of HA under physiological conditions and the specific influence of the tumor microenvironment will be critical. Such investigations will aid in optimizing the drug delivery system, enhancing its potential for clinical applications.
Cytotoxicity of electrospun PCL fibrous carrier loaded by PTX against pancreatic cell line
The utilisation of HA to coat electrospun PCL carrier loaded with PTX represents a strategic approach to enhancing targeted delivery against pancreatic cancer. This strategy is particularly relevant given the unique characteristics of pancreatic tumors, which often feature a dense stromal environment that complicates effective drug delivery. HA’s ability to specifically bind to CD44 receptors, frequently overexpressed on pancreatic cancer cells, facilitates the selective delivery of PTX directly to the tumor site while minimizing off-target effects. 11 CD44 is a transmembrane glycoprotein that serves as the principal cell surface receptor for HA. The binding occurs via the link domain of CD44, which recognizes and attaches to specific sequences within the HA polymer. This interaction follows a catch-bond mechanism, where the bond between HA and CD44 strengthens under mechanical stress, enhancing cell adhesion under dynamic conditions. 59 The HA-CD44 interaction is pivotal in cancer progression, particularly in tumors where CD44 is overexpressed. By exploiting this binding specificity, HA-coated nanocarriers can target CD44-expressing tumor cells, facilitating preferential tumor accumulation and increased cellular uptake of chemotherapeutic drugs. This targeted delivery mechanism is crucial for navigating the dense stroma of pancreatic tumors, as it can impede the effective penetration and distribution of therapeutic agents. By enhancing the interaction between the drug-loaded fibrous carrier and cancer cells, HA improves drug localization, promoting better therapeutic outcomes. Moreover, the incorporation of HA for targeting reduces systemic exposure to PTX, thereby decreasing potential side effects and improving patient safety—a significant consideration in cancer therapy. 60 The HA-CD44 axis plays a pivotal role in cancer biology, as CD44 is closely linked to cancer stem cell properties, metastasis, and resistance to therapy. Leveraging this interaction enables the development of targeted therapeutic strategies. For instance, HA-decorated nanocarriers can preferentially accumulate in CD44-overexpressing tumors, enhancing the delivery and efficacy of chemotherapeutic agents. 61 Furthermore, disrupting the HA-CD44 interaction has been explored as a therapeutic approach. Soluble CD44 or peptides that block HA binding can inhibit tumor growth and metastasis by preventing CD44-mediated signaling pathways. 62 In summary, the molecular interaction between HA and CD44 is a critical mediator of cellular functions and presents a valuable target for therapeutic interventions, particularly in oncology. Importantly, applying the HA-coated PCL fibrous carrier directly onto the surgical site after tumor removal may offer a dual benefit: inhibit the regrowth of residual cancer cells and directly deliver PTX to the affected area. This aligns with the principles of precision medicine, where therapies are tailored to the specific characteristics of the tumor and patient needs. This combined approach may represent a promising adjunctive therapeutic strategy that addresses the challenges of drug delivery in pancreatic cancer while prioritizing improved patient safety.
The results demonstrate that MiaPaCa cell viability remained relatively constant when treated with free PTX, ranging from 58% to 40% over 96 hours. This limited efficacy is like due to PTX’s poor solubility and rapid systemic clearance. In contrast, PTX-loaded electrospun PCL carriers exhibited a significant decrease in cell viability after 48 hours, attributed to the sustained release profile of PTX from the hydrophobic PCL carrier. Notably, HA-coated PTX-loaded electrospun PCL carriers demonstrated a more pronounced cytotoxic effect, reducing cell viability from 44% at 72 hours to 13% at 96 hours. This effect was significantly greater than that observed with non-HA-coated carriers, which exhibited a modest reduction from 27% at 72 hours to 22% at 96 hours, as illustrated in Graph 3. This enhanced cytotoxicity in HA-coated electrospun PCL carrier can be attributed to two key mechanisms: (i) HA’s ability to selectively bind to CD44 receptors, which are overexpressed on pancreatic cancer cells, thereby improving drug localization and uptake, and (ii) the HA coating’s role in modulating PTX release, mitigating the initial rapid release and promoting a more sustained therapeutic effect. Statistical analysis using a two-way ANOVA test confirmed significant differences in cell viability between HA-coated and uncoated PTX-loaded carriers, as well as in comparison to free PTX (p < 0.001, n = 3). Cytotoxicity of PTX-loaded PCL fibrous carriers against MiaPaCa pancreatic cancer cell monolayers. (a) Cell viability at varying exposure times, showing significant differences (p < 0.001, n = 3) between HA-coated and uncoated PTX-loaded carriers, as well as in comparison to free PTX, as determined by two-way ANOVA. (b) Representative optical microscope image of the MiaPaCa cell monolayer.
The distinct PTX release profiles of HA-coated and uncoated carriers further highlight the impact of HA on drug delivery kinetics. Uncoated PCL fibrous carriers exhibited a rapid PTX release within the first 48 hours, correlating with the observed reduction in cell viability at 72 hours (27%). While this immediate drug release provides an initial therapeutic response, the rapid clearance from the tumor site may limit long-term efficacy. In contrast, HA-coated fibrous carriers exhibited a slower, sustained release profile, with a significant increase in PTX release at 72 hours, corresponding to a more gradual but ultimately greater reduction in cell viability (13% at 96 hours). This prolonged exposure likely enhances PTX accumulation within the tumor microenvironment, ensuring more effective targeting of cancer cells over time. 63 Although this study does not directly investigate the HA-CD44 interaction via blocking assays, prior researches supports HA’s role in improving CD44-mediated drug delivery. This interaction facilitates internalization and intracellular drug activation, making HA-conjugated nanocarriers and HA-functionalized drug delivery systems highly effective. For instance, HA-decorated nanoparticles loaded with gemcitabine (GEM) or quercetin have successfully overcome chemoresistance with minimal systemic cytotoxicity.64,65 Similarly, HA-functionalized carbon nano-onion particles have shown enhanced encapsulation and delivery of GEM-derived prodrugs, improving treatment outcomes against chemoresistant pancreatic tumors. 66 These findings align with the current study, reinforcing the role of HA in facilitating targeted drug delivery and sustained therapeutic effects. However, nanoparticles dispersed in liquid form face challenges in achieving controlled and localized delivery to specific tumor sites.
Localized drug delivery systems, such as electrospun carriers, address this limitation by enabling sustained anticancer drugs release directly at the tumor site, minimizing systemic toxicity. While electrospun carriers have been investigated for various cancers, relatively few studies have focused on pancreatic cancer treatment. For instance, GEM-loaded core–shell electrospun fibers fabricated via emulsion electrospinning with HA hydrosol demonstrated sustained drug release for up to 3 weeks. By optimizing GEM load and hydrosol thickness, these fibers effectively inhibited pancreatic cancer cell proliferation and promoted apoptosis in both in vitro and in vivo models. Notably, GEM-loaded electrospun fibers outperformed systemic GEM administration by enhancing apoptosis rates and reducing liver toxicity. 67 Electrospun fibrous materials also show promise for delivering complex chemotherapies. Coaxial electrospinning has been employed to encapsulate 5-Fluorouracil and methotrexate within PCL fibers, achieving effective drug integration. However, due to PCL’s slow degradation, drug release was limited to 20% over 4 days. Despite this limitation, coaxial electrospinning remains a promising strategy for delivering combination therapies like FOLFIRINOX, commonly used in pancreatic cancer treatment. 68 In contrast, our HA-coated PCL carriers offer a more balanced degradation profile and sustained PTX release. Unlike previous studies, where slow-degrading PCL matrices resulted in limited drug release, our carrier ensures a more consistent and prolonged PTX release, improving therapeutic efficacy. Furthermore, while needle-based electrospinning has been widely used, it presents scalability and reproducibility challenges. Our approach, utilizing needle-less electrospinning technology, simplifies production, enhances adaptability, and improves scalability for potential industrial production. By integrating HA coating with electrospun PCL carriers for PTX delivery, our system provides a more efficient, scalable, and localized therapeutic strategy for pancreatic cancer treatment. Compared to other drug delivery approaches, such as GEM-loaded fibers or coaxial electrospun PCL fibers, our method addresses both drug release kinetics and industrial-scale production challenges.
Discussion
This study demonstrates the potential of HA-coated electrospun PCL carrier as a novel, scalable platform for localized PTX delivery in pancreatic cancer treatment. The integration of HA into the electrospun carrier allows for controlled, and more sustained drug release, addressing the rapid clearance challenges typically encountered with hydrophobic fibrous materials. The findings indicate improved drug delivery efficiency and prolonged cytotoxic effects, as evidenced by sustained reductions in MiaPaCa cell viability. The PTX release profile from the HA-coated PCL carriers revealed a dual-release mechanism. Initial rapid release, governed by matrix erosion, was characterized by the Hixson-Crowell model (R2 = 0.85, k = 0.001), reflecting degradation of the HA coating. Following the initial rapid release, a sustained diffusion-driven release occurred, characterized by the Higuchi model (R2 = 0.74, k = 2.9), ensuring prolonged drug availability. This dual-release profile may help ensure prolonged therapeutic concentration at the tumor site. However, the initial rapid release phase suggests potential areas for optimization, particularly to mitigate premature drug loss and enhance therapeutic precision. In comparison to conventional HA-based nanoparticle systems, which are often limited by short residence times in the target tissue, the HA-coated electrospun PCL carriers provide a stable and efficient platform for localized chemotherapy. These carriers offer several key advantages, including a high surface area for drug loading, control over drug release kinetics, and the ability to function as localized patches that stay in close contact with the target tissue. Unlike injectable HA-based nanoparticles, which depend on circulation and are often cleared rapidly, our carriers can be directly applied to surgical sites, ensuring prolonged local drug retention and minimizing systemic exposure. This localized drug delivery system could also optimize drug diffusion, maintain a consistent concentration gradient, and help address critical challenges such as preventing local recurrence. Additionally, the needle-less electrospinning technology NanospiderTM used in this study offers significant advantages for industrial scalability and clinical translation. Unlike traditional needle-based methods, this system enables efficient production of uniform fibers on a larger scale, addressing bottlenecks in fiber morphology and coating uniformity. Optimization of process parameters, such as solution viscosity and voltage, further ensures production consistency and quality. Our carrier also holds promise for further optimization through nanoparticle loading, as demonstrated in our previous study, which combined PCL, SF/PCL and SF electrospun carriers with nanoparticles, enhancing vaccine penetration and localized delivery. 36 This approach could further improve therapeutic efficacy by leveraging the synergistic effects of nanoparticles and electrospun carriers to enhance drug delivery and overcome the tumor microenvironment’s challenges. Looking ahead, we plan to conduct comprehensive in vivo studies to further evaluate the pharmacokinetics, tumor penetration, and therapeutic efficacy of the HA-coated electrospun PCL carriers. These studies will help validate the carrier’s safety and effectiveness, particularly in real-time tumor growth inhibition and recurrence prevention. Advanced imaging techniques will be employed to monitor the distribution of the carriers and assess their ability to maintain consistent drug concentrations at the target site. These studies will bridge preclinical and clinical translation, reinforcing the feasibility of our scalable approach for pancreatic cancer therapy.
Conclusion
This study presents a scalable and innovative approach for localized PTX delivery in pancreatic cancer treatment using HA-coated electrospun PCL carriers. By addressing challenges such as rapid drug clearance and scalability, this system enhances drug delivery efficiency and cytotoxic efficacy, as evidenced by significant and sustained reductions in MiaPaCa cell viability. Compared to conventional HA-based nanoparticle systems, HA-coated PCL carriers enables stable, localized chemotherapy with prolonged tissue contact and minimized off-target effects. The integration of needle-less NanospiderTM technology underscores the industrial scalability and clinical potential of this platform, overcoming the productivity limitations of traditional needle-based electrospinning methods. This HA-coated electrospun carrier offers a promising complementary approach to systemic chemotherapy, potentially improving therapeutic outcomes while reducing adverse effects. Future in vivo studies will be crucial to validating its safety, pharmacokinetics, and therapeutic potential, paving the way for clinical application in pancreatic cancer treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by the Ministry of Education, Youth and Sports of the Czech Republic and the European Union - European Structural and Investment Funds in the frames of Operational Programme Research, Development and Education - project Hybrid Materials for Hierarchical> Structures (HyHi, Reg. No. CZ.02.1.01/0.0/0.0/16_019/0000843).



