Abstract
Antipathogenic fabrics coated with different types of metallic nanoparticles were developed for the use in healthcare sector. The citrus plant waste was collected and processed to extract the bioactive molecules for the green synthesis of metal particles. Here, the citrus extract was used for a dual purpose, as a bio reductant and also as a bio dispersant (D-limonene). Subsequently, the green synthesis of a highly concentrated and stable colloidal dispersion of Silver nanoparticles (Ag-NPs), Copper nanoparticles (Cu-NPs) and Zinc Oxide (ZnO-NPs) was carried out using the self-assembled respective salts and green extracted reducing agents without using any other hazardous chemicals. Furthermore, the effect of the loaded D-limonene as a dispersant was justified by PDI, Zeta potential, particle size analysis and Dynamic Light Scattering (DLS). The synthesized particles were assessed for their morphology and geometric characteristics by Scanning Electron Microscopy (SEM), revealing the formation of particles with spherical and oval shapes. The justification for the formation of particles was also analyzed by using Fourier transform infrared spectroscopy (FTIR) and X-ray diffraction (XRD) techniques. In the next step, the synthesized Ag-NPs, Cu-NPs and ZnO-NPs were applied to cotton fabric and their antipathogenic properties (antibacterial, antiviral, and antifungal) before and after severe washing were characterized. It was observed that the nanoparticles applied fabric at higher concentration (5g) exhibit even covering of fiber surface, average particles sizes between 350 and 470 nm, with excellent antimicrobial activity against Escherichia coli (3.4 ± 0.35 mm) and Staphylococcus aureus (5.5 ± 0.19 mm). The untreated fabric had a log CFU/ml value of 5.43, indicating no antibacterial effectiveness of the control sample. However, the log values of all the treated samples were significantly lower. Moreover, the intensity of concentration of zinc silver and copper particles explains the 88.48 %, 85.01 % and 94.23% antifungal activity respectively. While antiviral activity (84% reduction) was also highest against copper nanoparticles coated fabric. The level of significance against antipathogenic activities among all particles coated samples was analysed by applying the statistical analysis of simple linear regression with paired t test. In addition the comfort parameters (air and water vapor permeability) for developed medicated textiles were also analyzed.
Introduction
The hospital-acquired infections (HAIs) or nosocomial infections (NI), are diseases that develop in patients while undergoing hospitalization, and are a constant source of problems for hospital staff. The risk factor of these infections is on the rise despite efforts to control them. 1 The cost of treating or managing these infections is high. People with these illnesses may become disabled or die.2,3 The bacteria and viruses found in the contaminated material could be the main cause of transmission of infection. Bed sheets, pillowcases, outlets, surgical drapes, covering, panels, curtains, patient gowns, doctor’s robes, surgical gowns and socks were among the most prevalent textile contributions in hospital locations (wards, operating rooms, rooms, ICUs, and surgical settings), as well as other items. 4 Pathogens thrive in textiles made of natural fibers because of the voids in the composition of the fabric, permeability, wetness and other natural materials. 5 To induce the antipathogenic properties in textiles, various kinds of agents, metal particles and treatments have been used. 6 Thus, the application of minute dimensional materials (having their sizes in accordance to the pathogens), may be applied over the fabric structure by using a variety of scientific methods (known as nano-science and nano-biotechnology). 7 Metal particles have been used as potential agents in a variety of healthcare fields including drug delivery, nanomedicine, and biotechnology applications. 8 The controlled amount of inorganic metallic nanoparticles presented as less toxic. 9 It is important to note that metals like Ag, Cu, and ZnO have broad antibacterial activity and are very often reported to show a harmful impact on mammals. Therefore, metal particles exhibit potential as an excellent antipathogenic mediator. Metal nanoparticles synthesized by artificial reducing agents typically pose a threat to human health or the environment. The metal particles made from synthetic sources were degraded and oxidized by various environmental factors. As a result, they are poisonous and pose a health risk. 10 Furthermore, there are evidences that engineered metal nanoparticles can allegedly cause adverse effects on humans, animals and plants as well as the environment. 11 Therefore, it is imperative that attention should be given to green antibacterial agents that are more hygienic, biodegradable, and affordable. 12 Moreover, green nanoparticles are said to have superior catalytic activity when compared to nanoparticles produced chemically. The green synthesis approach is much simple and easy, cost-effective, amenable to scale up, less toxic, and compatible with normal tissues for In vivo applications. A versatile range of organic agents have been used to extract the biomolecules, which are necessary to react with the metallic salt to produce the respective nanoparticles. Among them, the waste of citrus plant peels has a large variety of active phytochemicals, that act as bio reductants, bio dispersants and bio stabilizers. 13 Manal et al. 14 attempted to synthesize Ag-NPs using biological waste material from citrus lemon peels and characterized the produced green Ag-NPs for their antibacterial properties and cytotoxic effects. Spherical and irregularly agglomerated Ag-NPs development was reported according to a UV-visible spectrophotometer analysis of the synthesized Ag-NPs. The synthesized Ag-NPs had an average size of 59.74 nm. 14 Shahid et al. studied the effect of cuprous oxide nanoparticles coated cotton fabrics against various types of pathogens to deal with hospital-acquired infections. The Cu2O coated fabrics showed excellent antibacterial effects against E. coli and S. aureus. Moreover, various studies demonstrated the In vivo and In vitro effect of copper and silver nanoparticles against plant and human pathogenic fungus Aspergillus niger (A. niger)15,16. Usha et al. analyzed effectiveness of copper oxide nanoparticles coated fabrics against A. niger and exhibited about 100% reduction after 48 h of incubation. 17 Padma et al. attained the biosynthesis of silver nanoparticles by utilizing the lemon extract as a reducing agent. Subsequently, the silver particles were coated over cotton and silk fibers. The prepared bioactive fabrics showed strong antifungal activity. However, there are limited studies available for copper and zinc oxide nanoparticles coated textiles from citrus plants source.
The present study deals with the green synthesis of copper, silver and zinc oxide nanoparticles with a single source reducing agent (Citrus limetta). Also, the overall antipathogenic effect of copper, silver and zinc oxide nanoparticles coated textiles from citrus plants source has not been reported. 18 To fulfill the demand for a hygienic and green environment, the waste of orange peels was utilized. Moreover, the uniqueness of work was further reinforced by utilizing the citrus extracted bio dispersant (D-limonene) in combination with citrus peels extracted reducing agent to prepare the metal (Cu, Ag and ZnO) nanoparticles. Here, the citrus extract was used for a dual purpose, as a bio reductant and also as a bio dispersant (D-limonene). Antipathogenic fabrics coated with three different types of green synthesized metal particles (zinc oxide, copper and silver) were developed. The first step involved the green synthesis of different metal nanoparticles by using the obtained bio-reductant from citrus plants source. Subsequently, a mini-emulsion of each type of prepared nanoparticle was done using the dispersing agent D-limonene (obtained from citrus plants). In fact, the antibacterial properties of metal nanoparticle coated fabrics can be improved by using a dispersing agent. 19 A suitable natural dispersing agent plays a crucial role in stabilizing the nanoparticles and preventing their agglomeration. Hence, a dispersing agent was used to make sure the even distribution of metal nanoparticles over the fabric structure. The dispersant D-limonene (the major constituent of citrus) significantly inhibits the growth of Gram-negative and Gram-positive bacteria and exhibits inhibitory activity against fungi. 20 In the second step, the prepared nanoparticles were loaded on cotton fabrics. The chemical composition of citrus plant extracts and their role as bio-reductants in the synthesis of nanoparticles was also analyzed. The developed antipathogenic medical textiles were tested against various surface and functional parameters suited for the medicated textile. The antibacterial capabilities upon washing were also measured to check the durability of the coating. Moreover, comfort parameters (air and water vapor permeability) for developed medicated textiles were also analyzed. The passage of proper air and vapor through developed bandages indicates the breathability and permeability (of body fluid like perspiration, sweat and moisture) of developed samples. 8 The developed fabrics can be effectively used to fabricate bioactive health care fabrics used in bandages, surgical gowns, patient protective clothing, bedding textiles, coveralls, wound dressings, pillow covers and chair covers, etc.
Materials and methods
The cotton fabric generally used in hospital areas (dense weave and high GSM), was obtained from the weaving laboratory of the National Textile University, Faisalabad, Pakistan. The fabric was bleached, plane weaved, and had an area density of 150 g per square meter. The sodium hydroxide (NaOH), Zinc nitrate (Zn (NO3)2·6H2O), silver nitrate (AgNO3) and binder (ISYS MTX, binder) were provided by Sigma Aldrich. It is a type of organic/inorganic binder. The nanoparticles are attached to the fabric with pad-dry-cure method. The used binder does not diminish the antibacterial effectiveness of metal nanoparticles. While copper (II) chloride (CuCl2.2H2O) with 99% purity was acquired from Germany’s Riedel-de Haen. None of the compounds were further chemically treated or purified, they were all of analytical reagent grade. While the citrus extracted dispersing agent D-limonene (C10H16) more than 96.9 % purity, with general characteristics of M = 136.24 g/mol, can be used in a boiling point range of 170 °C–180°C, was purchased from Fluka, (Fisher Scientific, Leicestershire, UK).
Preparation of plant extract and phytochemical analysis
Experimental research and field studies on plants including the collection of plant material were carried out with relevant institutional, national, and international guidelines and legislation. The peels of Citrus limetta were properly washed and let to air dry as represented by Figure 1(a). Then, the dry peels were cut into flakes and chopped by using lab. scale pestle and mortar. Subsequently, 40 g of chopped peels were added in a round bottom flask followed by the addition of 100 mL of distilled water and refluxed at 80°C for 60 min. Then obtained extraction was heated at 80°C for 2 h and was left at room temperature to cool down. Afterward, the whole solution was filtered through Whatman filter paper to obtain the fine extract. The obtained extract i-e Figure 1(f) was then stored in the refrigerator at 4°C for further use. The phytochemical analysis of the obtained extract was also analyzed by using standard procedures as described previously.21,22 (a) Citrus limetta peels, (b) peels flakes, (c) chopped peels, (d) grinded peels, (e) filtrate, (f) reducing solution of orange extract (g) CuCl2.2H2O solution, (h) greenish black solution of copper nanoparticles, (i) calcinated obtained copper particles, (j) AgNO3 solution, (k) greenish grey solution of silver nanoparticles, (l) calcinated obtained silver nanoparticles, (m) ZnCl2.6H2O solution, (n) ZnCl2.6H2O solution after adding peel extract, (o) particles after calcination.
Green synthesis of Cu-NPs from Citrus limetta fruit peel extract
The copper chloride was used as a precursor salt for the green synthesis of copper nanoparticles. About 30 g/L of CuCl2.2H2O was dissolved in 100 mL of distilled water as shown in Figure 1(g). The solution was stirred at 60°C for 15 min followed by the slow addition (10 mL) of prepared extract into the solution. The color of the solution changed from blue to bluish orange after the addition of extract. Then it was stirred further for 30 min. The color of solution changes to greenish black, which confirmed the formation of copper nanoparticles (Figure 1(h)). Afterward, the solution was centrifuged at 8000 r/min for 10 min and placed in a furnace for calcination. The blackish-green colored copper particles were obtained after calcination (Figure 1(i)).
Green synthesis of Ag-NPs from Citrus limetta fruit peel extract
The silver nitrate (AgNO3) was used as a precursor salt for the green synthesis of silver nanoparticles. About 40 g/L of AgNO3 was dissolved in 100 mL of distilled water (Figure 1(j)). The solution was stirred at 60°C for 30 min followed by the slow addition of prepared extract (10 mL) into the solution. The color of the solution changed to brownish grey, which confirmed the formation of silver nanoparticles (Figure 1(k)). Then, the solution was centrifuged at 8000 r/min for 10 min. The obtained particles were then placed in a furnace for calcination. Greenish-grey colored silver nanoparticles were obtained after calcination as shown in Figure 1(l).
Green synthesis of Zn-NPs from Citrus limetta fruit peel extract
Zinc chloride (ZnCl2.6H2O) was used as a precursor. 15 g/L of ZnCl2.6H2O was dissolved in 100 mL of distilled water. The solution was stirred at 60°C for 15 min (Figure 1(m) followed by the slow addition (10 mL) of prepared extract into the solution. The solution or color altered from white to yellow after the addition of extract (Figure 1(n). The solution was stirred further for 15 min. The solution was then centrifuged for 10 min at 8000 r/min. The obtained particles were then placed in a furnace for calcination. Light brown-colored zinc particles were obtained after calcination (Figure 1(o)).
Application of prepared particles on cotton fabric
The application of particles was done on pure cellulosic cotton fabric by adopting the textile process of Pad-Dry-Cure. For this10 g of each prepared particle (Cu-NPs, Ag-NPs and Zn-NPs) were dissolved in 500 mL of distilled water with five different concentrations of dispersing agent (1g, 2g, 3g, 4g and 5g). Now, within every solution, 0.5 g of binder was dissolved and citric acid was used to keep the pH between five and 6. After stirring for 1h at room temperature (pre-emulsification), the mini-emulsion was prepared by ultrasonication for 15 min of pre-emulsion by sonication technique. The workflow of the dispersing process is described in Figure 2. Emulsion of particles by using citrus extract.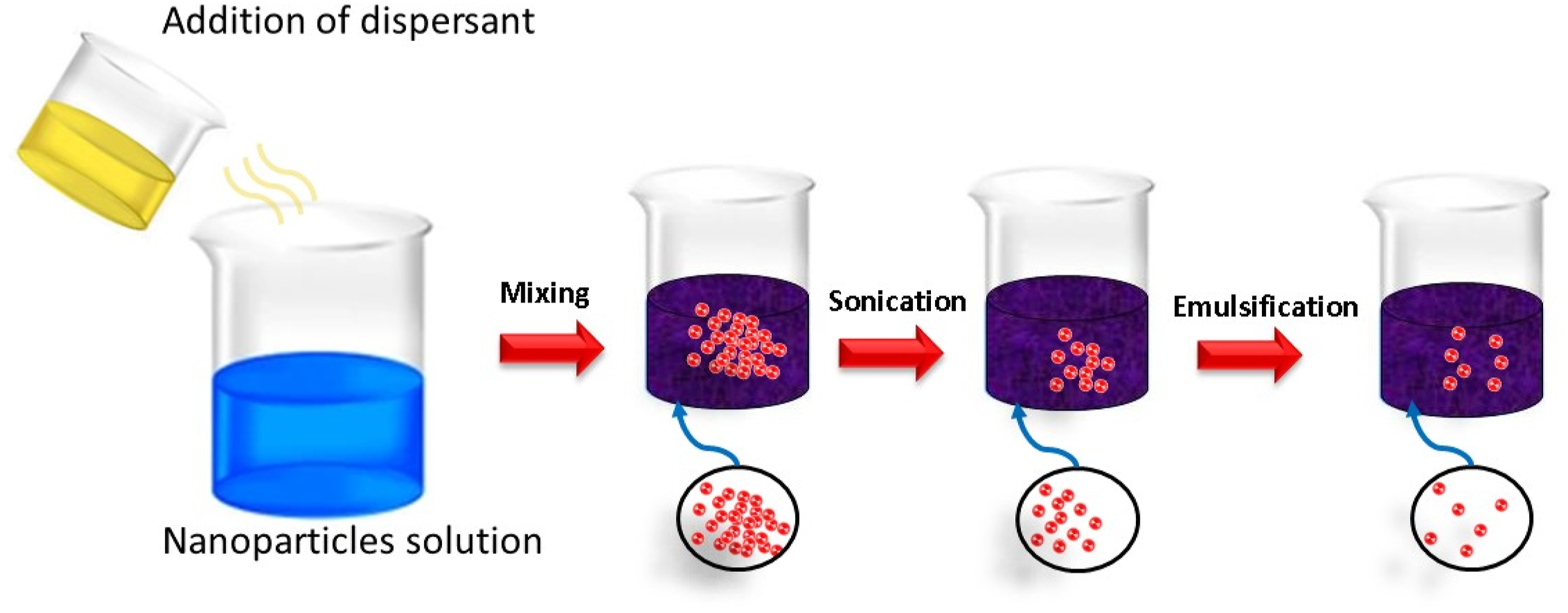
After being dipped within every solution for 30 min, the cotton cloth was pad-dried for 20 min at 90°C. For such procedures a 100% wet grab was achieved. The following procedure illustrates how prepared particles are applied over the structure of cotton fabric Figure 3. Several research works have given the preferences to pad-dry-cure method of applying particles over the fabric structure. The technique is relatively simple and low-cost and energy efficient technique.
23
The particles are attached over the fabric structure due to the combination of different forces and ionic attraction between the cotton anionic sites and cationic metallic particles.24,25 Secondly, the particles may attach over the fabric surface due to adsorption. In fact, the padding method provides uniform distribution, controlled application, compatibility with various fabrics. This coating technique is quite reliable in scaling up the methods for applying the particles over the fabric structure for industrial applications.
26
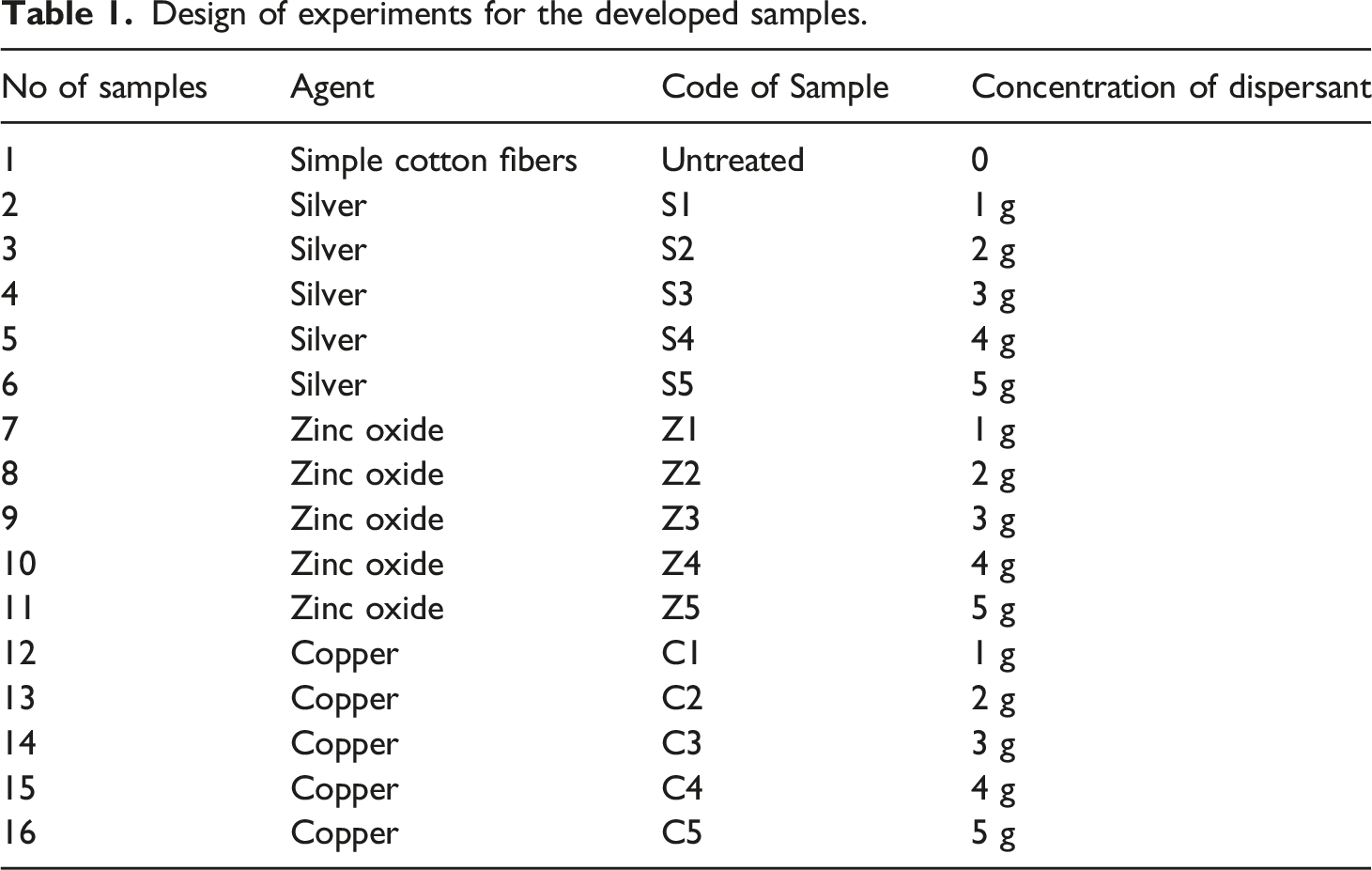
The experiment’s design for the developed samples is provided in Table 1. The process of coating the nanoparticles over the fabric structure. Design of experiments for the developed samples.
Characterization of the synthesized nanoparticles
Surface characterizations
DLS technology was used to estimate the dimension of the particles as synthesized. The particles were placed in a beaker and distilled water was used to make a diluted dispersion. The resultant solution was subjected to ultra-sonication for 20 min with an ultrasonic probe before testing. SEM was used to investigate the morphologies of samples of metal-coated fabric. The Tescan VEGA III SEM device was used to apply an accelerated voltage. The EDX analysis was used to calculate the elemental percentage by weight. The XRD study was performed using a diffractometer equipped with a conventional X-ray tube and Cu ka1 radiation (k 5 1.5406 A) voltage source (40 kV/30 mA). The X-ray diffraction patterns were measured in the 10–80 range with a 0.02 step size. The Percentage yield was calculated according to the formula given below
27
:
While the percentage of weight gain of particles coated textile was examined according to the following equations:

Antibacterial activity
The antibacterial activity was checked by the disc diffusion method. The bacterial strains of Gram-positive and Gram-negative bacteria were selected. Nutrient agar was used as a culture medium. The AATCC 147 standard method of inhibition test was used to assess the antibacterial activity of nanoparticles. For the antibacterial effectiveness of cotton fabrics coated with synthesized particles, both qualitative and quantitative measurement tests were applied.
Qualitative test (zone of inhibition measurement)
Determination zone of inhibition
The qualitative analysis was investigated against Gram-positive bacteria Staphylococcus aureus (CCM-3953) and Gram-negative bacteria Escherichia coli (CCM-3954). Always fresh bacterial suspensions were created by cultivating a single colony in a nutrient bath for the duration of the night at 37°C. The sample of turbidity was corrected to 0.1 optical density at (OD 600) before antibacterial testing agar plates were freshly prepared. Cells were evenly distributed on the agar plates after the cotton swab (which had been sanitized) was dipped in the culture mixture. These plates were used for antibacterial testing after being prepared.
Direct placement on inoculated agar plates was done with the coated cotton textiles (6 x 6 mm sqs) with particles. As a control test sample, cotton cloth that had not been treated was used. The samples and inoculated agar plates were then placed at 37°C for 24 h. Zone of inhibition was calculated as the entire diameter (mm) of the fabric coated with particles and the area where bacterial growth was inhibited. Each experiment was carried out in triplicate and mean values were computed.
Quantitative test (reduction factor)
The standard AATCC test (100-2004 procedure was used to conduct quantitative measurements). This quantitative approach describes the percentage decrease in inoculation bacterial concentration caused by the sample effect by using the reduction factor. It yields the number of surviving bacterial colonies (CFU) and from there the percentage of inhibition was computed. Consequently, a comparison of both treated and untreated samples is required (standardized). First, a sample that had been cut into 18 × 18 mm squares was placed in a sterile container for 30 minutes. Next, for each test, a specific bacterial strain (100 L) containing 105 CFU/ml was used. After 24 h of incubation at 37°C in a thermoset, the physiological solution was added. The results were determined using the total number of colonies across all three dishes.
Antifungal activity
The antifungal effect of all samples that had been treated was analyzed by the standard test method AATCC 100-2004 by a consistent way. In this experiment, A. niger, a type of fungus specie was used. Antifungal activity was estimated as a percentage change according to equation (1).
Antiviral activity
By using Behrens and Karber’s techniques the virus titer dropped from the initial viral concentration of infectivity (107) was calculated. The Vero-E6 cultures were kept maintained by using (DMEM) Dulbecco’s Modified Eagle Medium with 2% penicillin-streptomycin (PSA) and 9% fetal-bovine serum (FBS). Vero-E6 cultures were infected by the coronavirus. Gentle centrifugation was used to filter the supernatant for 30 min at 5 to 7°C temperature and 3700 r/min shaking speed. Vero-E6 cell lines were cultivated using standard test conditions (24 h at 37°C in 6% CO2) and stored in 96-well plates at a concentration of 2 x 105 to assess viral content. The coronavirus titer in cultivated cell lines was estimated by using the Behrens and Kerber’s techniques. Then by taking the fabric sample of size 20 mm and 20 mm this fabric sample were filled with the fabric sample vials. Through the test sample 100 μL of infection rate was passed, and any extractable viral loads in channels were removed using the filter. 101–108 repetitions were used to dilute infectious coronavirus. After having injections of each serial dilution the Vero-E6 cell cultures were grown under optimum environment at 37°C with 6% CO2 for 3 days. The Behrens and Karber method was used to assess the coronavirus titers in cultured cell lines. 28
Air permeability
Air permeability refers to the velocity of air flowing perpendicularly through a specific location under a particular air pressure differential between two surfaces of an object. The air permeability tests were conducted using an SDL air permeability tester, following the guidelines specified in ISO-9237. The air pressure differential between the two surfaces of the object was set at 100 Pa.
Water vapor permeability
The measurement of moisture (vapour) management parameters such as RWVP (%) and Ret [m2 Pa/W] was performed using the Permetest. This device is designed to replicate wet and dry human skin conditions in terms of thermal sensation and is commonly used to determine water vapour and fabric thermal resistance. The results obtained were expressed in units as specified in the ISO Standard 11,092.
Washing durability
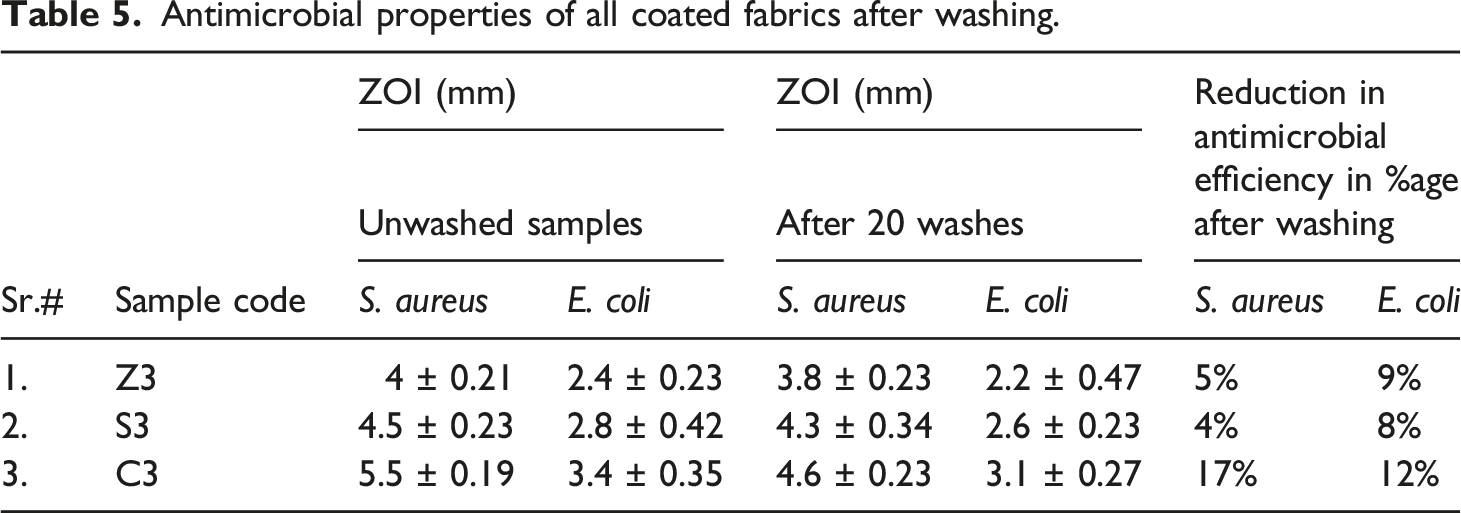
The study aimed to evaluate the washing durability of particles coated fabrics to determine their activity during use. To test the washing durability, the particles coated fabrics were mixed with a standard detergent in a 50:1 liquor ratio and subjected to washing at 40°C for 30 min while being stirred at 800 r/min. The washing test was conducted in accordance with ISO 105-C01. Before testing, the samples were dried and conditioned for 24 h in a normal environment with 65% humidity and 25°C temperature. The efficacy of particles on the fabric surface was confirmed by conducting antimicrobial activity before and after washing the samples.
Results and discussion
Phytochemicals screening analysis and percentage yield of each prepared nanoparticles
Phytochemicals present in peels of Citrus limetta. 29

Note: Present +, Absence -.
The obtained yield of each prepared nanoparticle was calculated by theoretical method. Where the initial salts of CuCl2.2H2O (30 g/L), AgNO3 (40 g/L) and ZnCl2.6H2O (15 g/L) were reduced by 10 g/L of reducing agent. The amount of particles obtained against CuCl2.2H2O (30 g/L), AgNO3 (40 g/L) and ZnCl2.6H2O (15 g/L) were 5g of copper, 9g of silver and 3g of Zinc oxide. Hence, their respective yields were calculated about 16.6% of Cu-NPs, 22.5% of Ag-NPs and 20% of ZnO-NPs respectively. So, the quick synthesis with maximum yield and stability was obtained with peels extracted reducing agent and dispersants.30,31
Moreover, the weight gain percentage of the copper, silver and zinc oxide nanoparticles coated cotton fabric was investigated. The effect of different applied concentrations (1 g, 2 g, 3 g, 4 g and 5 g) against weight gain of fabric was measured. It is clear from Figure 4 that the weight of the cotton fabric was increased in all three cases with an increase in concentrations of dispersing agents during the application of particles over the fabric structure. It was also observed that the trend of weight gain percentage against each concentration of copper, silver and zinc oxide was almost the same. However, the weight gain percentage in case of silver nanoparticles coated fabric was slightly higher as compared to copper and zinc. The same effect of higher weight gain percentage as compared to silver was noticed in a published work. There are multiple reasons for gaining more weight of fabric achieved by nanoparticles such as type and distribution of particles, fabric type and concentration. However, one main reason may be due the difference in actual densities of copper, silver and zinc oxide.
32
The percentage add on of nanoparticles to textile.
Particle size distribution, PDI and zeta potential
For determining the size of particles of Ag, Cu and ZnO, dynamic light scattering (DLS) was used which was based on Brownian motion of particles. The distribution of size of the particles is illustrated in Figure 5(a) and (b) which indicate a mono-modal size distribution ranging in nanometers. The particle sizes of Ag, Cu and ZnO were found to be 358 nm, 456 nm, and 470 nm, at a zeta potential of 35 mV, 30 mV and 35 mV respectively. This ensured that the particles were evenly distributed throughout the suspension and had a high negative potential from the nanometer to the micro range. The stability of particles is well analyzed by zeta potential, while the particle size distribution in nanoscience is more articulate with polydispersity index (PDI) values. The PDI of silver and copper nanoparticles was noted at about 0.312 and 0.258 respectively. The values show that the synthesized particles are highly polydisperse.
33
The particle size distribution of (a) Cu-NPs, (b) Ag-NPs and (c) Zn-NPs.
The observed change in particle size significantly depends on the concentration of dispersant (g) that is applied to the particles. As it is clear from Figure 6(b), the zinc oxide particles showed a remarkable increase in size when a certain quantity of dispersant was added. The particle size increased to 145 nm, 269 nm, 398 nm, 413 nm and 456 nm with dispersant concentrations of 1g, 2g, 3g, 4g and 5g respectively. A higher concentration of dispersant resulted in bigger nanoparticles. In the case of copper nanoparticles, their particle size also increased gradually with the increase in the concentration of applied dispersant as shown in Figure 6(d). Their respective graph represents a linear increase in particle size 178 nm, 207 nm, 305 nm, 456 nm and 470 nm with an increase in the dispersant concentration from 1g to 5g. However, there was a slight difference between the size of particles with dispersant concentrations of 4g and 5g. Similarly, the particle size of silver nanoparticles was also increased with the addition of dispersant. The graph in Figure 6(f) shows that the size of silver increased as 103 nm, 125 nm, 259 nm, 302 nm and 358 nm with the increase in dispersant quantity (1g – 5g). At different concentrations of dispersant (a) zeta potential and PDI of ZnO-NPs, (b) particle sizes of ZnO, (c) zeta potential and PDI of Cu-NPs, (d) particle sizes of Cu-NPs, (e) zeta potential and PDI of Ag-NPs, (f) particle sizes of Ag-NPs.
The graphs in Figure 6(a, c, e) represent the zeta potential and polydispersity index (PDI) of zinc oxide, copper and silver nanoparticles against each concentration of dispersant. Generally, the zeta potential indicates the stability or instability of particles within the solution. Zeta potential value of ±10 - 20 mV, ±20 - 30 mV and > ± 30 mV shows relatively, moderately and highly stable colloids respectively. 34 Figure 6(a) shows that the ZP of zinc oxide nanoparticles reached maximum stability (91 mV) with a dispersant concentration of 3g. When the concentration of dispersant was further increased to 4g and 5g, their ZP values began to decrease. While in the case of silver and copper nanoparticles, the highest stable values of zeta potential were recorded at the concentration of 4g of dispersing agent. On the other hand, a linear increase in the polydispersity index of zinc oxide particles was noticed. The maximum PDI value recorded was observed about 0.37. The nanoparticles with PDI value <0.1 are considered as highly monodisperse, while PDI value >0.4 and that of between 0.1 and 0.4 are indicated as highly polydisperse and moderately dispersed. The ZP of copper particles increased with the increase in dispersant concentration and it showed maximum value (87 mV) after the incorporation of 4 g of dispersant. At this stage, the particles of copper showed maximum stability and started to destabilize with more dispersant as indicated by the graph in Figure 6(c). The PDI value recorded at 5g of dispersant reached 0.34, which is quite an acceptable range under polydispersity. In the third graph, Figure 6(e) in the case of silver nanoparticles, the ZP of particles is easily reached above 40 mV, by use of only 1 g of dispersant. This means that the silver particles are already very close to the range of stability. With more dispersant, the zeta potential value increases and reaches its maximum i-e 89 mV at 4g dispersant concentration. Here the PDI value is also under good limit and indicates a linear pattern. Among these particles, silver nanoparticles are highly stable and that of zinc oxide and copper nanoparticles. The reason can be attributed by the fact of least oxidation state and less nucleation of silver ions as compared to copper and zinc. The statement can be further reinforced by a previous study conducted by Azam et al., 24 in which a comparative analysis between silver and copper nanoparticles was studied. The sizes of copper particles were big and had more clusters as compared to silver. Hence more nucleation means more agglomeration in structures and produces big-sized particles, which in turn causes less stability.
Mechanism of reductants and dispersants against metal salts
Figure 7 represents how biomolecules reduce the silver, zinc and copper ions to atoms for the synthesis of their respective nanoparticles. Amin et al.
35
explain in a study that different factors have an impact on the formation of silver, zinc and copper nanoparticles. These factors include plant source and the kind of organic compound in its extract. Such as in the phytochemical investigation of citrus plants extract, researchers found various acids including phenolic acids including p-coumaric acid and caffeic acid. Such biomolecules have carbonyl and hydroxyl group which may inactivate ions by chelating after binding with metals. Generally, this chelating property of phenolic compounds is majorly based on high nucleophilic ability of aromatic rings and not on the specific chelating groups that are present in the molecule. In caffeic acid, the reduction of silver ions in order to convert into silver nanoparticles is due to the presence of active hydrogen. In another study, Edison and Sethuraman
36
reported that the most important biomolecules found in fruit i-e Terminalia chebula are hydrolyzable tannins polyphenols, gallic acid and chebulagic acid. The exact reaction mechanism of reduction of Ag+ can include the formation of intermediate complexes with phenolic groups (–OH) that are usually found in hydrolysable tannins, that further undergo oxidation to quinone forms, followed by a reduction of silver ion Ag+ to silver metal Ag0 as depicted in Figure 7(a). A similar reaction mechanism occurs for the formation of zinc and copper nanoparticles shown in Figure 7(b, c). It is studied by Ahmad et al.
37
that luteolin is particularly a common flavone which is present in the upper aerial parts of the basil plants. The production of enol-form of the luteolin, releases reactive hydrogen, which is the main driving force behind the conversion of Ag+ into Ag0 and the ultimate formation of NPs. After that, the biomolecules reduce the silver ion into metallic silver as starting phase, and two other mechanisms are also seen for the synthesis of silver NPs. These two phenomena are referred as nucleation and growth. In the nucleation phase, particles of the metal plates have the ability to shift toward other metal particles, producing various agglomerations which generate clusters.
38
Moreover, these clusters further form tiny silver NPs. After that, the second phase (growth phase) starts where these tiny NPs spontaneously coalesce to transform into much bigger particles. Illustration for the mechanism of reduction through biomolecules against (a) silver, (b) zinc and (c) copper ions to atoms for the synthesis of their respective nanoparticles.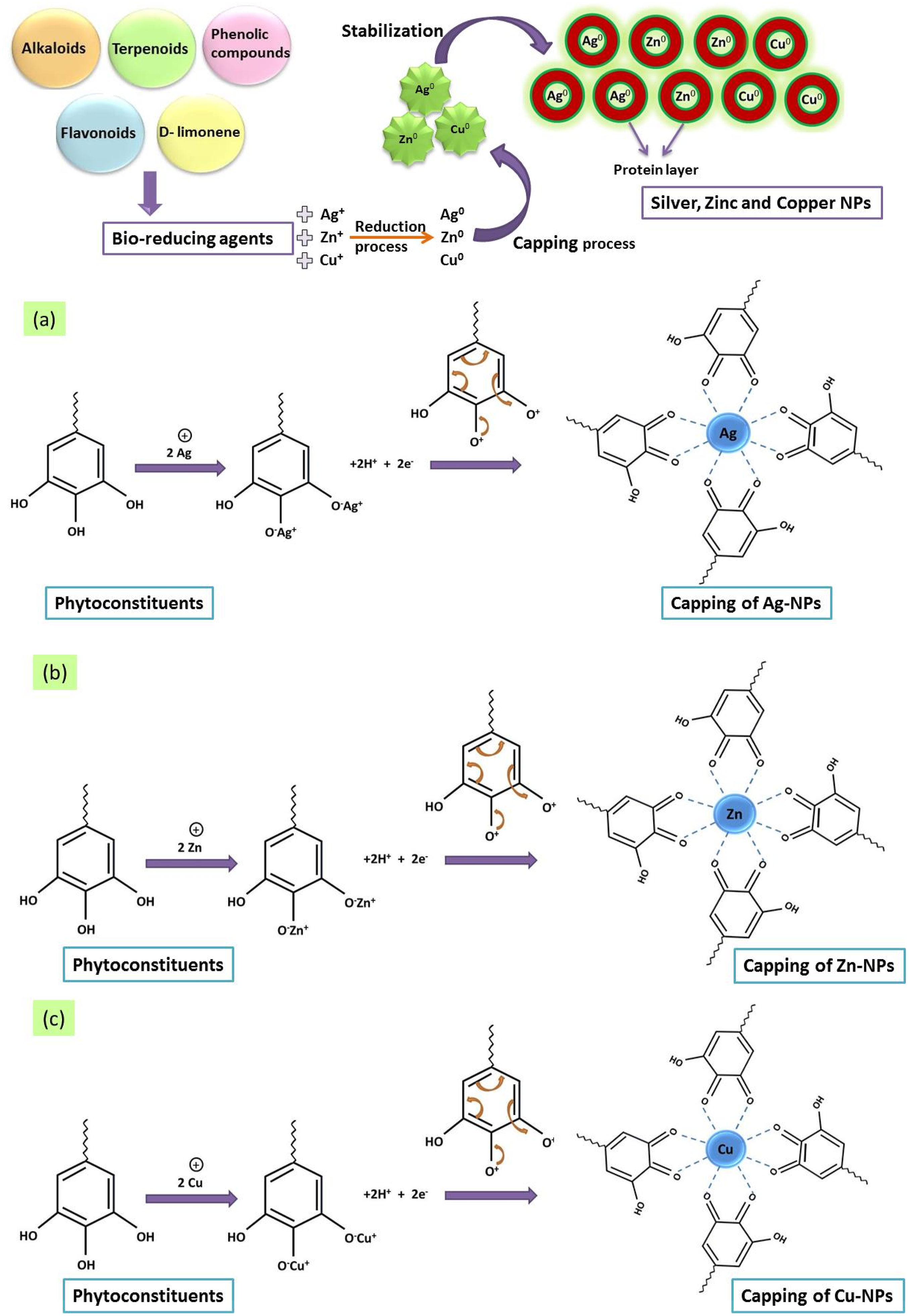
Surface characterizations
SEM analysis was conducted to examine the surface morphology of the samples including simple cotton fibers with and without treatment. Figure 8(a) illustrates the SEM analysis of the untreated cotton fibers. The SEM images of the fibers treated with zinc oxide (ZnO), silver (Ag), and copper (Cu) nanoparticles are presented in Figure 8(b), 8(c) and (d), respectively. These images demonstrate the formation of ZnO-NPs, Ag-NPs, and Cu-NPs at the nanoscale on the surface of the fibers. The deposition of nanoparticles appeared to be highly regular and intense. The structure of the zinc oxide nanoparticles indicated that they were synthesized at the nanometric scale, with rough surfaces, random clusters, and almost cylindrical shapes with tiny agglomerations. As a result, the microparticles were uniformly dispersed across the fabric surface. It should be noted that agglomeration was not observed in the majority of the prepared composites. SEM analysis of (a) untreated cotton, (b) Zinc oxide particles coated, (c) silver particles coated and (d) copper particles coated cotton fibers.
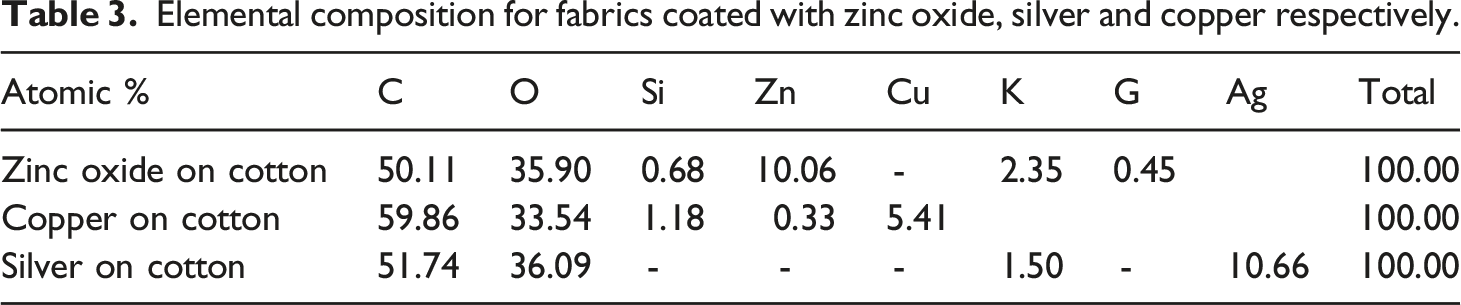
Elemental composition for fabrics coated with zinc oxide, silver and copper respectively.

(a)-(c) shows the TEM analysis of zinc oxide, copper and silver nanoparticles, (d-f) EDX spectra of zinc oxide, copper and silver nanoparticles.
Moreover, the TEM analysis was also performed to better clarify the sizes and morphologies of nanoparticles as shown in Figure 9 (a) to (c). The TEM analysis estimates the sizes of copper, silver and zinc oxide particles between 400 and 500 nm and reports the morphologies of silver and copper nearly spherical. However, TEM images shows the ZnO particles having hexagonal, a little bit in an oval plate and flatted morphology with different dimensions. Edges of the particles are not smooth and the existence of pores on the ZnO plates can also be seen. This is due to the evolution of gaseous products during the decomposition of zinc salt. The SEM and TEM analyses also prove the morphologies with the highly crystalline nature of all nano morphologies. As aforementioned, SEM analysis shows the nanoclusters of particles, ranging from 300 nm to 500 nm. During TEM analysis, it seems that the separate particles of copper, silver and zinc oxide are broken parts from their respective clusters. 39
X-ray diffraction analysis
XRD analysis was conducted in the range of 20-80° with a step degree of 0.02° to determine the phase composition of Ag, Cu, and ZnO nanoparticles deposition. The phase purity of manufactured Ag particles was confirmed by exact indexing of all the peak intensities to the silver structure, as shown in Figure 10(a). The four peaks for Ag nanoparticles appeared at 2θ values of 77.5, 64.5, 44.3, and 38.1, respectively, compared to cotton fabric that had not been treated. These peaks were attributed to cubic-shaped diffraction planes (3 1 1), (2 2 0), (2 0 0), and (1 1 1), according to data from the International Diffraction Centre (data number JCPDS 04-0783 card).
20
No significant peaks were observed for other impurities, such as silver oxide. XRD analysis (a) silver-coated cotton, (b) copper-coated cotton and (c) zinc oxide-coated cotton.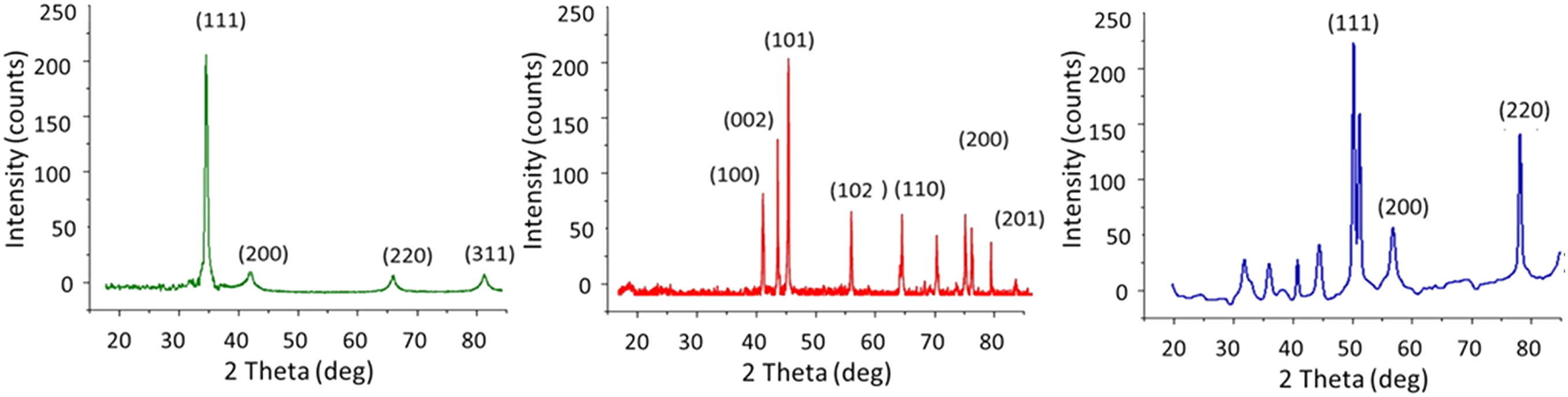
Figure 10(b) represents the XRD spectrum of Cu nano particles. A precise identification of every diffraction peak to the copper structure reveals the elemental composition of Cu particles. Cu diffraction planes (2 2 0), (2 0 0), and (1 1 1) are characterized by the occurrence of Cu diffraction pattern (2θ) at 74.2, 59.5, and 43.3°. From either the presence of peak position, the copper particle’s crystalline structure was investigated. Because no distinct impurity peaks were found, other than the development of the Cu2O peak (2θ) at 38°, the widening of the peaks instead indicated the synthesis of Cu particles at the nano range, respectively. 27
Figure 10(c) also displays the results of synthesized zinc oxide nanoparticles by X-ray diffraction pattern. The formation of zinc oxide nanoparticles is confirmed by the peaks of the X-ray diffraction peaks at 31.8°, 34.46°, 36.34°, 47.6°, 56.64°, 62.88°, 68.12°, and 72.6°, which correspond to the (100), (002), (101), (102), (110), (103), (200), (112), and (201) plane.
FTIR analysis
The extract of orange peels was used as a bio-reductant to synthesize the nanoparticles of copper and silver. Therefore, FTIR spectroscopy was employed to confirm this reduction process. An analysis of the FTIR spectroscopy analyzed the presence of functional groups on green synthesized silver and copper particles. The FTIR spectrum of orange peel, zinc oxide, silver and copper particles on cotton are plotted below in the form of a graph as shown in Figure 11. According to it, different regions/peaks (wavelengths) were taken to interpret which functional group is present at that specific wavenumber. These wavenumbers were in the range of 597 to 3350 cm−1. The peak at 1059 cm−1 wavenumber indicates the presence of alcohols or esters (C–O–H or C–O–R). The absorption peaks around 2950 cm−1, 3331 cm−1, 2115 cm−1, 1636 cm−1, and 597 cm−1 were recorded. On 2950 there is C–H stretching vibration absorption peaks in cellulose. While, the broad absorption band on 3670 cm−1 corresponds to the O-H stretching frequency, whereas at 1636 cm−1 depicts the C = O stretching of the carbonyl group. The peak of 1059 cm−1 was noted due to the link of alcohols or esters (C–O–H or C–O–R). The FT-IR spectra of orange peels extract, copper particles and silver particles coated fabrics.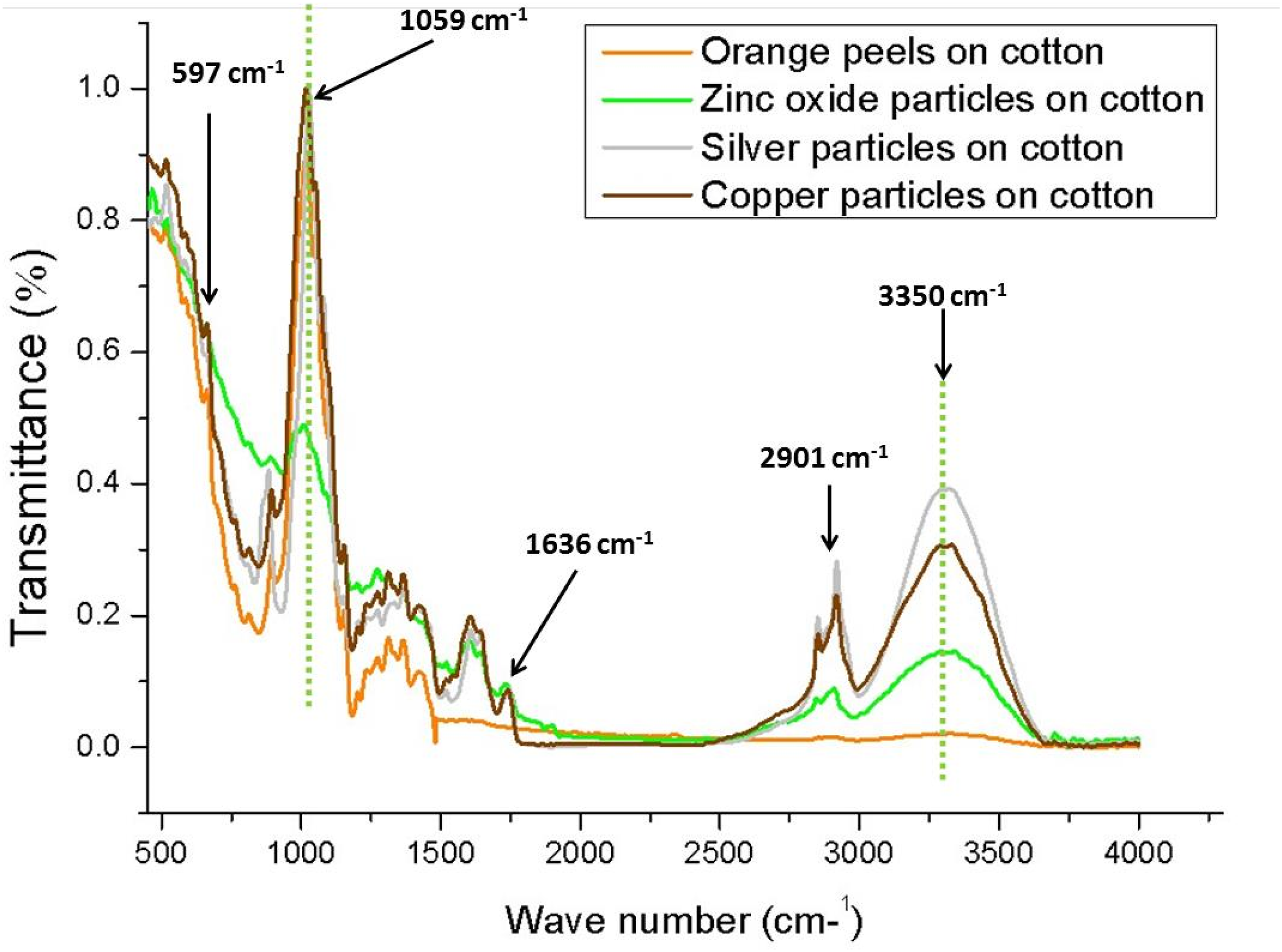
Antibacterial activity of the particles coated fabrics
The coated fabrics undergo qualitative and quantitative testing to determine their antibacterial efficiency.
Reduction factor (quantitative test)
The antibacterial activity of the samples was measured using the AATCC-100 technique, which quantitatively measures bacterial resistance to S. aureus and E. coli strains. The results are shown in Figure 12, which displays the reduction in log CFU/ml of the bacterial cultures on the treated and untreated fabrics. The untreated fabric had a log CFU/ml value of 5.43, which increased slightly to 5.44, indicating that the control sample was not effective against the tested microorganisms. However, the log values of all the treated samples were significantly lower, and the reduction in log values increased as the concentrations of ZnO, Cu and Ag nanoparticles on the fabric increased. The highest reduction was observed in samples with higher concentrations of ZnO, Cu and Ag NPs, where the log value decreased from 5.43 to 0, indicating a 99.99% reduction in the bacterial colonies of both S. aureus and E. coli. (a) Antibacterial activity in terms of log CFU/ml (b) Percentage reduction of fabrics treated with ZnO-NPs, Ag-NPs, Cu-NPs and untreated cotton fabric.
The growth of bacterial concentrations for treated (Z3, C3, and S3) and untreated (cotton fabric) samples is shown in Figure 13, which supports the aforementioned discussion. When compared to fabrics coated with zinc oxide, copper and silver nanoparticles particles, the untreated sample was found to be ineffective versus bacterial growth. Moreover, there was a noticeable improvement in colony reductions with a more than 99% efficacy for both species of bacteria at higher concentrations of each particle. Images of concentration of bacterial growth for the (a) untreated (pristine) cotton fabric, (b) for ZnO particles, (c) for Cu particles and (d) for silver particles.
Zone of inhibition test (qualitative measurements)
The ZOI values of ZnO, Ag and Cu nanoparticles coated fabrics.

As shown in given pictorial view in same table, all fabric samples exhibited clear inhibitory zones after being incubated for 24 h at 37°C in the dark. The fabrics treated with copper-coated nanoparticles (green synthesis) showed the strongest antimicrobial zones against both S. aureus and E. coli strains, while fabrics treated with ZnO nanoparticles had a smaller zone of inhibition. Each sample was tested three times and the average values were calculated.
The results indicated that the free-standing nature of the coated copper and silver particles led to significant disinfection of both bacterial strains, with S. aureus showing greater sensitivity than E. coli. For example, the area of inhibition for E. coli increased from 2.5 mm to 3.4 mm with copper, silver, and zinc particles, respectively, while the area of inhibition for S. aureus increased from 4 mm to 5.5 mm as shown in Figure 14(a) to (c). It should be noted that the annulus of the inhibition zone widened as the nanoparticle concentration increased, indicating that an increased nanoparticle concentration had a similar effect on the antimicrobial properties as reported in an earlier study.
8
Extent of concentration of nano particles categorized to (a) Zinc oxide, (b) silver and (c) copper influence the intensity of inhibition and obtained statistical analysis.
The particles of copper possess good antibacterial effects because the ionic activity associated with these copper ions (Cu2+) is greater (it is also less stable in transition state) as compared to silver or zinc ions. Therefore, this greater ionic activity helps copper to disrupt the membranes of bacterial cell and affect cellular processes, which increases antibacterial activity. Moreover, copper nanoparticles have higher surface area and unique structural morphology which enables them to affect many cellular components by different mechanisms and exhibit significant antibacterial activity.40,41 Also, the copper nanoparticles can easily dissolve in solutions to release more metal ions and indicate a higher antimicrobial effect with multiple antibacterial mechanisms at the same time. 42
The antimicrobial effect of coated fabrics is attributed to the combined physical and chemical action of bacteria with particles. The nanoparticles are taken up by cells through endocytosis processes. As the nanoparticles degrade, ionic species are released inside the cells, leading to an increase in ion absorption by the cells. 21 This causes an increase in intracellular concentration, resulting in more severe oxidative stress. Figure 14(a) shows the effect of the concentration of zinc oxide NPs on S. aureus and E. coli antibacterial inhibition properties. As the intensity of the Zinc oxide NPs increases the inhibition of S. aureus bacteria also increase and the same for the E. coli. The statistical analysis was performed to estimate the significance of the concentration on the inhibition efficacy of the S. aureus and E. coli bacteria’s suing simple linear regression analysis at 95% significant level (p < 0.05) where p (T<=t) two-tail value is p = 0.019. The Figure 14 shows that the amount of the increase of concentration of zinc-NPs, which explains the 97.96 % proficiency of the inhibition of S. aureus bacteria, while zinc particles explains the 100% proficiency of the E. coli bacteria. While the statistical analysis of Ag-NPs estimates the significance of the concentration on the inhibition efficacy of the S. aureus and E. coli bacteria’s suing simple linear regression analysis at 95% significant level (p < 0.05) where p (T<=t) two-tail value is p = 0.0181, which explains the 100 % proficiency of the inhibition for both type of bacteria (S. aureus and E. coli). In the same way the performed statistical analysis on Cu-NPs estimate the significance of the concentration on the inhibition efficacy of the S. aureus and E. coli bacteria’s suing simple linear regression analysis at 95% significant level (p < 0.05) where p (T<=t) two-tail value is p = 0.0996. The figure shows that the amount of the increase of concentration of copper NPs. Also it explains the 96.43 % proficiency of the inhibition of S. aureus bacteria while copper particles explains the 93.56% proficiency of the E. coli bacteria. 43
Antifungal activity of treated samples
The study utilized the AATCC-100 technique to assess the antifungal activity of uncoated and NPs coated fabric samples against A. niger fungi. Results are presented in Figure 15, where a percentage reduction in fungal spore germination was shown for all specimens. Both treated and untreated fabric samples demonstrated antifungal activity against the tested microorganism. However, it was found that particles-coated fabrics had better antifungal efficiency than untreated samples. Among all particles-coated samples, copper-coated fabrics showed the highest suppression of fungal growth. The statistical analysis was performed using simple liner regression analysis. The analysis portrays that the intensity of concentration of zinc particles explains the 88.48 % antifungal activity, while silver particles explains 85.01 % antifungal activity and copper particle shows 94.23% antifungal activity which is highest than rest of two particles performance. Reduction percentage of antifungal activity.
Antiviral effectiveness
Behrens and Karber’s method was utilized to calculate the decrease in viral titer from the initial viral titer of infectivity (108) for coronavirus. The virus infectivity titer log was demonstrated in Figure 16 for both 0 h and 60 min. All samples treated with copper exhibited greater antiviral activity, indicating that coating with particles has significant antiviral activity. The viral infectivity titer was significantly reduced in all specimens. The highest reduction in antiviral activity was observed in sample C3 (among all copper-coated samples), with a maximum reduction of approximately 84%. The lack of antiviral activity against the virus in untreated fabric indicated that the use of nanoparticles was responsible for the antiviral activity in all prepared materials. The interaction of particles with glycoproteins on the viral surface could be a source of the inhibitory effect on viruses and the antiviral activity of particle-treated fabrics.
11
(a) Reduction in viral infectivity titer (b) percentage reduction calculated from viral infectivity at a contact time of 0 and 60 min.
Washing durability of coated fabrics
Antimicrobial properties of all coated fabrics after washing.
Comfort properties
Effect of air permeability
The air permeability of bandages is an important factor that affects their comfortability. The exchange of air is facilitated by the production of body heat and perspiration. The results of air permeability for fabric specimens are presented in Figure 17(a). The findings indicate that incorporating extremely small nanoparticles into fabrics has a negligible effect on their air permeability. The air permeability of untreated fabric is approximately 876 mm/s, while the air permeabilities of particles coated fabric samples S3, Z3 and C3 were 834 mm/s, 808 mm/s, and 821 mm/s, the values are slightly reduced. Nanoparticle deposition only slightly reduces air permeability. There are two primary reasons for this phenomenon. First, repeated dips in solutions may cause relaxation shrinkage which could push the threads together and obstruct airflow. Second, during the application process, nanoparticles were deposited on the yarn framework and interstitial spaces, resulting in a reduction in the size of the cloth pores. The air permeability of a fabric is related to the size of its pores. As a result of the smaller pore size, the permeability of the fabric has decreased. However, the air permeability of the cotton bandages designed in this study was not affected. (a) Air permeability through particle-coated fabrics (b) Relative water vapor permeability through particle-coated fabrics.
Effect of water vapor permeability
The comfort of bandages is also affected by their water vapor permeability (WVP), which refers to the ability of the body to dissipate vapor from wound exudates. Figure 17 (b) shows that the average WVP of cotton bandages has not significantly changed. The untreated fabric had a relative WVP of 91%, while the samples coated with nanoparticles (S3), (C3), and (Z3) at various concentrations had relative WVPs of 88, 86, and 83 mm/s, respectively. There is a significant reduction in the relative WVP with nanoparticle deposition due to the relaxation of fabric chains from repeated dipping in the solution, bringing them closer together, and nanoparticle coverage in the fiber interspaces. The nanoparticle network on the surface partially covers the pores of the fabric, reducing porosity and minimally affecting vapor permeability. The textile’s air and water vapor permeability, which are essential comfort factors, remain unaltered. The ability of bandages to allow adequate air and vapor passage through them indicates their permeability and breathability, making them useful in preventing skin itching, irritation, and maceration from sweat and other body fluids.
Conclusion
Heavy GSM-weaved fabrics which are commonly used in hospital areas were utilized for the incorporation of green synthesized nanoparticles to cope with Healthcare-associated infections. The range of metallic particles in normal parts per million (to avoid concerns about leaching which leads to toxicity) can be coated over the textile structures to achieve antipathogenic properties. In the current research, efforts were made to endeavor green and sustainable resources (orange peels) for the synthesis of nanoparticles which in turn minimize the effect of toxicity. The beauty of the work becomes more noticeable, when we also employ a dispersing agent extracted from citrus fruits, ensuring proper dispersion and uniform application across the fabric structures. The effect of different concentrations of dispersing agents provided mini-emulsions of particles in sizes ranging from 300 to 600 nm and avoided unnecessary agglomeration, the effect was also justified by zeta potential and PDI analysis. A total of 16 samples were prepared against three different categories of nanoparticles. Surface structure and metal presence were examined using FTIR, TE, EDS, XRD, Dynamic light scattering, and Scanning electron microscopy. Next, qualitative and quantitative measurements were used to test the antibacterial activity of the particles coated fabrics. A higher reduction percentage of reduction about 99.99%, and higher ZOI values were found in the C3 prototype (copper particles coated fabric) as compared to the silver and zinc oxide-coated substrates. Moreover, the uncoated fabric sample showed 0% in reduction and no ZOI values. During the quantitative analysis sample C3 demonstrated the greatest decrease in bacterial colonies that survived, with the CFU values for E. coli and S. aureus reaching 0 from 7.34. However, the antibacterial activity of all particles coated substrates was noticed more significant than antifungal activity and antiviral activities. Furthermore, testing the coating’s antibacterial abilities after washing demonstrated its durability. The antibacterial activity of all treated fabric samples was insignificantly affected by numerous washing cycles, indicating that the particles remained on the fabric. Additionally, comfort parameters (air permeability and water vapor permeability) for the developed bandages were analyzed. The air permeability of untreated fabric is approximately 876 mm/s, while the air permeabilities of particles coated fabric samples S3, Z3 and C3 were 834 mm/s, 808 mm/s, and 821 mm/s. The same in the case of relative WVP about of 91% was recorded against uncoated textiles, while the samples coated with nanoparticles showed the relative WVPs of in the range of maximum 83 mm/s. Hence, the insignificant reduction in the relative WVP with nanoparticles was due the small interruption on open structures of fabric interspaces. However, the fabrics can be a potential candidate to fabricate the wound healing dressing which needs to absorb a suitable amount of exudates and also ensure the passage of air. The developed fabrics can be utilized to create bioactive sportswear or active wear, bioactive compression garments, as well as winter gloves and compression bandages such as head, wrist, or knee bandages.
Footnotes
Acknowledgment
The authors are thankful to the Government College University Faisalabad, Pakistan for providing the research facilities of this PhD work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research work received no funding.
Data availability statement
All data included in this study are available upon request through contact with the corresponding author.
