Abstract
The increasing demand for durable, eco-friendly clothing has led to the use of nanomaterials in textiles. However, concerns arise about the impact of engineered nanomaterials (ENMs). Studies have explored nanoparticle release from textiles during usage and washing. However, the existing data is fragmented, and a comprehensive grasp is absent. A systematic review is required to consolidate existing evidence and offer a thorough analysis of nanoparticle release from nano-enhanced textiles in commercial use and washing. This review assess the research aims to concerning the release of nanomaterials from commercial textiles during usage and laundering. The process involved defining objectives, crafting a search strategy, utilizing databases, and applying inclusion/exclusion criteria. After deduplication, articles were screened for eligibility, covering nanoparticle characteristics and release mechanisms. Searches spanned Scopus, PubMed, and Web of Science, using keywords like “Nano,” “Nanoparticles,” etc. Original articles on nanomaterial release from conventional textiles were included. Out of 1549 articles identified, and met the criteria for inclusion in the study. The results reveal that textiles employing nanotechnology can indeed release a significant quantity of nanoparticles. The characteristics of these released particles, including their quantity and composition, are influenced by various factors such as nanoparticle structure, adhesive properties, fabric type, and environmental interactions. While there exist variations between laboratory simulations and real-world conditions, these findings underscore potential risks associated with nanoparticle release, underscoring the necessity for toxicological assessments and additional research into particle behavior, particularly emphasizing the functional aspects of fibers and the environmental impact following nanoparticle release after washing.
Introduction
The rapid development and widespread adoption of nanotechnology have led to the incorporation of engineered nanomaterials (ENMs) into various consumer products, including textiles. 1 Nanomaterials (NMs), characterized by their size ranging from 1 to 100 nanometers, exhibit unique physical and chemical properties compared to their bulk counterparts, making them desirable for a range of applications. 2 The textile industry has embraced nanotechnology for over three decades, utilizing nanoparticles either through their incorporation into fabrics or their strategic arrangement within textile fibers. 3 Nano-textiles offer numerous functional benefits, such as enhanced chemical resistance, mechanical strength, water repellency, anti-aging performance, antibacterial properties, self-cleaning capabilities, and UV protection.4,5 Presently, the textile industry is highly focused on exploring the potential utilization of metallic nanoparticles (MNPs) to enhance fiber production and achieve fabrics possessing novel or enhanced attributes. 6
However, concerns have been raised regarding the potential release of nanoparticles, particularly metal nanoparticles (MNPs), from these nano-textiles during their use and washing processes. 7 During routine use and care, such as washing, this nanoparticles may be released into various matrices, including air, sweat, and effluent water. 8 Research shows that the total amount of nanoparticles released from investigated textiles ranges from less than 1% to nearly 100%, depending on the content of nanoparticles contained within the textiles.7,9 Geranio et al. in 2009 and Lorenz et al. in 2012 discovered that the amount of silver released from silver-functionalized textiles varied from not detectable to approximately 45%.10,11 These amounts depend on abrasion, mechanical influence, irradiation, water, sweat, washing detergents or temperature variations and it leads to the release of these nanoparticles into the environment (either to the human body, air, water, soil).12,13
In terms of sustainability, nanomaterials such as MNPs present both prospects and hazards. On one side, MNPs have the potential to replace harmful chemical additives, such as toxic flame retardants, or enhance material resource efficiency, thereby reducing environmental impacts. For instance, they could improve coloration processes. Conversely, several entities have raised alarms regarding potential health and environmental risks associated with MNPs exposure, emphasizing the absence of adequate regulatory frameworks.14,15
Exposure pathways for consumers include inhalation, dermal contact, and potential ingestion.16,17 For example, metals have the capability to be released from textile items and can infiltrate through the skin, accessing the bloodstream, and gathering in various organs or tissues. Recent research has indicated the potential permeation of MNPs into the skin, raising concerns about human health.18,19 Apart from the potential human health hazards associated with metals and metal NPs, the presence of these particles in textiles also raises environmental concerns, as numerous studies have highlighted their discharge from fabrics during the washing process.6,20 As the production of nanomaterials of various kinds continue to rise, there is growing apprehension regarding their potential release into the environment and the subsequent impacts on human and ecosystem health. 21 Yet, there is scarce information accessible concerning the factual emission of engineered nanomaterials and release coefficients for all life cycle stages and environmental compartments. 22 So, the release of nanoparticles have raised questions regarding their impact on human health and the environment. Previous research has demonstrated that certain nanomaterials can induce adverse health effects and alter biological behavior at cellular, subcellular, and protein levels.23,24 It is therefore crucial to understand the mechanisms, quantities, and potential risks associated with nanoparticle release from commercial nano-textiles during their use and washing. While some studies have investigated the release and migration of nanoparticles from textiles, the existing literature remains scattered, and a comprehensive understanding of this phenomenon is lacking.
Presently, a significant knowledge gap exists regarding the risks associated with engineered nanomaterials. Many of these uncertainties are foundational and are likely to endure for an extended period. Much of this uncertainty stems from the high diversity of nanoparticles in terms of size, purity, crystalline structure, surface area, porosity, density, surface modification methods, functionalization techniques, and so forth, leading to the emergence of diverse and varied physical and chemical properties. 14 Consequently, encountering or absorbing these nanoparticles in the body and their environmental effects becomes one of the most challenging issues. On the other hand, the impact of each of these parameters on the release of nanoparticles, as well as other influential factors such as the type of fabric, the arrangement of nanoparticles on them, washing conditions, and many other effective parameters, further complicates these challenges.
Thus, a systematic review is needed to synthesize the available evidence and provide a comprehensive analysis of nanoparticle release from commercial nano-textiles during their use and washing. The objective of this systematic review is to critically examine the studies on nanoparticle release from commercial nano-textiles during their use and washing, with a focus on the mechanisms of release, types of nanoparticles involved, release quantities, and potential health and environmental implications. By consolidating the existing research, this review aims to contribute to our understanding of nanoparticle release from nano-textiles and identify research gaps that require further investigation.
Materials and methods
Study design
This systematic review followed a rigorous methodology to assess the available literature on the release of nanoparticles from commercial nano-textiles during their use and washing. Commercial nano-textiles are textiles that are sold in the market and marketed as having undergone nanotechnological treatments, such as nano anti-sweat clothes or other textiles advertised with nano-enhanced characteristics. The study design involved several key steps outlined below:
Identification of research objective
The primary objective of this review was to evaluate and analyze studies that investigated the release of nanoparticles from commercial nano-textiles during their use and washing. By synthesizing the existing research, the aim was to gain insights into the mechanisms, types of nanoparticles involved, release quantities, and potential health and environmental implications.
Search strategy
To ensure a comprehensive and systematic approach, a detailed search strategy was developed. The search was focused on English-language research articles published between January 1999 and August 2021. Three prominent databases, namely Scopus, PubMed, and Web of Science, were utilized for the literature search. The keywords used in the search strategy included relevant terms such as “Nanostructures,” “Nano,” “Nanoparticles,” “Nanomaterials,” “Release,” “Migration,” “Textile,” and “Fabrics.” The search strategy aimed to identify studies that specifically addressed the release of nanoparticles from nano-textiles during use and washing. The specific details of the search strategy can be found in the Supplementary Materials (Table S1).
Study selection
The study selection process involved several stages to ensure the inclusion of relevant articles. First, the retrieved articles from the three databases were aggregated using EndNote 20 (Thomson Reuters, Toronto, Canada). Duplicate records were automatically identified and removed, resulting in a final set of unique articles. Then, the titles, abstracts, and full-texts of the remaining articles were screened by the reviewers to determine their eligibility based on the predefined inclusion and exclusion criteria.
The inclusion criteria for article selection were as follows: - Original research papers that investigated the release of nanoparticles from commercially available conventional textiles. - Language: English.
The following criteria were used to exclude studies: - Review articles, conference papers, letters to the editor, and book chapters. - Studies focused on the release of nanoparticles from products other than textiles. - Research articles primarily aimed at method development. - Studies examining nanoparticle release from nano-textiles during landfill disposal. - Studies exploring scenarios of nanoparticle release other than washing and use (e.g., contact with sweat).
Data extraction
Once the eligible articles were identified, a standardized data extraction form was developed. The form included various key parameters and characteristics of the studies, such as the first author, publication year, type of textile, textile material, country of origin of textiles, textile label, type of nanoparticle, confirmation of nanoparticle presence in textiles using scanning electron microscopy (SEM), location of nanoparticles in the fabric, nanoparticle shape and size, nanoparticle amount in textiles, nanoparticle release process, amount of nanoparticles released under different conditions, and the disposition of the released nanoparticles. Two independent reviewers systematically extracted the relevant data from the selected articles using Microsoft Excel 2013 (Microsoft Office).
By following this systematic study design, the review aimed to provide a comprehensive analysis of the literature on nanoparticle release from commercial nano-textiles during their use and washing. The structured approach ensured the inclusion of relevant studies and minimized bias, enhancing the reliability and validity of the findings.
Result and discussion
In 2021, an extensive search across databases (Web of Science, Scopus, PubMed) yielded 1549 studies. Manual searches on Google Scholar and reference lists added 7 more references. After removing duplicates, 1240 articles were screened based on title and abstract, meticulously assessing relevance.
Following the title and abstract screening, 46 papers were selected for a thorough full-text review. The reviewers meticulously examined each of these articles to assess their eligibility for inclusion based on the predefined inclusion criteria. Upon closer examination, 35 articles were excluded as they did not meet the specific criteria outlined for this systematic review, such as focusing on the release of nanoparticles from commercial nano-textiles during use and washing.
Finally, after the comprehensive screening and evaluation process, a total of 11 studies were deemed eligible and included in this systematic review (7, Geranio et al., 2009, 26, 2010, 16, 9, 27, 28, 29, 30, 2). These studies provided valuable insights into the release of nanoparticles from commercial nano-textiles during their use and washing.
To offer a visual representation of the study selection process, Figure 1 illustrates the sequential steps are involved in identifying and including the relevant studies for this systematic review. Flow diagram of the screening process and study selection.
Characteristics of the included studies.
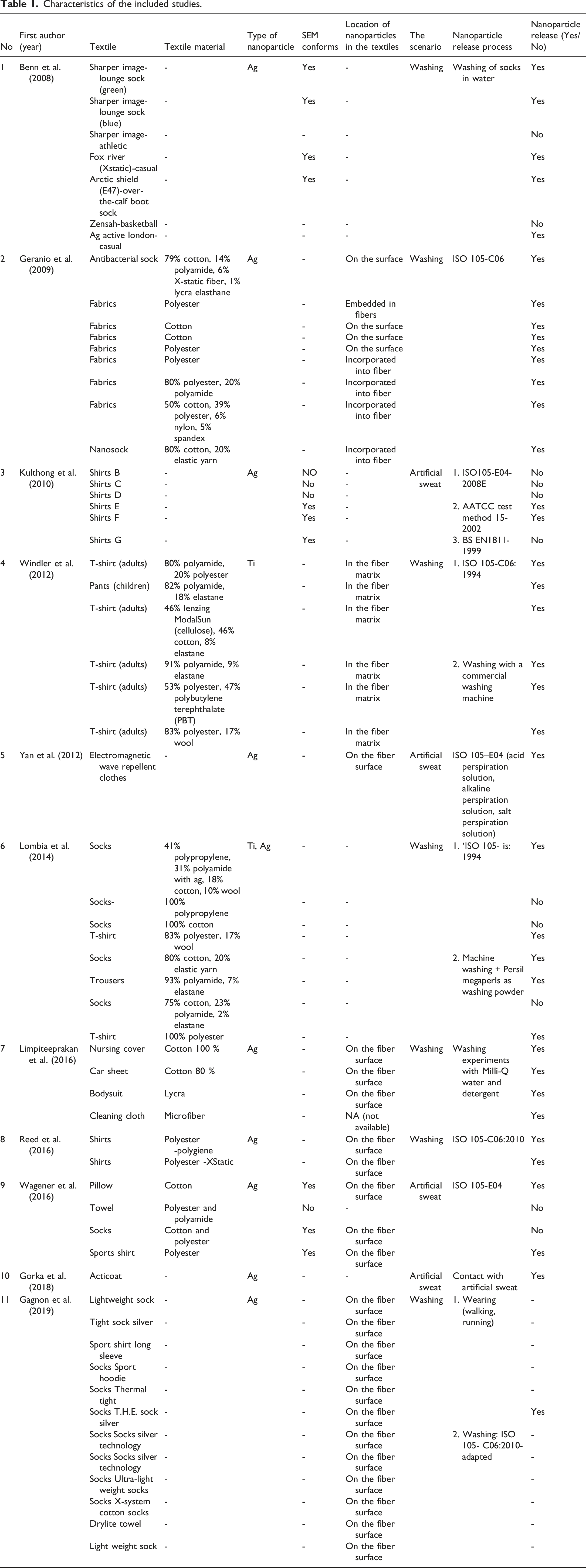
Nanoparticles incorporated into textile products
The initial step of investigating the release of nanoparticles from commercial textiles containing nanoparticles was the evaluation during usage and washing. As previously mentioned, the studies included in this systematic investigation focused on practical commercial textiles marketed as nano textiles. The results of this systematic review indicated that the primary origin of the studied textiles is 70% from Asia,13,14,19 24% from Europe,9,25 and only 6 cases have originated from the Americas. Nanotechnology, biotechnology, information technology, and cognitive science (NBIC) also reported that approximately 60% of the total nanotechnology products belong to Asian countries. 20 This distribution highlights the prominence of Asian countries in the production and advancement of nanoscience.
The textiles examined in this review encompassed a variety of products, including socks,7,9,10,26–28 fabrics, 10 shirts,2,26,29 T-shirts9,25 pants, 25 electromagnetic wave repellent clothes, 16 trousers, 9 nursing covers, 30 car sheets, 30 bodysuits, 30 acticoat, 31 cleaning cloths, 30 pillows, 26 and towels.2,26
The textile industry is currently focusing on the potential use of metal nanoparticles (MNPs) to enhance fiber construction and develop fabrics with improved properties.26,32 This has led to the emergence of a new field known as “nano-finishing” in textile finishing techniques. 33 The most important nanoparticles are zinc oxide (ZnO NPs),34,35 titanium dioxide (TiO2 NPs),24,36 cerium dioxide (CeO2 NPs),6,23,29 aluminum oxide (Al2O3 NPs),26,32,33 and copper oxide (CuO NPs),26,32,33 which have led to the introduction of new functionalities in textiles. These functionalities include self-cleaning properties,34,35 UV blocking activity,32,37 hydrophobicity,26,32,38,39 antibacterial properties,32,40,41 and electromagnetic wave shielding. 37 For instance, this study has demonstrated that titanium dioxide nanoparticles (TiO2 NPs) are commonly used as an example of nano-finishing to improve cleaning and bleaching processes in textile production. TiO2 NPs exhibit photocatalytic activity, which allows for the rapid degradation of natural dyes and pigments present in fabrics. Under light irradiation, photo-oxidant radicals such as OH, RO, and RO2 are formed on the surface of TiO2 NPs, leading to the oxidation and degradation of these compounds. This property makes TiO2 NPs beneficial for enhancing the cleaning and bleaching capabilities of textiles. 38
As previously mentioned, nanoparticles can be used to achieve various functionalities in textiles. In this systematic review, however, only two types of these nanoparticles were utilized in the textiles under investigation: silver nanoparticles and titanium nanoparticles. Among the textiles studied in this systematic review, silver nanoparticles (Ag NPs), which were used in 89% of the textiles, had the highest utilization.2,7,10,16,26,29–31,42 Silver particles are employed due to their protective antibacterial, odor-resistant, and electromagnetic properties. Titanium nanoparticles (Ti NPs) were found in 9% of the textiles and are primarily utilized for photocatalytic activities. 25 A small percentage of textiles (2%) also contained both silver and titanium nanoparticles. 9 These findings highlight the growing trend of using metal nanoparticles in textiles to enhance performance and introduce new prominent features.
As presented in Table 1, the results of the article reviews have shown that there is a discrepancy between the advertised nanoparticle content of commercial textiles and the actual presence of nanoparticles. In these studies, Nanoparticles underwent measurement utilizing inductively coupled plasma-mass spectrometry (ICP-MS). Also, the use of scanning electron microscopy (SEM) enabled a comprehensive examination of 23 textile products,7,29,42 revealing that only 15 of them contained nano-sized particles. This indicates that certain manufacturers may inaccurately represent their products or incorrectly label them as containing nanoparticles. The improper labeling of a product 42 lacking nanoparticles under the title of a nanoparticle-containing product highlights the significance of accurate labeling for these products. This issue underscores the need for validation methods beyond labeling to ensure the presence of nanoparticles in a product. This discrepancy raises concerns about transparency and the accuracy of information provided by textile manufacturers regarding nanoparticle presence. This issue is further supported by evidence; studies have indicated that certain products labeled as conventional items have actually contained nanoparticles. 25 As previously mentioned, this underscores the need for standardized terminology and labeling methods in the textile industry to ensure clarity and consistency in identifying and describing nanoparticle content.
The results of the studies reviewed in this review have demonstrated that the size and morphology of particles present in textiles varied in size from 60 nanometers 16 to 3.5 micrometers. 25 The particles exhibited predominantly round and irregular shapes,16,25,29,31 which is consistent with the characteristics of nanoparticles. Additionally, agglomeration was observed in one textile , 2 indicating that nanoparticles can form clusters or aggregates within the fabric structure.
Findings related to nanoparticle content in commercial textiles in the studies under investigation reveal a wide range of concentrations. Some textiles did not contain detectable levels of nanoparticles,2,7,9,42 while others exhibited significantly high concentrations (150,000 µg/g of textile). 9 In one of the studies, the unit presented for nanoparticles in textiles was different, and the amount of nanoparticles was 0.6-810 grams per textile. 25 The variation in nanoparticle content can be attributed to factors such as the type of textile material used and potential surface modifications or treatments applied to enhance nanoparticle binding. In addition, the results of the studies under investigation have demonstrated that textiles with synthetic fibers 9 tend to have higher nanoparticle content compared to those made from natural fibers. 2 Among the investigated textiles, 8% of the textiles were made of natural fibers,9,30,36,42 21% were made of synthetic fibers,9,30,36,42 and 11% were a combination of natural and synthetic fibers.9,10,25,42 The fiber type of 60% of the textiles was unknown.2,7,26,29 This could be due to the physical and chemical properties of synthetic fibers that allow for better interaction and bonding with nanoparticles. 30 The precise reasons for this trend would require further investigation.
One of the other items under investigation in this review has been the method of incorporating nanoparticles into textiles. Various methods have been considered for this purpose, with two primary approaches being the embedding of nanoparticles within fibers and onto the fiber surfaces during the textile manufacturing process. 43 The results indicated that approximately 31% of the textiles examined in these studies contained nanoparticles embedded within fibers. It seems that this method of incorporation has been chosen due to the uniform distribution of nanoparticles throughout the fabric compared to the surface application method.10,25 On the other hand, a significant portion of the textiles (40%) had nanoparticles present on their surface.2,10,16,26,30,42 This implies that nanoparticles were added to the textiles after the manufacturing of the base fabric, likely during the finishing stage. The nanoparticles applied on the surface can provide specific functionalities or enhance certain properties of the textile, such as antimicrobial properties or UV protection 2,10,16,26,30 It’s worth noting that for a considerable portion (29%) of the textiles in the reviewed studies the exact location of nanoparticles was not specified 7,9,29–31,42 This lack of information makes it challenging to determine whether the nanoparticles were embedded within the fibers or present on the surface. However, it is possible that in some cases, the nanoparticles may have been present in both locations, depending on the specific manufacturing processes and techniques employed.
Overall, these findings highlight the diverse methods of incorporating nanoparticles into textiles and demonstrate the versatility of nano-enhanced textiles in achieving desired functionalities and properties. The choice of embedding nanoparticles within fibers or applying them on the surface depends on the specific objectives and requirements of the textile application.
Health and environmental effect on nanoparticles incorporated into textile products
Given the growing application of nanoparticles in textiles, conducting research to assess the effects of these nanoparticles on human health and the environment is essential. Additionally, it is also crucial to explore effective methods for the safe disposal or recycling of textiles containing metal nanoparticles to minimize their potential environmental impact. Unintentional release of nanomaterials from these items could potentially influence both the environment and human health. Different environmental and health exposure scenarios are determined by the product’s life cycle and design. Unintentionally released from textiles might directly contact with people and ultimately enter wastewater systems.14,44
The release of nanoparticles during washing raises concerns about potential exposure to these particles. Individuals using nanoparticle-treated textiles may repeatedly come into contact with these released nanoparticles over time. Such exposure could have potential health implications, such as the long-term effects of prolonged exposure to nanoparticles on human health. Additionally, the environmental consequences of nanoparticle release during washing are significant. The wastewater generated from washing these textiles can carry the released nanoparticles into the environment, potentially leading to ecological impacts. The behavior and fate of these nanoparticles in wastewater treatment systems and their potential effects on ecosystems need to be studied further to better understand and mitigate any potential risks.
The major concern regarding nanoparticle exposure through textiles pertains to their potential absorption through the skin. Skin is regarded as an exceptional absorptive medium due to its abundant blood circulation, presence of tissue macrophages, dendrites, lymph vessels, and diverse sensory nerve endings. After entering the body, nanomaterials gather in the spleen, bones, kidneys, and liver.14,45
Currently, significant gaps in understanding remain regarding the potential hazards associated with nanoparticles46,47 Obtaining comprehensive insights into the hazards linked with nanoparticles remains a challenge. This is due to the diversity of nanomaterials, production methods, nanoparticle characteristics such as shape, dimensions, crystalline structure, porosity, agglomeration, aggregation, and a variety of other variables. 48
As mentioned earlier, the hazardous effects of nanomaterials in textiles on human health and the environment have not been extensively demonstrated, and available data are highly debatable, heavily contingent upon the stability, porosity, shape, size, surface dose, and ultimate use of nanomaterials. Consequently, accessible reports are not particularly compatible or comparable. When materials at the nanoscale are incorporated into textile products, especially those in direct contact with humans, environmental labeling becomes essential. Efforts to propose environmentally compatible fabrication stages through green chemistry, the development of less toxic nanomaterials, on-site preparation techniques resulting in reduced effluents, and enhancing the durability of nanomaterials bonded to textile substrates should be prioritized in future and forthcoming studies. 46
To investigate the hazards of nanoparticles on textiles, a method needs to be developed as their impact on the environment and human health is currently unpredictable. A review of the literature regarding the use of metal compounds in textile products demonstrates a rigorous regulatory framework in Europe. Textiles that have continuous contact with the human body, such as clothing, are typically regulated under the German Foods, Consumer Goods and Feedstuffs Act (LFGB). However, there are currently no explicit legal provisions concerning nanomaterials in textiles, nor guidelines for incorporating substances like TiO2. The European General Product Safety Directive requires manufacturers to assure the safety of their products, which means that textiles incorporating nanomaterials must offer no health concerns to consumers, according to the LFGB or the Product Safety Act. 49
The Biocidal Products Directive (1998/8/EC) governs the use of biocidal chemicals on textiles. The new EU Biocidal goods Directive (528/2012), which took effect in July 2012, requires a separate assessments of nanomaterials in biocidal and treated goods for possible concerns to human, animal, and environmental health. Biocidal product labels must include all biocidal components as well as the names of any nanoparticles present, followed by “nano” in brackets, as defined by the EU Commission.49,50
Regarding chemicals, the European REACH Act broadly embraces nanoparticles, although there are no explicit standards for their database or risk assessment. European discussions are underway to explore potential changes, and German federal authorities (BAuA, BfR, and UBA) have collaborated on a joint idea in this area.49,51
The EU Regulation 1007/2011 enforces restrictions on the concentrations of specific metals in textiles, such as cadmium, lead, chromium (VI), arsenic, and their compounds (2013). These limits aim to ensure consumer safety and minimize potential health risks associated with exposure to hazardous substances. Additionally, the regulation sets specific requirements for leather articles that come into contact with the skin, including a stricter limit on chromium (VI) content. On the other hand, the EU Ecolabel criteria, outlined in the European Commission directive (2009/567/EC), focus on environmental considerations for textile products (2009). This aims to promote environmentally friendly textiles by assessing various factors, including the use of hazardous substances and the overall environmental impact of the product. Regulatory measures and environmental criteria play a crucial role in ensuring consumer safety and promoting sustainable practices within the textile industry.
Overall, the study results under review emphasize the need for further research and evaluation of the release behavior, durability, and environmental impact of nanoparticles in textiles. It is crucial to develop appropriate testing methods, guidelines, and regulations to ensure the safe and sustainable use of nano-textiles while minimizing any potential risks associated with nanoparticle release during washing. Through, these results emphasize the need for comprehensive studies on the behavior, fate, and potential environmental consequences of nanoparticles released from textiles during washing. It is crucial to not only focus on the metallic nanoparticles themselves but also consider the different forms and compositions they may take during the washing process and subsequent wastewater treatment.
Regarding, potential hazards for children and pregnant women there is limited data regarding the presence of heavy metals in women’s underwear. Especially concerning are female briefs, which directly contact the vulva, potentially facilitating metal absorption by the mucous membranes of the vagina and vulva. So far, no research has specifically investigated women’s exposure to heavy metals in briefs via this distinctive pathway. However, it’s worth noting that the mere presence of metals in underwear doesn’t necessarily imply complete release. 52
Release of nanoparticles from textiles
Release scenario during washing
In general, the findings of the reviewed studies indicate that the release of nanoparticles from textiles during washing exhibits a complex nature, influenced by various factors. These factors include the nanoparticle content loaded into the fabric, fabric type, number of wash cycles, washing environmental conditions such as water quality, detergent type, and washing method (such as the use of a washing machine), methods of nanoparticle attachment to the fabric, etc. Additionally, the reviewed studies demonstrate the necessity for further research to comprehensively understand these factors and develop strategies to minimize nanoparticle release while preserving the desired properties of the textiles. 53 The results obtained from this systematic review demonstrated that a significant portion of these studies (69%) have focused on investigating the release of nanoparticles from textiles during washing.2,9,10,25,26,30
These studies utilized washing protocols based on ISO 105-C06:1994, a standard method for evaluating colorfastness in washing. The findings indicated a correlation between washing conditions and the release of nanoparticles from textile products. However, it is noteworthy that a recent study by Kowal et al. (2014) proposed an alternative method based on the tensile test (ASTM D4541) for assessing the adhesive strength of nanoparticles on fiber surfaces during washing. 54 This method offers a supplementary approach to evaluate the durability and adhesive strength of nanoparticles after washing cycles using adhesive tapes. 55 Interestingly, none of the studies included in this review have utilized the ASTM D4541 method, relying solely on the ISO 105-C06:1994 method for assessing the release of nanoparticles during washing.2,9,10,25,56 In addition, some studies employed washing machines to simulate real washing conditions and adopted a practical approach.9,25 This approach provides a more realistic representation of the release scenario during washing, considering factors such as mechanical agitation and water flow. Combining various testing methods seem to contribute to a more comprehensive understanding of nanoparticle release.
The findings from the studies under review demonstrated that the release of nanoparticles from textiles during washing can vary significantly depending on various factors. The percentage of released nanoparticles encompasses a wide range from less than 1% to nearly 100% of the total nanoparticles present in the textiles 7,9 This diversity indicates that the release behavior is influenced by multiple factors, and it cannot be generalized across all textiles. One of the important factors affecting the extent of nanoparticle release is the nanoparticle content within the textiles. Textiles with higher nanoparticle concentrations tend to release a greater content of nanoparticles during washing. This suggests that the loading capacity of the textiles play a role in determining the release behavior.
The nature of the fabric also emerged as a significant factor influencing nanoparticle release. Textiles with synthetic fibers, such as polyester, were found to release a higher amount of nanoparticles compared to textiles with natural fibers like cotton.9,10 This difference can be attributed to the surface chemical functions of synthetic fabrics, which are generally less durable than those of natural fabrics. 30 The weaker bonding between nanoparticles and synthetic fibers may contribute to their increased release. Furthermore, the manner in which nanoparticles are attached or embedded within the textile matrix affects their release. 25 Textiles with nanoparticles embedded inside their fibers demonstrated lower release percentages10,25 This can be attributed to the higher energy required to break the fibers and release the nanoparticles. On the other hand, textiles with nanoparticles loaded on the surface of fibers exhibited higher release rates, indicating that the bonding between nanoparticles and fibers, facilitated by adhesives, plays a crucial role in nanoparticle retention. Unfortunately, specific details of the adhesive formulations used in textiles are often proprietary, making it challenging to assess their effectiveness in retaining nanoparticles. This underscores the need for further research and transparency in understanding the role of adhesive formulation in nanoparticle release.
Another examined factor in the studies regarding nanoparticle release during washing was the size of these nanoparticles. The results indicated that a considerable portion of released nanoparticles from textiles during washing are in the form of larger particles (>450 nm) and tend to aggregate.2,9,25 This suggests that nanoparticles undergo degrees of aggregation or clustering, which potentially affects their behavior and interactions with the environment. However, it is noted that the behavior of these larger nanoparticles, particularly those that are not effectively removed by wastewater treatment processes, has not been extensively studied in the included research. This knowledge gap highlights the need for further investigation into the fate and behavior of these nanoparticles during wastewater treatment and in the environment. Understanding the behavior of nanoparticles, especially larger ones, is crucial for assessing their potential environmental impact and evaluating their risk. It is important to determine whether these nanoparticles persist in the water, sediment, or other environmental compartments, and if so, what effects they may have on ecosystems and human health. Future studies should focus on examining the behavior, fate, and potential toxicity of nanoparticles larger than 450 nm during wastewater treatment processes and in natural environments. This will provide valuable insights into their potential risks and enable the development of appropriate mitigation strategies to minimize their environmental impact.
The results of this systematic review showed that the process of nanoparticle release during washing can impact the efficiency and lifespan of nanotextiles. The reviewed studies demonstrated that nanoparticles are continuously released during multiple washing cycles, albeit with varying release patterns in different textiles. Benn et al. (2008) found that the highest nanoparticle release occurred during the third washing cycle, and in two types of studied textiles, all nanoparticles were completely liberated after four washing cycles. 5 In contrast, Geraniou et al. (2009) observed a reduction in nanoparticle release in the second cycle. 10 Similarly, Limpiteeprakan et al. (2016) reported a decreasing trend in release during consecutive washing cycles, stating that the release levels stabilize after the third washing cycle for most textiles, with approximately 50% to 70% of nanoparticles remaining even after 20 washing cycles in the fabric. 30 Furthermore, Gagnon et al. (2019) identified a diverse release pattern among different types of textiles. 2 The results from these studies indicated that the behavior of nanoparticles during washing is intricate and influenced by various factors, such as fabric type and specific nanoparticle properties.
Another influential parameter on nanoparticle release from textiles is the washing environment characteristics. The washing environment, including water quality, detergent, and washing machine usage, also affects nanoparticle release. The washing environment can significantly impact the amount of released metal ions. Benn (2008) 7 reported that up to 90% of silver nanoparticles are released as Ag+ in the washing environment, while Geranio et al. (2009) 10 reported lower values. Of course, the ordinary washing solution (urban water) has a high pH (10 and above) and contains a variety of other components such as chloride ions, which may interact chemically with nanoparticles. 53 Corrosiveness of water is reduced by increasing the buffer capacity and alkalinity. 57 As a result, the ratio of particles released in an ionized form from the fabric is influenced. This is despite the fact that most studies have used pure water to test the release of nanoparticles from textiles. Pure water has a pH of 7, which is more oxidizing than urban water, and as a result, it creates more dissolved ions in water. In experiments conducted using commercial n-Ag in ultrapure water, ionic silver in solution increased over time, indicating that n-Ag forms as the ion oxidized in solution. 7 Generally speaking, research reveals that tap water is less aggressive than ultrapure water in removing the silver from textiles. Therefore, these tests in ultrapure water may overestimate the content of nanoparticles that can be washed into domestic wastewater streams.
As earlier mentioned, the pH of the washing solution is particularly important, as it can influence the release of nanoparticles in their ionic form. The studies highlight that urban water, with its higher pH and presence of various components like chloride ions, can interact chemically with nanoparticles and potentially increase their release as ions. 53 In contrast, pure water, commonly used in experimental settings, has a more oxidizing nature and may result in higher levels of dissolved ions. It is worth noting that the corrosiveness of water is reduced by increasing its buffer capacity and alkalinity, which can impact the particle-to-ion ratio released from textiles. 57
Furthermore, results of studies suggest that tests conducted in ultrapure water may overestimate the content of nanoparticles that could be washed into domestic wastewater streams. 7 This indicates that real-world washing conditions, such as using tap water, may result in different release patterns. Overall, these findings emphasize the importance of considering various factors when assessing nanoparticle release from textiles during washing. Understanding the release behavior and the influence of different washing environments can inform the development of strategies to minimize the release of nanoparticles into the environment and promote the sustainable use of nano-textiles.
Another factor influencing nanoparticle release from textiles is the type and composition of the detergent used. The release and transformation of nanoparticles during washing is a complex process influenced by various factors. The chemistry of the washing environment, specific characteristics of the particles (such as capping agents), and methods of nanoparticle incorporation within the fabric all play a role in determining the rate and extent of dissolution and surface/morphological changes 58 Studies indicate that while complete dissolution of nanoparticles may not always occur, surface transformations and morphological changes can take place.42,59 The results of the studies examined in this review demonstrate a significant impact of washing detergents on the release of nanoparticles from textiles during washing. Detergents are formulated with various compounds to optimize the washing process, and among these components, fluorescent whiteners, ion exchangers (zeolites), and catalysts/activators for whitening can be mentioned.60,61 These compounds, especially highly oxidative constituents, can influence the behavior of nanoparticles embedded within the textiles. The presence of metal catalysts in conjunction with hydrogen peroxide (H2O2) enhances the bleaching efficiency by forming metal oxo intermediates with reactive oxygen species (ROS). 61 This process can lead to the transformation and release of nanoparticles from treated surfaces. 58 The studies highlight that the addition of oxidants, such as hydrogen peroxide and peracetic acid, during the washing process can lead to a notable release of Ag+ ions from textiles containing metallic silver nanoparticles. Additionally, when textiles containing metallic silver nanoparticles are exposed to detergents during washing, oxidation of silver from Ag(O) to Ag(I) occurs, resulting in the release of Ag+ ions. 10 The sensitivity of nano-Ag to oxygen is well-known, 62 and the chemisorbed Ag + formed during water or air exposure can be released when the textile is washed. While dissolution is critical in the nanoparticles and textile issue, the particles may not dissolve completely and instead undergo surface and morphological changes.59,63 According to the findings of the study Limpiteeprakan et al, 30 the amount of Ag that remained on the fabrics after washing with detergent was less than that after washing with pure water. In the study of GERANIO et al. (2009), 10 a significant release of Ag + was also observed only after the addition of an oxidant. In this study, H2O2 and PAA, formed in the perborate/TAED system, were two major oxidants during washing. First, perborate dissociates to H2O2. Then H2O2 reacts with Tetraacetylethylenediamine to form peracetic acid (PAA). PAA is a more active bleaching agent than hydrogen peroxide. H2O2 can oxidize metallic silver to Ag + while also producing hydroxyl radicals. The produced hydroxyl radical can oxidize metallic silver further. 61 The results obtained from the study by Lorenz et al. (2012) shed light on important factors concerning the impact of detergents on the release and behavior of nanoparticles, particularly silver nanoparticles, during washing. 33 One key finding is the potential precipitation of silver ions as silver chloride (AgCl) in the washing solution, particularly in the presence of chloride ions. In the Lorenz et al. (2012) study, 53 species calculations revealed that 8.7 mg L−1 of chloride in the washing solution could precipitate the large percentage of total Ag released from some textiles (more than 70%) if released in ionic form. This suggests that the fate of released silver nanoparticles may involve the formation of insoluble AgCl, which can have implications for their environmental impact. The study also identified the presence of silver sulfide (Ag2S) nanoparticles in the washing solution of a specific textile samples. While Ag2S is a common silver species found in wastewater, its occurrence in the washing solution was rare among the studied samples. The presence of Ag2S nanoparticles could be attributed to the composition of the washing powder used, which contained Na2SO4. The washing powder used in this study contained 21% Na2SO4, which can be the cause of the creation of nanoparticles containing silver sulfide. Reducing or removing Na2SO4 from the washing powder may help mitigate the formation of silver sulfide nanoparticles during washing. 33 Another significant finding in this study is the loss of antibacterial properties in textiles after washing, even when a substantial portion of silver nanoparticles remained post-washing. This textile also lost its antibacterial properties after washing, despite the fact that more than 85% of the silver was still present. This suggests that factors such as the low bioavailability of Ag2S or the presence of large AgCl particles, which dissolve at a slower rate compared to smaller nanoparticles, may contribute to the diminished antibacterial activity.
In the microscopic research, except for the silver sulfide particles released from one of the textiles, only agglomerated nanoparticles were found in the washing solutions, which shows that even in the washing solution with a high concentration of detergents, which are known to stabilize nanoparticles, aggregation is an important process. Because the wash water is directly discharged into the wastewater, the fate of these various forms of nanoparticles during treatment determines the potential release of nanoparticles into the environment. Therefore, research on the behavior and effects of silver nanoparticles should not be focused only on metal nanoparticles, but should also include studies of the compositions of these nanoparticles.
Another notable aspect to consider is the impact of mechanical stress as an influential factor in nanoparticle released from textiles during washing. The study by Lorenz et al. (2012) 53 demonstrated that the use of chemical detergents in machine washing, which involves agitation and mechanical stress, can lead to a greater release of nanoparticles compared to laboratory washing. This indicates that the synergistic effects of chemical factors and mechanical forces can contribute to increased nanoparticle release. Additionally, Gagnon et al. (2019) 10 investigated the impact of wearing textiles, specifically socks, prior to washing them. The study found that the act of wearing socks had a significant influence on the release of silver nanoparticles into the washing water. The mechanical stress experienced during wear, such as friction and movement, could cause the nanoparticles to become detached or fragmented from the fabric fibers, resulting in their release during washing.
These findings underscore the importance of understanding the various factors that affect nanoparticle release during textile washing. By considering both the chemical agents present in detergents and the mechanical stresses experienced during wear and washing, researchers can gain insights into the mechanisms underlying nanoparticle release and develop strategies to mitigate or control their release. This knowledge is essential for ensuring the safe and sustainable use of nanomaterials in textiles and minimizing their potential environmental impacts.
Release scenario during contact with artificial sweat
The results derived from the study in this review indicate that skin exposure provides a significant potential route for nanoparticle interaction and sweating can potentially facilitate the transfer of nanoparticles from textiles to the skin’s surface. Furthermore, it should be noted that damaged skin leads to heightened exposure, as released nanoparticles can traverse through compromised skin areas, enter the bloodstream via skin capillaries, and enable further interaction. 19 Artificial sweat has been widely used in assessing the release of nanoparticles from various materials, including textiles, and it serves as an intermediary in the skin’s nanoparticle absorption. 64 Approximately 31% of the studies included in this systematic review were focused on nanoparticle dispersion and the role of sweating.16,29,31,42 This highlights the significance of understanding the potential transfer of nanoparticles from textiles to the skin and the role of sweating in this process. The results indicate that the presence and release of nanoparticles in artificial sweat formulations from commercial textiles varied among the studies. In a significant portion of the textiles examined (57%), no nanoparticles were detectable in any of the artificial sweat formulations. This suggests that these textiles may have a lower likelihood of nanoparticle transfer to the skin during sweating. It is noteworthy that in 50% of the textiles studied in these investigations, the absence of nanoparticles in scanning electron microscopy (SEM) analysis aligns with the absence of nanoparticle detection in the artificial sweat formulation. This suggests that these textiles may either contain negligible amounts of nanoparticles or that nanoparticles are not readily released under simulated sweating conditions. However, in the remaining 43% of textiles investigated in this systematic review, nanoparticles were observed in the artificial sweat formulation. The levels of release varied depending on factors such as the specific composition of the artificial sweat formulation and the initial content of nanoparticles present in the textiles. The range of release levels spanned from approximately 0.01 to 5.5 mg/km, indicating significant diversity in nanoparticle dispersion among these textiles. Additionally, investigating the long-term effects and potential health risks associated with nanoparticle release from textiles during sweating is crucial to ensure the safety and well-being of individuals who come into contact with such textiles.
By using artificial sweat, researchers can simulate real-life scenarios and evaluate the extent of nanoparticle release from textiles. This knowledge is crucial for assessing the potential risks associated with dermal exposure to nanoparticles and understanding their impact on human health. Further studies using artificial sweat can provide valuable insights into the release behavior of nanoparticles from textiles and help in the development of appropriate safety measures and guidelines.
The use of standardized testing methods, such as ISO 105 E04, AATCC 15, and BS EN 1811:1999, plays a crucial role in assessing the release of nanoparticles from textiles in response to perspiration and skin contact. These standards provide a systematic approach to evaluate the behavior and potential risks associated with nanoparticle release in various scenarios. 62 ISO 105 E04 and AATCC 15 are widely recognized standards specifically designed to assess the resistance of textile dyes to human sweat. 63 They evaluate color stability and fastness under acidic and alkaline perspiration conditions. By subjecting textiles to these standardized tests, researchers can determine the release rate of nanoparticles when exposed to perspiration, providing insights into the potential transfer of nanoparticles from textiles to the skin surface. For example, the inclusion of BS EN 1811:1999 in the study conducted by Kulthong et al. (2010) 29 highlights the significance of assessing the release of specific elements, such as nickel, from textile products intended for direct and prolonged contact with the skin. This standard helps identify the amount of nickel released and contributes to the understanding of potential skin sensitization or allergic reactions caused by nanoparticle exposure.
The existence of such standardized testing methods not only ensures a consistent and reliable assessment of nanoparticle release, but also facilitates comparisons between studies, textiles, and different nanoparticles. They provide a foundation for evaluating the safety and quality of textiles and contribute to the development of regulations and guidelines in the textile industry. By employing these standards, researchers can gain a deeper understanding of nanoparticle behavior upon contact with sweat and the potential hazards associated with their release. This enables the identification of safer textile materials, facilitates the design of effective reduction strategies, and promotes the development of regulations aimed at minimizing potential health and environmental impacts.
Another influential parameter on nanoparticle release is pH and artificial sweat formulation, which has yielded interesting results in this context. The pH of the sweat formulation appears to play a significant role in determining the release behavior of nanoparticles.
Nanoparticles were not detectable in any artificial sweat formulation in 57% of the commercial textiles examined in these studies.29,42 This is not surprising for 50% of these textiles, as no nanoparticles were observed in the scanning electron microscope (SEM) examination of these textile. In contrast, nanoparticles were released at levels ranging from about 0.01 to 5.5 mg/kg from 43% of textiles in all synthetic sweat formulations, depending on the sweat formulation and the amount of nanoparticles in the textile.
In some studies, the ISO synthetic sweat formulation with a pH of 5.5 resulted in the lowest levels of detected nanoparticles, 29 which is advantageous considering its similarity to the pH of natural human skin. This suggests that textiles treated with nanoparticles may exhibit better stability and lower release rates when exposed to sweat with a pH closer to that of the skin. However, there are also studies where the highest release of nanoparticles occurred in the ISO synthetic sweat formulation with a pH of 5.5, indicating that the behavior of nanoparticles can be complex and may vary depending on other factors such as the specific textile and the composition of nanoparticles used. Additionally, the synthetic sweat formulation EN standard with a pH of 6.5 consistently showed the highest release of nanoparticles in all fabric samples.16,31,42
These findings highlight the importance of considering the pH and formulation of sweat when assessing the release of nanoparticles from textiles. It suggests that different sweat compositions may interact differently with the nanoparticles, leading to variations in their release. Further research is needed to better understand the underlying mechanisms and to develop standardized testing methods that more accurately reflect real-world scenarios of nanoparticle release from fabrics during contact with sweat. This knowledge can be valuable in designing textiles with improved nanoparticle retention and reduced potential for nanoparticle exposure to the skin and the environment.
The results of this systematic review indicate that the release behavior of metal nanoparticles from textiles is influenced by the composition of the simulated sweat environment. Different simulated sweat formulations, such as acidic, alkaline, and salty, have distinct effects on the release of nanoparticles. The acidic artificial sweat solution showed the highest release rate and content of nanoparticles, with a significant portion of them existing in an ionic form. This suggests that the acidic environment promotes the dissociation of metal nanoparticles into ions, leading to their release from the textiles. On the other hand, the alkaline artificial sweat solution exhibited a slower release rate and lower content of nanoparticles. In this case, the released nanoparticles predominantly remained in an ionic state throughout the process. The alkaline environment likely promotes the formation of metal ions through surface oxide reactions, resulting in a lower proportion of nanoparticles being released.
Surprisingly, the salt perspiration solution initially showed a slower release rate but accelerated over time, leading to a moderate amount of released nanoparticles. In this environment, the majority of released metal nanoparticles were in the particle form rather than as ions. The salt perspiration condition seemed to have a more sustained effect on nanoparticle release, possibly due to the mechanical forces exerted during the washing process.
The findings also highlight the potential risks associated with the release of metal nanoparticles in the salt perspiration environment. The higher exposure risk in this condition suggests that nanoparticles may be released into the surrounding environment and potentially enter the body. This underscores the importance of considering the specific conditions in which nanomodified textiles are used, as well as implementing appropriate safety measures to minimize exposure.
The results of the studies examined in this review indicate that the release of nanoparticles from textiles differs between artificial sweat tests and washing experiments. 24 In the artificial sweat solution, a higher percentage of metal particles were found to be dissolved compared to the washing liquid. This can be attributed to the presence of a relatively high concentration of chloride in the sweat solution, which forms complexes with the metal particles and keeps them in a dissolved state. On the other hand, the release of nanoparticles was generally lower in the artificial sweat solution tests compared to the washing tests. This suggests that there is a reduced release of particles in sweat or a greater tendency for nanoparticle precipitation in the washing liquids. The lower release of nanoparticles in the artificial sweat tests could be due to factors such as the composition of the sweat solution, including the presence of chloride ions that stabilize the particles, and the physiological conditions simulated by the artificial sweat. These findings highlight the importance of considering different exposure scenarios when assessing the release of nanoparticles from textiles. The presence of chloride in sweat can significantly affect the behavior of nanoparticles, leading to their dissolution and formation of metal-chloride complexes. Understanding these differences in release behavior between artificial sweat and washing liquids is crucial for evaluating the potential risks associated with nanoparticle exposure and guiding the development of safer nanomaterial-modified textiles.
Overall, these results emphasize the complex nature of nanoparticle release from textiles and the need for comprehensive studies to understand the behavior of nanoparticles in different environments. By gaining a better understanding of the release mechanisms and factors influencing nanoparticle release, researchers and manufacturers can work towards developing safer and more sustainable nanomodified textiles for everyday use.
Limitation
This systematic review has certain specific limitations that should be acknowledged. Firstly, despite the technological advancements in the textile industry and the increased utilization of nanomaterials in textiles in recent years, there are no studies available (from 2019 onwards) concerning the release of nanoparticles from commercial textiles. Additionally, certain aspects, such as the methods of nanoparticle loading into textiles which can potentially influence their release, have not been thoroughly investigated. Secondly, the textile production method and the incorporation of nanoparticles into their fibers have not been comprehensively examined, and the impact of the manufacturing process on nanoparticle release has not been evaluated. Thirdly, the studies included in this systematic review based on the inclusion criteria, despite the wide range of nanomaterials used in the textile industry, have predominantly focused on textiles containing silver and titanium nanoparticles, and their release has been the primary aspect under investigation. Fifthly, due to the limited number of studies available, other scenarios of nanoparticle release from textiles could not be explored. Sixthly, the data obtained from the studies were heterogeneous, making it impossible to conduct a meta-analysis or quantitative investigation of nanoparticle release from textiles. Seventh, considering the increased sensitivity of pregnant women and children, studies on the effects of nanoparticles released from textiles used by them should be prioritized. In essence, understanding the precise emission of nanoparticles from textiles in particular situations is crucial for accurately assessing human exposure. Consequently, it is essential to conduct a thorough examination of the occurrence release of nanoparticles in pregnant women, and children and their potential release. Eighth, While our study offers insights into nanoparticle release from commercial nano-textiles, it does not comprehensively address practical considerations regarding nanoparticle applications in textiles, such as how nanoparticles are utilized for these applications in relation to their final fabric size. These aspects were not directly discussed in any of the original studies included in our systematic review. To address these limitations in future research, it is crucial to examine all potential release scenarios throughout the life cycle of a product. Additionally, efforts should be made to replicate real-world conditions as closely as possible, estimate the variability in release among different products, and gather sufficient experimental data to facilitate robust statistical analysis in each case.
Conclusion
In various industries and fields, including the textile industry, nanomaterials have found extensive applications. However, there is also a growing concern regarding the potential hazards associated with nanotechnology and the use of nanomaterials in such products. This has led to a need for a systematic reevaluation of the release of nanoparticles from commercial textiles during their use and washing. Such a review can provide valuable insights into the distribution of size, concentration, speciation, and morphology of nanoparticles that may pose risks to human health and the environment.
Based on the analysis of relevant studies, which often simulated textile use and washing in laboratory settings, it is evident that textiles incorporating nanomaterials, especially those containing silver nanoparticles, can release a considerable number of particles in both particulate and ionic/soluble forms. The quantity and composition of nanoparticles released from textile fabrics are generally influenced by factors such as the nanoparticle structure, nanoparticle loading on the fabric, adhesive used to bind nanoparticles, fabric type, and the surrounding environment. According to the results of this study, by legislating and defining a standard instruction for the production and use of nano textiles, the release of nanoparticles from these textiles and exposure to them can be reduced as much as possible.
Although there are notable differences between laboratory simulations and real-world conditions, the results of these studies still provide valuable insights into the potential risks of nanoparticle exposure associated with nano-modified textiles for both humans and the environment. However, to comprehensively assess the safety of nano-modified textiles, it is essential to conduct toxicological studies on the released nanoparticles in future research.
Future studies should focus on gathering more data on the particle characteristics released during actual wear and washing of textiles. Additionally, it is crucial to conduct real-life wear and washing tests on various types of textiles treated with different forms of nanomaterials, assessing nanoparticle release in terms of concentration, size distribution, morphology, and characterization. Furthermore, understanding the dynamics of nanomaterial diffusion within textiles and evaluating the transfer of nanomaterials from textile fibers to the skin are important aspects to consider.
Supplemental Material
Supplemental Material - A systematic review of released nano-particles from commercial nano-textiles during use and washing
Supplemental Material for A systematic review of released nano-particles from commercial nano-textiles during use and washing by Zahra Beigzadeh, Malihe Kolahdouzi, Saba Kalantary and Farideh Golbabaei in Journal of Industrial Textiles.
Footnotes
Author Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Farideh Golbabaei, Zahra Beigzadeh, Saba Kalantry and Malihe Kolahdouzi. The first draft of the manuscript was written by Zahra Beigzadeh and Saba Kalantary commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Tehran University of Medical Sciences & Health Services Grant (Grant No.: 64788).
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
