Abstract
The contact layer of traditional medical dressing is easy to adhere to the new wound granulation tissue, which brings the deficiency of secondary wound injury. This work assumed that low-cost and soft mesh spunlace nonwoven was used as medical dressing contact layer. After micro-nano SiO2 modification, polydimethylsiloxane (PDMS)/n-heptane were uniformly sprayed on the surface of materials. PDMS modification was studied by using the three-factor three-level orthogonal experiment. The results showed that when the PDMS concentration was 1%, the heat treatment temperature and time were 120°C and 12 h respectively, material possessed the optimal hydrophobicity and its water contact angle (CA) was 151.0 ± 1.1°. Meanwhile, the hydrophobicity, microstructure, thermogravimetry, composition, tensile mechanics, adhesion resistance and durability of materials with/without treatment were compared. It was proved that PDMS was indeed grafted onto the material and could effectively improve the hydrophobicity in coordination with micro-nano SiO2 without affecting the mesh structure of material. The machine direction (MD) and cross-machine direction (CD) strength of material decreased but the elongation increased slightly. The MD and CD peeling energy was 129.7 ± 33.9 J/m2 and 98.1 ± 38.3 J/m2, respectively. Moreover, the water CA of material modified by micro-nano SiO2/PDMS was still 139.4 ± 1.4° after 10 friction stroke tests, indicating that the modified material possessed the great durability. Overall, this study can provide the research basis for the development of anti-adhesion medical dressing contact layer.
Introduction
Traditional cotton gauze had the characteristics of great absorption of wound tissue exudate, skin-friendly and low price. 1 However, the material possessed a large pore structure, which was easy to reverse infiltration after absorbing tissue exudate and adhere to new tissue after removal. Although soaking petroleum jelly could prevent the dressing from being adhered to the wound, there were problems such as low permeability, non-absorption of tissue seepage and being vulnerable to trauma when taken out.2,3 Although researchers had used grafting hydrogel and other methods for dressings to reduce adhesion and achieve a certain anti-adhesion effect, the cost would rise significantly and there were hidden dangers of losing functions for long-term storage.4–7 The tendency of the medical dressing contact layer to adhere to the wound has attracted considerable attention.
The hydroentangled fiber aggregation (spunlace nonwoven) was formed by the high-speed waterjet needle impinging on the fiber web, and possessed great softness, comfort, skin affinity, water absorption and low cost.8,9 In particular, the cellulose fiber spunlace nonwoven was very suitable for medical dressing substrate. However, when it was in direct contact with the wound, it still tended to adhere to new tissue.
As of now, studies showed that the rough surface morphology had anti-adhesion properties against non-specific proteins for cell growth and itself, and low surface free energy substrates could inhibit cell adhesion and diffusion.10,11 The rough surface of nano-silica (SiO2) could be precisely constructed on the cellulose fiber by means of soaking and baking to form hydrophobic fabrics. 12 The hydrolyzed nano SiO2 was adsorbed on the surface of cellulose fiber by hydrogen bonding, but the washing fastness of SiO2 nanoparticles on the surface of material was relatively low. 13 Zorko 14 and Vasiljević 15 used sol-gel and in situ growth method to immobilize micro-nano SiO2 particles on the surface of material. The nano SiO2 particles spontaneously polymerized to form the polysiloxane layer, slightly improving the fastness of particle fixation. However, the bonding fastness of multi-level micro-nano SiO2 structure on the material surface was unstable, and the superhydrophobic effect could not be achieved, which limited the anti-adhesion ability of material. 16
Polydimethylsiloxane (PDMS) was a type of safe and low surface energy silicone material, which had no irritation to human skin and mucous membrane. 17 Material surface coated PDMS could reduce the surface energy, which inhibited cell adhesion and diffusion. 18 Many scholars had found that PDMS could be evenly dispersed n-heptane or tetrahydrofuran solvents, and then grafted with nano SiO2 hydrolyzed polysiloxane silanol groups to generate stable slicon-oxygen bonds.19–21 Meanwhile, PDMS could be crosslinked with cellulose material by means of heating condition, and the finished material had great chemical stability and mechanical toughness.22,23 In the functional finishing of materials, spraying was a simple and low-cost operation method, which was suitable for spraying the surface of various materials and making the material possess special function. 24 Nevertheless, there has been no valuable research on the preparation of great hydrophobic dressing contact layer by using micro-nano SiO2 particles and low-surface substances to synergistically modify the spunlace nonwovens.
In this paper, 22-mesh viscose mesh spunlace nonwoven was used as the substrate of medical dressing contact layer, and SiO2 sol-gel and in situ growth method were used to modify the material to construct multi-level micro-nano SiO2 structure. Meanwhile, PDMS/n-heptane solution was evenly sprayed on the material surface. Orthogonal optimization experiments were carried out to obtain the optimal process condition. In addition, the hydrophobicity, microstructure, thermogravimetry, composition, tensile mechanical properties, adhesion resistance and durability of materials with/without modification were investigated. This can help to understand how to construct firm and stable structure on spunlace nonwoven to achieve hydrophobicity, and provide reference for the development of anti-adhesion spunlace nonwoven-based medical dressings.
Materials and methods
Materials
The 22-mesh viscose mesh spunlace nonwoven was kindly supplied by Hangzhou Hanford Technology Co., LTD. Tetraethyl orthosilicate (TEOS), anhydrous ethanol and ammonia were purchased from Sinopharm Chemical Reagent Co., LTD (Shanghai, China). Isopropyl alcohol, dimethyl silicone oil PMX-200 (viscosity 30000 mPa·s, 25°C), n-heptane were bought from Shanghai Aladdin Biochemical Technology Co., LTD (Shanghai, China).
Micro-nano SiO2/PDMS modification
Anhydrous ethanol as the solvent was put in a beaker, and 0.4 mol/L TEOS, 0.3 mol/L ammonia, and 4 mol/L water were added. The solution A was fabricated by stirring at 1000 r/min for 2 min and then at 360 r/min for 60 min in a 40°C water bath. On the other hand, the solution B was prepared by adding 0.4 mol/L TEOS and 0.5 mol/L ammonia water with isopropyl alcohol as solvent (Figure 1(a)). The condensation reaction process of TEOS was shown in Figure 1(b). When the solution A was used, it needed to be oscillated in ultrasound for 5 min to make the nano SiO2 particles evenly dispersed. The sample was cut to a size of 4 × 4 cm2 and then was impregnated with the solution A for 10 s. Next, the excess solution was removed by a small rolling mill, and the residual ratio was controlled between 160% and 180%. After soaking and rolling, it was placed in the oven at 100°C for 15 min. Finally, it was taken out and placed at room temperature for 24 h.
15
The previously treated samples were put into solution B and heated in a water bath at 50°C for 20 min and then taken out. The surface moisture of samples was dried with tissue paper and then samples were put into the oven at 100°C for 15 min. After being taken out, samples were placed at room temperature for 24 h.14,25 Finally, the samples modified by micro-nano SiO2 were obtained and recorded. Material handling process: (a) Preparation of solutions, (b) Condensation reaction process of TEOS, (c) Material modification, (d) Spraying device and (e) Friction testing equipment.
Factor level scheme.

Orthogonal experiment scheme.

Water CA measurements
The water CA measurements were performed according to ASTM D7334-08 (2013). 26 The water CAs of samples were acquired by the automatic CA measuring instrument (dataphysics OCA50Micro, Germany). During the test, the liquid output and velocity were 4 uL per time and 5 uL∙s−1, respectively. The test area should avoid the mesh and selected the place where the fiber assemblies were evenly distributed as far as possible. When the water droplets were stable, the water CAs of samples at three different positions were measured. Each position was tested twice, and the average value of the three valid test results was obtained.
Scanning electron microscopy (SEM) studies
The SEM images were obtained by using the cold field emission electron microscope (Hitachi SU8010, Japan). Samples needed to be coated with a thin gold layer to minimize the surface charge. Meanwhile, the sample morphology was observed under an acceleration voltage of 10.0 kV.
Thermogravimetry (TG) analysis
The sample TG weight was measured by the thermogravimetric analyzer (Seikosha TG6300, Japan) and the mass of tested sample was about 5 mg. All analyses were performed in a dynamic nitrogen atmosphere (100 mL∙min−1), between 30°C and 700°C. The scanning rate was set at 10°C∙min−1.
Fourier Transform Infrared (FTIR) spectroscopy
The composition analysis was carried out using the FTIR spectrometer (Shimadzu IRPrestige-21, Japan). The data collection was performed with a resolution of 4 cm−1, and its range is 4000-550 cm−1 with a total of 32 scans. According to the peak of FTIR spectra and the difference of bond vibration of samples, the composition was analyzed.
Covalent connection verification experiment
Samples were sprayed with PDMS according to the optimized process parameters. Three samples were divided into one group, and three groups of samples were fabricated. After spraying, all samples were placed in fume hood for 12 h. Notably, the second group was baked at 120°C for 12 h, and put into n-heptane solution after cooling. In addition, the third group was directly put into n-heptane solution. The n-heptane solution with sample was treated with a magnetic mixer at 300 r/min for 30 min and then placed in a fume hood for 12 h. Finally, three groups of samples were tested and analyzed by FTIR spectroscopy.
Tensile measurement
The tensile properties were measured by Instron 3365 universal material tester. The tensile interval, the pre-added tension and the tensile speed were set as 2 cm, 5 CN and 100 mm/min, respectively. Prior to testing, the sample should be balanced in laboratory conditions (the temperature was 20°C and the relative humidity was 65%) for 24 h.
Adhesion resistance test in vitro
Based on the Andrews and Kamyab in vitro gelatin model, the adhesive situation of wound seepage on the material was simulated and the anti-adhesive property was characterized by the peeling energy of gelatin-material.27–29 Firstly, the sample of 3 × 15 cm2 was immersed in deionized water for 5 min and tiled on the clean surface. A PTFE mold was placed on the surface (Figure 2(a)). Secondly, the gelatin was dissolved in deionized water at 70°C with a mass score of 40% and then poured into the mold window. The gelatin/sample module was placed in an environment of 25 ± 1°C and 65 ± 2% humidity for 24 h. Next, the material testing machine (Instron 3365, USA) was used to complete the stripping of gelatin from samples. When the gelatin was stripped from the sample, the gelatin and sample were at 180° and the stretching speed was 100 mm/min (Figure 2(b)). Eventually, Origin 2018 software was used to calculate the integration of the area surrounded by the curve and the (a) PTFE mold for adhesion resistance test and (b) Stripping test diagram.
Abrasion resistance measurement
The abrasion resistance measurements were implemented in reference to ISO 105-X16:2016. 30 The standard stick-lined cotton fabric was fixed at one end of the friction color fastness tester (Fang Yuan Instrument Y571 B, China). The sample was coated on the friction head and pressed with a pressure of 56 kpa in contact with the standard stick-lined cotton fabric, as shown in Figure 1(e). Then the sample was moved for 10 cm forth and 10 cm back as one friction stroke. After 10 friction strokes, the water CA of sample was measured, and the changes of sample hydrophobicity were compared and analyzed.
Statistical analysis
Three measurements were performed per sample, and the results were averaged. Data were expressed as the mean with standard deviation (SD). A 5% significance level was used for all analyses. The error bars in the figures were the standard deviation of data.
Results and discussion
Effect of PDMS spraying on materials
Orthogonal experiment results.

The bold values are the best experimental group analyzed in the subsection “Effect of PDMS spraying on materials”.
Surface morphology
Figure 3 shows the appearance of four types of mesh viscose spunlace samples (S1, S2, S3, S4), and they correspond to original sample, samples modified by micro-nano SiO2, PDMS or micro-nano SiO2/PDMS under the optimized process conditions. Notably, the viscose fibers in the material are entangled together and the whole material has the apparent mesh structure. The longitudinal surface of viscose fiber has obvious grooves (Figure 3 S1). Meanwhile, these modification methods have almost no effect on the macroscopic pore size of samples. Apparent morphologies of four types of samples (S1-S4).
After the in situ growth of nano SiO2, the fiber surface is filled with multi-scale micro-nano particles, which results in obviously rough fiber surface (Figure 3 S2). This may be due to the fact that nano SiO2 particles are combined in the fiber surface for in situ growth. In particular, the unoccupied hydroxyl group on the fiber surface and the adsorbed water molecules are both acted as reactants to promote the formation, nucleation and growth of new nano SiO2 particles. However, due to the different degree of polymerization, multi-level micro-nano rough structures are finally formed on the fiber surface.
After PDMS modification, the entanglement and cohesion among fibers are still obvious, and no bond is caused by being sprayed. It is noticeable that PDMS has been arranged on the surface of viscose fiber. The fiber surface is generally smooth, and the fiber grooves are filled with PDMS. Although the local area has been filled, the groove outline of viscose fiber is still visible (Figure 3 S3). The main reason is that PDMS can be effectively crosslinked with cellulose material. 19 Meanwhile, the specific surface area of the groove on the surface of viscose fiber is large, which leads to strong physical adsorption of PDMS.
Figure 3 S4 displays the morphologies of samples modified by micro-nano SiO2/PDMS. Notably, there is no adhesion or blockage of interfibrous pores due to modification, and the entangled fibers in the sample have still obvious entanglement and rough surface structures. Although the surface grooves of viscose fiber still exist, they are not obvious and are only visible at high magnification. Because there are a lot of gaps among the micro-nano SiO2 particles on the surface of viscose fiber. When the material is sprayed with PDMS, PDMS can easily fill these gaps, especially the surface grooves of viscose fiber. In addition, PDMS can be easily crosslinked with viscose fiber and micro-nano SiO2 particles after being heated.
Material TG analysis
Figure 4(a) (a) TG curves of samples and (b) Sample weight of each process.
Material chemical structure
Figure 5 FTIR spectra of four types of samples (S1-S4).
Covalent grafting of cellulose with PDMS
Figure 6(a) exhibits the FTIR spectra of samples treated by PDMS spraying. It is noticeable that the infrared transmittance of sample without being heat-treated and washed is low. After being washed, the overall bands of sample rise in the range of 500-1800 cm−1, and the infrared transmittance increases. Previous studies
33
show that the two peaks at 800 cm−1and 1260 cm−1 correspond to the vibration of Si-O and Si-C bonds, respectively. They present lower vibration compared with being washed sample according to the local magnified bands in Figure 6(b). It obviously indicates that part of the spray solution has been washed away. When the samples are washed, the bond vibration of samples without being heat-treated decreases more obviously than that of the treated samples. This indicates that the bonding strength of sprayed PDMS with cellulose is weak without heat treatment. No covalent bonding is formed between PDMS and cellulose, but a simple physical adhesion. After being heat-treated, PDMS not only relies on its own viscosity, but chemically reacts with cellulose to form covalent connection, which further enhances the bonding fastness. The covalent grafting mechanism of cellulose and PDMS can be seen in Figure 6(c). (a) FTIR spectra of PDMS spray modified samples, (b) Local magnified FTIR spectra and (c) Schematic diagram of covalent grafting of cellulose and PDMS.
Material tensile analysis
Figure 7(a) and (b) Sample tensile properties: (a) Breaking strength and (b) Elongation.
Adhesion resistance analysis
Figure 8(a) (a) Sample Photos after being peeled and (b) Peeling energy of samples.
Material durability analysis
Figure 9 describes the wettability diagram of four types of samples with/without friction. S1F, S2F, S3F and S4F correspond to S1, S2, S3 and S4 samples after friction respectively. It is obvious that S1, S1F, S2 and S2F have excellent wettability. On the contrary, S3, S3F, S4 and S4F possess low wettability. In addition, the wettability of samples has little change by friction. Meanwhile, the water CAs of S1 and S1F are 29.8 ± 2.1° and 28.7 ± 2.6° respectively. It is mainly due to the great water absorption and excellent wettability of viscose fiber. Meanwhile, the water CA of S2 and S2F are 25.9 ± 2.0° and 23.8 ± 2.1°, respectively, which are both slightly smaller than those of S1 and S1F. The difference in water CAs of samples is significant ( Sample wettability and water CAs with/without friction.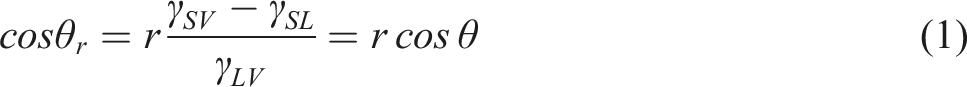

On the other hand, the water CA of S3 is 136.4 ± 2.9°, and the water CA of S3F is 130.8 ± 3.2°. Obviously, the sample water CA decreases slightly after friction treatment. According to previous studies, the natural property of solid surface 


Equation (5) can be obtained by combining the young-Dupre equation (3). In combination with the WSL of equation (4), equation (6) can be received.

Therefore, the cos 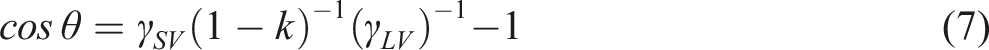
According to equation (7), when
After micro-nano SiO2/PDMS modification, the water CA of S4 is 151.0 ± 1.1°. Obviously, it indicates that micro-nano SiO2 and PDMS synergistically improve the material hydrophobicity, making the material have super-hydrophobicity. At the same time, the water CA of sample is still up to 139.4 ± 1.4° after friction treatment. Although the water CA of material decreases about 11.6°, it still has great hydrophobicity compared with S3. The main reason is that PDMS has great flexibility, and can be firmly cross-linked and grafted with viscose and SiO2, and micro-nano SiO2/PDMS synergistically improves the material durability. 20
Conclusions
In summary, the hydrophobic modification method of the contact layer of anti-adhesion dressing had been proposed. The mesh viscose spunlace nonwoven was used as the dressing contact layer, and the material was modified by sol-gel and in situ growth of nano SiO2. At the same time, PDMS was sprayed to modify material, and the optimal treatment conditions (PDMS concentration was 1%, heat treatment temperature was 120°C and treatment time was 12 h) were obtained by orthogonal experiment. Material possessed the optimal hydrophobicity and its water CA was 151.0 ± 1.1°. In addition, PDMS was demonstrated to be covalently grafted onto viscose fiber of material. The MD and CD strength of material decreased but the elongation increased slightly. The MD peeling energy was 129.7 ± 33.9 J/m2, and the CD peeling energy was 98.1 ± 38.3 J/m2, which fully met the requirements of anti-adhesion of dressings. After 10 cycles of friction movement, the water CA of material was still up to 139.4 ± 1.4°. Moreover, micro-nano SiO2/PDMS modification could be used to effectively construct hydrophobic and durable properties of material without affecting the mesh structure of spunlace nonwoven. This paper could provide reference for the development of anti-adhesion spunlace nonwoven-based dressing contact layer.
Footnotes
Acknowledgments
The author(s) wish to express their sincere gratitude to Zhejiang Hezhong Technology Nonwoven Co., Ltd and Hangzhou Hanford Technology Co., Ltd for their support in providing the means for conducting experiments.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by the National Natural Science Foundation of China Youth Found [grant number 51903156], the Zhejiang Provincial science and technology Department research and development project [grant number 2022C01SA691301], the Student Scientific Research Project of Shaoxing University [grant number Y20220702], the National Innovation and Entrepreneurship Training Program of College Students [grant number 202210349056S and 202310349040] and the Science and Technology Guiding Project of China National Textile Industry Council [grant number 2022027].
