Abstract
In this study, structural, thermal, mechanical and water uptake properties on the influence of alkaline treated cellulose nanoparticles (CNPs) filled starch-based bioplastic film is examined. Starch-based bioplastic films filled with alkaline treated CNPs at various concentration (0 to 5 wt%) were prepared by solution casting method. Thermal properties of film series are examined by Differential Scanning Calorimetry (DSC) and Thermogravimetry analysis (TGA) method. Alkaline treated CNPs addition induces crystallization to the bioplastic film. Degradation onset temperature (Don), 10% mass loss (T10) and 50% mass loss (T50) values show the positive effect of CNPs addition into the biopolymer matrix with improved thermal stability. Tensile result shows that about 2.5 times increased is observed in 2 wt% CNPs filled bioplastic films. Water uptake result shows that the alkaline treated CNPs starch-based bioplastic film limit the biopolymer dissolution time and extended the breaking down of the bioplastic film. Leading to about 2.5 times reduced equilibrium water mass uptake is observed in 3 wt% CNPs filled bioplastic film when compared with unfilled bioplastic film.
Keywords
Introduction
In recent years, much awareness has been created in developing eco-friendly biodegradable materials to mitigate environmental damage, pollution and preserve natural resources.1,2 Amylose/amylopectin-based starch polymers, lignocellulosic based plant fibers, poly(lactic) acid, chitin/chitosan-based polymers, etc. have gained prominent importance in the class of biodegradable polymeric materials.3–5 Cellulose is one of the commonly used biomaterials due to their high structural and mechanical properties. Cellulose is an extract from plant sources with a chemical structure that contains beta (1-4) glucose linkages. Cellulose can be processed into different kinds of nanostructures or nanoparticles (CNPs) such as bacterial nanocellulose, cellulose nanocrystals (CNC), cellulose nanofibrils (CNF), and electrospun nanocellulose.6,7 In general, cellulose nanoparticles (CNPs) are extracted by chemical treatments such as acid hydrolysis, steam explosion and micro-fibrillation methods, with and without the assistance of temperature and pressure. CNPs possesses interesting characteristics as nano-filler materials due to their high aspect ratio, defect free structure, low dense with high mechanical properties and structural properties.8–10 Starch-based polysaccharide polymer formed by alpha (1-4) glucose linkage, chemical structure consists of amylose and amylopectin. The ratio of amylose to amylopectin determines the mechanical, thermal and barrier thermal properties. Starch can be extracted from wide sources, namely, corn, potato, rice, husk, wheat, fruits, etc, which in turn provides various options in selecting amylose/amylopectin ratio.11–13
In starch based cellulose particles reinforced biocomposites, various work has been carried out on the effect of different types of starch (amylose/amylopectin ratios), plasticisers, nanocellulose, processing methods, chemical treatments and conditions.14–23 Improved physical, thermal, mechanical, barrier, antimicrobial and toxicity properties were observed.24–29 Application of these biocomposites on coating of packaging paper, muffin liner, biomedical vascular application, hemorrhage, therapeutics, implants, etc., has been recently demonstrated.19,20,22,30–32
However, few drawbacks of cellulosic fillers are its hydrophilic characteristics which induce incompatibility with hydrophobic matrix polymer, thereby affecting the dispersion and adhesion properties. One way of improving this effect is to enhance the compatibility by chemical treatments and fibrillation of cellulosic particles. Chemical treatment can induce improved filler matrix adhesion and compatibility mechanisms. The fibrillation process will reduce the particle size and therefore result in large number of smaller microfibrils with reduced diameter and increased surface area. Fibrillation could increase the dispersibility of the fillers in the polymer matrix. A recent study on alkaline treatment of cellulose particles has shown increased fibrillation of microfibers at controlled solute concentration.33–35 8% of alkaline treatment resulted in high fibrillation with decreased fiber diameter and retainment of its original cellulose-I structure. Whereas 16% alkaline treatment resulted in complete hydrogelation with conversion of cellulose structure from cellulose I to II. The rationale for this work is to improve the adhesion of bioplastic film by means of dispersion of alkaline treated CNPs in starch biopolymer. The CNPs fibrillation is achieved by 8% alkaline solution treatment. Although the alkali treated CNPs has been established, however, the alkali treated CNPs as filler in biopolymer is not yet been fully explored. Hence, this study provides an understanding on the effect of alkaline treated CNPs dispersed in starch-based biopolymer. This improves the biopolymer surface adhesion due to increased CNPs dispersion. This green and renewable materials have high potential in medical and automotive based textile industries where high strength and absorption properties are required.
Materials and methods
Raw materials
Commercial grade cornstarch (C6H10O, with chemical code S8250) dry powder and glycerol in liquid form were purchased from Laboratory Consumables Chemicals Supplies, South Africa. Dried banana fibers which were extracted from stem section of the plant was purchased from Reddcolt enterprises, India. The purchased fibers were in form of single long fiber of ∼1 m length. The fibers were stacked together and received in the bundle form. All other chemicals used for cellulose nanoparticles (CNPs) extraction, such as, sulphuric acid (H2S04) and Sodium hydroxide (NaOH) pellets were obtained from Merck Chemicals, South Africa.
Extraction of cellulose nanoparticles and further treatment
CNPs were extracted from banana fibers using acid hydrolysis process. Each long and dried banana fibers were cut into lengths of ∼5 mm, followed by CNPs extraction. CNPs extraction process involved 3 steps, namely, washing, bleaching and acid hydrolysis. In washing treatment, the chopped banana fibers, weighing 300 g were placed in hot water at 60°C for 0.5 h to disentangle the fiber structure. Fibers were then removed and washed in the running tap water to eliminate waxy substance and other unwanted phases. In the bleaching process, washed fibers were soaked and rinsed in 300 mL of 6.0% sodium hypochlorite solution. 3 wt% of fibers were soaked in the sodium hypochlorite solution for 1 h and then bleached in running tap water. Fiber variation was done to control the ratio of fiber to sodium hypochlorite solution. The fibers were then removed and placed in an oven at 60°C for 4 hours. In the acid hydrolysis process, 10% (35 mL) of sulfuric acid (H2SO4) was mixed with 365 mL of distilled water in a beaker. Thereafter, 7 wt% bleached fibers were taken from the initials 3 wt % treated with sodium hypochlorite solution, then soaked into the acid solution, stirred using a mechanical stirrer for 30 min. Thereafter, the excess sulphuric acid liquid solution was extracted by a vacuum process. The acid hydrolysed fibers were then washed with distilled water and dried in an oven at 60°C for 4 h. In the above processes, cellulosic phase in the banana fiber was extracted and all other non-cellulosic phases such as lignin, hemicellulose, etc., were eliminated.
Following the cellulose extraction, the CNPs were further alkaline treated using NaOH solution. 24 g of NaOH pellets was dissolved in 300 mL (8:100, NaOH to distilled water content ratio). After which 5 wt% were taken from the overall prepared CNPs and deposited in ∼250 mL NaOH section of the solution in a beaker. The beaker with CNPs and NaOH solution was then placed in an ultrasonic agitator (MRC laboratory Equipment, UK. Model: DC-150H, operating at 40 kHz with ultrasonic power 150 W) at 45°C for 4 h. The agitated CNPs were filtered from NaOH solution using filter paper. Following this, the CNPs were washed with running water and dried in oven at 60°C for 4 h to eliminate moisture. The dried CNPs were further subjected to manual grinding to break the agglomerated particles and improve the dispersion in starch polymer. Grinding was carried out manually using ceramic mortar-pestle to reduce the diameter from micro to nano-size at 15 minutes different time interval, for a period of 3 h.
Processing of starch based composite films
Unfilled and treated CNPs filled corn starch biofilm composite were prepared by plasticization method. A total of five set of biocomposite films with different concentration of CNPs in starch (0.5, 1, 2, 3 and 5 wt%) and neat unfilled starch film was prepared. The processing of biofilm was similar to the method adopted in our earlier studies.36,37 Cornstarch powder (5 g) was dissolved in 80 mL distilled water. The solution was maintained at 80°C and stirred at 500 r/min using a temperature-controlled magnetic stirrer for about 10 min. Following this, glycerol (1.5 g) was added to the solution. Thereafter, treated CNPs with desired concentration to that of the combined weight of cornstarch and glycerol) was individually added into the heating mixture. The whole mixture was stirred for about 45 min, and after the formation of a white viscous solution the mixing was stopped. The solution was then poured into an open-end metallic mould and left in an oven at 60°C overnight. During this process, the water molecules evaporated leaving behind a thin film on the mould. The mould was then removed from the oven and the film was hand peeled from mould for characterization and testing. Wax was used as the release agent. A uniform film thickness (0.2 mm ± 5%) was maintained in all the unfilled and CNPs filled biopolymer film series.
Characterization
A scanning electron microscopy (SEM) was used to analyse the fracture surface morphology of biofilm series. Fracture surfaces were examined by Zeiss Environmental SEM (ESEM: model EVO HD 15 operating at controlled pressure conditions at 20 kV). Before conducting the actual SEM surface analysis, specimens were surface coated with gold using Quorum-150R ES model thin film coating equipment. A high-resolution transmission electron microscopy (TEM) was used to study the dispersion of CNPs in the starch polymer matrix. A brightfield TEM image mode was carried out on an ultrathin microtomed nanocomposite specimen using JEOL HR-TEM (JEM-2100 series), operating at 120 kV.
Thermogravimetric analysis (TGA) and Differential Scanning Calorimetry (DSC) were together measured using TA instruments SDT Q600 model. In TGA, thermal properties of nanocomposites such as weight loss, decomposition temperature and degradation were evaluated. In DSC, the crystallization temperature during first heating was measured. In TGA/DSC analysis, ∼5 mg of the sample was placed in an alumina crucible of TA apparatus and the sample was scanned from RT to 600°C at a scanning rate of 10°C/min under atmospheric condition.
FTIR (Nicolet) analysis was carried out for CNPs, neat starch film and CNPs filled starch biocomposite film series. Scanning was carried out in an attenuated total reflectance (ATR) mode to study the functional group, transmittance peak intensity and bonding characteristics of film series.
Testing
The tensile test of film series was conducted to study the tensile modulus, strength and elongational properties. The tensile test was conducted as per ASTM D3039 standard test, with specimen dimension of 5 cm gauge length × 1 cm width × 0.2 mm thick. The tensile test was conducted using an MTS UTM Tensile Tester (Model LPS 304 – 424,708 series) with a crosshead speed of 1 mm/min and 1 kN load cell. A mean value of tensile property of three specimens was selected and considered for analysis.
Water uptake properties of nanocomposites was studied by a water immersion test method, as per ASTM D570-98 (2005) test procedure at 25°C. Three test specimens with each of dimension 3 cm × 3 cm × 0.2 mm was chosen for this study. In order to eliminate surface or subsurface entrapped moisture and retain actual solid mass in the specimen, which will allow consistency in weight measurement during water absorption test. The test samples were dried at 60°C for 4 h in an oven. During the 4 h drying, samples were taken out of the oven at an interval of 1 h and immediately transferred into the airtight desiccator. The rapid cooled sample, having reached room temperature (RT) was weighed and ensured that the mass loss remained constant until the 4 h heating cycle.
The actual solid mass sample was then immediately taken out of the desiccator and fully immersed in a distilled water medium which was placed in a temperature-controlled water bath set-up. The bath temperature was constantly maintained at 25°C for entire duration of the water immersion test. The water-soaked specimen was taken from the water bath at different time intervals and wiped using a paper towel to eliminate surface water. The sample was then weighed in an electronic balance and then immediately transferred back into the water bath set-up. This weighing procedure was repeated until the water-soaked sample showed no or negligible increase in the water mass uptake (i.e. equilibrium water uptake content, We). The We was measured as per equation (1)
Water contact angle (WCA) testing of unfilled and CNPs filled biofilm series was carried out using Kruss Drop Shape Analyser (DSA) 100 (Hamburg, Germany), as per ASTM D5946. Stainless steel needles (diameter: 0.5 mm) were purchased from Kruss. Distilled deionized water (conductivity: 0.333 μS/cm) from CSIR (SA) water filtration system (Evoqua Water Technologies, Pittsburg, USA) was used throughout the experiments. Sessile drop methodology was used to analyse the WCA of the materials.
Results and discussion
Structure and morphology
Figure 1 shows the SEM images of fibrillated and grinded. According to the SEM images, Figure 1(a) shows the surface feature of non-alkali treated fibrillated CNPs, while Figure 1(b) shows alkali treated fibers fibrillated CNPs. The diameter of an extracted fibrillated CNPs varies from 0.1 μm (100 nm) to 0.5 μm (500 nm), while the length of the fibrillated CNPs varies from 0.5 μm (500 nm) to 5 μm (5000 nm). Whereas alkaline (NaOH) treatment reduced the fibrillated CNPs size (diameter and length). The diameter of alkaline treated fibrillated CNPs varies from 0.05 μm (50 nm) to 0.2 μm (200 nm). Length of this fibrillated CNPs also varies from 0.1 μm (100 nm) to 1 μm (1000 nm). This also indicated high level of fibrillation in the alkaline treated CNPs. The alkaline treatment of CNPs also affected the aspect ratio (ratio of length to diameter) of the fiber. As extracted CNPs shows average aspect ratio of 12, while the alkaline treated CNPs shows average aspect ratio of 24. The improvement in the aspect ratio was increased by 50% after alkaline treatment. Aspect ratio was measured using image analysis. The increased aspect ratio of alkaline treated CNPs suggests higher fibrillation, less defect and improved structural properties. Likewise, in the same Figure 1, the surface morphology of the grinded cellulose nanoparticles CNPs was examine by SEM. Figure 1(c) shows the surface feature of non-alkaline treated CNPs while Figure 1(d) shows the alkaline treated CNPs. This fibrillation effect tends to positively influence dispersion properties of CNPs when they are filled in starch biofilm matrix.
38
In addition, high aspect ratio (length to diameter) and surface area of alkaline treated CNPs is expected to improve particle matrix interface adhesion and load transfer characteristics. SEM image of fibrillated CNPs, (a) untreated, (b) alkaline treated, (c) grinded untreated and (d) grinded alkaline treated.
EDX elemental values of grinded untreated and grinded alkaline treated CNPs.

Note: CK = carbon and OK = oxygen.
TEM images of untreated CNPs and alkaline CNPS are shown in Figures 2 and 3 respectively. Although their morphology may look similar but not in their entirety. The CNPs were embedded in the polymer matrix at the similar percentage weight variations (0 – 5 wt %) with improved compatibility. The improvement of the bioplastic with the addition of CNPs shows that Alkaline treated CNPs morphology are much better than the untreated CNPs. Figure 2 shows the TEM image of different weight concentration of as extracted CNPs filled starch biofilm composite. The bright continuous phase represents matrix phase while dark discontinuous phase represents CNPs phase. The dispersion of CNPs differs as a function of its concentration in matrix polymer. Figure 3 shows the TEM image of different weight concentration of alkaline treated CNPs filled starch biofilm composite. On comparing Figures 2 and 3 with their corresponding fillers weight concentration, the result clearly shows that the alkaline treated CNPs fillers resulted in much improved dispersion with highly fibrillated particles when compared with an extracted CNPs filled biofilm composite series. The result also shows the dispersion depends upon the concentration of the fillers, as the higher content CNPs (> 2 wt%) fillers tend to cause agglomeration in the matrix polymer. Forthcoming thermal and mechanical properties are carried out only for alkaline treated CNPs filled biofilm composite series and their results are discussed. TEM image of corn starch (CS) film filled with (a) 0.5 wt%, (b) 1 wt%, (c) 2 wt%, (d) 3 wt% and (e) 5 wt% untreated CNPs. TEM image of cornstarch film filled with (a) 0.5 wt%, (b) 1 wt%, (c) 2 wt%, (d) 3 wt% and (e) 5 wt% alkaline treated CNPs.

Figure 4 shows the FTIR image of treated CNPs, neat cornstarch film and treated CNPs filled starch films. The characteristics peaks obtained is typical to glucose molecular structure. Treated CNPs shows characteristics peaks at 3289 cm−1 (due to -OH hydroxyl stretching vibration at inter/intra molecular chain), 2924 cm−1 and 2851 cm−1 (due to CH, HCH and COH symmetrical or asymmetrical vibrations), 1645 cm−1 (due to water or moisture molecule) in treated CNPs, cellulosic characteristics peak at 1456 cm−1, 1152 cm−1 and 781 cm−1(due to C-O-C glycosidic linkage in cellulose), 1008 cm−1 to 1054 cm−1 (due to C-O strain of cellulose) and 862 cm−1 C-H strain of beta-glucose linkage of cellulose.
39
Starch film shows characteristics peaks at 3291 cm−1 (due to hydroxyl -OH vibration), 2919 cm−1 and 2849 cm−1 (due to CH, HCH and COH symmetrical or asymmetrical vibrations), 1639 cm−1 (due to water or moisture molecule), typical characteristics of starch molecule at 1408 cm−1, 1335 cm−1, 926 cm−1 and 730 cm−1, 1150 cm−1 and 1052 cm−1 (due to C-O vibration of COH and C-O-C), peaks at 981 cm−1 due to C-C vibrations, 850 cm−1 due to alpha-glucose linkage of starch.40,41 FTIR results further shows that the peaks of starch and cellulose are similar as the prime molecular structure are emerging from glucose molecules. A little shift in wavenumbers between these two molecular vibrations could be due to nature of structural characteristics. FTIR spectrum of treated CNPs, neat cornstarch and CNPs filled biofilm series.
FTIR spectrum of all the composite film series appeared similar with little change in peak intensity values. All treated CNPs filled biocomposite film shown similar peak value, in particularly -OH vibration peak indicating increased fibrillation due to NaOH treatment. This fibrillation would have induced improved CNPs and starch polymer matrix adhesion and expose more CNPs surface area and ultimately providing more hydroxyl interaction site with polymer matrix. At the -OH hydroxyl peak, the percentage transmittance value for all CNPs filled bioplastics at 0.5, 1, 2, 3 and 5 wt% resulted in percentage increase of 6%, 11%, 13%, 6%, 9% respectively when comparison to cornstarch bioplastic film -OH hydroxyl peak. Whereas, at higher CNPs content, the -OH hydroxyl peaks tend to be reduced, this suggests lower fibrillation and CNPs-matrix interface adhesion. Moreover, in all composite series, cellulosic peak of 1152 cm−1 due to the absence of glycosidic linkage, including the breaking of this bonding and interaction of CNPs and matrix phase, suggest improved interaction between filler and matrix phases (as also observed from TEM result).
Thermal properties
Figure 5 shows the DSC heating curve of unfilled and treated CNPs filled starch biofilm series. Unfilled starch biofilm shows no exotherm or endotherm up to 180°C, which suggests polymer is in amorphous phase. The absence of crystallization effect could have resulted in no melting. It is well noted that the melting point of starch is around 110°C to 120°C.
42
The processing effect due to plasticizer and water molecules would have hindered crystal formation and hence possibly resulted in amorphous polymer. However, in treated CNPs filled biocomposite film, exothermic crystallization peak is observed at 55°C. It can be observed that variations in T
g
of 55°C, 57°C, 58°C, 56°C and 54°C were found in 0.5, 1, 2, 3 and 5 wt% treated CNPs filled bioplastic films respectively. This could be due to the presence of CNPs particles. Possibly CNPs may act as nucleation site and induce crystallization of starch molecules. A broad exothermic peak was also observed from 105°C suggesting melting of crystallized phase of starch polymer. DSC heating curve of unfilled and treated CNPs filled biofilm series.
Figure 6 shows the TGA curves of the treated CNPs, unfilled and treated CNPs filled biofilm series with their TGA properties shown in Table 2. The thermal stability of CNPs is higher than that of unfilled and CNPs filled starch series up to degradation on-set temperature (Don). However, beyond degradation end-set temperature (Dend), CNPs resulted in highest mass loss. As the CNPs being nano-fibrillated, more amount of surface area would have exposed to heat and resulted in higher level of mass loss. Similar result on nanocellulose degradation was observed when compared with its initial plant fiber source,
43
due to increased surface area. The CNPs addition has positive effect on improving thermal stability (Don and Dend) of starch polymer. Whereas the improvement depends upon CNPs concentration. 2 wt% CNPs filled biofilm resulted with better and improved TGA properties when compared with other series. Possibly, a well dispersed and interfacial properties might had caused this improvement. Table 1 also shows 10% (T10) and 50% (T50) mass loss temperatures of unfilled and CNPs filled biofilm series. T10 temperature of CNPs filled biopolymer was lower when compared with unfilled biofilm, which could be due to the presence of higher moisture content in these materials. CNPs and starch are prone to high water/moisture absorption due to their large amount of hydroxyl group and might had resulted in this effect. Biofilm with 0.5 wt% CNPs and 2 wt% CNPs appeared out of range from the other film series results. As T10 temperature also depends upon so many other factors such as initial debonding between fillers and matrix, initial moisture content and initial dissociation, etc., which could have all resulted in this uncertain trend. However, T50 temperature of CNPs filled biofilm series shows improved mass gain when compared with unfilled biofilm. All biofilms are expected to have lost mass in terms of weight, related to the temperature exerted. Higher temperature values in CNPs filled biofilm is an indication of mass gain, which could be possibly due to high thermal stability of CNPs. Moreover, the trend of these properties (T50, Don and Dend) appear to be in sequence. TGA curve of unfilled and treated CNPs filled biofilm series. DSC and TGA properties of CNPs, unfilled and CNPs filled biofilm series.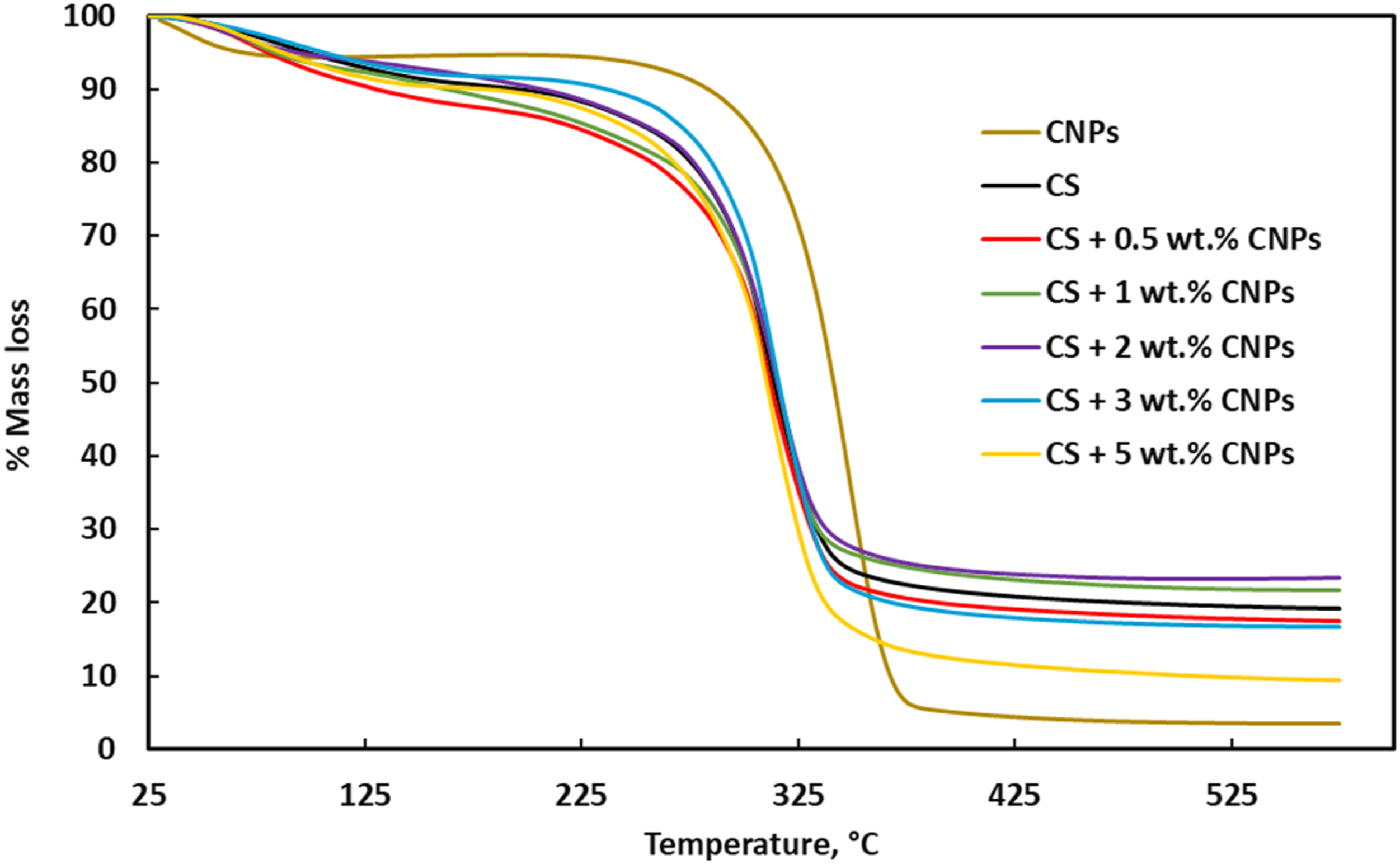

Tensile properties
Figure 7 shows the tensile stress-strain curves of unfilled and treated CNPs filled film series. The results shows that the tensile properties such as modulus, strength and elongation at break of biofilm were affected due to the CNPs addition. In general, a positive improvement is observed at around 2 wt% CNPs content, whereas at 0.5 wt% and 5 wt% the modulus and strength values were decreased. The result on failure strain values resulted in mixed trend. Table 3 shows the values of tensile properties of unfilled and CNPs filled biofilm series. About 2.5 times increased tensile modulus and strength values was observed at 2 wt% CNPs filled biofilm. This effect could be due to effective particle distribution, particle-matrix interface adhesion and high level of fibrillation as observed in TEM and FTIR studies. Tensile stress-strain curves of unfilled and treated CNPs filled biofilm series. Tensile properties of unfilled and CNPs filled biofilm series.

The tensile fractography analysis of biofilm series was examined by SEM analysis. Figure 8 shows the tensile fracture surface of unfilled and CNPs filled biocomposite film series. Neat unfilled biofilm shows extended and deformed fracture surface. Possibly the plasticising effect of glycerol, hydroxyl group of starch and water molecules would have resulted in large deformation to failure. This trend continued even for 0.5 wt% CNPs filled biofilm. However, 1 wt% to 3 wt% CNPs filled biocomposite film resulted in decreased elongational properties. The fractography (Figure 8(b)-(d))) shows different kind of fracture surface than that of neat unfilled polymer fracture surface. The CNPs phase (as seen by discontinuous bright phase) possibly reduced the elongational characteristics of polymer due to their rigid phase morphology. Clear visibility of diversion of crack propagation at fiber-matrix interface in CNPs filled biofilm was observed. This suggests improved strength and arrest of deformation resulting in reduced elongational values. Biofilm with 2-3 wt% CNPs content resulted in optimum tensile properties. At 5 wt% CNPs content, possibly the agglomeration of CNPs had exposed more matrix phase and resulting in the similar failure pattern of unfilled biofilm. This is further confirmed by SEM image of 5 wt% CNPs filled biofilm (Figure 8(e)) which shows similar fracture surface as that of unfilled biofilm. SEM of tensile fractography image of (a) unfilled biofilm, (b) 0.5 wt%, (c) 1 wt%, (d) 2 wt%, and (e) 5 wt% treated CNPs filled biofilm series.
Water uptake property
Figure 9 shows the water uptake characteristics of unfilled and treated CNPs filled starch biofilm series. The water uptake curve shows three stages of absorption, namely, initial, mid and end absorption. During initial stage, there was a linear increase in water uptake corresponding to the period of 5 h1/2. Following this, mid stage occurred, where a constant water uptake (or negligible water mass uptake) was noticed. This mid-stage presented until the period of 11 h1/2. In the end-stage, a rapid increase of water content with respect to time and followed by reduction in mass suggesting the dissolution of polymer in water. The rapid increase in water content was observed in smaller time unlike the initial phase time periods.
44
During the dissolution stage, CNPs addition has positively enhance the starch polymer against mass loss. The water uptake curve provides an insight of water permeation into the biofilm samples. The linear increase suggests a gradual flow (or permeation) of water molecules into the sample. The mid stage suggests that the CNPs particles acts as a barrier medium while resist the further water uptake over a certain time (i.e., until the end of mid stage). There upon, possibly CNPs might yield to the permeation of water molecules and resulted in rapid increase of water uptake and subsequent dissolution. Water uptake curves of unfilled and treated CNPs filled biofilm series.
Water uptake properties of unfilled and CNPs filled biofilm series.


Water contact angle (WCA) of: (a) unfilled starch, (b) 0.5 wt % (c) 1 wt % (d) 2 wt % (e) 3 wt % (f) 5 wt % treated CNPs filled biofilm series.
Conclusions
The main objective of this work is to improve the dispersibility and adhesion properties of CNPs in starch polymer matrix, by inducing NaOH treatment into the CNPs. The following conclusion can be drawn based on the present study: • Increased surface area and fibrillation is observed in NaOH treated CNPs when compared with as untreated CNPs. Furthermore, effect of treated CNPs on thermal, mechanical and water uptake properties of starch polymer is examined. Although there are limitations in term of its hydrophilic characteristics which induce incompatibility with hydrophobic polymer, thereby affecting the dispersion and adhesion properties. The importance of this work is to improve the dispersibility of the alkaline treated CNPs in the biopolymer matrix by means of creating highly fibrillated CNPs. • DSC result shows that the alkaline treated CNPs addition induces crystallization effect into the starch polymer at the peak of 55°C. Hence, showing improved thermal stability. • TGA result shows that T10 temperature of CNPs filled biofilm series is lower than of the neat starch film, except for 2 wt% CNF filled biocomposite film. The T50 temperature resulted in marginal effect due to the CNPs addition in biofilm. However, improvement was observed in Don temperature of CNPs filled biofilm, suggesting improved higher temperature processibility. About 50°C improved degradation on-set temperature (Don) was observed in 3 wt% CNPs filled biocomposite film. Whereas CNPs effect on Dend shows negligible effect on biocomposite film, due to the exposure of large surface area of fillers and hence aiding heat flux and degradation. • Tensile result shows that ∼2.5 times increased modulus and strength values at 2 wt% CNPs filled biocomposite film. Moreover, comparable elongational values obtained at 2-3 wt% CNPs filled biocomposite film. • Water uptake properties of biofilm series suggests that fibrillated CNPs reduces equilibrium mass uptake of starch biofilm by 50% at 3 wt% CNPs filled biofilm and with no dissolution at 2 wt% and 5 wt% CNPs filled biofilm. The addition of CNPs also limited the dissolution time of starch polymer. • WCA result shows the hydrophilic characteristics of CNPs filled biopolymer film composite. Hence, the addition of CNPs fillers decreased the water contact angle with biofilm surface. Following that the CNPs is in nano size, more amount of hydroxyl group would have exposed on the biofilm surface.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the National Research Foundation of South Africa (grant number 137949).

