Abstract
Oil sorbents play a significant role in the treatment processes of oil spills, therefore, enhancing the oil-water separation capacity of sorbents has been charming the consideration of scientific researchers. To improve the properties of oil sorption and simplify the process of oil recovery, many advanced oil sorbent devices have been projected recently. Different types of materials are used as sorbents for oil-water separation, however, up to now, there is no review paper focused on textile (woven, knitting, and nonwoven) sorbents, and compared with them. In this review paper, the classification of textile fabrics, properties of surface wetting, fabrication methods of textile sorbents for oil spill cleanup, their properties, and recent applications in the field of oil cleanup are studied. We believe this review can serve as an essential reference for the formation of textile sorbents, their properties, and their applications in the field of oil spill cleanup.
Keywords
Introduction
The rapid-global growing energy has enthused the fast expansion of using oil in industries, resulting in an increase in the number of oil spill accidents during oil production, transport, exploration, and storage.1,2 The awareness of scientific researchers about offshore oil spills is increasing due to substantial economic losses and environmental worries that have caused human health dangers.3,4 These spills occur during exploration, production, transport, usage, storage, operational failures tools failures, and natural disasters.5–7
Different factors affect the ability of an oil spill to spread including surface tension, viscosity, and specific gravity. The higher the oil’s surface tension and viscosity, the more likely a spill will remain in place. Since water is heavier than most oils, they float on top of it. However, the specific gravity of an oil spill can increase if the lighter substances within the oil evaporate. Heavier oils, vegetable oils, and animal fats may sink and form tar balls or may interact with rocks or sediments on the bottom of the water body.8,9
The “Deepwater Horizon” oilrig in 2010 is a good example of the risks of an oil spill. Almost 4.9 million barrels of crude oil spilled into the Gulf of Mexico due to uncontrollable crude oil blowout.10,11 This disaster damaged ecosystems of local marine life resulting in changing in the ecological and eliminating of rare species.
12
Besides numerous oil spill accidents, the increase in the quantity of oily wastewater generated by different industrial processing (i.e, food,
13
daily chemical,
14
petrochemical,
15
leather,
16
textile,
17
metal finishing,
18
steel processing
19
) and the seepage of organic solvents (i.e, benzene, toluene, chloroform, dichloromethane, cyclohexane) also threaten the earthly ecosystem.
20
Furthermore, a wide range of fresh water can be polluted by organic solvents or oil leading to the freshwater crisis21,22 (Figure 1). Consequently, using advanced methods for oil cleanup has become a necessity and rapidly expanding issue in both of research community and industry area. Different traditional methods have been used to solve the environmental problems caused by oil spills, industrial oily wastewater, and organic pollutions such as mechanical,23–26 chemical,27,28 and biological techniques.
29
Real images of the effect of oil spill accidents.
Mechanical techniques include barriers, skimmers, oil booms, and synthetic as well as natural sorbents create an ideal mechanical cleanup system, which is the major line of protection against organic solvents or oil spills.30–32 Nevertheless, it is difficult to remove the entire released oil using those devices owing to their large size and insufficient separation efficiency. Regarding chemical techniques, they include in situ burning, chemical solidification, and chemical dispersants which are commonly used to decrease oil spreading.33,34 In situ burning is a successful method to remove oil from the water surface, but it harms air and water by releasing toxic dioxins, resulting in increased cancer risks of residents via the food chain and inhalation.35,36 Also, the pollution generated by chemical solidification and chemical dispersants cannot be neglected.37–39 For biological techniques, the used oil-eating bacteria has the ability to breakdown, transfer, and alter the oil hydrocarbons into other non-toxic materials, such as carbonic gas, fatty acids, and water. 40
In the last century, starting from the exceptional advantages of membrane separation technique (i.e, simple, 41 energy efficient, 42 not harmful for the environment, 43 fire resistant, 44 anti-corrosion, 45 dye degradation in water, 46 anti-fouling, 47 and cost effective 48 ), it becomes the most commonly used method in numerous fields such as gas separation, 49 water purification, 50 seawater desalination, 51 energy harvesting,52–54 and so on. 55
In general, fibers membrane is considered the most significant membrane for oil-water separation owing to their numerous exceptional advantages for separation. (I) fibers with different physical and chemical properties can be used to fabricate separation membranes with specific specifications such as cotton fibers, 56 cellulose fibers, 57 carbon fibers, 58 metal wires, 59 electrospun fibers,60–62 and so on. (II) The ability to adjust the pore size of membranes that have cross-linked fibers structure. 63 Large pores are used for removing immiscible oil in water, whereas small pores are usually used for emulsion separation. 64 (III) Outstanding specific surface areas play an important role in separation efficiency.65,66 Textiles including woven fabrics, knitted fabrics, and nonwoven fabrics are considered good materials in the field of oil-water separation owing to their low density, low cost, good flexibility, and mechanical stability.67–71
The main objective of this review is to spot the light on the importance of textile sorbents in the field of oil-water separation. To the best of our knowledge, this is the first review paper that compares the significance of textile sorbents (woven fabrics, knitted fabrics, and nonwoven fabrics) for oil cleanup applications. This review summarizes the classification of textile fabrics, guidelines for testing sorbent materials, surface wetting properties, classification of textile sorbents, and their performance in the field of oil spill cleanup. We believe this review can be served as a good reference for using textile sorbents in the field of oil-water separation.
Classification of textile fabrics
A fabric is defined as any thin, flexible material made from yarn, directly from fibers, polymeric film, foam, or any combination of these techniques used for different applications.
72
Textile fabrics can be classified into woven, knitted, and non-woven fabrics. Woven fabrics are fabricated by interlacing two sets of yarns (warp yarn and weft yarn). Knitted fabrics which are classified into warp-knitted fabrics and weft-knitted fabrics are produced by the intermeshing of loops of yarn. The Knitting process can be defined in simple words as the interloping of yarn. The bending of yarn provides better stretchability, extensibility, comfort, and shape retention properties. However, they tend to be less durable than woven fabrics. The manufacturing process of knit fabrics is easier than woven fabrics. Therefore, the cost of knit fabrics manufacturing is cheaper than woven fabrics. Nonwoven fabrics are produced by connecting staple or filament fibers with gummy or bonded materials. Nonwoven fabrics have many advantages such as lightweight, cost-effective, good physical properties, water repellent and breathable, and so on. However, nonwoven fabrics have poor strength and durability compared with woven and knitted fabrics and they can’t be washed like them (Figure 2).73–76 Table 1 compares woven, knitted, and nonwoven fabrics.
Properties of surface wetting
Wetting at liquid and solid interfaces is a common phenomenon that is controlled by surface roughness and surface chemistry.81–87 With the development of the surface wettability phenomena, some exceptional wetting phenomena enthused by the natural phenomenon like the self-cleaning and superhydrophobicity of lotus leaf, superoleophobicity in water, and superoleophilicity in air of fish scales have caused extensive concern (Figure 3(a)).88,89 In general, the material might be water hated (superhydrophobic) and oil loved (superoleophilic) or water loved (superhydrophilic) or oil hated (superoleophobic).90–93 For example, superoleophilic/superhydrophobic webs can absorb only oil from oil/water mixtures, whereas superhydrophilic/superoleophobic webs can absorb only water from oil/water mixtures.93,94 Superhydrophobicity means the surface with a sliding angle <10° and static water angle >150°. The surface wettability is assorted by water contact angle (WCA) in the air, represented by superhydrophilic surfaces (<5°) and superhydrophobic surfaces (>150°) has been confirmed to be effective in the field of oil-water separation mixture (Figure 3(b)).95,96 Superhydrophobic surfaces can absorb oil while repelling water in the field of oil cleanup. Constructing the superhydrophobic surface in the air compared with the underwater superoleophobic surface is distinct with the advantages such as direct separating of oil-water mixtures and easy fabricating process.97–99 The underwater superoleophobic surface phenomenon in nature can be found in lotus plants.100,101 Scientific found that the superhydrophobic effect of the lotus leaves is ascribed to their rough structure and the presence of the waxy layer with low surface free energy.
93
In general, creating superhydrophobic surfaces imitative to the lotus effect can be got by two methods: enhancing the roughness surfaces of materials and decreasing their surface energy.102,103 Superoleophilicity/superhydrophobicity is an exceptional wettability for oil-water mixture separation owing to the difference between the surface tension of water and oil.
104
It is worth mentioning that woven fabrics, knitted fabrics, and nonwoven fabrics with superhydrophobic surfaces were produced successfully.105–108 (a) Examples of natural superhydrophobic surfaces. (b) schematic diagram illustrating WCA of hydrophilic, hydrophobic, and superhydrophobic surfaces.
116

Different methods have been used for generating superhydrophobic surfaces including spray-coating, 109 electro-assisted chemical deposition, 110 dip-coating, 111 sol-gel, 112 chemical vapor deposition, 113 polymer grafting, 114 and chemical etching. 115
Fabrication methods of textile sorbents for oil spill cleanup
Different techniques have been used to change the wettability of textile sorbents toward superoleophilic and superhydrophobic. Herein, recent techniques for the construction of superoleophilic and superhydrophobic are discussed briefly. These techniques include sol-gel, coating, bottom-blown stirring, UV curing, plasma irradiation, pad-dry-cure, stitch bonding, needle punched, melt blown, electrospinning, and immersion electrospinning. Generally, generating superoleophilic and superhydrophobic surfaces can be obtained via (I) using low surface energy materials, (II) making rough surfaces, or both. The textile sorbent performance is affected by significant features, such as surface energy, wettability, and pore size.
Sol-gel method
It is a wet chemical method also named chemical solution deposition. It is a cost-effective and mature strategy for modifying surface chemistry and surface morphology. This technique is mainly suitable to prepare metal oxide nanoparticles, functionalized metals, and composites. This technique includes the following steps: precursor preparation, hydrolysis, condensation, growth of particles, and agglomeration. 117
Coating method
It is a technique for covering the surface of substrate materials, usually referred to as the substrate owing to functional, decorative, or both. There are different types of coating including dip coating, spray coating, and spin coating. Regarding the dip coating, a substrate is dipped in the coating solution at an adjusted immersion speed; and the coating solution sticks to the surface of the substrate because of the capillary effects. Then, the substrate is drained to remove an extra coating film. Finally, the gel layer is formed on the surface of the substrate after evaporating solvents. 118 Spray coating is a low-cost and effective method that produces thin layers of organic materials films. This process starts with atomization where the surface of substrates is coated via sprayed solution. Herein, during the spraying process, the temperature of the surface should be constant to get a uniform coating composition. 119
Spin coating generates also uniform thin films but on a rotating substrate. In this technique, the coating thickness is controlled via repeated cycles. The thickness of the film depends on the spinning speed polymer concentration, surface tension, and evaporation rate of solvents. 120
Plasma irradiation
It is the most commonly used flexible technique for surface modification. Plasma is a partially ionized gas, containing ions, free electrons, and neutral species such as atoms, molecules, and radicals that are produced by electron generation through subjecting a gas to energy. The electrons can be sped in high electric fields by eliminating them from neutral molecules, resulting in the generation of free radicals, ions, and atoms. Random fragmentation on the surface can be triggered by subjecting surfaces to ion bombardment with high energy levels resulting in depositing chemicals onto the adsorbent surfaces or etching.121–123
Bottom-blown stirring
It is an energy-saving and gentle technique for generating superhydrophobic surfaces. The blown air floats up because of buoyancy and circulates the mixture. The air stirring consumed energy 60 times less than mechanical stirring. Generally, oil-water separation sorbents produced via this method had excellent mechanical abrasiveness and high efficiency.124,125
Brush-grafted
It is an adaptable technique to achieve surface modifications easily. It is used to alter the inherent surface chemical/physical properties of materials. In addition, it produces functional polymer brushes with fine-defined chemical configurations, thicknesses, architectures, and densities on the surface of materials based on surface-initiated polymerization technique.126–128
Pad-dry-cure technique
It is one of the most common application techniques for durable press finishes and easy care. In this method, the softener, catalyst, crosslinking reactant, and other components are dried on the surface of fabrics before the crosslinking reaction happens during the curing step.129,130
Stitch bonding
Stitch bond is a nonwoven construction where the fabric is formed by stitching or knitting the fibers to form a fabric with the appearance of a knit fabric. It is a process that holes through flexible substrates in different forms such as nonwovens, papers, films, foams, and/or their mixture with a series of sharp needles placed close to each other. Then, the needles stitch or knit either a complex or simple network of textile threads into the substrate(s) to produce nonwoven/textile hybrids for multiple applications. The stitching either reinforces substrates or strengthens or provides various alternative properties to the resulting products. 131
Needle punched
It is a technique for producing nonwoven fabrics from different fibrous webs using a needle loom where fibers are mechanically bonded together via fibers frictions and entanglement after good needle barbs frequently penetrated through the fibers web. The properties of these webs can be modified by controlling the characteristics and length of fibers resulting in making it preferable for many applications. 132
Melt blown
It is a conventional technique for producing micro/nanofibers. Herein, the melted polymer is extruded from tiny nozzles surrounded by gas blowing at high speed. The advantages of this method are solvent-free, simplicity, and high productivity. Starting from the flexibility of this technique represented by optimizing surface properties and polymer combination rheological, it has been used in many applications such as sorbents, filtration, drug delivery, and apparel. 133
Spun bond
It is a widely used technique for producing nonwoven fabrics. This process combines the process of spinning with the process of sheet formation by adding the device of bonding in the same continuous line. This process consists of several related steps: polymer feeding, extruder, metering pump, die assembly, filament spinning, drawing, and deposition system, collecting belt, bonding zone, and winding. These webs are used widely in many applications such as filtration, packaging, geotextile, civil engineering, and so on. 134
Electrospinning
It is an efficient technique for generating micro/nanofibers with desired features, such as surface morphology and fiber diameter and structure.135–137 This method consists of a syringe pump to emit a polymer solution from the top of the needle (spinneret) to the collector at a constant flow rate.138,139
Polymer solutions can be formed by dissolving polymers in suitable solvents, then, a polymer solution is ejected by a syringe at a constant flow rate, resulting in generating a droplet at the top of the needle. Appling voltage between the syringe needle and collecting electrode leads to generating an electric field resulting in accelerating and elongating an electrically charged polymer jet (forming Taylor cone).78,140 Subsequently, a nearly straight jet develops from the cone. Concurrently as the jet flies on the way to the collector, the polymer solution undergoes stretching and whipping while the solvent evaporates. Even though the jet is stable close to the tip of the spinneret, it quickly enters an unstable bending step with further elongating of the solution jet by the electrostatic forces in the solution as the solvent evaporates. In addition, the coils’ perimeter of each turn grows monotonically. 141 Finally, the accumulation of the dried jets on the collector forms an electrospun ultrathin fibers web. In general, solvent from the jets is evaporated before reaching the collector.
The properties of materials can be altered through engineering surfaces of electrospun fibers.142–144 In addition, the properties of materials can be enhanced by electrospun deposition on them. 145 There are different types of electrospinning such as near-field electrospinning, far-field electrospinning, immersion electrospinning, and so on.42,146,147 Electrospun fibers have shown great potential in many applications such as oil-water separation,148,149 air filtration,150,151 keeping food fresh,62,152 and so on.153–155
Applications of textile sorbents for oil spill cleanup
Textile fabric is a two-dimensional plane-like structure consisting of textile materials that have reasonable strength, flexibility, elongation, etc., used for multiple applications. 72 There are different kinds of textiles used in the oil/water separation which can be grouped into woven fabric, knitted fabric, and nonwoven fabrics. Fabrics are considered an exceptional candidate for the creation of oil spill cleanup materials owing to their advantages such as good mechanical properties of woven fabrics, worldwide range of applications, cost-effectiveness of nonwoven fabrics, and so on.
Woven oil cleanup sorbents
Any kind of textile produced by interlacing two or more yarns at right angles to one another is called woven fabric. These fabrics are formed on a loom, and made of many yarns woven on a weft and a warp. These fabrics can be made of synthetic fibers, natural fibers, and a mixture of both. 156 As discussed previously, superhydrophobic surfaces can be obtained by the presence of micro/nano hierarchical topographies on substrate surfaces, and the coating or attachment of them with low surface energy materials. Generally, the surface of textile fabrics has naturally hierarchical structures. The hierarchy of textile fabrics differs according to the pattern and nature of knitting and weaving processes which play an essential role to improve superhydrophobic textiles. Nevertheless, the polar groups present on the textile surfaces make them highly hydrophilic. To make them hydrophobic, their surface chemistry should be altered either by attaching hydrophobic material on them or by modifying them chemically. 157 Many methods have been used to obtain textiles with superhydrophobic surfaces to be used as oil-cleanup sorbents.
Cao et al.,
158
prepared superhydrophobic, durable, and eco-friendly sorbent for oil-water separation by coating woven cotton fabric via poly(dimethylsiloxane) (PDMS)–ormosil (silica aerogel) (Figure 4(a)–(c)). The sorbent showed high superhydrophobicity with a WCA >160° due to its hierarchical surface. In addition, it can withstand 5 cycles of washing and 100 cycles of abrasion without decreasing superhydrophobicity. Furthermore, it showed high stability for alkaline and acidic solutions. Moreover, it does not affect the physical properties of woven cotton fabric, such as flexibility, air permeability, and strength. The oil separation efficiency of this sorbent was more than 98% even after 10 separation cycles. (Figure 4(d) and (e)). (a) Schematic diagram illustrating the formation process of the sorbent. (b) SEM image of the treated fabric. (c) Digital images of the selective absorption (I–III) and speedily continuous absorption (IV–VI) process of oil-water mixture. (d) Effect of recycling numbers on the separation efficiency. (e) Schematic diagram illustrating the mechanism of oil-water separation mixture.
158

Wang et al.,
159
developed a new technique to prepare oil-water separation sorbent from Janus cotton fabric using coating and cross-linked method (Figure 5). They coated the sorbent with PDMS-b-PCEA and poly(N,N-dimethylaminoethyl methacrylate) PDMAEMA. They used PDMS due to its low surface tension and its ability to make superhydrophobic surfaces. They selected PDMAEMA owing to its ability to ionize in water and coalesce emulsion droplets by screening the electrostatic repulsion. They concluded that their sorbent has high and rapid oil separation efficiency for different kinds of oils. Schematic diagram illustrating the formation process of the sorbent.
159

Su et al.,
160
prepared a cost-effective, simple, and high-efficiency oil-water separation sorbent consisting of woven polyester (PET) treated by PDMS + Tetraethyl orthosilicate (TEOS) via vapor−liquid sol−gel technique (Figure 6(a) and (b)). The sorbent showed an outstanding WCA of 160° owing to its high roughness. Furthermore, the sorbent exhibited excellent oil-water separation efficiency which is 94.1% and 95.6% for dichloromethane and hexane, respectively. Moreover, the sorbent showed a high WCA (∼150°) and the separation efficiency was above 96% at 30 separation cycles (Figure 6(c)). (a) Schematic diagram illustrating the formation process of the sorbent. (b) SEM image of the treated fabric. (c) Effect of separation cycles on the separation efficiency and contact angles of different oils/water mixture.
160

Chen et al.,
161
fabricated oil cleanup sorbent with smart surfaces using sustainable and green paths. They prepared their sorbent which is made of woven cotton fabric by coating it with treated polyurethane via UV light irradiation (Figure 7(a)). The sorbent exhibited outstanding superoleophobicity in water and superhydrophobicity in the air after treatment with the acid aqueous solution because of the protonation of LEDA chains within PU (Figure 7(b) and (c)). The results showed that the sorbent has a high WCA of (151.3°) after 30 times of washing (Figure 7(d)). In addition, it showed high separation efficiency above 99% for light oil and heavy oil from an oil-water mixture after 30 times of washing owing to the good mechanical stability of cross-linked polymers (Figure 7(e)). (a) Schematic diagram illustrating the formation process of the sorbent. (b) Digital images illustrating the separation process of oil (red)/water (green) mixture of pristine fabric. (c) Digital images illustrating the separation process of the oil-water mixture of the treated fabric. (d) Effect of washing cycles on WCA. (e) Effect of oil type on the separation efficiency.
161

Guo et al.,
67
developed a stable and robust coating on woven cotton fabric using a sol−gel method with PDMS and functional polymers polyvinylpyrrolidone (PVP)-PDMS-FA for oil-water separation (Figure 8(a)–(f)). The results showed that the sorbent has outstanding separation efficiency of heavy oil and light oil (above 99.9%) from oil water (Figure 8(g)). In addition, the separation efficiency of CH2Cl2 was above 96% even after 30 cycles of washing. Furthermore, the separation efficiency of CH2Cl2 in harsh conditions such as salty solutions, alkaline, and acidic was above 97.3% after 30 cycles of washing (Figure 8(h)). (a) Schematic diagram illustrating the formation process of the sorbent. SEM images of (b), (c) pristine fabric, and (d), (e) treated fabric. (f) Schematic diagram illustrating the mechanism of the separation process. (g) Separation efficiency of different kinds of oil. (h) Effect of cycle times on the separation efficiency and WCA.
67

Chu et al.,
162
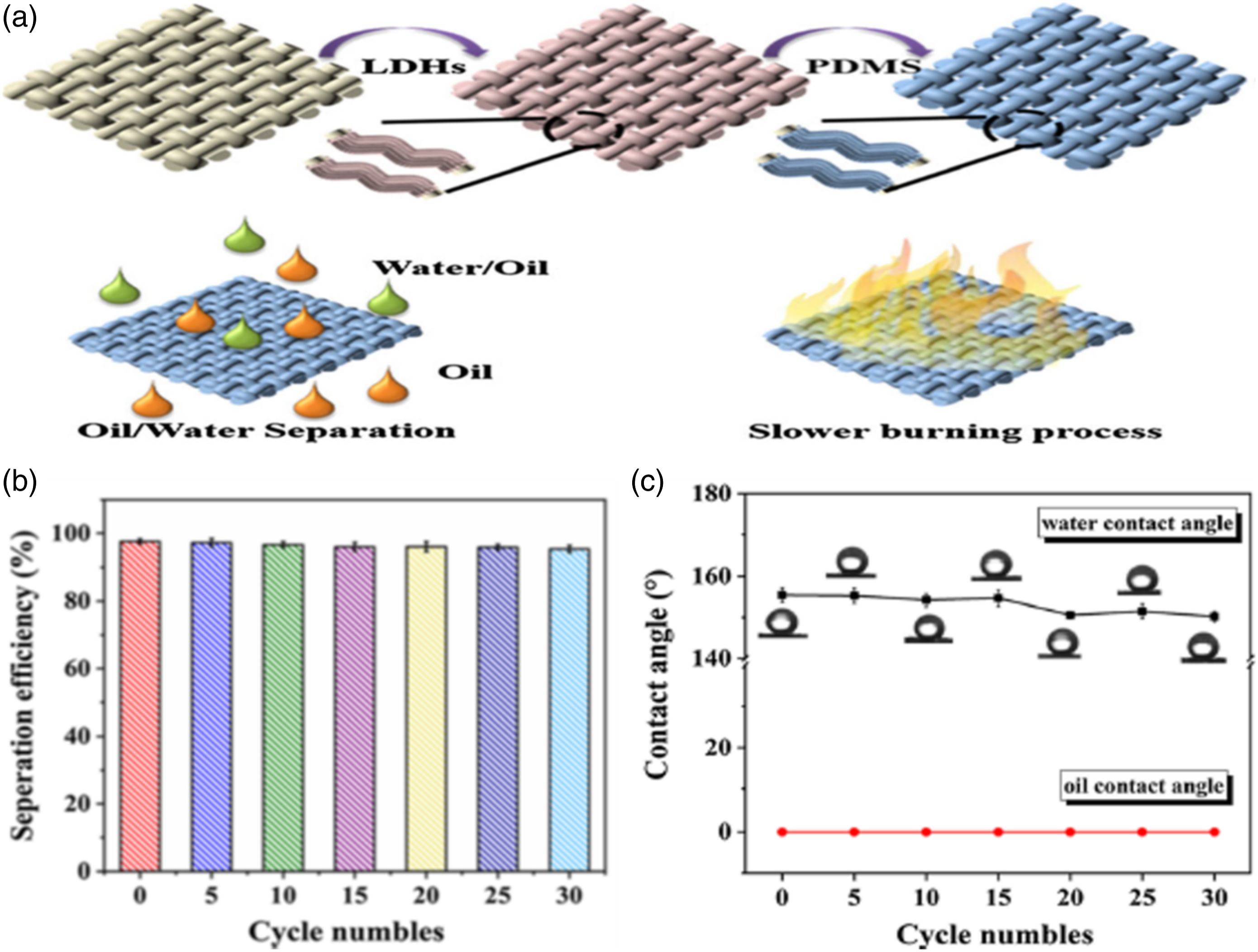
fabricated flame retardant, superoleophilic and superhydrophobic sorbent of woven ramie fabric for oil spill cleanup. This sorbent was fabricated through hierarchical layered double hydroxide (LDH) and PDMS composite coatings (Figure 9(a)). They found that by treating samples for 6 h, the Ramie@LDH6h@PDMS sorbent has an outstanding WCA of ∼155.4° ± 1.7° because of extra air pockets trapped between the pillars of the hierarchical structure. In addition, their sorbent had outstanding efficiency for oil-water separation which is above 96.3% for six kinds of oils. Furthermore, they tested the separation efficiency for different mixture ratios of oil to water. Importantly, this sorbent exhibited high recyclability of oil-water separation efficiency (above 95.36%) and hydrophobicity after 30 cycles (Figure 9(b) and (c)). These results were attributed to the superoleophilic and superhydrophobic performance of the sample surface of Ramie@LDH6h@PDMS. (a) Schematic diagram illustrating the formation process of the sorbent. (b) Effect of cycle numbers on the efficiency of oil/water separation. (c) Effect of cycle numbers on oil contact angle and WCA.
162
Huang et al.,
125
used a novel bottom-blown stirring method to fabricate superhydrophobic and superoleophilic woven PET fabric (Figure 10(a)). Herein, the fabric was dipped in an anhydrous ethanol solution containing TEOS (4 v/v %) and Hexadecyltrimethoxysilane (HDTMS) (5 v/v %). After that, ammonia was added with stirring for 2 h at room temperature. Then, the sample was baked in a vacuum oven after washing it with anhydrous ethanol. The treated sample showed an outstanding WCA of 152° ± 7°. Furthermore, the sample exhibited outstanding oil-water separation efficiency (above 99.5%) and high oil flux (29433 ± 2403 m2/h) after 10 cycles because of the uniform roughness surface (Figure 10(b) and (c)). (a) Schematic diagram illustrating the formation process of the sorbent using a bottom-blown stirring technique. (b) The capacity of oil absorption of different kinds of oils. (c) Effect of cycle numbers on oil flux and separation efficiency.
125

Toteva et al., 163 discovered a novel sorbent for oil spill cleanup based on natural textile products. This sorbent consists of woven cotton fabric modified with a layer of cross-linked chitosan treated by ZnO particles which control the swelling degree of the chitosan layer. They studied the effect of the treated thickness layer by fabricating samples with two layers and samples with three layers. They concluded that there is a negative relationship between the oil sorption capacity and the thickness of the treated layer as well as the amount of ZnO particles.
Zhang et al.,
164
produced super-absorbent, superhydrophobic, and self-cleaning woven cotton sorbent for oil-water separation. The sorbent surface was decorated with nano- and microstructures to mimic the rose petals effect in nature (Figure 11(a)). Microsheets of noncorrosive, harmless, non-toxic magnesium hydroxide were placed in the shape of a rose on the surface of the fabric after modifying it with cetyltrimethoxysilane (Figure 11(b) and (c)). The sorbent showed high hydrophobicity with a WCA of ∼165° owing to the presence of magnesium hydroxide in a specific morphology and cetyltrimethoxysilane. In addition, the sorbent showed excellent efficiency for oil-water separation ∼99.9% (Figure 11(d)). Furthermore, the rate of oil absorption was ∼100%, whereas the rate of oil storage ranged from 115.86% to 558.62% with a good capacity of oil storage for different kinds of oil (Figure 11(e)). (a) Schematic diagram illustrating the formation process of the sorbent. (b, c) SEM images of the modified fabric at different magnifications. (d) The separation efficiency of the sorbent for different kinds of oils. e) The storage efficiency of the sorbent for different kinds of oils.
164

Hossain et al.,
96
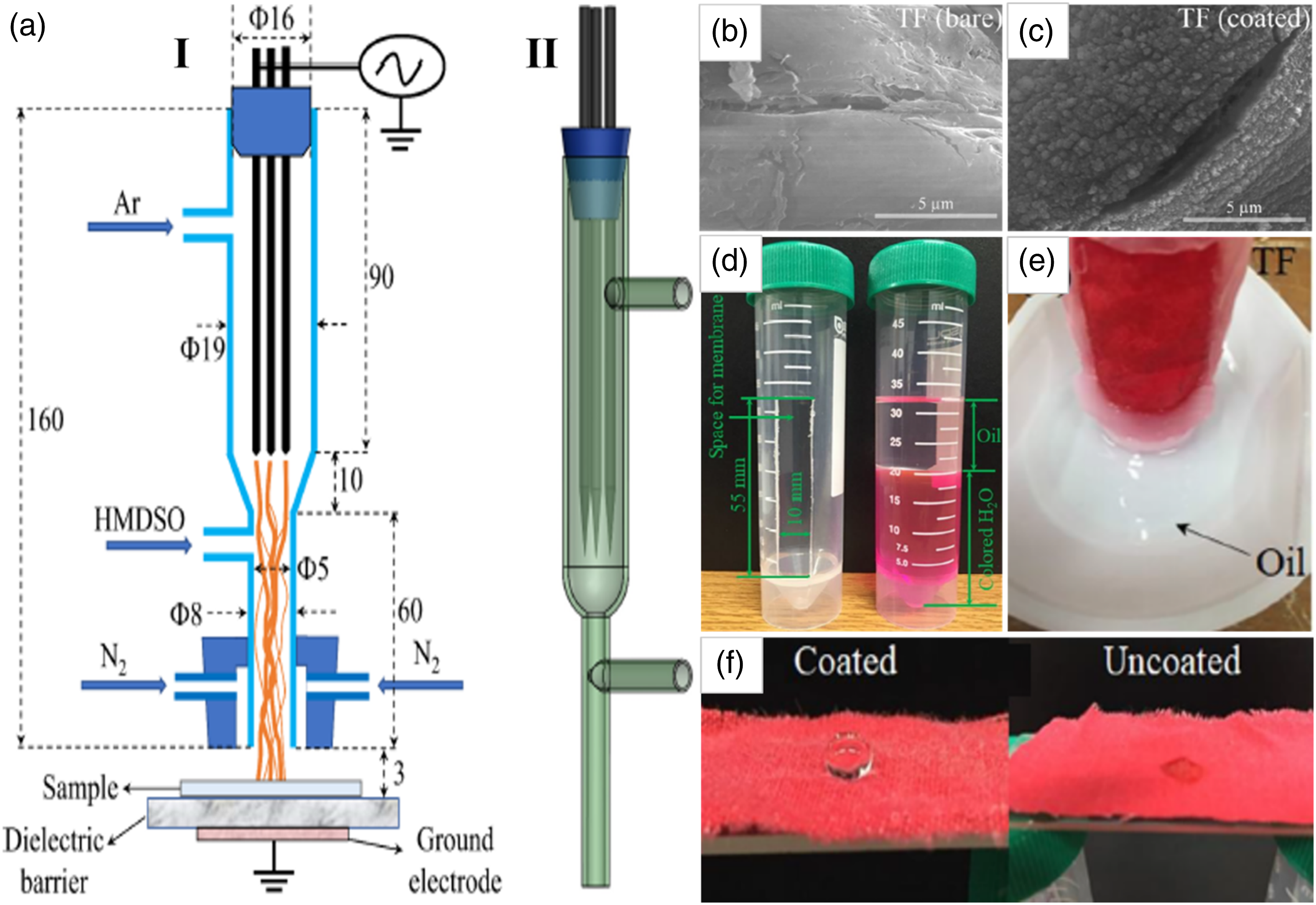
prepared efficient superoleophilic and superhydrophobic sorbent for oil-water separation via one-step plasma polymerization of hexamethyldisiloxane (HMDSO). For depositing the functional thin films onto textile fabric, a dielectric barrier discharge plasma jet with argon was used (Figure 12(a)–(c)). The sorbent showed high durability and outstanding hydrophobicity with a WCA of ∼165° and a sliding angle of ∼2° after 5 cycles. Furthermore, it showed excellent oil separation efficiency of ∼98.1% and good capacity for oil absorption of ∼68.2%. These results were ascribed to the presence of HMDSO which has low surface energy and hydrophobic nature (Figure 12(d)–(f)). (a) Schematic diagrams of (I) the plasma polymerization system of the reactor, and (II) its 3D view. SEM images of (b) pristine fabric, and (c) treated fabric. (d) Digital photo illustrates the membrane space and labeling of oil and water using (e) the sorbent. (f) Digital image compared the WCA of untreated sample and untreated sample.
96
Knitted oil cleanup sorbents
Knitted fabric is a textile fabricated by inter-meshing of loops or interloping of yarns. Generally, knitted fabric is more flexible compared with woven fabric, therefore, some researchers preferred to use it in the field of oil-water separation. It is worth mentioning that the oil cleanup capacity of knitted fabrics is enhanced by surface engineering or by chemical modification. Only a few studies have investigated the importance of knitted fabrics in the field of oil cleanup.
Islam et al.,
165
coated 3D weft-knitted spacer fabric with SiO2 aerogels to get oil cleanup sorbent via the sol-gel method (Figure 13(a)). Their results showed that the oil weight gain (%) of uncoated fabric and coated fabric was 562 ± 2.65% and 763 ± 2.31%, respectively, for vegetable oil. Whereas, the oil weight gain (%) of uncoated fabric and coated fabric was 521 ± 2.31% and 698 ± 2.65%, respectively, for engine oil. They concluded that the weight gain (%) of vegetable oil is the highest for both compared sorbents owing to its low viscosity (Figure 13(b) and (c)). (a) SEM image of I) uncoated fabric, and II) coated fabric. (b) Digital images illustrating the process of absorbing oil, recovering oil, and reusing procedure of the sorbent. (c) Schematic diagram illustrating the mechanism of oil-water separation.
165

Saha et al.,
166
fabricated a new sorbent of knitted cotton fabric for oil-water separation via the pad-dry-cure technique (Figure 14(a)). Herein, the fabric was coated with C6- Fluorocarbon (FC)-dendrimer (Figure 14(b) and (c)). The sorbent showed excellent water repellency, self-cleaning performance, and outstanding separation efficiency of ∼97–99% for the oil-water mixture (Figure 14(d)–(f)). Furthermore, it showed antimicrobial activity, UV ray absorber, flame retardant properties, and acid resistance. These results were ascribed to the presence of FC-dendrimer which provides on the surface of fabric extra functional groups. (a) Schematic diagram illustrating the formation process of the sorbent. (b), (c) SEM images of untreated and treated fabric, respectively. (d, (e) WCA of untreated and treated fabric, respectively. (f) The process of oil-water separation.
166

Nonwoven oil cleanup sorbents
Nonwovens have been attracting more attention in developing new oil-water separation sorbents due to their porous 3D fiber networks resulting in having high absorbability and performance compared with traditional fabrics.167–169 However, unmodified nonwovens have poor selectivity to water and oil and cannot separate them with high quality. 170 To solve this problem, many studies have focused on providing superhydrophobic structures into nonwovens surfaces because superhydrophobic surfaces have a great repulsive force to water, resulting in enhancing the capacity of nonwoven sorbents for oil cleanup at a large extent.79,171 In addition, nonwoven materials are cost-effective, high productivity, and have outstanding physical and mechanical properties which enable the fabrication of products with unique properties for multiple applications. 172
Ortega et al., 173 fabricated highly capable nylon nonwoven sorbent for oil-water separation via the spunbond method. The results showed that the sorbent has the ability to absorb >26 times its mass in high-viscosity oil (gear oil) and >16 times its mass in low-viscosity oil (crude oil). In addition, the sorbent can separate >95% of gear oil from the oil-water mixture. These results were ascribed to the oleophilic nature of nylon and the structure of the sorbent.
Neznakomova et al.,
174
investigated the probability of using non-woven sorbents from different waste fibers for oil spill cleanup. Herein, 4 samples were fabricated, sample 1 consists of wool 15%, cotton 18%, viscose 20%, PET 17%, polyacrylonitrile (PAN) 30%, and stitch fiber–PET 100%. Sample 2 consists of wool 5%, cotton 20%, viscose 17%, PET 23%, PAN 35%, and stitch fiber–PET 100%. Sample 3 consists of wool 20%, cotton 20%, viscose 20%, PET 20%, PAN 15%, and polyamide 5%. Sample 4 consists of 100% recovered wool (Figure 15(a)). The type of samples was stitch-bonding for samples 1 and 2, while needle punched for samples 3 and 4. They found that the stitch-bonded samples have higher efficiency for oil cleanup than needle-punched samples, owing to their rich free fibrous surface (Figure 15(b)). In addition, they concluded that the efficiency of oil cleanup increased by increasing the ratio of PAN. (a) Digital images of used samples I)Sample 1, II) Sample 2, 3) Sample 3, IV) Sample 4. (b) The oil adsorption ratio for studied samples.
174

Brindha et al., 175 investigated the effects of needling density, fabric weight, and blend ratio of polypropylene (PP) and nettle needle-punched nonwoven fabrics on crude oil and engine oil Sorption. They found that there is a negative relationship between oil sorption efficiency and needle density because increasing the needle density leads to a decrease in the voids which banned the sorbent to absorb more oil. In addition, there is also a negative relationship between oil sorption efficiency and fabric weight because increasing the fabric weight leads to increasing the available amount of fibers to the needle barb. Furthermore, they concluded that oil sorption efficiency increases by increasing the ratio of PP owing to its low density. Therefore, their sorbent which is designed at PP/nettle (70/30) with low fabric weight and needling density can absorb 23.59 g/g of crude oil and 17.02 g/g of engine oil.
Zhang et al.,
176
fabricated eco-friendly, simple, and cost-effective facile PP nonwoven sorbent for oil-water separation via the melt-blown method (Figure 16(a) and (b)). The sorbent showed outstanding superoleophilicity with an oil contact angle of 0 and superhydrophobicity with a WCA of >150° owing to the rough surface of fibers and porous structure of the sorbent (Figure 16(c)). In addition, the sorbent showed outstanding efficiency for oil spill cleanup (>99%) for various oils including crude oil and pump oil from different solutions such as saline media, alkaline, and strongly acidic (Figure 16(d) and (e)). Furthermore, the sorbent exhibited exceptional durability in harsh environments where the oil separation efficiency is still >99% even after 20 cycles. (a) Schematic diagram illustration the melt-blown process for forming PP nonwoven sorbent. (b) SEM image of the sorbent. (c) WCA and oil contact angle of the sorbent. (d) The efficiency of oil–water separation of the sorbent. (e) The oil flux of the sorbent.
176

Pakdel et al.,
177
produced a new nonwoven sorbent for oil-water separation. The sorbent was formed from carbon fiber waste and polyamide 6 (PA6) using carding and needle-punching methods. Then, the PA6 was eliminated by HCL treatment, subsequently coating surfaces with fluorine-free comprising zeolite imidazole framework-8 (ZIF-8) particles and PDMS (Figure 17(a) and (b)). The sorbent showed outstanding superhydrophobicity with a WCA of 153.5° owing to the presence of ZIF-8 particles. In addition, the sorbent exhibited an excellent capacity for oil absorption which reached 24.5 g/g for dichloromethane (Figure 17(c)). Furthermore, they concluded that the absorption capacity of their sorbent was better for heavy oils compared with organic solvents and light oils. (a) Schematic diagram illustrating the formation process of the sorbent. (b) SEM image of treated carbon fiber. (c) The oil absorption capacity of the sorbent.
177

Singh et al., 178 obtained a novel blended nonwoven sorbent compound of (10/90, cotton/kapok) fibers using needle-punching and carding techniques. Their results showed that the WCA of the sorbent was high (∼147°) due to the high ratio of more hydrophobic kapok fibers. In addition, they found that the oil sorption capacity of their sorbent which has a density of 150 g/m2 was 39.92 g/g, 43.97 g/g, and 45.36 g/g for diesel oil, vegetable oil, and engine oil respectively, owing to brilliant oil sorption of kapok, contributed by its hollow lumen structure and superior oil wettability.
Zaarour et al.,
179
investigated the importance of the surface morphology of electrospun poly (vinylidene fluoride) (PVDF) nanofibers for oil spill cleanup (Figure 18(a)). They found that the sorbent based on the macroporous structure has the highest oil sorption capacity compared with grooved and rough structures which are 23.96 ± 2.68 g/g, 37.74 ± 4.33 g/g, and 50.58 ± 5.47 g/g for olive oil, motor oil, and silicon oil, respectively (Figure 18(b) and (c)). They attributed these results to the high specific surface area of macro-porous fibers and the high viscosity of the silicon oil. (a) SEM images of sorbents with different surface morphology: I) porous, II) rough, III) grooved. (b) Digital images for the process of oil-water separation. (c) The oil absorption capacity of sorbents.
179

Zhu et al., 66 enhanced the capacity of oil cleanup by electrospinning porous nanofibers webs with spindle porous bowl-like beads. They found that the sorbent has a high WCA (∼152°) because of tiny voids between the nanofibers which can enhance hydrophobicity by trapping air. Furthermore, the sorbent has outstanding oil cleanup capacity which is 35.17 ± 6.21 g/g, 49.21 ± 7.13 g/g, and 62.98 ± 7.45 g/g for olive oil, corn oil, and silicon oil, respectively, due to the existence of spindle porous bowl like beads and the tiny diameter of fibers which trap the oil droplets.
Li et al.,
180
generated novel crumpled polystyrene nanofibers sorbent for oil spill cleanups. The sorbent was formed via immersion electrospinning, where the collector and nozzle were deepened together in a coagulation bath (Figure 19(a) and (b)). The sorbent showed significant capacities of oil sorption (72, 77, and 83 g/g for mineral, canola, and silicone oils, respectively) compared with electrospun conventional fibers (24, 30, and 41 g/g for mineral, canola, and silicone oils, respectively). Furthermore, this sorbent exhibited a quicker oil uptake rate compared with the sorbent based on conventional fibers (Figure 19(c) and (d)). These results are attributed to the zigzag structure of crimped sorbent which trapped oil. (a) Schematic diagram illustrating the electrospinning device used for forming crumpled nanofibers. (b) SEM image of crumpled nanofibers. (c) Digital images of I) comparing the behavior of oil sorption of the traditional-electrospun sorbent (left) and the crumpled-electrospun sorbent (right), II) digital describing the removing oil from water. (d) The capacity of oil sorption of the sorbents.
180

Huang et al.,
181
fabricated pitch/PAN composite nanofibers sorbent for oil-water separation via the electrospinning method (Figure 20(a)). The WCA of this sorbent was ∼139.4°, and the oil contact angle of ∼0 (Figure 20(b) and (c)). The capacity of oil absorption of this absorbent reached 42.47 g/g within 5 min. The sorbent showed outstanding reusability, where the capacity of oil absorption was >70% even after 10 cycles of absorption-desorption (Figure 20(d) and (e)). The results exhibited this sorbent showed more surface roughness, superior hydrophobicity, and higher oil absorption compared to the sorbent based on pure PAN nanofibers. These results were attributed to the presence of petroleum pitch which has excellent oil absorption and hydrophobicity nature. (a) Schematic diagram illustrating the formation process of the sorbent. (b), (c)WCA and oil contact angle of the sorbent. (d) Reusability of the sorbent for different kinds of oil. (e) Digital images showed the oil absorption process of the sorbent: I) oil-water mixture; II) before absorption; III) during absorption; IV) after absorption.
181

Summary and future outlook
In the review, different textile sorbents including woven sorbents, knitted sorbents, and nonwoven sorbents for oil spill cleanup are reviewed. This paper reviews the materials, sorbent types, fabrication methods, and applications of textile sorbents for oil-water separation. The classification of textile fabrics, surface wetting properties, fabrication techniques of textile sorbents for oil spill cleanup, their properties, and recent applications in the field of oil cleanup are discussed in detail. It can be concluded from our discussion: 1. The efficiency of textile sorbents is enhanced by regulating the surface roughness and surface chemistry. 2. Increasing the specific surface area of textile sorbents leads to enhancing their oil sorption efficiency. 3. Surface morphology of nanofibers sorbents play a vital role in enhancing their oil sorption efficiency. 4. Different techniques can be used for generating superhydrophobic textile sorbents such as sol-gel, coating, bottom-blown stirring, UV curing, plasma irradiation, pad-dry-cure, stitch bonding, needle punched, melt blown, electrospinning, and immersion electrospinning. 5. The low-cost and high-productivity nonwoven sorbents showed the highest oil spill cleanup efficiency compared with other textile sorbents owing to their excellent properties such as high specific surface area, high porosity, and so on.
In recent years, textile sorbents for oil spill cleanup have shown brilliant accomplishments in fundamental understanding and technological developments. Concerning the future of textile sorbents, some essential issues should be addressed: 1. Investigating the effect of woven structures (plain, satin, and twill) on the oil-water separation efficiency of woven sorbents. 2. Exploring the relationship between various knit structures (single jersey, double jersey, tricot, rip, pearl structure, and interlock) of sorbents and their oil-water separation efficiency. 3. Studying the relationship between the nonwoven types (spunlace, heat-bonded, air-laid, wet-laid, spunbond, meltblown, acupuncture, stitch, and electrospinning) of sorbents and their oil-water separation efficiency. 4. Innovating new methods that can increase the oil sorption capacity of textile sorbents.
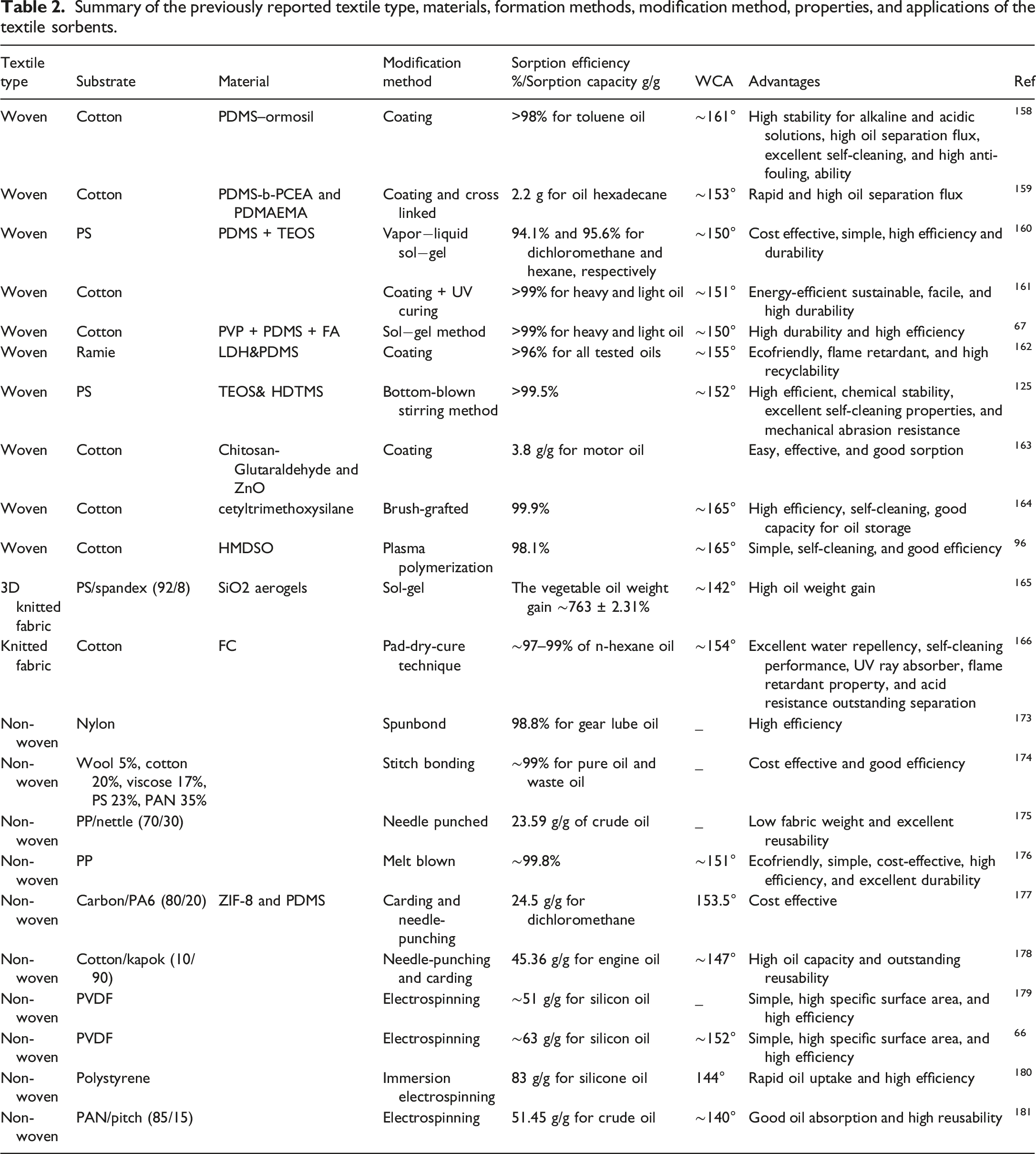
Summary of the previously reported textile type, materials, formation methods, modification method, properties, and applications of the textile sorbents.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

