Abstract
Polyacrylonitrile (PAN) carbon fibers are often used to prepare high-performance paper-based materials owing to their high strength, good electrical and thermal conductivity, and superior comprehensive properties. In this study, a novel method for preparing PAN-based carbon fibers by centrifugal spinning was developed, and a stable and homogeneous PAN carbon fiber paper was successfully obtained. Subsequently, the formation process, microscopic morphology, electrical conductivity, electrochemical performance and hydrophobicity of the PAN carbon fiber paper were studied and evaluated. The results showed that the electrical conductivity of the PAN carbon fiber paper prepared via this method reached 43.250 s·cm−1, resistivity was as low as 0.023–0.033 Ω·cm, and contact angle exceeded 140°. This study adopted a new method to prepare PAN carbon fiber paper, which provided another method for the preparation of high-performance fiber paper.
Keywords
Introduction
Carbon fiber paper is a new type of energy material, which is used in many fields due to its excellent performance. At present, carbon fiber paper can be widely used in supercapacitors and fuel cells areas. Supercapacitors have much higher capacitance values than other capacitors, but have lower voltage limitations, bridging the gap between electrolytic capacitors and rechargeable batteries to suitable for applications that require many fast charge/discharge cycles. Fuel cells are clean, efficient and simple green energy device that uses renewable resources to replace conventional fossil fuels and are considered to be the most promising environmentally friendly energy source that will simultaneously solve the dilemma of energy depletion and reduce environmental pollution.1–3 The gas diffusion layer is an important component of fuel cells for proton exchange. Carbon fiber paper is typically used as the basic conductive material of gas diffusion layer.4–6 However, the conductive performance of traditional carbon fiber paper can no longer meet the requirements of the modern industry, of which the production process is complicated and expensive. 7 One solution is to modify the fibers by grafting highly conductive materials to improve the electrical properties. Another solution is to use high-performance fibers as raw materials.
Among the high-performance fibers, Toray Industries, Inc., located in Japan, has developed a super-fiber by mixing a dispersion medium containing short fibers that could be carbonized and own self-bonding properties. After pulping, the fibers were bonded to each other and carbonized. Suktha et al. s used acid treatment to activate carbon fiber paper to improve its electric capacity. 6 Zhang et al. used a high-temperature carbonization method to prepare an activated carbon fiber paper electrode with a maximum capacitance of 750 mF·cm−2. 8 Wang et al. used electrochemical deposition to significantly improve the capacitance of carbon fiber paper, and no obvious capacitance decay was exhibited even after 20,000 cycles. 9 Yan et al. synthesized a carbon fiber paper/polyaniline composite electrode at room temperature using a hydrothermal method, and the capacitance reached 638 F·g−1 at a current density of 2 A·g−1. 10 To achieve a wider range of applications, considerable research has been conducted on modifying carbon fiber papers to enhance its electrical or mechanical properties. Hung studied the effect of different mass fractions of phenolic resin, an impregnating agent, on the properties of carbon fiber paper. The carbon fiber paper with a mass fraction of 15% of phenolic resin achieved the best performance. 11 Maheshwari et al. impregnated carbon fiber paper with a colloidal graphite-modified phenolic resin impregnation solution to prepare carbon fiber paper for graphite-modified proton exchange membrane fuel cells. When the volume fraction of graphite was 7.1%, the carbon fiber paper exhibited the best performance, with a flexural strength of 62.5 MPa, and resistivity was 10 mΩ·cm. 12 Du et al. prepared a carbon fiber paper with high hydrophobicity for fuel cells by growing carbon nanotubes on the surface of the carbon fiber. 13
In contrast, a dense three-dimensional (3D) network structure tends to improve the electrical conductivity of carbon fiber paper. 14 Therefore, a new spinning method, i.e., centrifugal spinning, which is efficient, safe, and environmentally friendly, may be a good method for preparing carbon fiber papers.15,16 In the centrifugal spinning process, micro- and nanoscale fibers are produced from a polymer solution or melt using a vessel with a spinneret that spins at high speeds. After the solution or melt is shaken out, fibers are formed after stretching, evaporation of the solution, and solidification. 17 In recent years, fiber-based materials fabricated by centrifugal spinning have been used in various fields, such as energy storage, thermal insulation, and medical dressings.18–23 Moreover, the productivity of centrifugal spinning exceeds 1 g·min−1 per nozzle, which is significantly higher than that of laboratory-scale electrospinning (0.1 g·h−1). 24 However, most centrifugal spinning equipment can only collect fibers using columns. This method works well, but achieving the continuous fabrication of large-scale fiber mats is still a challenge. 25
In view of the above facts, the purpose of this study is to develop a facile strategy to prepare polyacrylonitrile (PAN) carbon fiber paper with high electrical conductivity and near-super-hydrophobicity via a planar centrifugal spinning device. The special morphology of the fiber paper was measured, formation mechanism of the PAN carbon fiber paper (PCFP) was examined, and key parameters such as electrical conductivity and resistivity of the PCFP were evaluated in this study. The results showed that the PCFP prepared using this method had broad application prospects in the field of fuel cells.
Experiment
Materials
PAN powder (average molecular weight: 85,000 and 250,000) was supplied by Shanghai Macklin Biochemical Technology Co., Ltd. (Shanghai, China). N, N-dimethylformamide (DMF, anhydrous, ≥99.5%) was purchased from Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China).
Preparation of polyacrylonitrile solution
Main preparation process parameters of each PAN fiber papers.

Preparation of polyacrylonitrile fiber paper via centrifugal spinning
Figure 1(a) showed the schematic illustration for fabrication of the PAN fiber papers via centrifugal spinning, and the centrifugal spinning equipment was independently designed and developed. Its spinning rate was 4000 r/min, and the output rate was 1.1–1.4 g/min, which was much higher than electrospinning. Figure 1(b) showed a screenshot of spinning cup from the spinning process. At the bottom of the spinning cup there were two conical nozzles, from which the spinning solution was thrown out and finally became fibers. The pore diameter of conical nozzle was 0.16 mm, and the vertical height of the nozzles from planar collector was 5 cm. During spinning, the ambient temperature was always maintained at approximately 40°C, and the ambient humidity was maintained at 30–50%. The PAN fiber felts were first dried at 60°C for 6 h and then hot pressed for 5 min at 10 MPa to transform them into PAN fiber paper (PFP). The corresponding relationship between PFP and PAN solution was summarized in Table 1. (a) Schematic illustration for fabrication of the PAN carbon fiber Paper, (b) A screenshot from the spinning process, (c) Optical photo of PAN fiber paper, (d) Optical photo of PAN carbon fiber paper, (e) SEM images of the PAN carbon fiber paper.
Preparation of polyacrylonitrile carbon fiber paper by pre-oxidation and carbonization
Main preparation process parameters of each PAN carbon fiber paper.

Characterization
The surface micromorphology of the PAN fiber papers and carbon fiber papers were observed using scanning electron microscopy (SEM, JSM-IT1300A, JEOL, Japan) at an acceleration voltage of 5.0 kV. For the measurement of fiber diameter, the image processing software Image J (ImageJ 1.53c, USA) was used, and each sample was tested five times. Wide-angle X-ray diffraction (WXRD) patterns were recorded using an X-ray diffractometer (X’pert PRO, PANalytical, Netherlands) at a voltage of 40 kV and current of 40 mA. The FTIR analysis of samples was performed using a FTIR spectrometer (VERTEX 70, Bruker Technologies Ltd., Germany) with the KBr technique. All spectra were taken in the spectral range of 4000–400 cm−1 using an accumulation of 64 scans with a resolution of 4 cm−1. The static contact angle was measured using an optical contact angle measuring system (OCA 15EC, DataPhysics Instruments GmbH, Germany) with water droplets of 3 μL. Each sample was tested three times and the results were averaged. The electrical conductivity and resistivity of the PFP were measured using a dual electrical measurement four-point probe tester (RTS-9, Guangzhou 4 PROBES TECH, China). Each sample was tested 50 times at different points and the results were averaged. The electrochemical performance of the carbon fiber papers (1 cm × 2 cm) was tested on a CHI440 C electrochemical quartz crystal microbalance (Shanghai Chen Hua Instrument Co., Ltd., China), in which the resultant electrode served as the working electrode without current collectors; moreover, platinum was used as the counter electrode and Ag/AgCl in a 3 M KCl aqueous solution was used as the reference electrode. In this study, 1 M H2SO4 was chosen as the electrolyte to investigate the electrochemical performance of the electrodes. Cyclic voltammogram tests were performed in a voltage window of −0.4 to 0.2 V at room temperature.
Results and discussion
Morphological analysis of polyacrylonitrile fiber paper and PAN carbon fiber paper
PAN fibers were obtained from the PAN spinning solution by centrifugal spinning; then, the PCFP were prepared by hot pressing, pre-oxidation, and carbonization. The morphology of the PFP prepared at different mass ratios of PAN to DMF were studied via SEM. Figures 2(a) and (b) showed the SEM images of PFP-1, PFP-2, PFP-3, and PFP-4. The disorderly arrangement, smooth surface and uniform morphology of all PAN fibers could be clearly observed. Figure 2(c) showed the diameter distributions of PFP-1, PFP-2, PFP-3, and PFP-4. The diameters of all fibers were within 7 μm. The average diameter of PFP-1 was the smallest at approximately 2.790 μm, whereas the average diameter of PFP-4 was the largest at approximately 5.973 μm. Moreover, Figure 1(d) showed that the fiber diameter increased with the increase in the PAN mass ratio. Similarly, the morphology of the PFP prepared at the same mass ratio and different average molecular weights was studied by SEM. The surface and cross-sectional morphologies of these fiber paper were not significantly different from those of the fibers prepared with different mass ratios. Figure 2(c) showed the diameter distributions of PFP-5 and PFP-6. The average diameters of PFP-5 and PFP-6 were larger than those of PFP-2 and PFP-3 by approximately 4.392 μm and 4.660 μm, respectively. Figure 2(e) showed the relationship between the fiber diameter and the average molecular weight, and it can be observed that the fiber diameter increased as the average molecular weight increased. SEM images of the PAN fiber paper and fiber diameter distribution with different mass fraction and average molecular weight of the PFP-1, PFP-2, PFP-3, PFP-4, PFP-5, PFP-6: (a) the surface, (b) the single fiber, (c) fiber diameter distribution and relationship between fiber diameter and (d) mass fraction, (e) average molecular weight.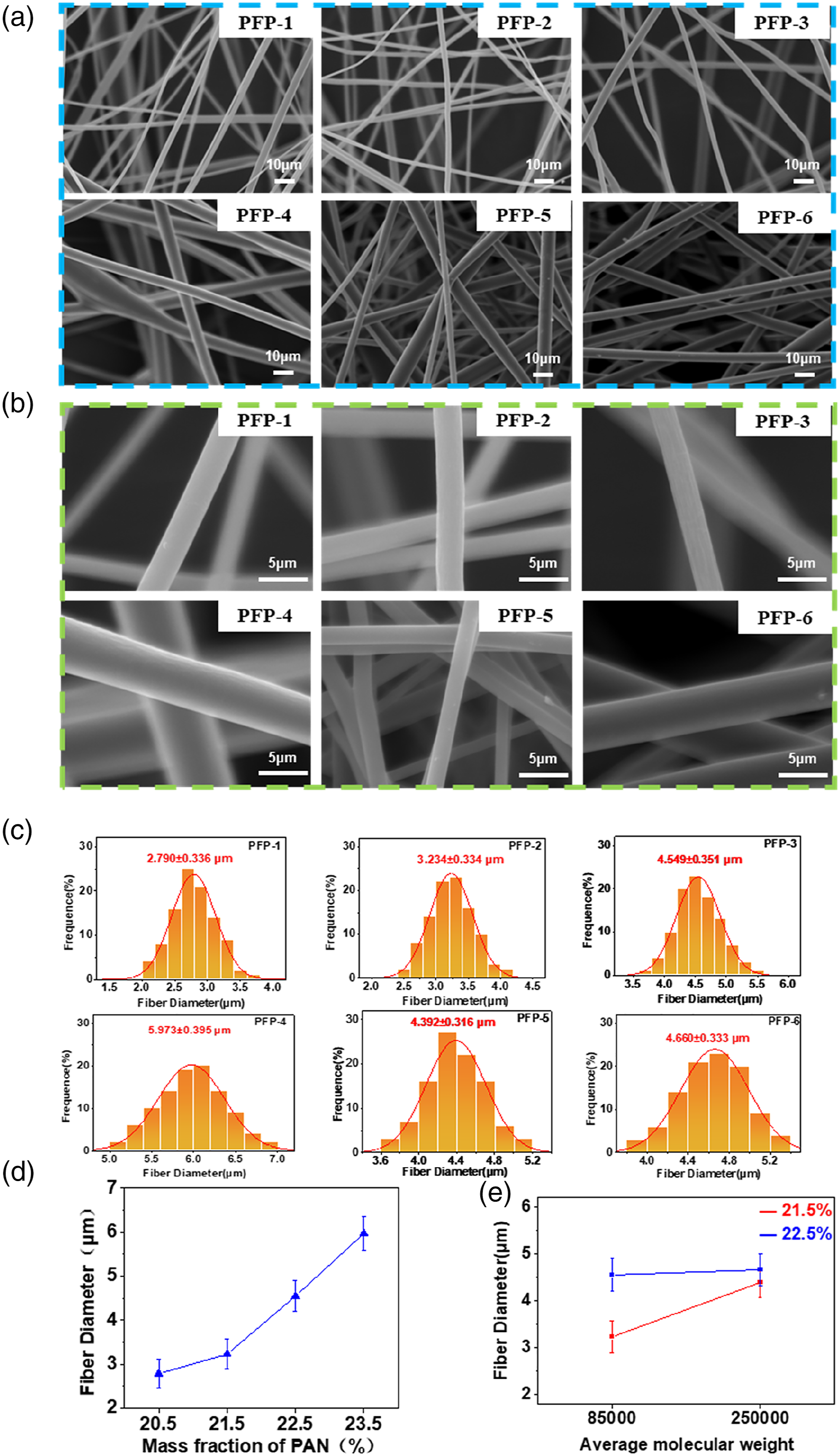
Figure 3(a) showed the SEM image of the PCFP; the fiber surface was smooth, with no wrinkling or twisting phenomenon, similar to that of PFP. Figure 3(d) and (f) showed that the fiber diameter distribution was in accordance with the above rule, and the PAN carbon fiber diameter was smaller than the PAN fiber diameter. The average diameter of PCFP-1 was the smallest at approximately 2.106 μm, whereas the average diameter of PCFP-4 was the largest at approximately 4.078 μm. As the average molecular weight increased, the fiber diameter increased from 2.990 μm for PCFP-2-1200 to 3.481 μm for PCFP-5-1200, and from 3.688 μm for PCFP-3-1200 to 3.983 μm for PCFP-6-1200. This occurred because thermal cracking removed certain small molecular products from the fibers, and the carbonization process limited the axial shrinkage of the fibers during the carbonization process.26,27 Thus, the fibers exhibited a large radial shrinkage macroscopically, and the fiber diameter decreased. Figure 3(e) showed the effect of different carbonization temperatures on the diameters of the carbon fibers. The sample with the lowest carbonization temperature had the largest fiber diameter of 3.997 μm. As the carbonization temperature increased, the fiber carbonization process became more complete, and the diameter decreased. SEM images of the PAN carbon fiber paper and fiber diameter distribution with different mass fraction, carbonization temperature and average molecular weight of the PCFP-1-1200, PCFP-2-800, PCFP-2-1000, PCFP-2-1200, PCFP-3-1200, PCFP-4-1200, PCFP-5-1200, PCFP-6-1200: (a) the surface, (b) the single fiber, (c) fiber diameter distribution; and relationship between fiber diameter and (d) mass fraction, (e) carbonization temperature, (f) average molecular weight.
Chemical structure of polyacrylonitrile fiber paper and polyacrylonitrile carbon fiber paper
The X-ray diffraction patterns of the different PFP (PFP-1 to PFP-6) were shown in Figure 4(a). It was observed that the PFP had two peaks at approximately 2θ = 16° and 2θ = 25°. The peaks around 2θ = 16° represented the crystalline structure of the fibers, and those around 2θ = 25° were corresponded to the amorphous peaks. As shown in Table 3, the crystallinity of PFP-1, PFP-2, PFP-3, PFP-4, PFP-5, and PFP-6 were 31.2%, 35.4%, 35.7%, 36.6%, 39.2% and 35.9%, respectively. It was observed that, as the mass fraction increased, the crystallinity of the primary PAN fibers increased.
28
At the same mass fraction, as the average molecular weight increased, the crystallinity of the fiber increased. At a molecular weight of 250,000, the crystallinity of the high-mass-fraction sample was lower than that of the low-mass-fraction sample. The reason for this phenomenon may be that the viscosity of the high-molecular-weight PAN spinning solution was too high, and the spinnability decreased with the increase in the mass fraction. (a) WXRD patterns of the PAN fiber paper: PFP-1, PFP-2, PFP-3, PFP-4, PFP-5, PFP-6 and (b) WXRD patterns of the PAN carbon fiber paper: PCFP-1-1200, PCFP-2-800, PCFP-2-1000, PCFP-2-1200, PCFP-3-1200, PCFP-4-1200, PCFP-5-1200, PCFP-6-1200. Crystallinity of each PFP.

Crystallinity of each PCFP.

Figure 5(a) and (b) showed the FTIR spectra of PFP and PCFP, respectively. For PAN, the observed peaks at 2241, 2929, 1728, 1662, 1450 and 1360 cm−1 could be assigned to the -C≡N stretching vibration, -CH3 stretching vibration, -C = O stretching vibration, C = C stretching vibration, -CH2 bending vibration, and -CH3 bending vibration. For PCFP, only peaks appeared obviously around 800 and 1370 cm−1 and there were almost no characteristic peaks in the range of 1600–4000 cm−1. These phenomena indicated that decomposition reactions occurred during the carbonization process, and most of the organic matter was completely decomposed, leaving only carbon. FTIR spectra of (a) PFP and (b) PCFP.
Electrical properties of polyacrylonitrile carbon fiber paper
The electrical performance index was the most important performance parameter of the carbon fiber paper and it directly affected the normal usage of the carbon fiber paper. Figure 6 showed the effects of different mass fractions, carbonization temperatures, and molecular weights on the electrical conductivity and resistivity of the carbon fiber paper. Figure 6(a) showed that the electrical conductivity of the carbon fiber paper gradually increased and the resistivity gradually decreased with a continuous increase in the mass fraction. PCFP-4-1200 exhibited the best conductivity and resistivity of 29.286 s·cm−1 and 0.0345 Ω·cm, respectively. As the conductivity increased, the resistivity decreased and the electrical conductivity increased. The reason for this phenomenon may be that the increase in the fiber diameter and greater crystallinity lead to a larger space for the movement of charges among the fibers, which in turn led to better electrical conductivity.1,5 As shown in Figure 6(b), the carbonization temperature also had a significant impact on the electrical properties, and as the carbonization temperature increased, the electrical properties were improved. In particular, when the carbonization temperature was 800°C, the resistivity reached 1.830 Ω·cm and there was almost no electrical conductivity. When the carbonization temperature was less than 1000°C, the carbon fiber paper exhibited a great resistivity; conversely, when the carbonization temperature reached 1200°C, the electrical conductivity of the same samples exhibited approximately four-fold increase (PCFP-2-1200 to PCFP-2-800). This was because the carbonization process was divided into two stages: low-temperature carbonization (300–1000°C) and high-temperature carbonization (1000–1600°C).
29
Because PCFP-2-800 and PCFP-2-1000 were not carbonized at high temperatures, the carbonization process was incomplete, and the carbon content and crystallinity of the samples simultaneously became significantly low, resulting in weak electrical properties. A higher molecular weight also resulted in better conductivity. Under the same process, the conductivities of PCFP-5-1200 and PCFP-6-1200 were twice those of PCFP-2-1200 and PCFP-3-1200, as shown in Figure 6(c). From the previous fiber diameter distribution and WXRD diagrams, it can be observed that the samples with higher molecular weights had larger diameters and higher crystallinity, and the microstructure of the samples was more stable, which was conducive to the mutual conduction and flow of electrons. Therefore, high-molecular-weight PAN led to better performance of the carbon fiber paper. PCFP-6-1200 exhibited the highest conductivity, reaching 43.250 s·cm−1. Figure 7 showed that the prepared PAN carbon fiber paper had a higher conductivity than the reported carbon fiber paper,6,8,30,31 and also proved that the preparation of carbon fiber paper by centrifugal spinning had a good prospect in related fields. (a) Resistivity and conductivity of the PAN carbon fiber paper with different mass fraction: PCFP-1-1200, PCFP-2-1200, PCFP-3-1200, PCFP-4-1200, (b) Resistivity and conductivity of the PAN carbon fiber paper with different carbonization temperature: PCFP-2-800, PCFP-2-1000, PCFP-2-1200 and (c) Resistivity and conductivity of the PAN carbon fiber paper with different average molecular weight: PCFP-2-1200, PCFP-3-1200, PCFP-5-1200, PCFP-6-1200. Comparison of the electrical properties towards conductivity with reported work.

Electrochemical performance of the carbon fiber paper
The electrochemical characterization of the PAN carbon fiber paper-based electrodes was conducted in a three-electrode system with an electrolyte of 1 M H2SO4. Figure 8(a) showed that the cyclic voltammetry (CV) tests of the electrodes under different mass fractions of PAN exhibited a quasi-rectangular shape at a scan rate of 50 mV·s−1 and the rectangular area increased positively with an increase in the mass fraction of PAN. It can be observed that, with the increased mass fraction of PAN, large-area CV curves was obtained, which showed better areal capacitance. This phenomenon occurred because the porous structure of the carbon fiber paper provided large amounts of space for electron transport channels. Electrochemical performance of carbon fiber papers. (a) CV profiles of Carbon Fiber Paper electrodes at 50 mV s−1 and (b) CV profiles of PCFP-6-1200 electrode at various scan rates.
CV tests were also performed on the PCFP-6-1200 electrode at various scan rates ranging from 1 to 200 mV·s−1, as shown in Figure 8(b). The results showed that the CV curves remained approximately rectangular at this broad scan rate, indicating that the PCFP-6-1200 electrode possessed a superior rate capability.
Hydrophobicity of polyacrylonitrile carbon fiber paper
Owing to the working environment of the carbon fiber paper, strong hydrophobicity was essential, which should be sufficiently high to achieve a superhydrophobic effect.
32
Figure 9 showed the contact angles of the PCFP with water for different processes. The figure showed that the contact angles of the carbon fiber papers with different mass fractions and carbonization temperatures were maintained at approximately 140°, which was almost superhydrophobic in nature. Even if the carbonization temperature was too low, the contact angle remained high. Figure 8(a) showed that the contact angle first increased and then decreased, but the range of increase and decrease was not limited by the increase in the mass fraction. PCFP-3-1200 had the highest contact angle of 142°, but it was only 2.1° higher than that of PCFP-1-1200, which exhibited the lowest contact angle. As shown in Figure 9(b), when the carbonization temperature increased, the contact angle also increased marginally. (a) OAC to water of the PAN carbon fiber paper with different mass fraction: PCFP-1-1200, PCFP-2-1200, PCFP-3-1200, PCFP-4-1200 and (b) OCA to water of the PAN carbon fiber paper with different carbonization temperature: PCFP-2-800, PCFP-2-1000, PCFP-2-1200.
Conclusion
In conclusion, this study demonstrated a novel and efficient method for the preparation of high-performance PCFP. The PCFP prepared by centrifugal spinning exhibited a dense 3D structure and controllable fiber morphology. Consequently, the obtained PCFP had the highest electrical conductivity of 43.250 s·cm−1, and all the PCFP exhibited significantly strong hydrophobicity. Thus, the PCFP prepared in this study had considerable application prospects in the field of fuel cells. It was hoped that this work will provide guidance for the preparation of high-performance PCFP in the future, which can be applied in the energy field to meet the needs of the rapid developments in the current energy industry.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Thanks to the Analysis and Testing Center of Wuhan Textile University for providing the characterization instruments. This study was financially supported by National Natural Science Foundation of China (No. 52173062, U21A2095) and The Key Research and Development Program of Hubei Province (No. 2020BAB080, 2020BED004, 2021BAA069, 2021BAA068).
