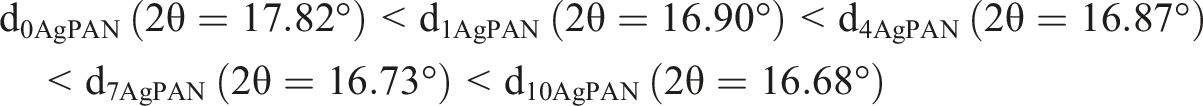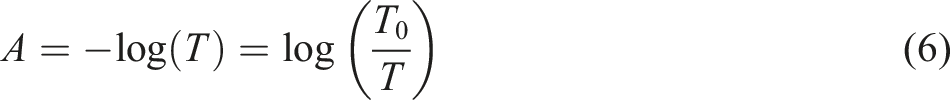Abstract
Research problem
The demand for polymeric materials is increasing day by day, provided that they have excellent virtues to meet desired needs. The best way to obtain high-quality polymeric materials was surface treatment in multiple methods. All the methods that have been used were expensive, multi-stage, harmful to the environment, and other shortages. Therefore, we sought in our paper to invent and develop a surface metallization protocol under green and safe conditions, based on integrating two safe methods for preparing nanomaterials (electrospinning/ultrasound) in one-pot.
Aim of this work
The main objective of this work is to answer a question that has been neglected in other papers: how does in-situ metallization affect the crystalline behavior of the semi-crystalline phase of the PAN matrix depending on different amounts of MA NPs and their synergy for the rest of the properties (morphological and structural) is presented, in first degree. In the second degree, an explanation of the synergy mechanism between the mechanical/hydrophilic properties and the modified structural properties of metalized fibers has explained.
Results
Here, an unaccustomed and green methodology to metalize the polyacrylonitrile, PAN, surface is reported with the intention of fashioning of a stable fibrous structure and the best specifications. The in-situ metallization process is essentially based on a loading of various amount of silver nanoparticles, as MA NPs “MA NPs”, (1 wt%, 4 wt%, 7 wt% and 10 wt%), which prepared by ultrasound irradiation assisted with a little amount of a reducer system (hydrazine/NaOH), on the PAN surface. The physiochemical characterization of as-synthesized fibrous samples was consisted of two parts: (i) characterization of sample nature (utilizing of XRD and UV-Vis. techniques), (ii) the chemical structure/morphological characterization (utilizing FESEM, TEM and AFM techniques) with performing all relevant measurements. From crystalline and UV-Vis. assessments, the observed shift in the diffraction peaks towards lower angles and slight shifts in the absorption bands of PAN (1 0 0) in the 1AgPAN and 4AgPAN samples, containing smaller-sized nanoparticles, are overt results of an improvement in the crystalline traits of the parent matrix. The FT-IR data revealed the intensification and overlapping of the IR-bands “C=N, C≡N, N-H, C=O and O-H” without band shifting for fibrous samples with low content of MA NPs (1AgPAN and 4AgPAN samples). The intercalation of the MA NPs between the chains of the molecular matrix and the strong interconnection/hydrogen bonds between both components explain not only the change of the crystalline properties of the crystalline matrix but also the homogeneous distribution and creation of a stable layer of the MA NPs on the 0AgPAN surface, confirmed by the FESEM and TEM images. The results of the study of mechanical properties showed that the loading of small spherical particles onto the polymeric matric, in particular 1AgPAN and 4AgPAN samples, was positively reflected on the improvement of the mechanical performances and the related values, not in the 7AgPAN and 10AgPAN samples. Because the MA NPs have a hydrophobic character, loading them onto the bare polymeric fiber surface made the metalized fibers a stronger hydrophobic than that of hydrophilic character, which is consistent with differences in surface roughness (increasing Ra values). The main focus of this paper was to study the synergy of structural properties with each other. So, the results of the synergistic relationship study showed that the low amount of the MA NPs led to a comprehensive and positive correlation between all structural properties and in turn affected the mechanical properties-hydrophobic/hydrophilic properties. Noting that the fibers of the 1AgPAN and 4AgPAN samples had the best structural stability, compared to the fibers of the 0AgPAN, 7AgPAN and 10AgPAN samples, when they were: decreasing in Z value, increasing fiber diameter/particle size “P.S.”, increasing RCR and C.D. values and decreasing D.D, alteration in values obtained of the mechanical behavior and the increasing of contact angle “CA” values for water “H2O” droplets and decreasing for ethylene glycol “EG”. As a final result, the changes observed in the aforementioned measurements were reasonably regular and gradual for fibers with low MA NPs content, while the same measurements were significantly deteriorated in the case of fibers with higher MA NPs contents.
Keywords
Introduction
In a world in which environmental, energy, health, economic, industrial and sustainability problems are expanding, research teams in all disciplines around the world are constantly working to keep pace with developments in science and technology, hoping to reach the best results.1–6 The continuity of keeping pace with development lies in three fundamental aspects: (1) finding effective solutions to the crises facing developing countries, (2) investing time in the best way by selecting good protocols for the production of materials with high quality specifications and (3) observing the conditions of green chemistry to prevent the creation of any environmental danger to the environment or health on the work staff.2,6–10 Today, among the materials that occupy a large space in the field of technological developments and research studies, are polymers (both industrial and natural), due to their abundance, ease of handling, distinctive properties and other prominent virtues. Since Hermann Staudinger won the Nobel Prize in 1920 for his important discovery in polymeric materials to the present century, countless numbers of polymeric materials have been produced using a variety of manufacturing techniques.11–13 In this context and based on the statistical reports issued by the economic research centers, the indicators of the global polymeric materials market are witnessing a huge growth for the purchase and production of these materials, assuring that these indicators will reach their highest summit in the near future.11,14 For example, not to be limited to, in the United States of America, the purchase of polymeric materials has been estimated at a whopping sum of ˜ 530$ billion, that the rate of purchase will increase by ˜ 5% during the forecast period (2020–2030), to become ˜ 830$ billion by 2030.11,14 Recently, in order to reduce the high costs of securing these important materials and using those on a larger scale in various fields, the role of nanoscience and nanotechnology has come to pave the way for designing polymeric surfaces with different dimensions and shapes (fibers, composite powders, etc.) with new properties.11,13,15–18 Among these formulations, polymeric fibrous structures, which are classified under the group of one-dimensional structures, have a prominent position in the list of nanomaterials, because they have very significant structural properties materials (good mechanical properties, distinctive hydrophilic properties, good electrical and thermal properties, etc.) compared to ordinary fibers or polymeric raw materials.13,19–21 A variety of protocols has been applied for fabrication of the fibrous structures which differs from each other in terms of their applied conditions and mechanisms, including: phase separation, 22 chemical vapor deposition (CVD), 23 template synthesis, 24 self-assembly, 25 wet-chemical route, 26 and electrospinning 27 and so forth. Among them, electrospinning has aroused the interest attention by polymeric researchers in the past two decades and can be classified among the most promising green methods for producing large quantities of long fibers, textiles, ceramics and composites with high continuity within a short time .19,21,28 The origin of this method is to prepare a viscous polymeric solution then stretch it by Taylor cone jetting under high voltage electrostatic forces on the syringe tip . 21 This method contributes to the treatment of alignment and modeling of fibers that become attractive features (high specific surface area, high surface area-to-volume ratio, porosity).27,29,30 These specifications make the resulting fibers applicable in the fields of energy, biotechnology, absorbent materials, and filters . 31 One of the most important technical observations in electrospinning are that the desired fibrous structures (with variable diameters (ultra-thin or micron-diameters) and various morphological surfaces (e.g. porous fibers, cactus-shaped fibers, creased fibers, etc.) can be obtained by controlling the experimental conditions such as the viscosity of the polymeric solution, the applied voltage, the regulation of the distance between the collector and the syringe.27,31,32
Although polymers are commonly used materials, it is not recommended to use them in their virgin form, because of their weak mechanical properties, re-polymerization reaction and purification (especially if they are of natural origin), their morphological structure, bulk structure, poor electrical and thermal conductivity. 33 It is necessary to find other more effective treatment methods that include utilization of polymeric waste, more efficient treatment of natural polymers, utilization of biomolecules, more feasible protection of weak properties of polymers, therefore, the concept of “surface modification” of electrospun fibers has been defined and widely used.33,34 So far, various strategies (mechanical strategies 34 and chemical strategies , 35 that possess a direct influence on the behavior, stability, and physicochemical properties of the polymer (wetting, mechanical properties, bio-properties, etc.), 35 are available to accomplish modification of a large number of polymeric fibrous surfaces such as polycaprolactone “PCL”, poly (L-lactic acid) “PLLA”, poly (styrene-alt-maleic anhydride) “PSMA”, Poly (vinylidene fluoride) “PVDF”, etc.29,34 The basis of these methods is based on dramatically chemical or physical reaction between polymer and chemical additives.34,35 With the beginning of 21th century, the method of modifying polymeric fibrous surfaces by metallization, so-called surface metallization “either with an in-situ or ex-situ path”, has received indescribable interest from surface engineering specialists and polymer science researchers.19,20,36–38 More precisely, it is possible to define the term of “metallization of polymeric surfaces” as follows: all operations in which natural, synthetic origin or hybrids polymers, in their various formulas (solutions, fibers), are shaped or combined with micro or nano-scaled inorganic compositions (alloy, zero-valent metal, metal-organic frameworks, oxides) to obtain a new product characterized by its function of use and its ability to meet the desired goals .36–38 The process of electrospun nanofibers metallization is implemented through three essentially stages: thermal treatment of the polymeric solution in order to activate the polymer surface, adding inorganic material (either prepared using one of the synthesizing ways or adding the mineral raw material used) for the metallization procedure, spinning the fibers.33,36,37 After the metallization of the fibers, secondary treatments such as calcination of the fibers at high temperatures, or soaking in an alkaline solution 35 can be followed in order to further activate the surface of the fibers.37,39 The fiber produced by this process can be called metalized fiber and metalized agents, respectively.13,31 When the metalized material is blended with the polymer, there are two possibilities for the metalized fiber form.33,39 Either a homogeneous fibril structure is generated by completely miscible of phases or a fiber with heterogeneous structure is produced by immiscible phases. The final product must possess specifications that outweigh the properties of each of the constituent materials. The polymeric matrix, or as-known as a continuous phase, ensures a suitable environment for uniformly loading the metallic agent along the metalized fibers and preventing agent from accumulating on the fiber surface, because it contains anchor centers along its main chain.31,33,38,39 However, the metalized agent, or as-known as a discontinuous phase , 13 plays the role of the enhancer of the properties, improving the orientation and crystallization of the polymer chains in the parent matrix, increasing the cohesion and stability of the fibrous structure. Among the factors affecting the properties of the metalized fibers: the nature of the polymeric matrix, its surface charge, the type of functional groups in the matrix backbone, the interface quality, the dimensions of the metalized agent and its nature .31,33 This type of fibrous structures affects in many filed of distinguished applications, namely: manufacturing of aircraft/ships and automobiles, anti-bacterial textile, bio-scaffolding and others.33,40 It must be emphasized that, according to the foundations of green chemistry, some of the metallization strategies (Laser-induction, thermal spray, ion-exchange self-metallization, CVD and so on)33,39,41 are undesirable for application, due to the release of toxic gases, the use of toxic reactants, the formation of pollutants and residues of toxic solvents and harsh condition applied. As an alternative, green chemistry researchers always recommend searching environmental-friendly alternatives, which may create safer conditions such as use of non-toxic solvents, to reduce energy consumption and time required to perform the metallization method and so on to replace those applied in the aforementioned methods.33,39,40 According to these surveys, it can be confirmed that this prevalent strategy is a remedy for many current and future challenges in several areas, through which it will save developing countries billions of dollars.33,39
Amongst the polymeric fibrous materials, polyacrylonitrile nanofibers (PAN NFs) have recently been increasingly popularized, due to their interesting physical and chemical properties, unusual specific surface, outstanding morphological features, abundant active sites, etc.42–44 Despite its good structural advantages, these fibers have drawbacks, including: insufficient crystalline organization (semi-crystalline material), limited mechanical properties, orientation, thermal and electrical conductivity.42–44 The importance of these distinct fibrous structures is attributed to the PAN chemistry, not only due to the high carbon content (up to 56%, compared to other polymers), but also due to this polymer being highly malleable to changing its chemical structure under the reactive conditions of different strategies.42,45 The most important characteristic of PAN from the rest of its counterparts is the possibility of conducting cyclization and dehydrogenation reactions using different treatments (heating in nitrogen atmosphere, oxidative heating in the presence of air, heating at very high temperatures in the abundant oxygen-containing atmosphere, gamma rays, microwaves).33,41,42,46 These two reactions generate distinct structures with different formulas, depending on the type and conditions of treatment, for example, cyclic, ladder and graphitic structures. 33 Each of these structures has different physicochemical characteristics from the other, and the mechanism of their interaction with many materials differs. The cyclization reactions mean the transformation of a large part of the nitrile groups (C≡N) in the linear polymer structure into double bonded nitrile groups (C=N).33,43 The gradual or complete disappearance of the infrared-bands of the nitrile groups at 2242 cm−1 and the appearance of the double bonded nitrile groups located at about 2200 cm−1 clearly indicate the formation of a crosslinked structure in the fibrous structures.43,44,46 However, the dehydrogenation reaction is directly related to the loss of hydrogen atoms from the polymer backbone with progression transformation time.42,44 The intensification of the adsorption infrared-bands of the methylene (CH2) groups around 2920–2939 cm−1 inferred to the loss of H-atom. Related fibers are carbon-fibers with a high content of aromatic structures of a ladder or graphite formula.33,43,47 The difference between both reactions is that the cyclization reaction takes place in an easier way and does not require harsh conditions to occur.19,33,42 It can occur by exposing the target polymer to any simple heat treatment, unlike the dehydrogenation reaction. Among the differences mentioned in other papers, related to changing the structure of PAN, it has been highlighted that the structure resulting from the cyclization reaction is more chemically active, as it contains more effective functional groups, compared to the structure resulting from the other reaction .33,43 The common notes between both reactions are: both reactions occur together, caused the structure stability, changing in the color of the polymer solution or the color of the suggested the reaction occurrence and release of toxic volatile gases (NH3, HCN, CO2 etc.).19,42,43,46,48 The polymer researchers relied on the method of thermal oxidation and subsequent carbonization to produce surface-modified PAN fibers with a stabilized structure.49,50 More than nearly 90% of the world’s commercial carbon fiber production has benefited from this method.19,42 Because of the frequent use of this method, the complexity of the stages, the inability to control many of the chemical reactions, the generation of by-products with different molecular weights, the thickening of very dangerous gases during heating at high temperatures, the metallization treatment of the fibrous surface of PAN provides an advanced avenue to transport their properties to the best levels.43,51,52 When performing this method, it is likely: the properties of the metalized-PAN fibers are much better than those of the fibrous surfaces modified by the thermal oxidation methods, and there is greenness in the work stages .19,39,40 Particles of noble metals such as Pd NPs, Ag NPs, Au NPs, Pt NPs, etc. are in the forefronts of metallic compositions used in the PAN metallization strategy, which is very popular with surface engineering researchers.6,53–56 In general, the use of these metals gives the ineffable improvements to the structural properties of polymeric fibrous structures, allowing that the metalized fibers were used in many prospect applications.51,57,58 Silver nanoparticles “Ag NPs”, as a cheap material, prepared by different green methods, anti-bacterial properties, orientation and alignment reinforcements for structures (particularly polymers), high thermal stability, crystallization with face-centered cubic system, attractive shape-controlled, surface plasmon resonance property, small nanoscale, high reactivity with polymeric substrates, shrinkage reducers for polymeric chains, are considered as valuable sensitizing activities for metallization surface of PAN and expanding its usage in various fields.4,16,59–61 Likewise, Ag NPs produced by many preparation methods have high surface energy and tend to aggregate unless properly bonded with capping agents as polymers.60–61 Numerous studies have shown that manipulating the surface morphology of Ag/PAN fibers by establishing appropriate bonds between Ag and PAN allows to reduce or improve the crystalline phase, mechanical properties of PAN, etc. 62 that have considerable impact in their applications. 63 Kara’s group has shown how the different morphologies of the three noble metallic particles affected the structural properties of PAN. They deduced the positive synergistic link between these properties and the mechanical/wetting properties. 19 Zhang et al. postulated grafting the surface of the PAN with the amino hyperbranched polymers (HBP) via a chemical reaction pathway between the cyanide groups in the PAN and the amine groups in the HBP. 64 So that the resulting composite fiber (PAN-G-HBP) becomes superficially active that plays as complexing/reducibility agent for Ag ions. In the later stage, the surface of the PAN-G-HBP fiber has been metalized by coating it with a uniform layer of Ag NPs under hot steaming condition. 64 They also, based on the obtained data, proved that each of the two components protected each other’s properties. The produced fiber showed high antibacterial susceptibility against Escherichia coli and Staphylococcus aureu 64 . Jang et al. proposed that the good miscibility between the silver/graphene oxide “Ag/GO” and PAN nanofibers is as a detectable factor for enhancing wetting. 65 Pan 66 and Chen 67 an antibiofouling forward osmosis (FO) membrane and a reusable/ant-toxicity membrane by facial chemical activation/metallization approaches of PAN-based nanofibers under electrospinning conditions in the presence of Ag NPs and Myoporum. bontoids Ag/g-C3N4, respectively, which were as stabilizer/reinforcement agents. Ma et al. 68 Mai et al. 69 successfully synthesized electrospun Ag-containing nanofibers from solutions of hyperbranched polyethyleneimine grafted polyacrylonitrile/graphene oxide (PAN/DMF/HPEI-GO) and polyacrylonitrile/N,N-Dimethylformamide/zeolite imidazolate framwork-8 (PAN/DMF/ZIF-8), respectively. After transformation of Ag ions to Ag zero-valent by reduction reaction, the modified membranes demonstrated improved remediation and recovering of Ag(I) 68 and p-nitrophenol . 69
In this paper, for the first time, bare PAN fiber and metalized PAN NFs in the presence of various amounts of Ag NPs were prepared by safe protocol, as shown in Figure 1. PAN was selected as the reference polymeric matrix with poor mechanical properties, wetting and crystal orientation due to its susceptibility electro-spinnability and good tensile strength after electrospinning route. More importantly, PAN is an ideal silver ion carrier, making it an outstanding support for the in-situ metallization surface. Silver nanoparticles, because of its high structural properties (crystalline, small-scale of spherical morphology, orientation enhancers) can modify the surface of bare matrix fibers. The novelty of this paper revolved around some remarkably points, as following: (i) Designing a modern/facial green protocol (referred to the greenness of the protocol in the 1.2.2. section “Supporting information”) to metallize the surface of PAN through the integrating of two efficient methods (electrospinning/ultrasound irradiation) in the preparation of nanomaterials and to replace conventional/expensive chemical sensitizing-modification PAN surface. (ii) Unlike previous report,17,45,64 this paper mainly focused on showing the effect of different additions of Ag NPs on all structural properties of the polymeric matrix (crystalline, morphology, structural and surface roughness). (iii) Within the same context, the stability of the polymeric matrix structure was studied by some calculations made using related data. (iv) On the other hand, because of the limited and paucity of research in which the synergistic property of silver-metallic polyacrylic nitrile fibers was discussed. The synergistic relationship between crystallization and the rest of the structural properties was defined in detail. (v) The effect of each fibrous structure with its own content of Ag NPs was separately determined on the values of Young’s modulus, ultimate tensile strength, elongation-at-break and contact angle values of two liquid droplets of different nature (H2O and EG). (vi) The relationship between the results of the last two properties and the structural properties was investigated, and it was found that there is a positive relation between them. It is noteworthy that this research is a continuation of our previous work
20
and the work of Karbownik,
70
but with a new point of view. The current work is expected to provide a potential approach for fabricating highly hydrophilic/hydrophobic PAN nanofibers as well as mechanical features utilized in the wastewater treatment and oil/water separation process. In this paper, all as-synthesized fibrous samples were analyzed by X-ray diffraction (XRD), field emission scanning electron microscopy (FESEM), transmission electron microscopy (TEM), atomic force microscopy (AFM), UV-Vis spectroscopy and Fourier-transform infrared spectroscopy (FT-IR). The CA values and tensile parameters were determined to ascertain the effect of metallization PAN surface on the hydrophobicity/hydrophilicity and mechanical behaviors of un-metalized sample. Schematic diagram of the experimental protocol used to fabricate metalized nanofibers: Step1: Preparation of MA NPs by ultrasonic irradiation method, Step 2: Preparation of metalized nanofibers by electrospinning method.
Experimental section
This section, including (materials, methods, importance the protocol used, apparatuses, mechanical and wetting assessments), detailed are supplied in the Supplementary materials. The results of the relevant measurements of the prepared fibrous materials are organized into the appropriate tables (from Table S1 to Table S4, Supplementary materials).
Results and discussions
Physical Characterizations
Study of XRD patterns and UV-Vis spectroscopy
The XRD patterns of all electrospun polymeric fibers “(0AgPAN, 1AgPAN, 4AgPAN, 7AgPAN and 10AgPAN) samples” and MA NPs powder were introduced in the following patterns (Figures 2(a) to (f)) and were processed via X’Pert HighScore Plus 2.1 software. Furthermore, the crystalline nature of all samples was more confirmed by explanation appropriate reasons for their formation and performing the crystal calculations that were recapitulated in Table S2. The XRD pattern of 0AgPAN has been given in Figure 2(a). According to this pattern, the observed strong diffraction peak at 17.84° with a crystal plane (1 0 0), compared to another one located at 27.06°, divulged the long-range arrangement of crystalline phase of the polymer molecules in these fibers .
70
The second peak was centered at 27.06° with a lower intensity and a wider peak, (0 2 0) crystal plane, than another peak associated with the irregular state of the cyclized formula of matrix polymer in the PAN macromolecule chain, as clearly seen in Figure 2(b), in good agreement XRD findings by Zhang et al.
71
The formation of intramolecular cross-linking or hydrogen bonding between PAN molecular chains, the presence of hydrogen bonds between functional groups such as nitrogen groups, α-H, O-H, C=O, etc., weak intramolecular interactions of dipole-dipole type between the nitrile groups and the short time of the desired method to form the electrospun polymer fibers are remarkable clues to explain the irregular nature observed of the bare PAN NFs at both diffraction angles (17.84° and 27.06°).71–73 The XRD pattern of MA NPs was given in Figure 2(c). The appeared strong peak at 38.15°, having the orientation of (1 1 1), was closely indexed to the face-centered cubic “FCC” regime (JCPDS file (96–110-0137) with space group of Fm XRD patterns (at the rang of 2θ: 10–80°) of 0AgPAN (a), showing the shape of the diffraction peak in the rang 2θ of 10–34° (b), Ag NPs (c), 1AgPAN (d), 4AgPAN (e), 7AgPAN (f) and 10AgPAN (g).
It should be noted that after metallization process, the permeation of the MA NPs to internal surface or their loading on the external surface of polymeric matrix created a kind of a spatial retardation among the PAN macromolecular plane, depending to Bragg’ equation, via the formation of powerful interconnections between nanoparticles and polymer.
The crystal size (D) and the degree of crystallinity (Z %) of PAN for (1 0 0) crystal plane was calculated by following equations (Eqs. (1) and (2)) using Debye-Scherrer’s formula and using the Hinrichen’s method, respectively70,74
In equation (1), “β” is the full at width half maximum (FWHM), “λ” is the wavelength of Cu Kα (1.5406 Å) of radiations, “θ” is the Bragg’s angle and “D” is the mean crystallite size of the sample. While, in equation (2), “Ic” is the integral of the peak corresponding to the crystalline phase of the polymer, and “Ia” the integral of the peak corresponding to the amorphous phase of the polymer. According to Table S2, the size of the PAN crystals gradually increased with the gradually addition of the MA NPs level, because the crystallization approach of the polymeric matrix was accelerated by fortifying the level of the crystallized metalized agent. Thus, that component has a positive effect on improving the primary crystals sizes and the crystalline phase of the polymeric matrix. Lastly, from the outcomes of all patterns in Figure 1, it can be extracted that all samples displayed various intensity diffractions and the width of peaks at 2θ values of PAN (1 0 0) and MA NPs (1 1 1), because of the difference the crystalline phases, formation multiple crystallites and different components in such blended regimes . 75 According to resulting data, all peaks were in agreement with the standard peaks of both components in their fibrous samples (1AgPAN, 4AgPAN, 7AgPAN, 10AgPAN) without any impurities.
MA NPs loading within the polymeric matrix, after in-situ metallization process, was studied by comparison between the UV-Vis spectra of pure as-prepared materials (Ag NPs and 0AgPAN) and the UV-Vis spectra of all fibrous structures. The UV-Vis spectra of all the resulting solutions given in Figure S1(a)-(c). On the UV-Vis spectrum of 0AgPAN/DMF solution (Figure S1(a)), two absorption bands were visible. The first band at the range of 285-250 nm was associated with π- π* electronic excitations of the C=N conjunctions. 48 The second broad band at the wavelength range toward 395-355 nm. This absorption band was assigned with the n- π* transitions of C-O and C-N groups in the polymeric matrix. 76 From the spectrum of the same sample (Figure S1(a)), it can show that weak bands at wavelength of ˜ 247 nm and ˜ 60 nm represented a π- π* transition and n- π* transition of C=O and C=C bonds,48,77 respectively. Whitford et al. 48 reported that an absorption peak in the ultraviolet region of 265–275 nm with varying intensity attributed to the PAN chromophore. The UV-Vis. spectrum of the nano-metalized agent/DMF solution (Figure S1(b)) exhibited a strong/beamy absorption band with a sharp maximum at ˜ 427 nm. This band indicated the phenomenon of surface plasmonic resonance (SRP) of the MA NPs with spherical shape. Also, another narrow absorption band of the small particles of nano-metalized agent appeared in the spectral range of 385-310 nm and in 260 nm. These UV-Vis observations suggested the formation of MA NPs with the large scale and with appearance some of the clot-shaped nanoparticle.29,78 The typical UV-Vis absorption of the (metalized nanofibers/DMF) solution was shown in Figure S1(c). Two distinct absorption peaks were observed at ultraviolet and visible regions, that means the presence of both components in these fibrous samples. Unlike the rest samples, an intense peak appeared around 220 nm and a weak shoulder located around the wavelengths of 320 nm and 260 nm clearly explained the blue-shift of the absorption band of the polymeric component of 1AgPAN sample (Figure S1(c)). While in the last three samples, Figure S1(c) exposed a noticeable blue shift for the polymeric component with various highly intense absorptions around the entire violet region at 260 nm, and disappearance its absorption band in the UV region at 285 nm. Also, the band absorption of the nano-metalized agent was overlapped with the band of the polymer on the band absorption at 260 nm and became narrower. This may be due to the formation of crystal with bigger diameter from the polymeric matrix under the induction by the nano-metalized agent. It is clear that the metalized agent absorbed at the wavelength of 430 nm, so, a negligible shift in their absorption edges was observed, which explains that these particles have preserved the SRP characteristic in the corresponding samples. 29 In other words, the size of the formed particles did not change as a result of the stabilization of the particles by the polymer chains. 29 The reason behind all the changes in the absorption peaks related to the polymeric matrix was due to the complex formation between the functional groups in the polymeric matrix and the metalized agent. 79 It was evident from Figure S1(c) that, as the amount of MA NPs, there was a progressive increasing in the intensity of the SPR band. There were no additional bands around the visible region, indicating the obstruction of the agglomerated shapes of the metallic agent particles by diffusion on the outer surface and in the inner surface of the polymeric matrix, and the absence of silver oxide formation of these particles This, in turn, increased the characteristic absorption band width of the MA NPs. There is a good concordance between the results of the study of the UV-Vis spectra and the X-ray diffraction patterns. These results are in good agreement with the results of many references29,78–81.
FT-IR spectra
The FT-IR spectra of all fibrous structures (0AgPAN, 1AgPAN, 4AgPAN, 7AgPAN, 10AgPAN) were sketched in Figures 3(a) to (c). The IR data of 0AgPAN in the region of 4000-400 cmˉ1 was given as follows: FT-IR spectra of 0AgPAN, 1AgPAN, 4AgPAN, 7AgPAN, 10AgPAN samples (a), the spectral changes in the IR-bands of C=N, C=C in the region of (1900-1300 cmˉ1) (with brown rectangle in the spectrum) (b), and the spectral changes in the IR-bands of C≡N in the region of (2500-2200 cmˉ1) (with yellow rectangle in the spectrum) (c).
The 0AgPAN possessed four sharp IR-bands at 2242 cm−1 of triple-nitrile groups (C≡N), indicating the presence of some unchanged acrylic units, at 1652 and 1593 cmˉ1 of doubled-nitrile groups (C=N) and 1635 cm−1 of (C=C), carboxylic acid groups (C=O) at 536, 1646, 1732 cm−1 and C-O at 1237 cm−1, which attributed to monomeric unites of methyl acrylate, methyl methacrylate and itaconic acid82,83 with a weak or medium intensity), which were in good agreement with other papers.84–86 From the series of IR-bands shown in Figure 3(a) (purple line), the structure of PAN evolves gradually from linear structure to the cyclized architecture. 70 This structural change during two reactions (cyclization and dehydrogenation reactions84–86) was well attributed to decreasing of the intensity peak at 2242 cmˉ1 and appearance of the other discriminatory absorption bands such as (C=N), (C=N) n , C=O and C=C.
The conversion process in the PAN structure aims to activate its surface and increases its stability structure, which has been extensively studied in several references [46.47,70]. We present a reference overview about the most important FT-IR studies of PAN fibers prepared in different conditions to prove the validity of our results and our designed laboratory method. According to the results of Karbownik ‘s paper, 70 the pre-heat treatment of the PAN/DMF solution, before adding Ag NPs, under air at 45°C, caused form a cyclic structure of the polymer thanks to the dehydrogenation reaction but not the cyclization reaction. Study of FT-IR spectrum of PAN nanofibers revealed the presence of C=C and C=N bands. The results of the infrared and ESR study, in a paper presented by Park, 87 proved that as a result of important interactions like intra-molecular interactions and hydrogen bonds within the compound the aromatic structure of polyacrylonitrile was formed via the dehydrogenation pathway. The aforementioned pathway, which took place under aerobic conditions, was a reason for the emergence of oxygen functional groups. Wang 88 confirmed the formation of a hexagonal-ring structure of the same polymeric matrix mentioned above by heating the polymer solution at low temperature under oxidation conditions, which helped to obtain the cyclized structure of PAN by cyclization pathway. Therefore, it is also necessary to indicate the contribution of oxygen or air in the transformation of PAN (from linear structure to hexagonal-ring structure). Furthermore, as mentioned in Friedlander’s explanations, 89 after the cyclization reaction, the polynitron-ring units of PAN was formed as a by-product of oxygen uptake by the naphtyridine-type ring. According to the results of the research by Houtz, Zhang and Park87,90,91 the limited amount of oxygen affected the acceleration of the PAN transformation through the cyclization reaction. When the cyclization reaction was done in a limited amount of oxygen, the methyl acrylate and methyl methacrylate groups play as the active centers to initiate cyclization reaction through a nucleophilic or electrophilic attack of the carbon of the C≡N groups. Whitford has studied the stability and remarkable coloration of undoped PAN nanofibers prepared under oxidizing conditions. 48 Whiteford his coworkers ported that the coloration process of PAN involves cyclization of the adjacent nitrile groups and gives rise to the conjugated, oxygen-absorbing carbon–nitrogen C=N sequences, which form a chromophore structure of polynitron (-C=N (+O)) n comprised of the random conjugated imine and nitrone bonds. Whitford’s research team 48 proved that β-ketonitrile, as an oxygen group, had the greatest role in the occurrence of partial cyclization and the formation of a stable conjugation system of chromophore imines. As verified by our FT-IR analysis of 0AgPAN NFs, in which the electrospinning solution was heated for 24 h at 70°C under aerobic conditions, vibrations of C=C, C=N, C-N with C=O, O-H, CH and C-N in the backbone of the polymer matrix it secures more stability of the related fibers by creating some sensitive interactions such as intramolecular interactions and hydrogen bonds of the polymeric matrix chain such as (OH…OH, CN…HO, etc.). Conjugate system development of chromophore nitrogen groups and the formation of polyimine and polynitron, during the cyclization pathway under oxidative conditions, and the presence of acidic comonomers, which enhance the ionic mechanism of the conversion structure of PAN. It can assist creation C = C, C=N chromophores in hexagonal-ring units, strong intramolecular bonding and strong intramolecular cross-linking. This explains the change in the hue of the polymeric solution during the heating process (see Figure 1, step 2). Our results are consistent with the results of the studies of Karbownik 70 and Zhuo 92 who confirmed that the mentioned transformation process of PAN can be occurred at a relatively low temperature under aerobic conditions. Based on FT-IR spectra of the different metalized fibrous samples, as shown in Figure 3(a)&c, the intensities of the C≡N bands at 2242 cm−1 were significantly decreased, in particularly for 1AgPAN sample. In addition, the lack of the amount of MA NPs in this sample made its spectrum largely similar to that of the 0AgPAN sample. The same IR-band was gradually vanished for the 4AgPAN, 7AgPAN and 10AgPAN samples, as shown in Figure 3(a)&b marked with yellow rectangle. The IR-band intensities of the C≡N groups at the wavenumber of ˜ 2347 cm−1 were also significantly reduced and the peak width at the same region was simultaneously enhanced. From the enlarged spectrum of Figure 3(c) (marked with brown rectangle), it can be found that the IR-bands at 1652, 1593 and 1635 cm−1 were also more intense in the metalized nanofibers samples than those of the 0AgPAN sample. At the same time, both gradual hike in the intensities, overlapping with other bands in the range 1735-1590 cm−1 and no shift of -C=O- bands were observed for these different fibrous samples relative to 0AgPAN sample (as shown in Figure 3(c)). The hydroxyl bands of the metalized fibers appeared as a sharp, strong and wide band at 3100-3700 cm−1, and overlapped with the -NH2 bands at the same range. Any shift of aliphatic groups (δC-Hωc-H, νC-HωCH2 and δC-HωCH2) and other groups (C=N, C=O, (C=N) n , and C≡N) were detected. This could probably be explained many reasons and are in line with the above-mentioned fact concerning the intensity changes of the characteristic IR-bands of the metalized nanofibrous samples. The first reason is related of the interaction types, whether a physical or chemical interaction, between the functional groups. For the prepared MA NPs (Figure S4), there are hydroxyl functional groups at 3300-3440 cm−1 on their surfaces. These hydroxyl functional groups engage in the formation of strong hydrogen bonds with their counterparts in the cyclized structures of the polymeric matrix.19,93 It is also expected that these same bonds can form more firmly with the carboxyl groups, these groups (O-H and C=O) tend to change in their intensity and become wider (particularly for the hydroxyl band at wavenumber 3330 cm−1). Sukarova et al. 94 attributed this to changing the order of those bonds. Interface/interphase interactions occur between the polymeric component and the inorganic component via van-der Waals and strengthen of the hydrogen bonding between adjacent polymer molecules.56,95 Many surveys represented by Li, 96 Tham 8 Cao 95 and Wang, 79 physico-chemical crosslinking affects the prominent change in the intensities and shapes of the IR-bands, and the stability of final fibril structure. In the 0AgPAN sample, the functional groups in the naphtyridine ring,87,91 as the main modified structure of the polymeric matrix after the cyclization reaction, tend to create a suitable environment for the formation of physical cross-links8,96 between the chains of the polymeric matrix, resulting in a rather weak cross-linked fibrous structure of the 0AgPAN sample. Chemical cross-links arose after adding the MA NPs in the presence of hydrazine and sodium hydroxide. According to the views presented by Tham, 8 hydrazine acts as a bridge, as-called cross-linker, 8 which connects all the components in the metalized nanofibers structure through its amine groups (N-H). These amine groups react with the hydroxyl groups available on the surface of the MA NPs with strong hydrogen bonds. The task of sodium hydroxide comes through the hydroxyl ions, which stimulate the functional groups in the mother polymeric matrix to form more powerful chemical cross-links. In addition, the hydrogen bonding between the amine groups of hydrazine can be formed with the induced functional groups of the polymeric matrix (hydroxyl, carboxyl, amine and nitrilo). 8 By comparing all spectra of different fibrous samples with 0AgPAN sample, due to the physicochemical cross-linking and due to the nucleophilic addition of hydrazine to the polymeric matrix in the presence of hydroxyl ions, 8 it is noticeable that the intensity of some bands decreased, the intensity of others increased, and some shapes for those bands widened. The amount of MA NPs controls the strength and type of these cross-links and the interface/interphase interactions between two components. Increasing the amount of metallic agent contributes to reducing the regulation of these interactions or cross-links, which directly affects the IR-bands.8,95 The second reason is the steric hindrance effect and the surface charge of the two components. Indeed, each substituent group in an organic substrate has its own steric position, which appears at its own absorption peak in the infrared spectrum. Citing the opinions of Whitford, 48 Van veen 97 and Jiang , 98 it can be deduced that the intramolecular repulsion and intermolecular attraction mechanisms of the nitrogen groups (C=N, C≡N and N-H) and the oxygen groups (C=O and OH), and even the dipole-dipole interactions are affected by the loan electron pairs on the oxygen and nitrogen atoms. Therefore, these create a steric hindrance that controls the interaction mechanism between them and changes the vibrational frequencies of groups.97,98 The results of these studies48,97,98 can be linked with the results of our study in the following: The MA NPs do not have a steric substitution, but cause relatively large intermolecular distances during metallization process, and they have a crystal structure containing on its surface hydroxyl substituent groups with a specific steric position. The steric hindrance effect in the metalized nanofibers samples appears through the coordination between nitrogen and silver. Because of the different nitrogen group formulas and their respective steric positioning, therefore, each group in the hexagonal ring structure of the polymeric matrix hinders the donation of the electron pair from the nitrogen atom to the vacant orbitals of the metalized agent in a different form than the other group. 98 With the increase in the particle size and equivalence with the increase in the amount of metalized agent, the surface adsorption capacity agent, of the smaller particles in the samples are more organized and stronger than the larger particles. This is consistent with that the intensity of the wavenumber shifts was noticeable and more detectable in the oxygen groups than in the nitrile groups, forming hard hydrogenous bonds between the oxygen and nitrogen substitutions in the polymer and the hydroxyl groups of the metalized agent, which have signalized by Matsumura et al. 99 It is also known that the electronegativity of the nitrogen atom is less more than that of the oxygen atom and based on the effect of the steric obstruction, the chemical bonding and coordination between the oxygen groups and the MA NPs are easier and more robust than it is with the nitrogen groups. As it was seen that the absorption band of oxygen groups merged with that of other groups.98,100 While a decrease in the intensity of the absorption bands of nitrile groups was found, due to the different bond order of those groups and their new spatial positioning after interaction with the metalized agent. 98 To clarify the influence of the surface charge of two components, attraction electrostatic forces arise between the particles of the metalized agent, having a positive charge, and the polarized functional groups (carboxylic, nitrile, nitrilo and hydroxyl), having a negative charge, which promote the strength of the bonds and a change in the intensity of their absorption band in the infrared spectra. 70 Various outstanding styles of interactions take place when MA NPs interact with polymeric matrix molecules. These interactions encourage intermolecular crosslinking of the polymer chains and provide greater stability to sustain high mechanical and wettability behaviors.
These detectable observations in the frequencies of (1AgPAN, 4AgPAN, 7AgPAN, 10AgPAN) samples might be due to interactions between Ag NPs and functional groups of the polymeric matrix.70,101 Based on the results obtained from FT-IR spectra and using the equations (3)–(5), the cyclization degrees (C.D.) and the dehydrogenation degree (D.D.)
70
and the percent of the relative cyclization reaction (RCR)50,102 were calculated. The possibility of the reaction occurring in the cyclization reaction is measured through the cyclization index, or the so-called C.D, and the other reaction is measured through the hydrogenation reaction index, the so-called D.D.. The discrepancy between the values of two indicators indicates the mechanism of transformation of PAN according to one of the two mentioned paths, as indicated by Karbownik.
70
Where: A1593 is a maximum absorbance of C=N bond at T%=1593 (a.u.), A2244 is a maximum absorbance of C≡N bond at T%=2244 (a.u.) and A1635 is a maximum absorbance of C=C at T%= 1635 (a.u.). For performing these calculations, firstly the absorbance was determined from the related transmittance of each bond. The absorbance was determined by equation (6).
By interpretation the data of cyclization (C.D.) and dehydrogenation (D.D.) degrees70,103 (Table S3), it can be pointed out that not only the MA NPs increased the rate of cyclization reaction of polymer chains but also blocked the dehydrogenation reaction (increasing the C.D. values and decreasing the D.D. values). Ali 29 and Wang 103 reported that how these types of particles can contribute to the cycling reaction. During the heating of the polymer solution for a period of 24h at 70°C under aerobic conditions, a primary transformation of the polymer matrix occurred that leads to produce many active centers of functional groups. In Wang’s study, the nitrogen groups donate their electron pairs from their occupied 2p-orbitals to empty s orbitals in Ag NPs, as resulting form Π-bonds.88,101 In this present study, it can be confirmed that during metallization process of the polymeric matrix were cyclized to a more extent than the un-metalized polymeric matrix. In contrast, another reaction requires harsher conditions (high temperatures and oxygen availability) to occur.70,86 On the other hand, the presence of MA NPs leads to increase a stable cyclized formula of polymeric matrix, because RCR (%) values of metalized fibrous samples were higher than the RCR (%) values of 0AgPAN sample. The FT-IR results support the findings from UV-Vis. and XRD, where the absorption and position diffraction peak showed that the ordering of crystallization takes place and hexagonal-ring crystal grows as observed from d values.
The morphological and surface roughness study of nano-fibrous structure
The FESEM technique was used in order to monitor the morphology of the Ag NPs powder and the shape of the loaded Ag NPs layers on the surfaces of all the metalized fibrous samples, as shown in Figures 4(a) to (f). More FESEM images were observed in the material support file (Figure S2(a)-l). On the other hand, the sizes of Ag NPs and the average diameter of the nanofibers were estimated by Image J software and the change of average diameters with changing the content of the metallic agent nanoparticles loaded on the surface of the polymeric matrix was studied. Figure 4(a) and Figure S2(a)-(c) declared that the shape of the as-received Ag NPs powder is mainly spheroidal particles stacked to each other with an average diameter of 20 nm. Because of the massive surface attraction forces of the spheroidal particles, these particles converge with each other to form coagulated large spherical particles of MA NPs with generation of deep voids between these coagulated particles. It was observed that the surface morphology of the fibers containing no Ag NPs were prepared with cylindrical shape and without any bead (Figure S2(d)-(f)). The same sample also displayed some longitudinal and narrow splits in different regions of its superficial crust (Figure 4(b)). By reviewing the Lis’s study ,
104
the mechanism of vapor-induced phase separation can be essential clue to explain this kind of morphology in the fibrous surfaces. All the FESEM images (Figures 4(c) to (f) and Figure S1(g)-r) related to the shape of the surface of the polymeric fiber covered with a layer of nano-metalized agent showed that there were obvious differences in the surfaces of the mineral fibers relative to the surface of the 0AgPAN. As shown in Figure 4(c)&d and Figure S2(g)-l, the micrographs of the 1AgPAN and 4AgPAN samples disclosed that the small spheres of Ag NPs were uniformly distributed as thin layer over the entire surface of the polymeric matrix. The same images of the 4AgPAN sample, taken on the other area for this fiber and at high magnification of 200 nm, revealed that the thin layer of nano-metalized agent at 4wt% was a layer of little roughness with superficial folds and zigzags. Whereas, as depicted in Figure 4(e)&f, a non-homogeneity layer of irregular large/small spherical-shaped of Ag NPs with furrowed edges was more identifiable in the 7AgPAN and 10AgPAN samples, compared with the other fibrin structures. By careful examination of the fibrous network in the metalized fibers (Figure S2(g),(j), m, p), it was found that their fibers were long, well-orientated and similar to the branches of bushes intertwined with each other. Notably, the average diameter of the 0AgPAN sample was 208.2 nm and it was altered to 214.72, 228.65, 266.14, 274.68 nm for 1AgPAN, 4AgPAN, 7AgPAN and 10AgPAN samples, respectively, as the level of MA NPs was increased. Despite of the agglomeration phenomenon in high amount of the MA NPs, it can be seen that the fiber diameter of the metalized fibrous samples (1AgPAN, 4AgPAN, 7AgPAN and 10AgPAN) was higher than that of 0AgPAN sample, because of difference in the viscosity of the final solution spinning (Ag/PAN/DMF).105,106 The second clue for the increase in the fiber diameter of the metalized nanofibers samples is the increase in the thickness layer of the MA NPs.
19
Various high magnification FESEM images of Ag NPs (a), the external surface of 0AgPAN (b), and the external surface of metalized fibrous samples with different concentrations of Ag NPs, 1AgPAN (c), 4AgPAN (d), 7AgPAN (e) and 10AgPAN (f).
To acquire information about the change in the internal/outer structure (thickness and morphology) of the fibril samples and the size/morphology of the nano-nano-metalized agent, we executed transmission electron microscopic (TEM) examination, as described in Figure S3(a)-l. The TEM images of the nano-metalized agent (Figure S3(a)&b) illustrated that the diameter of its nanoparticles varies in ranging (20–35) nm with almost spherical in shape, well compacted with voids between these particles, and nearly monodispersed in nature. The FESEM and TEM images of these particles indicated that the distribution size of the nano-metalized agent was narrow and was in accord with Gaussian distribution. Figure S3(c)&d showed the TEM images of 0AgPAN sample. It can be distinctly seen from that images that both external surface and outer surface of this sample were bare from any other constitutes. This surface has other morphological features, including: absence of any beady structure, small cracks across the outer surface “marked with yellow circles”, low thickness. The TEM graphs in FigureS3(e)-l displayed the morphological virtues of the metalized fibril structures coated with various content of the nano-metalized agent. When the content of nano-metalized agent= 1 wt%, either the spread of a small number of large-sized particles on the outer surface or the filling of the inner surface of this metalized fibrous structure with a greater number of particles, suggesting an increase in the fiber thickness of this sample (Figure S3(e)&f). When the nano-metalized agent content was increased from 1wt% to 4 wt%, the surficial thickness of the 4AgPAN sample remarkably increased (Figure S3(g)&f) and the number of MA NPs dispersed on the external and outer surfaces also increased (Figure S3(f)). When the amount of nano-metalized agent= 7 w% (Figure S3(i)&j), Larger spherical-liked particles were abundantly seen in this sample. The outer surface of 7AgPAN sample was densely packed with large particles that were lined up together with smaller particles. Among the important observations of this sample is that the inner section of the surface of this metalized nanofibrous sample was more occupied with small particles than large particles (Figure S3(j)). Meanwhile, a greater increased in the fiber thickness was noted compared to the previous two samples. Whilst, when we further enhanced the amount of nano-metalized agent to 10 wt%, it was detected that not only the outer layer of the 10AgPAN sample was completely covered with nano-metallic agent particles, but also the inner layer was filled with small size particles alone, as shown in Figure S3(k)&l. This indicated the deletion of the smooth surface of the basic fibrous polymeric structures. The fiber of 10AgPAN sample became the thickest relative to the rest of the samples. Kara 19 and Ali 29 pointed out in their research the possibility of actual interconnections between carbon-nitrogen groups and small particles of metalized agent or the tightening of hydrogen bonds, due to the presence of high reactive areas in the places where the functional groups spread on the surface of the polymeric matrix. Such observations are not observed in large particles. According to the above-mentioned, the difference between the 0AgPAN sample and the rest of the samples is that: (1) The 0AgPAN sample did not possessed a cover or a thick shell to form a core, (2) The particles resided on the outside surface of the polymeric basis contributed to construct the shells for this structure. So, we believed that all metallic nanofibers completely coated with metalized agent (whatever the amount of metalized agent) undoubtedly led to the formation of a coexistence structure. On the basis of abundant literature sources on this topic,10,107,108 the combination of one structure within another structure constitutes the coexistence structures of great importance in the environmental applications of materials compounds, biological applications of fibers, chemical sensitivities and others.
AFM observations in Figure 5(a) revealed that the 0AgPAN surface was slightly unsmooth with Ra value of 12.21 nm, probably due to having many elongated incisions. The roughness values (Ra= 19.78 nm “1AgPAN”, Ra= 25.35 nm “4AgPAN”, Ra= 42.65 nm” 4AgPAN” and Ra= 65.12 nm “10AgPAN”, see Table 1) of the different metalized fibrous samples had an upward slope that may be due to the various dense layers of MA NPs, which the thickness of the metalized agent layer depending on their levels, as shown in Figures 5(b) to (e). 2D-AFM images and 3D-AFM images of 0AgPAN (a), 1AgPAN (b), 4AgPAN (c), 7AgPAN (d) and 10AgPAN (e).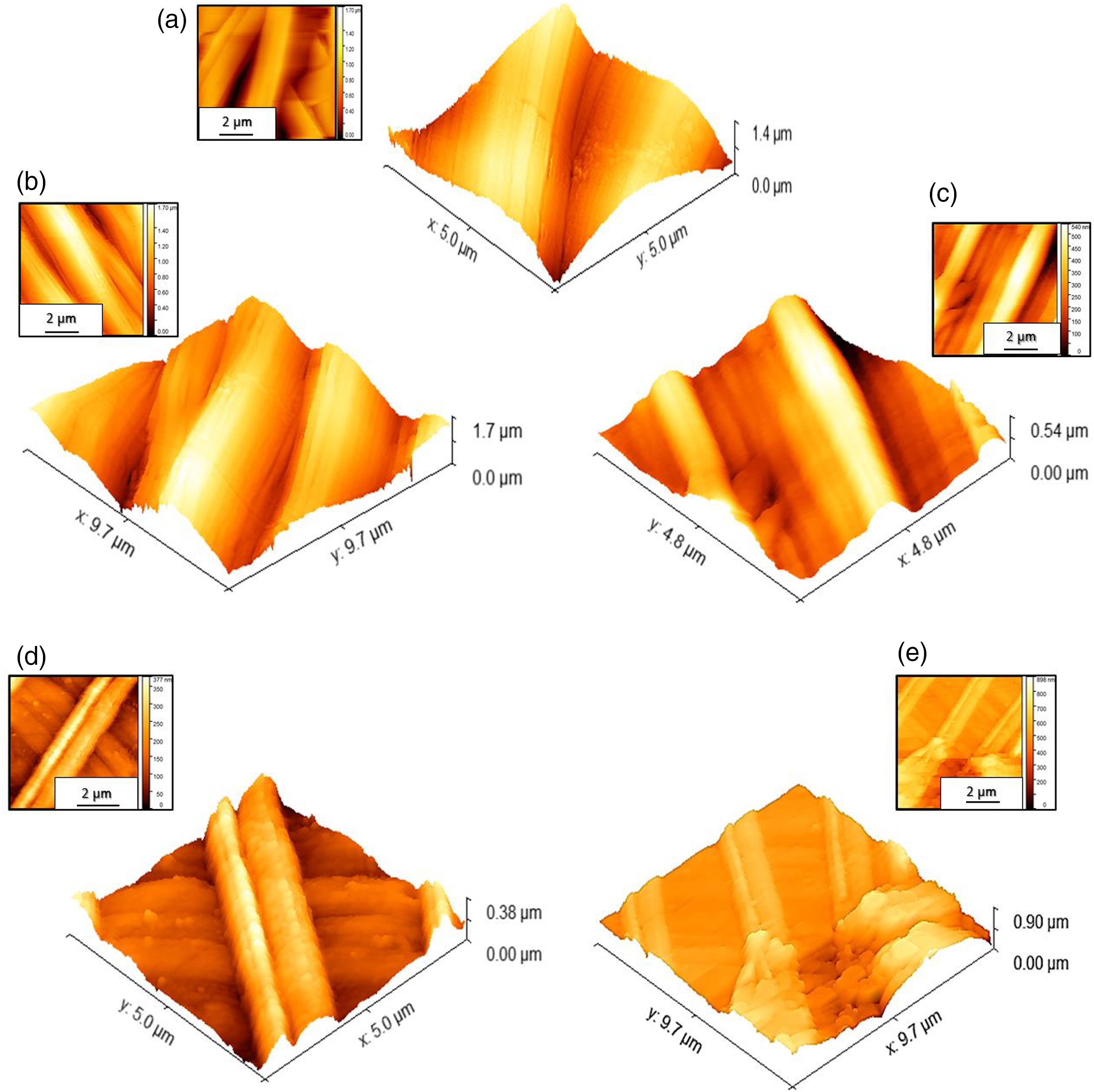
Study of tensile and hydrophobicity/hydrophilicity properties
A brief of theoretical calculations of the metalized PAN NFs.


Curves of changes of σ values (a), ε values (b) and Υ values (c) for all prepared fibers (**** p values < 0.0001).

Comparison of CA values for H2O (a) and EG (b) droplets at various retention times (10 and 20 s), showing a significant difference in CAs between the 0AgPAN sample and other metalized fibers (* p values < 0.05, ** p values < 0.01, *** p values < 0.001, and **** p values < 0.0001).
Discussion
In the discussion section, synergistic relation is introduced by two formulas: in the first formula, a study of the (Z/P.S. vs. fiber diameter/RCR) and (fiber diameter/Z vs. Ra) correlations are adopted (Figure 8(a)). The correlation study (Z vs RCR/CD/DD) (Figure 8(b)) is the core of the second formula to answer an important question, which is how MA NPs affect the stability of the chemical composition of the polymeric matrix. Later, the effect of the structural synergistic correlation on the wettability/mechanical properties of metalized nanofibers was concluded. Detailed discussion of the synergistic relations was arranged in the supporting materials. As reported in Table 1, the Z% value of 1AgPAN, 4AgPAN, 7AgPAN, 10AgPAN samples were smaller than 0AgPAN sample, which is decreased by approximately 16.48% “for 1AgPAN sample”, 22.21% for “4AgPAN sample”, 48.23% “for 7AgPAN sample” and 55.59% “for 10AgPAN sample”. This reducing in the Z% values can be interpreted by two parameters. The first reason is the effective interactions between the functional groups of the polymer and the MA NPs and the formation of strong bonds. The strength of these bonds increases with the decrease in the amount of the MA NPs. In the MA NPs amounts of 7 wt% and 10 wt%, larger particles caused disturbances in the crystalline structure of the polymer and led to a reduction in the Z. The second cue is the small nano-size and good dispersion of the MA NPs on the fiber surface induced the orientation the polymer molecular chains around the surface of MA NPs. This output closely belonged to expansion the Z% values of the polymeric matrix .19,21 The presence of this MA NPs offered nucleation centers for the best crystallization of the polymeric matrix crystals and enhance of enthalpy reaction, also facilitated the cyclization reaction by absorbing the heat generated during the heating of polymeric solution, augmenting RCR values of 1AgPAN, 4AgPAN, 7AgPAN, 10AgPAN samples relative to 0AgPAN sample (Figure 8(a)). According to results arranged in Table .1 and Figure 8(a), the high values of both Z and RCR in the small nano-sized MA NPs of 1AgPAN and 4AgPAN samples, compared to large size of those for 7AgPAN and 10AgPAN samples, explained to form stabilized composition of (Ag-PAN) with strong intermolecular bonds.
70
By varying the loading amount of MA NPs (from low to high content) and their sizes, the metalized fiber diameter was gradually increased along with reducing the Z and RCR values, suggesting a modification in the orientation of the polymer chains. An interesting observation is that the Ra values increased with the increase in the P.S. values of MA NPs, as shown in Figure 8(a). Phan et al. explained that the silver nanoparticles scales was proportional to excess of their migration rate to the outward surface of the fibers and the rate of DMF evaporation. The results of tensile test showed that the σ values have downward tendency of 1AgPAN and 4AgPAN. Consistent with the previous explanations, the large-sized of MA NPs, particularly in 7AgPAN and 10AgPAN sample, was responsible for fabrication of a fibrous structure with lower interfacial merits than the rest of the other metalized fibers (comparison the values of Z, RCR, mean fiber diameter (Figure 8(a))). The increase in the values of RCR and C.D. and the decrease in the values of D.D. (Figure 8(b)) indicated that the cyclization reaction occurred and the cross-links between the chains of PAN macromolecules were formed. It is expected that the dissipation of mechanical forces in such cases is inhomogeneous and insufficient over the entire cross-sectional area of the fibril surface and mechanical interlocking is weak, therefore, alterations in the tensile values was observed (Figure 8(c)). From Figure 7(d)&e and Table 1, it can be confirmed that P.S, average diameter and Ra were as the convincing structural factors to analyze the variations of CA values. As a result, the CA values of metalized fibers fortified proportionally to the fortified particle size and Ra. The coarseness surfaces of 10AgPAN, having silver granules, trapped many of the air pockets, which acted as hinders for enhancing the hydrophilicity properties. Additionally, the small surface area in the large diameter of fibers can be helped to decline the (liquid-fiber surface) interaction.31,118 The results support the existence of synergy between all structural properties and the great influential on the mechanical, hydrophilic/hydrophobic properties. Correlation between Z variations and other structural measurements (RCR, P.S, fiber diameter and Ra) of the fabricated fibrous structures (a), stability of polymeric matrix by studying the effect of Z values variation on the RCR, C.D. and D.D. values (b), the correlations between structural parameters for all fibers and engineering features (c), hydrophilicity performance at 10 and 20 s (b) hydrophobicity performance at 10 and 20 s.
Conclusions and Challenges
Briefly, we presented a theoretical/experimental investigation of in-situ surface metallization of 0AgPAN fibers with four amounts of MA NPs (1 wt%, 4 wt%, 7 wt% and 10 wt%), and tested their effect on their mechanical properties and hydrophilic/hydrophobic properties using two drops of H2O and EG. On the basis of the above results and discussions, a summary of the essential conclusions can be gained as below: (1) New series of the metalized nanofibers (1AgPAN, 4AgPAN, 7AgPAN and 10AgPAN) were successfully prepared by combining two eco-friendly and efficient ways (ultrasound/electrospinning) simultaneously as a one-pot protocol. At the beginning, spherical formula as well as single crystal of Ag NPs with (1 1 1) facet as dominant planes were synthesized by safe reduction reaction using (NaOH/N2H4.H2O) and applying ultrasound irradiation for 30 min. In the second step, cyclized structure, as a result of cyclization reaction, of polymeric matrix with activated functional groups of nitrogenous and oxygenic characters was formed by heating polymeric solution (PAN/DMF) at 70°C under aerobic conditions to be ready to react with the MA NPs. By FESEM and TEM images, it can be speculated that the surface of the 0AgPAN sample was clear without any beady defect and the muted MA NPs particles on the surface of the fiber were spherical in shape. Also, the number of these spheres and their size increased with increasing of the amount of the loaded MA NPs. The small MA NPs were embedded onto the 0AgPAN surface, whereas the larger ones were muted on its external surface. Regarding to fiber diameters, the fiber diameter increased with the increasing thickness of the MA layer. The obtained AFM data (3-D images, and Ra values) of the metalized nanofibers affirmed that the 10AgPAN surface was more coarseness than others. (2) Loading of MA NPs, which are as the primary nucleation centers, is a direct effect in changing the crystalline properties of the polymeric matrix. With the addition of such agents, all crystalline features of the polymeric matrix were dramatically changed, depending on shapes, and scales of MA NPs. On one hand, the improve of Z values, in turn, affected on the fiber diameters by changing the molecular orientation and on the particle distribution by forming potent H-bonds with functional groups of cyclized structure of polymeric matrix. FTIR analysis exposed that the MA NPs with small size and polymer chain were physically interconnected, which led to a good distribution of MA NPs on electrospun fibers. The strength of this interconnection is governed by many factors such as charge density of the functional groups and their capacity to create a hydrogen bridge with the small size of the MA NPs. In addition to that, after metallization, the shift in the wavenumbers is also attributed to the size and shape of the MA NPs. Another positive contribution of the MA is the enhancement of the stability of the cyclized structure of PAN matric, confirmed by promotion of RCR. (3) Discrepancy between the tensile strength of different nanofibers was tested using a tensile test machine. The nonlinear elastic performance and poor mechanical properties of the 0AgPAN was altered by its metallization surface. In the outcomes of this test, the metalized fibers possessed higher mechanical modulus compared with electrospun 0AgPAN sample. The blending of a soft phase, represented by the polymeric matrix, with a hard phase, represented by the MA NPs, leads to an increase in the rigid interconnected of the polymeric network; and the role of the mechanical percolation in the couple of “macromolecules of the polymeric matrix/the MA NPs spheres”, indicating a greater resistance of the fibrous sample in relative to mechanical tensile forces. In addition to what was noted, the ε, Υ, σ values were remarkably influenced by the synergism correlation of (fiber diameter/P.S. vs. Z). As noted of all samples, it was found that the mechanical behaviors of both fiber’s small diameter with either external edges or internal surfaces supported by small spherical particles (1AgPAN and 4AgPAN) are better than those of larger diameters and loaded with agglomerated particles (7AgPAN and 10AgPAN). This observation may be due to elevation of the Z% values and molecular orientation with depreciation of MA NPs size. (4) Hydrophilicity/hydrophobicity assessments revealed the CA value of H2O droplet for the 0AgPAN sample (CA= 36.82°) at 20 s was lower than that of CA value of EG droplet (CA= 51.04°), confirming the hydrophilic surface. The H2O CA of the metalized fibers increased with increasing the MA NPs amounts and retaining time of the H2O droplet on each surface of them, indicating poor hydrophilic. But, the EG CA for the same sample decreased, suggesting hydrophobic surface. (5) In all measurements, significantly increasing amounts of MA NPs were not conducive to improving the properties of the polymeric matrix, as they cause structural disturbances. (6) Interestingly, there is a close synergistic correlation between the whole structural features, making improvement of mechanical/hydrophilicity, hydrophobicity properties of the as-prepared metalized fibers. (7) In the near future, this kind of research can be expressed as a new platform to provide qualitative research in the field of surface mining of different polymers using other metal materials. Also, this research will benefit researchers to identify applications of these metalized polymeric surfaces in various fields such as tissue engineering, mechanical engineering, surface engineering and textile engineering, medicine and chemistry.
Supplemental Material
Supplemental Material - A comparative investigation of the synergistic correlation (mechanical-hydrophobicity/hydrophilicity vs. structural behaviors) of a series of surface-metalized polyacrylonitrile fibers by silver nanoparticles (AgPAN): An in-situ surface metallization protocol
Supplemental Material for A comparative investigation of the synergistic correlation (mechanical-hydrophobicity/hydrophilicity vs. structural behaviors) of a series of surface-metalized polyacrylonitrile fibers by silver nanoparticles (AgPAN): An in-situ surface metallization protocol by Gheffar Kher al-deen Kara, Elaheh Kher al-deen esmaeili, Mousa Kehtari, Rayhaneh Kher al-deen Ghafourian, Azadeh Tadjarodi in Journal of Industrial Textiles
Footnotes
Acknowledgements
We gratefully acknowledge Iran University of Science and Technology, and Stem Cell Technology Research Center for providing materials and facilities.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
The raw/processed data required to reproduce these findings cannot be shared at this time due to technical or time limitations.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.