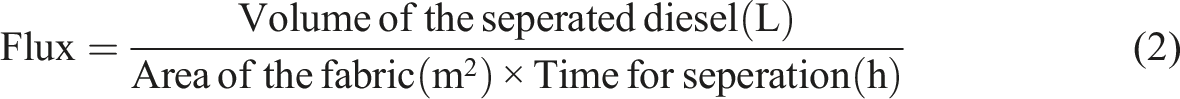Abstract
A simple, durable, superior UV-shielded, self-cleanable, hydrophobic/superoleophile cotton fabric was developed using SBA-15 mesoporous silica and epoxy coating (SE). The cotton fabric surfaces after coating with SE showed the highest water contact angle value of 146° and lowest surface free energy of 13.9 mN/m. As a filtration membrane, the developed SE/cotton delivered 95.8% oil–water separation efficiency with a notable flux value of 6800 L/m2h against diesel–water mixture. The presence of an epoxy and silica hybrid layer on the surfaces of cotton fabric induced self-cleaning behavior and notable durability against UV light (UPF-29), pH resistance, washing fastness, and mechanical abrasion. Hence, the present coating developed using mesoporous silica and epoxy can be applied to different types of textile substrates to construct durable low-cost membranes.
Introduction
The natural properties of lotus leaves have sparked the interest among academics and industrial researchers to develop superhydrophobic surfaces.1–4 A superhydrophobic surface can be created either by reducing surface free energy (SFE) or by introducing roughness.5–8 Superhydrophobic materials have proven to be useful in applications such as anti-icing, 9 self-cleaning, 10 antifouling, 11 anticorrosive, and oil–water separation.12–14 There is a high demand for superhydrophobic membranes to address the utmost challenges of oil spills in all parts of the world.15,16 For this purpose, superhydrophobic membranes made of metal, polymers, and textiles have been developed.14,17–19 A number of woven and nonwoven textile fabrics have recently been explored as suitable substrates for separating oil-water mixtures.20,21 Cotton is a promising woven fabric that offers a variety of benefits such as biodegradability, flexibility, and superior absorbency. As a result of the presence of polar hydroxyl groups, oil and water or both absorbed by this cotton. 22 Researchers’ advancements in altering the surface properties of cotton fabric through coatings have made it possible to selectively absorb oil and water and recognize it as a potential oil–water separator.23,24 Nevertheless, the prolonged usage could also cause pollutants adsorption and subsequent exposure to UV degrades both the adsorbed pollutants and cotton membranes.18,25 In addition, oils and water at different pH may also cause corrosion to the coatings along with the mechanical deterioration. Hence, durable coatings are still in high demand to overcome cotton membrane’s limitations.
Several thermoset polymers have recently received attention for their use in developing industrial textiles, including polyester, 26 polyurethane, 27 polybenzoxazine,28,29 and epoxy resins.31–33 The epoxy resin is one of the most widely used engineering polymers because of its versatile chemical reactivity and processing properties. 34 It is used for coating, composites, sealants, and adhesives in addition to the conventional applications like electrical insulators.35,36 The molecular network of the epoxy can be altered depending on the applications due to the reactive nature of the oxirane ring with selective cross linking agents and modifiers.37,38 Owing to the presence of very high surface functionalities such as peripheral hydroxyl (−OH) groups and ether (−O−) bonds, epoxy has strong adhesion to various substrates. 39 It has been evidenced from the report of Laban and Mahdi, 40 due to strong adhesion of epoxy with cotton, a square composite tube prepared was found to have enhanced load-carrying capacity and energy-absorption capabilities. Dan et al. 41 found that the durability of antibacterial coating was enhanced by forming a strong chemical bond between thiol-functionalized cotton and epoxy-functionalized quaternary ammonium salt. A recent report by Kamble et al. demonstrates that the composite developed using epoxy, cotton waste fibers, and graphite can be used to develop furniture materials and automotive components. The presence of cotton and graphite offered enhanced properties in terms of high mechanical strength, thermal stability, and low water absorption properties. 42 Similarly, noise-reducing and cost-effective composite panels were made using epoxy and cotton. The strong interfacial interaction between fibers of cotton and the molecular structure of epoxy provided enhanced mechanical strength. 43 In addition, because of a wide scope for the epoxy/cotton material, recently a modal study has been conducted by Lu et al. 44 to develop a real-time 3D needled composite. However, no significant studies have been done about the surface behavior of epoxy-coated cotton and about its application in oil–water separation.
Porous material with large surface area is used in a number of different applications including adsorption, chromatography, catalysis, gas storage, drug delivery, and sensors.45,46 Materials that possess pores with diameters between 2 and 50 nm are known as mesoporous materials. 47 Since the discovery of ordered mesoporous silica in 1992, 48 significant progress has been made in understanding the morphology, pore size, composition, and applications of these materials.47,49 Various mesoporous structures have been synthesized during the last two decades and can be roughly classified into three categories based on the pore types: almost spherical cage, cylindrical channel, and bi-continuous channel. 50 Among different ordered mesoporous silicates, SBA-type silicates (SBA = Santa Barbara Amorphous) have more advantages particularly in terms of textural properties. The main textural components of SBA-15 are high specific surface areas (more than 1000 m2/g), uniform pores (with a size range of 4 to 30 nm), a strong silicate framework, and crystallite walls.51,52 Various studies have found that composites of silica with different polymers can improve functional behavior, particularly low SFE, water repellency, and UV-shielding tendency.53,54 As a result, the combination of mesoporous silicates and epoxy is expected to give more water repellency and UV protection.
As previously mentioned, epoxy materials have been used as a coating materials for textile applications.35,36 In contrast, no significant emphasis has been placed on studying UV-resistant nature, surface behavior, or oil–water separation efficiency. In a similar context, the development of mesoporous materials for oil–water separation has not been explored. Inspired by the properties of both epoxy and SBA-15, an attempt has been made to explore the surface properties and UV-blocking nature of SBA-15/epoxy (SE)–coated cotton along with its oil–water separation efficiency.
Experimental
Materials
Grey plain woven cotton fabric with warp and weft count of 50S Ne × 36S Ne having areal density of 120 GSM, with thickness 0.35 mm, Ends per inch (EPI), and Picks per inch (PPI) 80/68 was purchased from Jeyanthi Textile Products Private Limited, Coimbatore, Tamil Nadu, India. Araldite epoxy (LY 556; DGEBA) and diaminodiphenylmethane (DDM) were purchased from Javanthee Enterprises, Chennai, Tamil Nadu, India. Mesoporous silica (SBA-15) was purchased from Turiyam Research Laboratories (TRL), Pondicherry, India. NaOH and acetone were purchased from Sisco Research Laboratories (SRL), Chennai, Tamil Nadu, India.
Methods
Coating of cotton fabric using epoxy (E) and mesoporous silica (S)
The cotton fabric was desized with 5% NaOH solution and subsequently washed with distilled water till the pH became neutral and dried at 80°C. The pretreated cotton fabrics of dimension of 10 × 12 inch were treated with epoxy, mesoporous silica, and synergism of both. In this content, 10 wt% epoxy and 1 wt% mesoporous silica with respect to the weight of the fabrics were chosen as coating agents. An appropriate amount of DDM as hardener, equivalent to that of epoxy was added to catalyze the curing and silane coupling agent of three equivalents to that of SBA-15 was added to enhance the coupling between epoxy and silica network. The resulted mixture was further allowed to dissolve and disperse in acetone for 1 h. After soaking for 30 min, cotton fabrics were subsequently allowed for padding at 2 kg/cm2 pressure. The process was repeated till the coating attained homogeneity. Afterwards, the fabric was dried at 80°C for 3 h and subjected at 180°C for 5 min only in order to cure the epoxy completely. The formation of SE/cotton is illustrated in Figure 1. Coating of resulted mixture with epoxy more than 10 wt % cotton fabrics lead to the formation of a rigid network, so cotton fabric loses its flexibility. On the other hand, only 1 wt % SBA-15 was chosen because of its low denser nature. In addition to the coating of synergistic mixture containing epoxy and SBA-15 over cotton (SE/cotton fabric), cotton fabrics were separately coated with 10 wt% epoxy (E/cotton) and 1 wt% SBA-15 over cotton fabric (S/cotton) for comparison. Schematic representation of formation of ES coating over cotton.
Material characterization
The Fourier Transform Infra-Red (FTIR) analysis of uncoated and coated cotton fabrics were carried out in a Thermoscientific Nocolet iS5 spectrometer via attenuated transmittance resonance technique.The SBA-15 procured was fully characterized for its surface area, porosity, and morphology using a N2 adsorption–desorption analyzer from Autosorb iQstation-1(Quantachrome Instruments, United States).The morphology and elemental analysis of the SBA-15, E/cotton, S/cotton, and SE/cotton fabrics were conducted using the Zeiss Sigma field emission scanning electron microscope (FESEM) attached with a Bruker XFlash EDS detector. Microstructure of SBA-15 was recored using a JEOL JEM-2100 high-resolution transmission electron microscope with an Oxford energy-dispersive X-ray spectrometer. The crystalline state of the uncoated cotton, E/cotton, S/cotton, and SE/cotton fabrics were determined using an X‐Ray Diffraction (XRD) analysis using Malvern Panalytical X’Pert3Powder. The surface roughness of the uncoated and coated fabrics was measured using an Atomic Force Microscope (AFM) from NTEGRA spectra II (NT-MDT Spectrum Instruments). The areal density of the fabrics was measured using a GSM cutter, Mag-Circucut XT, as per ASTM Standard D3776-20. Further, the thickness was determined in accodrance with ASTM Standard D1777-96(2019), withadigitial thickness guage(Wira; UK). The tensile properties of the fabrics were measured using a universal testing machine (Tensile Tester Z10; Zwick/Roell, Germany), with a constant rate of extension as per ASTM Standard D5034-21.To measure the stiffness, the bending length of fabric was measured using a Wira bending length tester as per ASTM Standard D1388-96.In accordance to the ASTM Standard D737-18, the air permeability of the uncoated cotton and the treated fabrics was tested using an air permeability tester (Textest, FX3300-IV; Switzerland).ACapilary Flow Porometer (CFP 1500 AEX; Porous Materials Inc) was used to verify the changes in the textural properties of fabrics after coating. Bursting strength was determined using a Mag-Digi Burst as per ASTM Standard D3786-18. The whitness index was measured using Carry 60 UV-visible spectrocopy (Agilent). A UV2000 F UPF tester (Labsphere) was used to analyze the UV-shieldingability of cotton fabrics as per AATCC TM183-20 in the ultraviolet wavelength region from 250 to 450 nm. Washing fastness of the coated fabric was evaluated using a MAG Laundrometer as per AATCC:16-2A.19,55 To predict the durability after mechanical abrasion (Figure 2), the fabric was tested using 200g load over the coated fabric and kept in contact with sandpaper (1000 mesh), and tested with a moving speed of 4 cms−1.
30
Schematic representation mechanical abrasion test.
Oil–water separation
The oil–water separation experiment was performed without the aid of pressure using cotton fabrics (diameter = 0.04 m, area = 0.00125 m2) and 50% (v/v) oil–water mixtures.
56
Different oil–water mixtures were prepared using diesel and water to study the seperation efficiency. The separation efficiency (%) was calculated using equation (1). About 10 times, the fabric was cycled to separate diesel–water mixture to verify its separation efficiency. After each cycle, the fabric was washed with ethanol and dried at 45°C for 1°h, before subjecting to subsequent oil–water flux experiments. Equation (2) was used to calculate the flux value in (liter/area in m2 × time in h).
Results and discussion
Characterization of SBA-15
In the present work, SBA-15 was initially characterized using XRD and FESEM. The Braggs diffraction pattern of resulted SBA-15 observed from wide angle XRD is presented in Figure 3(a). Accordingly, the mesoporous SBA-15 shows only a single broad reflection at 2θ = 23°, which is attributed to the amorphous nature of silica. The microstructures of the SBA-15 obtained from field emission SEM analysis at different magnification are illustrated in the Figure. 3(b)-(c). Figure 3(b) and (c) shows the existence of micrometer sized one-dimensional channels of ordered SBA-15 particles with regular rod shapes.
38
These results suggest that the silica network is channelized with pores.
32
Further, the HRTEM analysis of the SBA-15 was recorded and presented in inset of Figure 3(c), which shows the existence of porous network. In addition, elemental mapping of SBA-15 was performed and presented in the Figure. 3(d)-(f). Initially, the general scanning result is presented in Figure 3(d), with yellow colour, which shows the presence of Si and O. However, the individual elemental scanning of Si and O elements clearly demonstrates their homogenous distribution as presented in Figure 3(e) and (f), red and green respectively. (a) XRD, (b-c) field emission scanning electron microscope, Inset of c shows the HR-TEM (d-f) elemental mapping images of SBA-15.
In addition, the low angle XRD preformed for the SBA-15 (Figure 4(a)) shows diffractions signals at 2θ = 0.98° and 1.63°, which confirms the existence of ordered two-dimensional hexagonal structure. Further, the BET surface area of 527.8 m2/g and pore volume of 0.907 cm³/g (Figure 4(b)), was also noticed for the SBA-15 used in the present work. (a) Low angle XRD, (b) N2-adsorption/desorption isotherm of SBA-15.
Basic properties of uncoated and coated fabrics
Basic properties of uncoated and coated fabrics.

In addition to the basic properties of the fabric, the mechanical properties such as bursting, tensile strength, bending, and flexural rigidity were also examined. It is known that the bursting strength refers to the multi-dimensional force required to break the fabric, which is a crucial parameter needs to be verified for any filtering medium. The bursting strength values shown in Table 1 suggest that the coated fabrics showed a moderate increase in the strength than the uncoated fabrics. The increase in strength could be attributed to the strong adhesion of epoxy polymer with the cotton surfaces. Thus, the coated fabrics can even use as filtering medium under pressure in addition to gravity separation process. The tensile strength of the coated fabrics showed a significant load bearing capacity, which can be evident from their respective tensile strength presented in Table 1. In both directions (warp and weft), the increase in tensile strength is due to the presence of three-dimensional array of polymers by coating and subsequent curing. To ascertain the stiffness, bending length and flexural rigidity were also analyzed. The increase in the bending length for the coated fabrics than the uncoated fabric was due to the increase in stiffness. However, increase in the bending length was not significant enough to reduce the stiffness unduly. The fabrics tended to show flexibility due to the optimal concentration of epoxy coating. Further, the results are in accordance with those observed for flexural rigidity (Table 1). The slight increase in the bending length and flexural rigidity is due to the following reasons. The coating of either epoxy or silica or both occurs only at a molecular level due to a lower concentration that could probably penetrate in between yarn/fibers. Thus, the coating results in raise in the overall mass of the fabric without altering the mechanical and other desirable properties required for membrane filters. Finally, for the coated fabrics, whiteness index was calculated to match the surfaces with uncoated fabric. It was noticed that S/cotton (680.6) showed a higher whiteness index value than the uncoated (666.1) and other coated fabrics namely E/cotton (622.9) and SE/cotton (621.7). This could be attributed to the presence of a white opaque silicate network over the fabric. However, the epoxy- and silica/epoxy-coated fabrics showed comparatively closer whiteness values, suggesting that the coating layer was very thin.
Surface analysis
To confirm the surface wetting nature, the treated fabrics were subjected to water contact angle (WCA) analysis and the results are shown in Figure 5. The WCA of the uncoated fabric was observed to be 0° as water was completely absorbed, displaying its hydrophilic nature (Figure 5). However, the E/cotton fabric surface was turned hydrophobic WCA, 138 ± 5° after coating with epoxy.58,59 The WCA of the cotton treated with SBA-15 was observed to be 136±5° expressing an equivalent hydrophobicity similar to E/cotton. However, there was a phenomenal increment in the hydrophobicity displaying WCA. The WCA was observed to be 146±5° approaching toward superhydrophobic surface (WCA > 150°), when coated with both epoxy and SBA-15. In addition, the sliding angle for the SE/cotton was found to be around 12°. The synergistic influence of the SBA-15/epoxy contributed impressively toward increment of hydrophobic nature, which is in accordance with results of epoxy-coated different substrates reported earlier.60–62 Hence, it is strongly believed that the addition of SBA-15 along with epoxy could create a porous channel distribution uniformly, which could subsequently able to reduces the contact between the water droplets to and the surface of cotton. Water contact angles (WCAs) of (a) uncoated cotton, (b) E/cotton, (c) S/cotton, and (d) SE/cotton.
Surface free energy (mN/m) and elemental atomic % of the treated fabrics.

Morphology and elemental analysis
The FESEM images of the coated fabrics are shown in Figure 6. The uncoated fabric shows smooth, flat, and clear fiber surfaces, as shown in Figure 6(a)–(c) at different magnifications (1000×, 2500×, 5000×). The epoxy-coated cotton, E/cotton (Figure 6(d)–(f)), shows randomly distributed rough and protuberance texture, confirming the coating of epoxy on the fibers of cotton and contributing to the origin of hydrophobicity displaying WCA (138°). Figure 6(g)–(i) shows the FESEM images of fabric coated with SBA-15. The presence of whisker-shaped particles over the surfaces confirms the coating of microvillus SBA-15, contributing to dominating surface roughness similar to other inorganic coating constituents.63–65 In addition, the porous nature of SBA-15 as discussed in low angle XRD and BET (Figure 4(a)-(b)) contributes to the equivalent air permeability similarly to the uncoated cotton (Table 1), though it has been coated over the surface in between the interphases of cotton. However, in case of cotton fabric coated with SBA-15/epoxy mixture (Figure 6(j)-(l)), the surfaces show optimum distribution of protuberance morphology, which causes roughness (Figure 6(l)), contributing to the increase in the WCA (146°).
9
Field emission scanning electron microscope images of (a–c) uncoated cotton, (d–f) E/cotton, (g–i) S/cotton, and (j–l) SE/cotton.
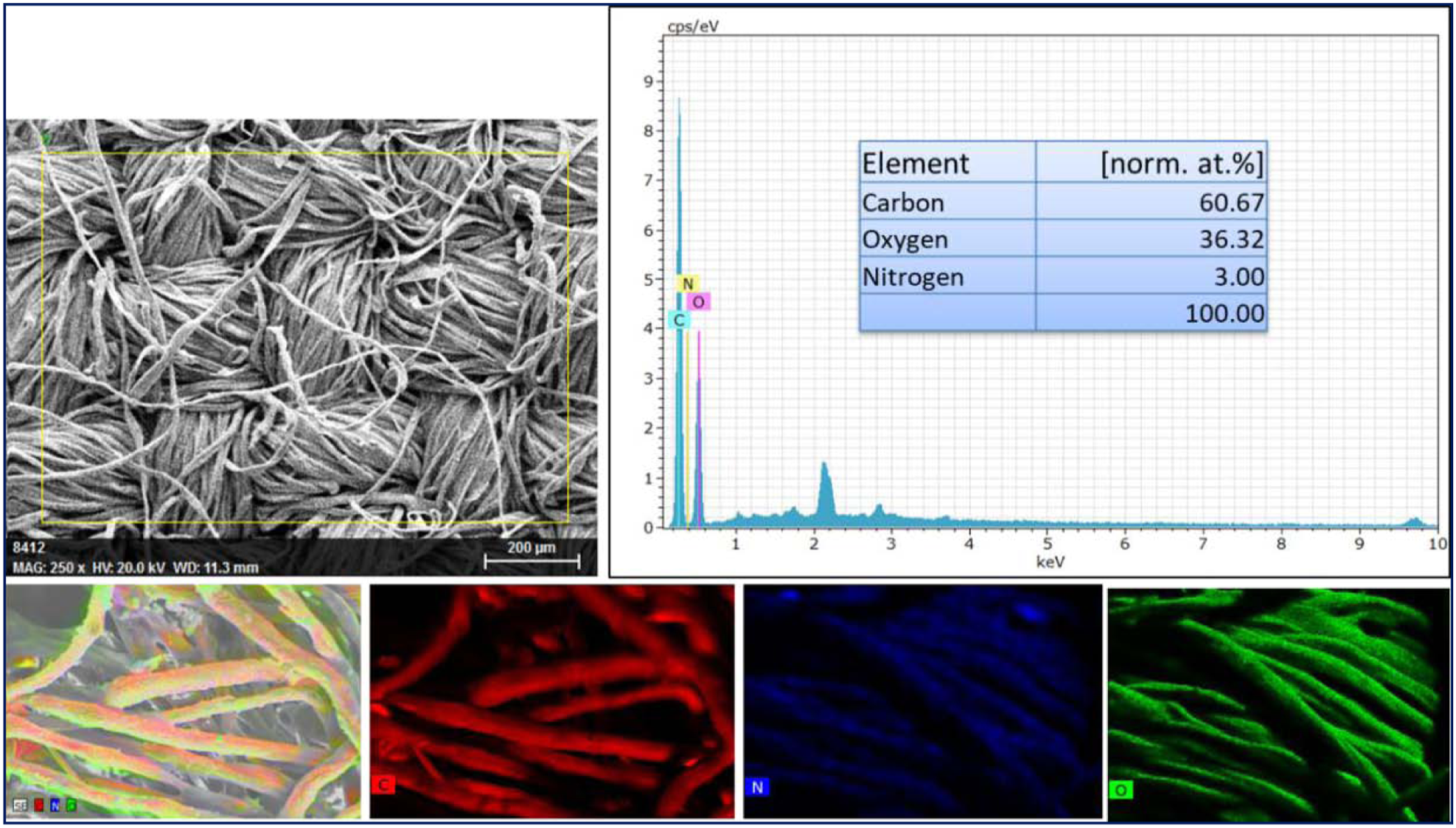
The elemental mapping was performed for all the fabrics coated along with uncoated fabric. The observed elemental ratios for respective fabrics are given in Table 2. The EDS and mapping results of uncoated fabric, E/cotton, and S/cotton are shown in Figures 7, 8, and 9, respectively, The uncoated cotton (Figure 7) shows only the presence of carbon (52.19%) and oxygen (47.81%), corresponding to the carbohydrate molecule of cellulose. However, epoxy-coated fabric shows (Figure 8) the presence of nitrogen (3.0%) in addition to carbon (60.67%) and oxygen (36.32%). The increase in the carbon wt% could be attributed to the presence of cured epoxy coating and the presence of nitrogen is due to the addition of DDM hardener during the curing process. On the other hand, the EDS and elemental mapping of S/cotton in Figure 9 shows the presence of Si (8.63%) in addition to carbon (55.90%), oxygen (33.16%), and nitrogen (2.31%) attributing to the presence of SBA-15. Figure 10 shows the EDS spectra and elemental mapping of SE/cotton. The presence of carbon, oxygen, silicon, and nitrogen from both the EDS spectra and mapping confirms the existence of epoxy and silica in the coatings applied over the cotton (Figure 10). With respect to the binding energy peaks observed for C, O, Si, and N, their amount was found to be 69.31, 24.32, 3.82, and 2.55 wt%, respectively. The mapping of SE/cotton is also shown in Figure 10, which highlights the presence of elements through individual colors. The appearance of red, blue, green, and brown corresponds to the existence of carbon, nitrogen, oxygen, and silicon, respectively, confirming that the coating was applied using both epoxy and SBA-15. EDS and elemental mapping of uncoated fabric. EDS and elemental mapping of E/cotton. EDS and elemental mapping of S/Cotton. EDS and elemental mapping of SE/cotton.


AFM topography
Among the developed fabrics, SE/cotton showed hydrophobic nature; hence it is highly desirable to study its textural properties in detail. Accordingly, AFM analysis of SE/cotton fabrics was performed in comparison with uncoated cotton. The resulted 3D images of the uncoated, E/cotton, S/cotton, and SE/cotton fabrics are shown in Figure 11(a)–(d). The average surface roughness values of the uncoated fabric, E/cotton, S/cotton, and SE/cotton fabrics were 21.47, 35.30, 50.80, and 42.79 nm, respectively. The surface roughness of the cotton fabric observed to increase after coating with the both epoxy and SBA-15. However, SBA-15-alone coated fabric tended to possess more predominant surface roughness compared to those of other coated samples. This phenomenon can be attributed to the presence of the Si–O–Si network over the surfaces of the fabric. These results are in accordance with the observation noticed in the WCA behavior. The synergistic contribution of SBA-15 and epoxy, the SE/cotton fabric tends to improve surface roughness. AFM topography 3D images of coated fabrics in comparison with uncoated fabric.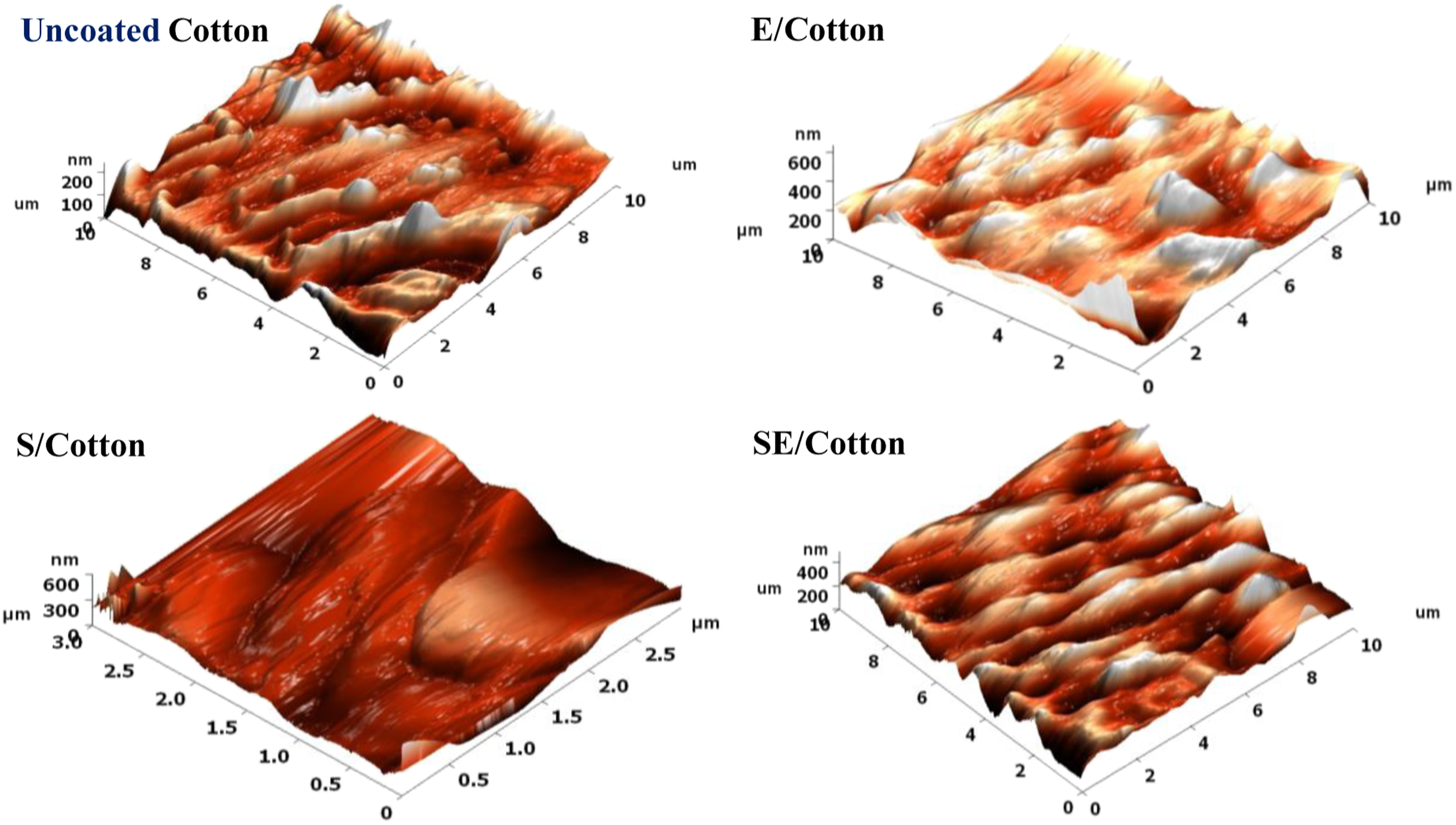
To ascertain the coating of epoxy and SBA-15 over the cotton fabrics, FTIR analysis was performed in comparison with the uncoated cotton. Initially, the FTIR spectra of epoxy (E), SBA-15 (S),and mixture containing SBA-15/epoxy (SA) were recorded and are shown in Figure 12(a). The pure epoxy and SBA-15/epoxy show similar vibrational spectra. The band at around 567 cm−1 corresponds to the para-substituted phenyl ring of the epoxy moiety. The band at 828 cm−1 corresponds to the aromatic–CH out-of-plane deformation. Further, the band at 913 cm−1 corresponds to the oxirane ring vibration and confirms the presence of epoxy. Further, the bands at 1027and 1235 cm−1 correspond to the asymmetric C–O stretching, whereas the band at 1178 cm−1 corresponds to the aliphatic C–O stretching vibrations. Further, the band at 1287 cm−1 could be attributed to aliphatic CH2 asymmetrical deformation. The minor bands at 2928 and 2976 cm−1 correspond to the symmetric and asymmetric stretching vibrations of aliphatic –CH2- group, respectively. A characteristic band of SBA-15 was observed from the broad band appearing at 1074 cm−1, which corresponds to the Si–O–Si stretching vibration. FTIR spectra of (a) epoxy (E), SBA-15 (S), SBA-15/epoxy (SE), and (b) uncoated and coated cotton fabrics.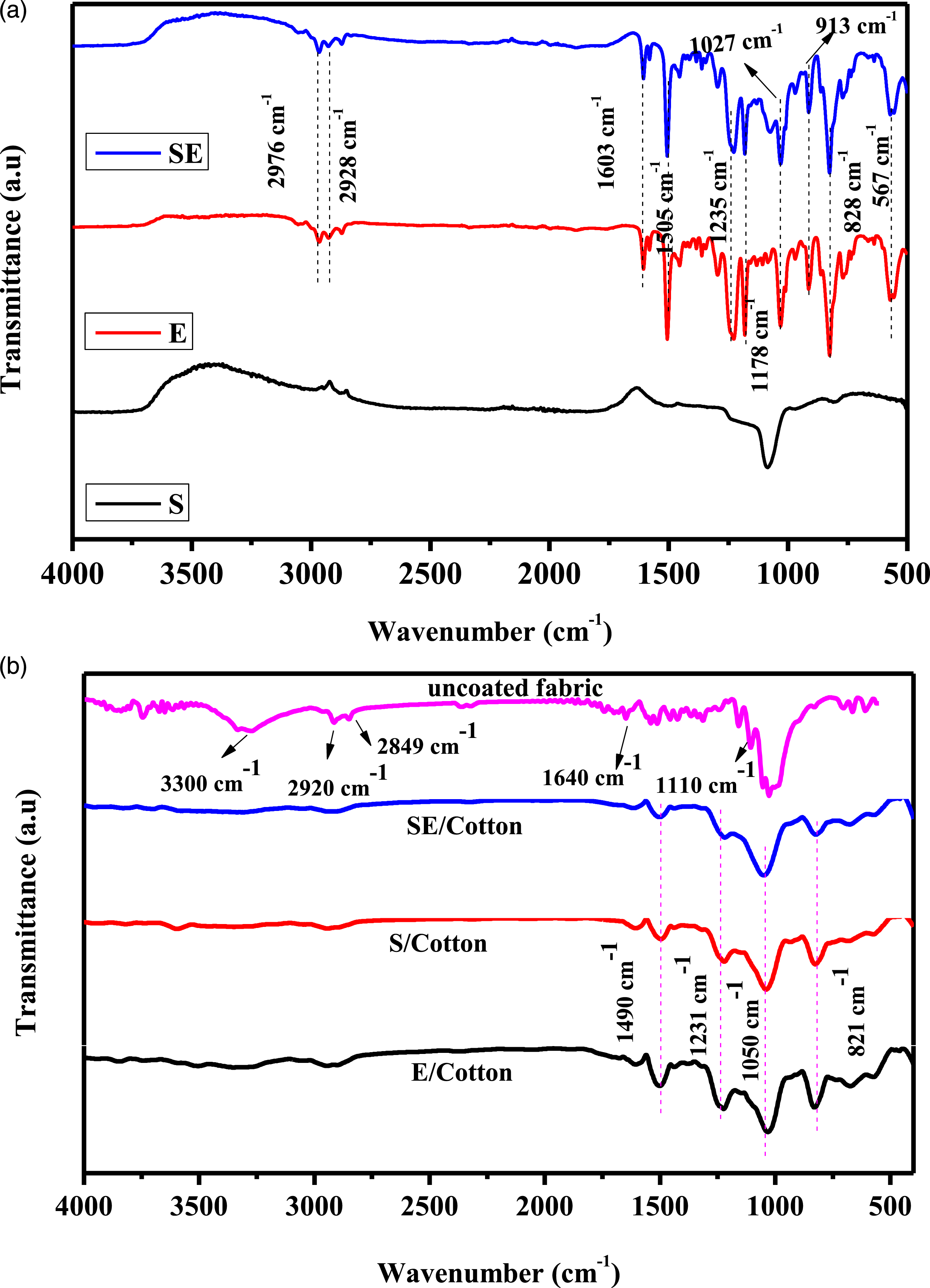
In addition, the FTIR spectra of the uncoated fabric are shown in Figure 12(b) and compared with those of fabrics coated with E, S, and ES. For the uncoated fabric, the appearance of bands at 1640and 1110 cm−1 correspond to the vibrations of carboxylic acid functional groups.66,67 In addition, the peaks around 2920 and 2849 cm−1 correspond to the aliphatic–CH2- vibrations of cotton fabric. The broad peak around 3300 cm−1 corresponds to the vibrations of the hydroxyl functional groups present in the cotton fabric. However, after coating, the broad band corresponding to the hydroxyl functional groups was not recorded. This confirms the occurrence of a strong interaction between the cotton and coated materials. In addition, the absence of vibrational bands at 918 and 1675 cm−1 confirms that the coated epoxy has undergone ring opening polymerization on the surfaces of the cotton fabric.68–70 Also, no distinct peak for (–Si–O–Si-) was observed, which confirms the coating of silica is done with low concentration. 71 These results confirm the coating of epoxy layer over the surfaces of the cotton fabric along with silica in the form of SBA-15.
XRD analysis of the coated fabrics
Figure 13 shows the Bragg X-ray diffractogram patterns of the uncoated cotton in comparison with the treated cotton fabric. As noticed in Figure 13, the uncoated and coated fabrics, namely E/cotton, S/cotton, SE/cotton, show characteristic peaks at 14.5°, 16.5°, and 22.7° having Miller indices of (110), (110), and (200) corresponding to the crystalline structures.72,73 The results suggest that even after coating with epoxy and SBA-15, the formation of a molecular symmetry between carbohydrate molecules of cellulose is disturbed. XRD pattern of the uncoated cotton and treated cotton fabrics.
UV protection behavior
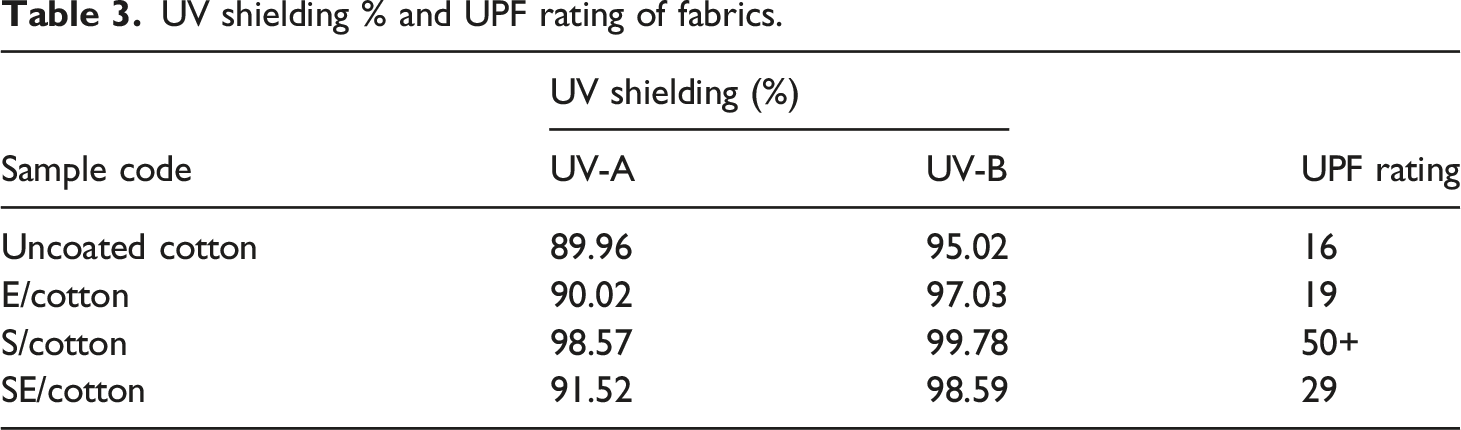
The UV transmittance spectra of the uncoated cotton, E/cotton, S/cotton, and SE/cotton fabrics are presented in Figure 14. The mean UPF values and UPF rating observed are also presented in Table 3. The UPF rating value of the uncoated cotton was observed to be 16, and its corresponding UV transmittance spectra (Figure 14) confirmed a poor UV-shielding behavior. However, after coating with epoxy resin, i.e., the E/cotton, the UV-shielding behavior was meagerly enhanced, as perceived from the UV spectrum, and accordingly the UPF rating increased to 19. Surprisingly, in case of SBA-15-coated cotton (S/cotton), the UV-shielding behavior increased tremendously, as represented in the UV spectrum and the UPF rating, thus the obtained value was as high as 50+(234). This could be attributed to the strong UV-shielding nature rendered by the Si–O–Si network. In case of SE/cotton, the UV-shielding effect was comparably higher than E/cotton and the resulting UPF value was observed to be 29. These results suggest that the coatings present over the cellulosic fiber provide a complete UV protection. SBA-15-coated cotton fabric showed excellent UV protection among this series of coating materials. However, SE/cotton also provided constant protection for both the UV-A and UV-B regions (Figure 14) in addition to its greater hydrophobicity. The synergistic effect contributed by the presence of SBA-15 in the continuous carbon network of bisphenol epoxy resin protects the cellulose structure of the fabric from UV radiations. UV–visible transmittance spectra. UV shielding % and UPF rating of fabrics.
Oil–water separation of the coated fabrics
The process of oil–water separation was examined using a mixture containing equal volumes of distilled water and commercially available diesel dissolved in dichloromethane (DCM) (Figure 15, inset a). The oil–water separation setup equipped with SE/cotton fabric was initially used to verify the test because SE/cotton had superior surface properties. Later, the separation efficiency of E/cotton and S/cotton was also compared. Once, the oils–water mixture was fed over the SE/cotton membrane, the diesel dissolved in DCM immediately started to penetrate through the fabric and got collected in the bottom collection flask under gravity (Figure 15, inset b). After that the water layer was observed to retain over the fabric surface (Figure 15, inset b). This process confirms that the fabric coated with SBA-15/epoxy resin possesses a superior hydrophobic behavior contributed by the low SFE and surface roughness, as witnessed from the WCA and SFE studies. The values of separation efficiency along with respective flux values were calculated (Equations (1) and (2)) and are presented in Figure 15. The separation efficiency of SE/cotton fabric was observed to be about 95.8% (black line) and flux value was found to be 6800 L/m2h (blue line), which was comparatively equivalent to those of thermoset polymeric resin coated fabric.28,30 To explore the separation efficiency and flux values of the other samples, the oil–water experiment was performed by replacing the SE/cotton membrane with E/cotton and S/cotton, respectively. The observed results of separating efficiency and flux values E/cotton and S/cotton are presented in Figure 16 and Figure 17 respectively in order to compare with the results of SE/cotton. The oil–water separation results of E/cotton and S/cotton are not too impressive than those of SE/cotton and the flux values are inconsistent, which could be attributed to the clogging of oils. These results suggest that a synergistic contribution of both epoxy and SBA-15 is highly desired to separate the oil–water mixture. The observed results are in accordance with those of different types of thermoset polymers reported earlier.13,18,29,56 Diesel–water separation efficiency (%) (black) and flux (L/m2h) (blue) of SE/cotton fabric at different cycles (inset: (a)diesel–water mixture; (b) diesel–water separation process, diesel separated through fabric, water retains over the fabric; (c) separated diesel). Diesel/water separation efficiency (%) (denoted in black colour) and flux (L/m2h) (denoted in blue colour) of S/Cotton. Diesel/water separation efficiency (%) (denoted in black colour) and flux (L/m2h) (denoted in blue colour) of E/Cotton.


Self-cleaning properties
The self-cleaning performance of SE/cotton fabrics was studied using commercially available turmeric powder. The SE/cotton fabric surface was scattered with turmeric powder [Figure 18(a)], followed by dropping the water, which allowed the powder to roll down and left no stains [Figure 18(b)]. This behavior shows that the SE-coated fabric shows the self-cleaning surface in addition to the superior hydrophobic nature similar to fluorine-containing polymers.7,74,75 (a) SE/cotton with turmeric powder, (b) self-cleaning performance of SE/cotton fabric.
Durability studies
To predict the durability, the SE/cotton fabric was subjected to laundering and mechanical abrasion analysis. After the laundering and mechanical abrasion, the WCA was taken to interpret the durable nature of coating present over the fabric. The WCA and SFE after 10 wash laundering were observed to be 140° [Figure 19(a)] and 19.3 mN/m, respectively. Similarly, the WCA value was found to be 138° even after 100 cycles of mechanical abrasion, suggesting that the surfaces still retained hydrophobic. These could be attributed to the strong adhesion of epoxy layer along with SBA-15 by chemical bonding, contributing to an enhanced interfacial interaction. In addition, the FESEM images recorded after laundering and mechanical abrasion are also illustrated in Figure 19(c)–(d). The SE/cotton fabric micrographs show the existence of roughness over the open surfaces as well as at the interfaces [Figure 19(c), (d)]. In addition, the WCA value of SE/cotton fabric was measured in different pH liquids and the obtained values are shown in Figure 20. The WCA values were found to be 134° and 132° against acid (pH2) and alkali (pH12) liquids, respectively (Figure 20). The durability of hydrophobic nature of fabric might be due to the presence of acid–alkali resistance epoxy coating. WCA after (a) laundry durability, (b) mechanical abrasion, and FESEM after (c) laundry durability and (d) mechanical abrasion. WCA of SE/cotton at different pH.

Conclusion
This study describes a facile approach to develop low-cost, durable, and hydrophobic/superoleophilic coatings for cotton fabric using epoxy resin and mesoporous SBA-15. The FESEM images showed the microvillus-aggregated morphology of the surface-modified cotton fabrics, with greater surface roughness. The resultant improved in hydrophobicity of the SE/cotton fabric was confirmed from the highest value of WCA (146°) and with the lowest SFE, accounting for its high hydrophobic performance. Subsequently, the SE/cotton fabric was verified for its separation efficiency, which showed tremendous separation efficiency (95.8%) and flux value (6800 L/m2h) against diesel–water mixture. In addition, the UV-shielding ability of the SE/cotton fabric was confirmed from its strong UPF-rating value (29), which is highly desired for membranes in real-time application. Thus, the prepared SBA-15/epoxy resin–coated cotton fabric (SE/cotton) can serve as an effective, low-cost, and durable hydrophobic/oleophilic membrane for oil–water separation applications.
Footnotes
Acknowledgements
This research received no specific grant from any funding agency in the public, commercial, or industrial sectors. The authors thank the PSGTECHS COE INDUTECH, Coimbatore, Tamil Nadu, India, for providing essential instrumentation and characterization support. The authors also thank Turiyam Research Laboratories, Pondicherry for their support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.