Abstract
Herein, inorganic silicon dioxide (SiO2) nanoparticles are deposited on the surface of continuous basalt fibers (CBFs) to enhance the interfacial interactions between CBFs and epoxy matrix (EP) and the tensile strength of composites. According to results from scanning electron microscopy and atomic force microscopy, the surface morphology of CBFs changes from smooth to rough with the increase of SiO2 deposition content. The rough surface plays a role of chock to make CBFs implant into the EP better, offering strong mechanical engagement effect to the interface between CBFs and EP. Therefore, the interfacial shear strength (IFSS) between CBFs and EP and the tensile strength of composites both increase. However, depositing excessive SiO2 nanoparticles also brings some porous structures on CBFs surface, becoming defects and stress concentration points to weaken the IFSS and the tensile strength of composites. Finally, a comparison between grafting organic aliphatic chains and depositing inorganic rigid nanoparticles about their effects on the surface roughness of modified CBFs and the interfacial interactions between modified fibers and matrix is carried out to clarify the reinforcing mechanisms of interfacial adhesion and the tensile strength of composites.
Keywords
Introduction
As an environment-friendly natural material, basalt rock composed of SiO2, TiO2, Al2O3, Fe2O3, CaO and MgO is used to fabricate continuous basalt fibers (CBFs) from a 1450–1500°C of melting process and a high-speed drawing achieved by platinum/rhodium alloy slip plate.1–3 Due to its sound mechanical properties, good low- or high-temperature resistance, high chemical stability, non-toxic, non-combustible and corrosion resistance, CBFs has great potential to be used as fiber reinforcing materials to prepare fiber reinforcing polymer (FRP) composites.4–7 Compared with conventional high performance filaments (carbon fiber, glass fiber and aramid fiber), CBFs not only has comparable mechanical properties and higher strain to failure, but also has some advantages in the terms of low-cost, environmental cleanness and source abundant.8–11 However, the smooth morphology and the chemical inert of CBFs surface lead to a poor interfacial adhesion between CBFs and resin matrix, still limiting the reinforcing effect of CBFs in FRP composite.12–14 On the one hand, poor interfacial adhesion results in an insufficient infiltration process between CBFs and resin matrix, then bringing lots of interspaces and voids at interface. These structural defects will accelerate the fracture of composite in the form of fiber fallen-off. On the other hand, poor interfacial adhesion also depresses the load transfer from resin matrix to fibers and the energy dissipation of applied load during transmission. Therefore, enhancing the interfacial adhesion between CBFs and resin matrix is a prerequisite to maximize the reinforcing effect of CBFs.15,16
Until now, the interfacial adhesion between fiber reinforcing materials and resin matrix has been improved by many methods, mainly including chemical grafting modification,17,18 plasma modification,14,19 irradiation modification,20,21 acid-alkali etching modification, 22 and nanoparticles modification.23–26 Saline coupling agent (SCA) as an active molecule not only reacts with the hydroxyl groups on CBFs surface, but also offers intermolecular interactions with matrix chains based on the existence of aliphatic chains. 18 Therefore, SCA is widely applied to activate the surface of CBFs, allowing to achieve a good infiltration process with resin matrix and improve interfacial adhesion. Meanwhile, the hydroxyl and the amino groups existed in SCA molecules also play a role of bridge to link CBFs and long organic chains. Then, these long organic chains not only reduce the formation of structural defects at interface by improving the affinity between fibers and matrix, but also offer stronger intermolecular interactions to corresponding interface. Yu et al. published that grafting more amino groups and longer aliphatic chains in SCA can provide stronger interfacial adhesions between CBFs-g-SCA and polyamide 66 matrix. 27 Based on Kelly-Tyson model, they found that the change trend of experimental tensile strength of composites with increasing the aliphatic chains in grafted SCA follows a similar trend with fairly good agreement as the change trend of theoretical tensile strength with increasing the interfacial shear strength. This just further confirmed that grafting SCA molecules on CBFs surface can enhance the tensile strength of composites via strengthening the interfacial adhesion between fibers and matrix. Our previous investigations also found that sufficient aliphatic chains offer homogeneous interfacial interactions at all interfaces to carry out the load effective transfer from resin matrix to CBFs, and finally achieve the enhancement of mechanical performances. 28 And limited or excessive aliphatic chains grafted on CBFs surface also resulted in structural inhomogeneity (or structural defects) to depress the reinforcing effects of CBFs for the mechanical properties of composites. Apart from chemical grafting modification, some active functional groups (including hydroxyl, carboxyl, alkoxy, amino, fluorine-containing group and so on) were also decorated on the surface of CBFs via plasma modification, ionic liquid modification and irradiation modification.29,30 These active functional groups also play a similar role as grafted organic aliphatic chains for enhancing the interfacial adhesion between CBFs and resin matrix. Compared with organic aliphatic chains, although the interfacial interactions derived from active functional groups are lower, the corresponding surface becomes more active, offering more possibilities to link various organic chains and then better infiltration process with resin matrix. 31
Increasing the surface roughness of CBFs is also a method to enhance the interfacial adhesion with resin matrix via strengthening mechanical engagement effect between CBFs and resin matrix.25,32,33 When organic aliphatic chains or active functional groups are grafted on CBFs surface, BFCs surface also becomes rough.27,29 However, the mechanical engagement effect provided by flexible aliphatic chains and functional groups is too weak to enhance the interfacial adhesion. Compared flexible aliphatic chains, inorganic rigid nanoparticles deposited on the surface of CBFs embed into resin matrix and play a role of chock to bring stronger mechanical engagement at interface. 25 For example, silica nanoparticles have been widely used in glass fiber-reinforced epoxy resin composite, and the tensile fracture behavior and reinforcing mechanism had been investigated in detail.34–36 From the tensile fatigue behavior of a silica nanoparticle-modified glass fiber reinforced epoxy composites, the particle de-bonding and plastic void growth mechanisms can suppress matrix cracking and reduced crack growth rate, contributing for the observed enhancement of the fatigue life in the GFRP with the nanoparticle-modified matrix. 26 Meanwhile, from the molecular dynamics simulations investigations, the well dispersion of nanoparticles on the surface of fibers and high interfacial binding energy (IBE) between nanoparticles and epoxy matrix can promote tight bonding to the interfaces and provide effective load transfer between the matrix and fibers. The agglomeration phenomenon and the large steric hindrance of nanoparticles will accelerate the crack growth of the composites under stress. 37 In addition, the rigid roughness surface of CBFs is also achieved via acid-alkali etching modification. 38 Although rigid inorganic nanoparticles have been widely studied for enhancing the interface and the mechanical properties of fiber-reinforced composites, their applications in CBFs-reinforced epoxy composites are still limited.39,40 Meanwhile, the relationships among inorganic nanoparticles content, surface roughness, interface adhesion and tensile strength are still waiting to be clarified. According to the results from Li et al., the tensile strength of CBFs/EP composites was enhanced significantly via making CBFs surface rough after being modified by La3+ ions. However, with the further increase of surface roughness, the tensile strength of CBFs-La3+/EP composite suddenly decreased. 29 And from our previous investigation, on the one hand, the intermolecular interactions between CBFs and EP matrix increased with the increase of the grafting content of organic aliphatic chains. On the other hand, the more roughness of CBFs surface derived from grafting excessive organic aliphatic chains also deteriorated the tensile strength of composite. The effects of roughness from being modified by inorganic rigid nanoparticles on the mechanical engagement effects and the mechanical properties of composites need to be further studied and clarified. Furthermore, the contributions of introducing rough surface (rigid) or bringing more intermolecular interactions for enhancing the interfacial adhesion and the performance of composites also need further comparison for ensuring the mechanism of surface modified CBFs reinforced epoxy resin.
Taking these into consideration, inorganic silicon dioxide (SiO2) nanoparticles were used to bring CBFs surface roughness and strengthen the mechanical engagement between CBFs and EP matrix. There are two reasons for selecting SiO2. Firstly, as inorganic nanoparticles, their participations do not bring obvious intermolecular interactions (compared with organic aliphatic chains) to the interface between CBFs and EP matrix. It not only eliminates the interference of intermolecular interactions on the enhancement of composite, but also is helpful to study the effects of roughness and mechanical engagement effect on the reinforcement of composites separately. Secondly, based on the bridge effect of silane coupling agent (KH550), SiO2 nanoparticles can be deposited on the surface of CBFs-g-KH550 via the dehydration to ether reactions among the silanol groups on KH550, the silano groups on SiO2 surface and the silanol groups on CBFs surface. In order to investigate the effects of roughness and mechanical engagement effect on the interfacial adhesion and the performance of composites, a series of CBFs-g-KH550-d-SiO2 with different SiO2 deposition content were fabricated via changing the concentration of KH550 molecules on CBFs surface. And CBFs-g-KH550-d-SiO2/EP composites were obtained from laminating process. The effects of SiO2 deposition content on the surface morphology of CBFs, the surface performances of CBFs, the interfacial shear strength (IFSS) between CBFs and EP matrix, and the tensile strength of composites were characterized by scanning electron microscopy (SEM), atomic force microscopy (AFM), surface energy measurement and tensile test. Finally, the relationships among the surface roughness, the intermolecular interactions, the IFSS and the tensile strength were discussed in detail via comparing the related consequents from depositing inorganic SiO2 nanoparticles with grafting organic aliphatic chains. And a clear mechanism from different methods about strengthening the interfacial adhesion between CBFs and EP matrix and the mechanical properties of composites was also clarified.
Experimental section
Materials
Continuous basalt fibers (CBFs) plain-weave fabric with an areal density of 225 g/m2 were obtained from Sichuan Fiberglass Group Co., Ltd. (Deyang, China). Considering the corrosion resistance and the radiation resistance of the resin matrix, a novel silicon-based modified epoxy resin (EP) with an epoxy value of 32 was obtained from Chengdu Priest New Material Co., Ltd (Chengdu, China). Silicon dioxide (SiO2) nanoparticles (mean diameter: 7–40 nm) was purchased from Evonik Degussa Co., Ltd (Shanghai, China). Methyltetrahydrophthalic anhydride (MeTHPA, industrial grade) as curing agent was purchased from Jining Huakai Resin Co., Ltd. (Jining, China). Dimethylimidazole (analytical grade) as accelerator and 3-aminopropyltriethoxysilane (KH550, analytical grade) as silane coupling agent were bought from Aladdin Biochemical Technology Co., Ltd. (Shanghai, China). Acetone (analytical grade) and ethanol (analytical grade) were obtained from Chengdu Kelong Chemical Reagent Factory (Chengdu, China).
The deposition of SiO2 nanoparticles on continuous basalt fiberss surface
The sizing agent existed on the surface of CBFs plain-weave fabric prevented the interfacial reactions among CBFs, KH550 and SiO2. Therefore, in order to eliminate the size agent, CBFs plain-weave fabric (20 cm × 20 cm) was immersed into acetone with an ultrasonic treatment for 3 h. This process was carried out twice, and then obtained CBFs plain-weave fabric was dried at 80°C for 24 h. 28
KH550 solutions with different concentrations (1 wt%, 2 wt%, 3 wt% and 4 wt%) were prepared via adding a specific weight of KH550 into a mixture of deionized water (50 wt%) and ethanol (50 wt%). And, some SiO2 nanoparticles was also dispersed in the same solvent system to obtained 5 wt% SiO2 dispersion. Subsequently, CBFs plain-weave fabric was immersed into KH550 solution. After 30 min, these CBFs plain-weave fabric was immersed into SiO2 dispersion for 2 h. In order to carry out the interfacial reactions among KH550, SiO2 and CBFs, CBFs plain-weave fabric was put into oven under 110°C for 2 h. As shown in Figure 1, the -OC2H5 groups linked with KH550 converted into the silanol groups due to the hydrolysis reaction. Then, the dehydration to ether reactions among the silanol groups on KH550, the silano groups on the surface of SiO2 nanoparticles surface and the silanol groups on CBFs carried out the depositing of SiO2 nanoparticles on CBFs surface based on the bridge effects of KH550 molecules. Finally, CBFs plain-weave fabric was washed twice in acetone with an ultrasonic treatment for 3 h to remove free SiO2 nanoparticles as well as free KH550 molecules. A series of CBFs grafted with different KH550 contents and different SiO2 contents were named as CBFs-g-x%-KH550-d-SiO2, where x represented the concentration of KH550 solution. The grafting contents of KH550 and SiO2 on a series of CBFs-g-x%-KH550-d-SiO2 were calculated from weighing method, and related data was listed in Table 1. Before experiments, a series of CBFs-g-x%-KH550-d-SiO2 were dried at 50°C for 24 h. The interfacial interactions among CBFs, KH550 and SiO2 nanoparticles. The grating content of KH550 and the deposition content of SiO2 on the surface of CBFs-g-x%-KH550-d-SiO2.

The fabrication of CBFs-g-x%-KH550-d-SiO2/EP composites
The fabrication process of CBFs/EP composites was presented in Figure 2. Firstly, according to the weight ratio of 100:60:1, EP, MeTHPA and dimethylimidazole were weighed respectively. Then, dimethylimidazole was mixed with MeTHPA liquid in a glass beaker with mechanical stirring under 80°C. After complete dissolution, the mixture of dimethylimidazole and MeTHPA was blended with EP together with mechanical stirring to obtain EP solution. After that, prepared EP solution was coated uniformly on the surface of polyester film. Secondly, based on the final weight ratio between CBFs and EP matrix in composites (75 wt%: 25 wt%), a specific weight of CBFs-g-x%-KH550-d-SiO2 was placed on the top of EP solution. According to the density of EP matrix (1.25 g·cm−3) and the density of CBFs (2.80 g·cm−3), the volume fraction of CBFs-g-x%-KH550-d-SiO2 in final composite was calculated to be 57.3%, and the volume fraction of EP matrix correspondingly to be 42.7%. After being coated by another polyester film, CBFs-g-x%-KH550-d-SiO2/EP solution mixture was rolled several times by laminating machine for achieving a full impregnation between CBFs-g-x%-KH550-d-SiO2 and EP solution. Then, the bubbles existed in CBFs-g-x%-KH550-d-SiO2/EP solution mixture were removed via vacuuming. The fabrication process of CBFs/EP composites.
Thirdly, after removing the polyester film on the surface, 10 layers CBFs-g-x%-KH550-d-SiO2/EP prepregs were stacked vertically and then placed between two smooth and flat steel plates for carrying out the hot press forming process. At the beginning, the temperature was set at 90°C, and the prepregs were pre-cured for 0.5 h without applied pressure. Then, the applied pressure was increased to 1 MPa for 0.5 h. After this, the curing process was carried out at 120°C with a 3 MPa of applied pressure for 1 h. At last, temperature increased to 150°C and applied pressure was set at 5 MPa to achieve the complete curing of CBFs-g-x%-KH550-d-SiO2/EP prepregs. And a series of composites with different SiO2 deposition contents were named as CBFs-g-x%-KH550-d-SiO2/EP, where x presented the concentration of KH550 solution.
Measurements and characterizations
The surface morphology and the surface roughness of CBFs, CBFs-g-x%-KH550 and CBFs-g-x%-KH550-d-SiO2, and the cross-section morphology of composites were observed via an Inspect F50 field emission scanning electron microscopy (Field Electron and Ion Co., OR, USA). The cross-section of composite was obtained from the mechanical test. Before observation, all samples were coated by gold. And the acceleration voltage was 20 kV.
Atomic force microscopy (AFM) (Smart SPM, AIST-NT Inc, CA, USA) was also used to observe the surface roughness of CBFs, CBFs-g-x%-KH550 and CBFs-g-x%-KH550-d-SiO2. Meanwhile, a 3D image of observation area was drawn by aist-3.3.112 software. And the root mean square roughness (Rms, unit: nm) of observation area was also calculated by this software.
The wettability and the interfacial properties between CBFs and EP matrix were analyzed from the contact angle measurement carried out via an optical contact angle meter (SDC-100, Shengding Precision Instrument Co., Ltd., Dongguan, China). The contact angles of three substances (water, glycerol and epoxy solution) on a series of CBFs surface were measured. According to Young-Dupre equation, the interfacial energy 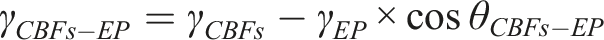



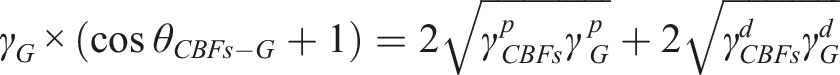
The IFSS between CBFs-g-x%-KH550-d-SiO2 and EP matrix was characterized via droplet microdebond test
43
based on a composite material interface microscopic tester (Beijing Fuyouma Technology Co., Ltd., Beijing, China). The length of CBFs-g-x%-KH550-d-SiO2 embedded in EP droplet was set about 10 µm. And the loading rate was set at 0.02 mm/s. IFSS was calculated from following equation
The tensile strength of CBFs-g-x%-KH550-d-SiO2/EP composite was measured by a universal tensile testing machine (Instron 5967, Instron Corporation, MA, US) based on GB/T 1447-2005. The sample size for tensile tests were 150 mm × 10 mm × 2 mm. The tensile rate was set at 5 mm/min, and each sample was tested five times and averaged.
Results and discussions
Surface morphology
According to our previous investigations,
28
KH550 molecules were grafted on the surface of CBFs successfully via the dehydration to ether reactions between the silanols on KH550 and the silanols on CBFs at 110°C. KH550 molecules also played a role of bridge to link CBFs and SiO2 nanoparticles by the dehydration to ether reactions among the silanol groups on KH550, the silanol groups on SiO2 nanoparticles and the silanols on CBFs (Figure 1), which was confirmed by Kuzmin et al. based on FTIR spectrum.
40
The morphology changes of CBFs surface before and after grafting KH550 as well as depositing SiO2 nanoparticles were observed from SEM images (Figure 3). For neat CBFs, its surface is smooth, dense and non-porous. After being grafted KH550 molecules, the surface morphology of CBFs-g-1%-KH550 becomes rough and rugged, which is consistent with previous publications.
44
With further depositing SiO2 nanoparticles, many SiO2 nanoparticles are observed on the surface of CBFs-g-1%-KH550-d-SiO2, and the morphology becomes more rough and like pebble-pavement. Excluding free SiO2 nanoparticles, these tiny particles deposited on CBFs surface are regarded as the grafted SiO2 nanoparticles.
40
The scanning electron microscopy images of the surface morphologies of neat CBFs, CBFs-g-1%-KH550 and a series of CBFs-g-x%-KH550-d-SiO2.
With the further increase of SiO2 deposition content, more and more SiO2 nanoparticles are observed on the surfaces of CBFs-g-2%-KH550-d-SiO2, CBFs-g-3%-KH550-d-SiO2 and CBFs-g-4%-KH550-d-SiO2. And the deposited SiO2 nanoparticles becomes a coating layer gradually covering on the surface of CBFs. The surface morphologies are still rough, and some gullies and pores are observed on the surface of SiO2 coating layer. These gullies, pores and rough morphologies offer a large specific surface area to CBFs surface, which promotes the infiltration process between CBFs and EP matrix. 40
The surface morphology of various samples is also observed via AFM images (Figure 4). The results are similar as the results from SEM images. The surface morphology of neat CBFs is smooth without obvious grooves or salient. For CBFs-g-1%-KH550 and a series of CBFs-g-x%-KH550-d-SiO2, their surface morphologies of become rough and rugged. And some raised areas are also observed. It not only confirms the existence of KH550 molecules as well as SiO2 nanoparticles on CBFs surface, but also indicates that introducing KH550 molecules and SiO2 nanoparticles also bring a rough morphology to CBFs surface. Meanwhile, with the increase of SiO2 deposition content, not only the surface morphology of CBFs becomes more rough, but also the number of raised areas existed on CBFs surface increases. It indicates that the roughness of CBFs surface increases with increasing the deposition content of SiO2 nanoparticles. The atomic force microscopy images of a series of CBFs before and after the modifications of KH550 as well as SiO2 nanoparticles.
Surface roughness
The roughness of CBFs before and after a series of modifications can be quantified by the root mean square roughness (Rms, unit: nm) of surface. And this value can be calculated via specialized software based on AFM images. The Rms of CBFs grafted KH550 molecules, CBFs grafted KH550 and organic aliphatic chains (HDE), and CBFs grafted KH550 as well as deposited SiO2 are given in Figure 5. For neat CBFs, its Rms is 3.65 nm. After being grafted KH550 molecules, the Rms of CBFs-g-1%-KH550 increases to 21.12 nm. With further increasing the concentration of KH550 solution from 1 wt% to 4 wt%, the Rms of CBFs-g-x%-KH550 decreased from 21.12 to 9.85 nm. With the increase of KH550 grafting content, the grafting area of KH550 molecules on CBFs surface changes from island-like morphology to sea-like morphology, and finally becomes a uniform coating layer covering on CBFs surface. Therefore, the Rms of CBFs-g-x%-KH550 decreases with the increase of KH550 grafting content. When CBFs-g-1%-KH550 is deposited SiO2 nanoparticles, the corresponding Rms decreases from 21.12 to 7.03 nm. It might be related with the size of SiO2 nanoparticles as well as their limited deposition content of CBFs surface. Compared with the length or size of KH550 molecules, the size of SiO2 nanoparticles is larger. For CBFs-g-1%-KH550-d-SiO2 and CBFs-g-2%-KH550-d-SiO2, the deposition contents of SiO2 nanoparticles are only 0.33 wt% and 0.99 wt%. The deposition of limited SiO2 nanoparticles on CBFs surface might make up the height differences between the area aggregated more KH550 molecules and the area grafted less KH550, resulting in a reduction of surface roughness. The root mean square roughness of the surface of a series of CBFs grafted with different substances (black: CBFs grafted different KH550 content; red: CBFs deposited different SiO2 content; blue: CBFs grafted different HDE content).
With the increase of KH550 molecules content on CBFs surface, sufficient KH550 molecules offer more active cites to deposit more SiO2 nanoparticles on CBFs surface. Therefore, the SiO2 deposition contents of CBFs-g-3%-KH550-d-SiO2 and CBFs-g-4%-KH550-d-SiO2 increased to 2.89 wt% and 6.07 wt%, respectively. And, the corresponding Rms increases sharply to 59.87 and 81.14 nm, respectively. When sufficient inorganic SiO2 nanoparticles deposits on CBFs surface, SiO2 nanoparticles offer higher surface roughness to CBFs compared with KH550 molecules. 45
According to our previous investigations, grafting 1,6-hexanediol diglycidyl ether (HDE, organic aliphatic chains) also brought some changes to the surface roughness of CBFs-g-x%-KH550. When the grafting content of KH550 molecules existed on CBFs surface was low, the adding of HDE chains accelerated the transmission of grafting area from island-like to sea-like. With further increasing the grafting content of KH550 molecules and HDE chains, more grafting areas (new islands) appeared on the surface of uniform grafting coating (sea), which resulted in new roughness again. Therefore, the surface roughness of CBFs-g-3%-KH550-g-HDE and CBFs-g-4%-KH550-g-HDE further increased. Finally, due to the size differences among KH550 molecules, HDE chains and SiO2 nanoparticles, the increase of surface roughness derived from the deposition of SiO2 nanoparticles is higher than that from grafting HDE chains, and these two methods are both higher than the case from grafting KH550 molecules.
Surface energy
The contact angles of different droplets on a series of CBFs surfaces.

The surface energies of a series of CBFs and the interfacial energies between CBFs and EP matrix as well as adhesion works.

After grafting KH550 molecules and depositing SiO2 nanoparticles, the surface energies of a series of treated CBFs increase. From Table 3, the increase of CBFs surface energy after modification mainly derives from the increase of polar components. The hydroxyl groups and amine groups in KH550 molecules as well as SiO2 nanoparticles increases the polar component on the surface of CBFs, which leads to the increase of CBFs surface energy. Meanwhile, the adhesion work as a parameter to characterize the energy required for interface separation between fibers and matrix, also increased gradually after grafting KH550 molecules and depositing SiO2 nanoparticles. The increases of CBFs surface energy and adhesion work between CBFs and EP matrix indicates that the interfacial adhesion between CBFs and EP matrix can be improved by grafting KH550 molecules and depositing SiO2 nanoparticles. And compared with grafting KH550 molecules, depositing SiO2 nanoparticles on CBFs surface can offer stronger interfacial bonding with EP matrix based on stronger mechanical engagement effects. In addition, the aggregation of excessive SiO2 nanoparticles also reduces the adhesion work between fibers and matrix as well as the related interfacial bonding with EP matrix.
Interfacial adhesion
In order to further investigate the effects of depositing SiO2 nanoparticles on the interfacial adhesion between CBFs and EP matrix, the cross-section morphologies of composites before and after depositing modification are observed and analyzed from SEM images (Figure 6). For neat CBFs/EP composite, some obvious interfacial de-bonding and fibers fallen-off phenomena are observed from the fracture morphology of composite. Meanwhile, for the fallen-off CBFs, their surfaces are smooth and there shows less EP resin attached to them. The results indicate that the interfacial adhesion between reinforced fibers and EP matrix is weak before surface modification. After grafting KH550 molecules, even though the interfacial de-bonding and fibers fallen-off phenomena are still found on the fracture section of composites, there are some EP resin attached to the surface of the fallen-off CBFs. According to the investigations from LIU et al., the existence of attached matrix resin revealed the improvement of interfacial adhesion between reinforced fibers and matrix.
46
This also indicates that grafting KH550 molecules on CBFs surface enables a certain degree of improvement to the interfacial adhesion between CBFs and EP matrix via increasing the roughness of CBFs as well as the interactions between CBFs and EP matrix.
47
The cross-section morphologies of neat CBFs/EP, CBFs-g-1wt%-KH550/EP and a series of CBFs-g-x%-KH550-d-SiO2 composites.
When SiO2 nanoparticles are deposited on the surface of CBFs-g-x%-KH550, the interfacial de-bonding and fibers fallen-off phenomena disappear gradually. Instead, a flat and compact fracture morphology is observed. Meanwhile, more and more EP resin adheres on the surface of CBFs-g-x%-d-SiO2. This indicates that the interfacial adhesion between CBFs and EP matrix is further enhanced with the increase of the SiO2 depositing content. 48 The SiO2 nanoparticles deposited on CBFs surface can play a role of chock to make CBFs implant into the EP better. When CBFs are implanted into the EP matrix better, the mechanical engagement between CBFs and EP matrix offers stronger interfacial adhesion. However, when excessive SiO2 nanoparticles are deposited on CBFs surface, the interfacial de-bonding and fiber fallen-off phenomena appear again. This indicates that the further increase of surface roughness of CBFs deteriorates the interfacial adhesion between CBFs and EP matrix. When excessive SiO2 nanoparticles are deposited on CBFs surface, some porous structures are obtained with the stacking of SiO2 nanoparticles (Figure 2). These pores at the interface become structural defects and stress concentration points to reduce the interfacial adhesion between CBFs and EP matrix.
The surface roughness obtained from different modification methods also brings different IFSS to the interface between CBFs and EP matrix. Figure 7 presents the IFSS of different interfaces obtained from different modification methods. When KH550 molecules are grafted on the surface of CBFs, the IFSS between CBFs-g-KH550 and EP matrix increases from 43.4 to 47.3 MPa with the increase of KH550 grafting content. When SiO2 nanoparticles are deposited on the surface of CBFs-g-KH550, the IFSS between CBFs and EP further increases. For example, the IFSS between CBFs-g-1%-KH550-d-SiO2 and EP matrix increases to 47.6 MPa compared with CBFs-g-1%-KH550. Meanwhile, with the increase of SiO2 deposition content, the IFSS increases gradually. For CBFs-g-3%-KH550-d-SiO2, its IFSS with EP matrix reaches 58.6 MPa. It indicates that the chock effect of SiO2 not only makes CBFs implant into EP matrix better, but also strengthen the mechanical engagement effect between CBFs and EP. Therefore, the interfacial adhesion between CBFs and EP matrix increases significantly after depositing SiO2 nanoparticles. However, when excessive SiO2 nanoparticles are deposited on CBFs surface, the porous structures and gaps derived from the mutual stacking between nanoparticles also become weak points to deteriorate the IFSS. The interfacial shear strength of between CBFs and EP matrix in a series of composites obtained from different modification methods.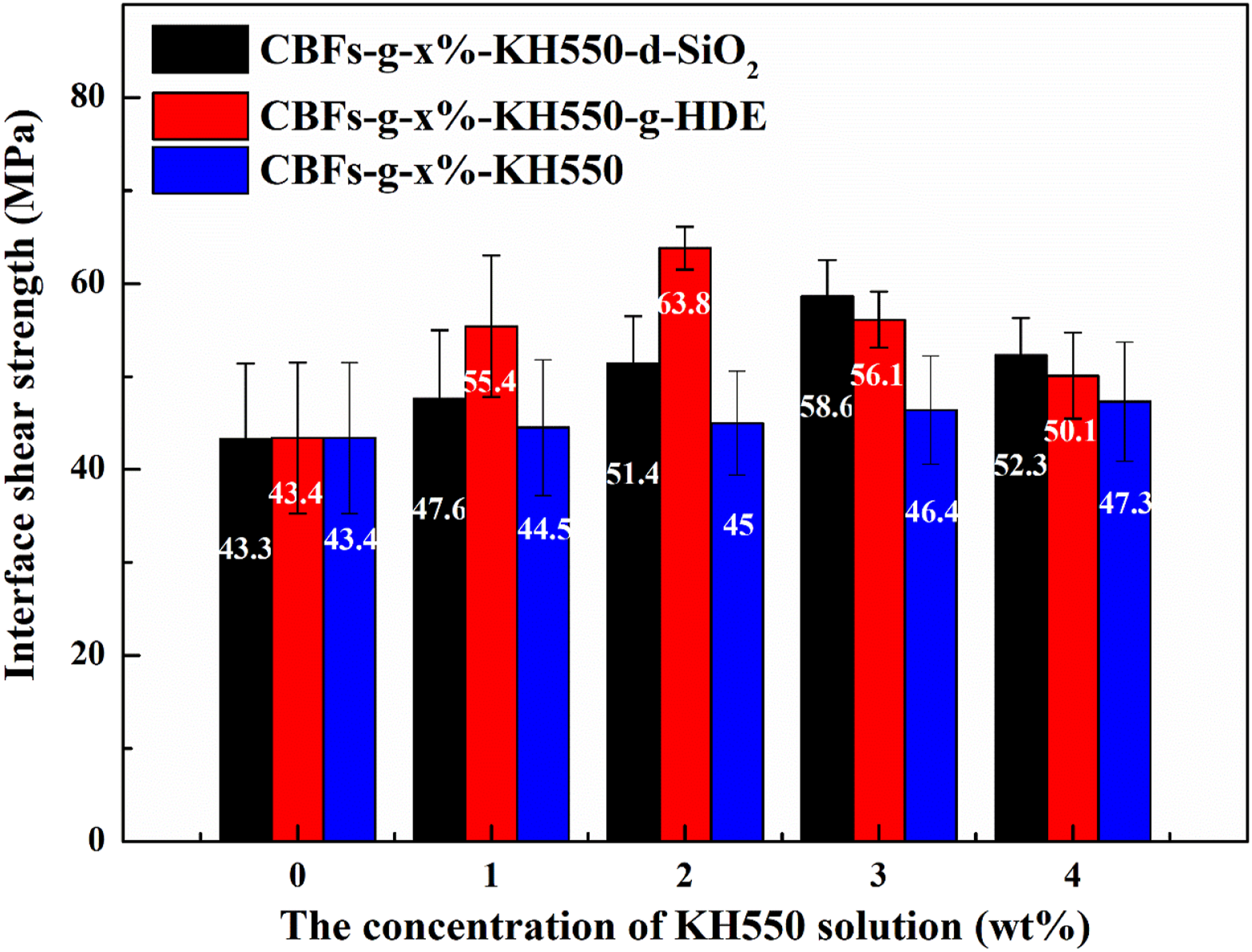
Apart from inorganic nanoparticles, the IFSS between CBFs and EP matrix is also strengthened via grafting organic aliphatic chains. When the grafting content of HDE chains is low, although the surface roughness of CBFs-g-1%-KH550-g-HDE (or CBFs-g-2%-KH550-g-HDE) was low, its IFSS with EP matrix was higher the situations from CBFs-g-1%-KH550-d-SiO2 and CBFs-g-2%-KH550-g-SiO2. On the one hand, it is difficult for limited SiO2 nanoparticles deposition content to provide sufficient mechanical engagement to enhance the IFSS. On the other hand, the intermolecular interactions between HDE chains and EP matrix offered stronger interfacial adhesion to reinforce the IFSS. With further increasing the grafting content of HDE, the IFSS between CBFs-g-3%-KH550-g-HDE (or CBFs-g-4%-KH550-g-HDE) and EP matrix decreased. It was related with the non-homogeneous location of excessive HDE chains on the surface of CBFs-g-KH550-g-HDE. The differences of intermolecular interactions at different interfaces resulted in some weak points to deteriorate the IFSS between CBFs and EP matrix. When sufficient SiO2 nanoparticles are deposited on the surface of CBFs, it also provides an improvement in IFSS comparable to that was achieved by grafting organic aliphatic chains. Meanwhile, for CBFs-g-3%-KH550 and CBFs-g-4%-KH500, depositing SiO2 nanoparticles can obtain higher IFSS between CBFs and EP matrix compared with grafting HDE chains. Finally, the maximum of IFSS obtained from grafting HDE chains is higher than that of from depositing SiO2 nanoparticles, indicating that grafting organic chains has larger potential to improve the IFSS between CBFs and EP matrix.
Tensile strength
Finally, the tensile strengths of a series of composites fabricated from different modified CBFs are given in Figure 8. When KH550 molecules were grafted on CBFs surface, the tensile strength of CBFs-g-x%-KH550/EP composites increased gradually with the increase of KH550 grafting content. For example, compared with neat CBFs/EP composite, the tensile strength of CBFs-g-4%KH550/EP composite increased from 251 MPa to 293 MPa. It was related with the activation of CBFs surface after grafting KH550 molecules as well as the improvement of the intermolecular interactions between CBFs and EP matrix. When SiO2 nanoparticles were further deposited on the surface of CBFs-g-x%-KH550, the tensile strength of CBFs-g-x%-KH550-d-SiO2/EP further increased. For example, compared with CBFs-g-1%-KH550/EP composite, the tensile strength of CBFs-g-1%-KH550-d-SiO2/EP composite increased from 263 MPa to 292 MPa. And when CBFs-g-3%-KH550 was deposited SiO2 nanoparticles, the tensile strength of composite reached maximum (358 MPa). It indicated that the mechanical engagement effect between CBFs-g-x%-KH550-d-SiO2 and EP matrix not only further strengthened the IFSS, but also the tensile strength of composite via offer a path to carry out the load transfer from matrix to fiber. However, with the further increase of SiO2 nanoparticles deposition content, the tensile strength of CBFs-g-4%-KH550-d-SiO2/EP decreased to 315 MPa, which was related with the porous structures derived from the random stacking of excessive SiO2 nanoparticles on CBFs surface. These porous structures existed on CBFs surface became some structural defects or stress concentration points to accelerate the interfacial exfoliation between CBFs and EP matrix as well as the fracture of composite. The tensile strength of a series of composites derived from different modified CBFs.
Meanwhile, according to our previous investigations, the tensile strength of CBFs-g-x%-KH550/EP composite was further strengthened via grafting organic aliphatic chains on CBFs-g-x%-KH550. Compared with depositing SiO2 nanoparticles, the maximum tensile strength of CBFs-g-x%-KH550-g-HDE/EP was higher, and reached 393 MPa when 2 wt% KH550 solution was used to prepared CBFs-g-KH550-g-HDE. It indicated that the intermolecular interactions between CBFs and EP matrix provided from grafting organic aliphatic chains had stronger ability to achieve the load effective transfer from matrix to fiber compared with mechanical engagement effect derived from depositing SiO2 nanoparticles. Although excessive HDE chains grafted on the surface of BCF weakened the tensile strength of composite, their improvement effects on the tensile strength were still stronger than depositing SiO2 nanoparticles. It also indicated that the structural defects or stress concentration points derived from the non-homogeneous location of the intermolecular interactions between CBFs-g-x%-KH550-g-HDE and matrix were less than that from the random stacking of excessive SiO2 nanoparticles. In addition, compared with grafting organic aliphatic chains, although the higher roughness from depositing SiO2 nanoparticles was obtained, but the related improvement effects on tensile strength was still lower. Therefore, it can conclude that enhancing the intermolecular interactions from grafting organic aliphatic chains played a more important role in reinforcing interfacial adhesion as well as the tensile strength of composite compared with increasing the surface roughness of CBFs via depositing SiO2 nanoparticles.
Conclusions
In this paper, SiO2 nanoparticles were deposited on CBFs surface successfully via the bridge effect of KH550 molecules. After depositing SiO2 nanoparticles, the surface morphology of CBFs became rough. The existence of SiO2 nanoparticles on CBFs surface played a role of chock to strengthen the mechanical engagement effect between CBFs and EP matrix, which not only improved the interfacial adhesion, but also the tensile strength of composites. In addition, the porous structures derived from the random stacking of excessive SiO2 nanoparticles also weakened the interfacial adhesion and tensile strength.
Based on the results from grafting KH550 molecules and organic aliphatic chains, and depositing SiO2 nanoparticles, the interfacial adhesion between CBFs and EP matrix and the tensile strength of composite mainly depended on the surface roughness of CBFs, intermolecular interactions between CBFs and EP matrix and the structural defects (or stress concentration points) at interface. Although both strengthening intermolecular interactions and increasing surface roughness can reinforce the interfacial adhesion as well as tensile strength, the non-homogeneous dispersion of intermolecular interactions at interfaces and the porous structures derived from the rough surface also became weak points to deteriorate the interfacial adhesion and tensile strength. Meanwhile, compared with increasing the mechanical engagement effect by depositing rigid nanoparticles, strengthening intermolecular interactions with EP matrix from grafting compatible chains on CBFs surface had greater potential to reinforce the interfacial adhesion and the tensile strength of composites. Finally, achieving the homogeneous dispersion of strong intermolecular interactions at all interfaces was also an important factor to maximize the related enhancement effects on interfacial adhesion as well as tensile strength.
Footnotes
Acknowledgements
The authors were grateful to the Sichuan Science and Technology Program (No. 2022YFG0272) for financial supporting.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Sichuan Science and Technology Program (No. 2022YFG0272).
