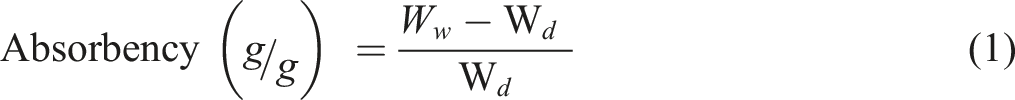Abstract
Sodium alginate and hyaluronic acid are being used extensively in wound care applications for their exceptional properties, including gelation, biocompatibility, exudate absorption, and the ability to provide a moist environment to the wound bed that accelerates healing. This research work presents the potential of developing silver and zinc ions loaded bioactive fibers for wound care applications with improved absorption, swelling (gelling), and antibacterial properties. The effect of loading silver and zinc on alginate/hyaluronic acid biofiber absorption, swelling (gelation), tensile and antibacterial activity were analyzed. It was found that the addition of silver and zinc salts improved the absorption, tensile, and gelation properties of alginate/hyaluronic acid fibers. Zinc-containing fibers exhibited superior properties to silver-containing fibers. The presence of hyaluronic acid influenced the release of silver and zinc ions in various liquid media with a maximum of 26 g/g absorption was observed which suggested the good wound exudate absorption capacity of the developed fibers. The developed fibers showed good antibacterial activity against Staphylococcus aureus and Escherichia coli strains.
Introduction
Over the last few years, growing environmental concerns have extended the use of naturally occurring polymeric materials. 1 Those are the polymers that undergo microbially induced chain degradation leading to mineralization and thus produce no harm to environments.2,3 Researchers have been mainly focusing on developing green biopolymeric materials involving the use of non-toxic and environmentally friendly manufacturing processes.4,5
Natural polymers have significant applications in medical and healthcare sectors concerning their favorable biological functions, including biocompatibility, antibacterial, antioxidant, bioactive nature, and anti-inflammatory activities with minimum toxicity.4,6 Polysaccharides are one of the most considerably known natural materials on earth. They are carbohydrates and are comprised of repeated small glucose molecules that are affixed with each other. They have distinct molecular structures and have found suitable applications in various fields.6,7 Owing to their chemical characteristics, they exhibit polyfunctional nature, chelation activity, and absorption ability that enables them to be modified biochemically.8,9
Alginate and hyaluronic acid are naturally occurring polysaccharides.7-9 These polysaccharides are biocompatible, non-toxic, bio-degradable, and non-allergic. They also exhibit excellent gelling and absorption properties.4,7 Alginate possesses two main components in its structure, that is, α-L-guluronic acid (G) and β-D-mannuronic acid (M).
10
The structure of alginate is shown in Figure 1. High guluronic acid content fibers have better dimensional stability and excellent “calcium-binding ability” compared to mannuronic acid contents.
11
Structure of sodium alginate.
Alginate originates from brown algae and has properties of biocompatibility and biodegradability. 12 It has been found non-toxic to the human body and mainly used to develop wound dressing fibers. It has no antibacterial activity; however, it effectively provides a moist environment on wounds due to its unique gel-forming properties. 13
Hyaluronic acid is polyanionic and naturally occurring in connective tissues of vertebrates.
14
The molecular weight of hyaluronic acid ranges from 103-107 Da (Figure 2).
11
It is also present in connective tissues of the body and has a high molecular weight.
14
The exceptional viscosity of hyaluronic acid makes it an ideal lubrication provider.
15
Hyaluronic acid absorbs moisture from surroundings and possesses viscoelastic characteristics that broaden its applications. Due to its unique properties like biologically compatible anti-inflammatory, hyaluronic acid stimulates the growth of anti-bodies, promotes the body’s immune system, and retains the moist environment. Hyaluronic acid plays a vital role in the repairing of tissues and act as a hydrating agent.
16
Furthermore, hyaluronic acid plays important role in providing strength and cushioning effects to the skin.
17
Structure of salt of hyaluronic acid.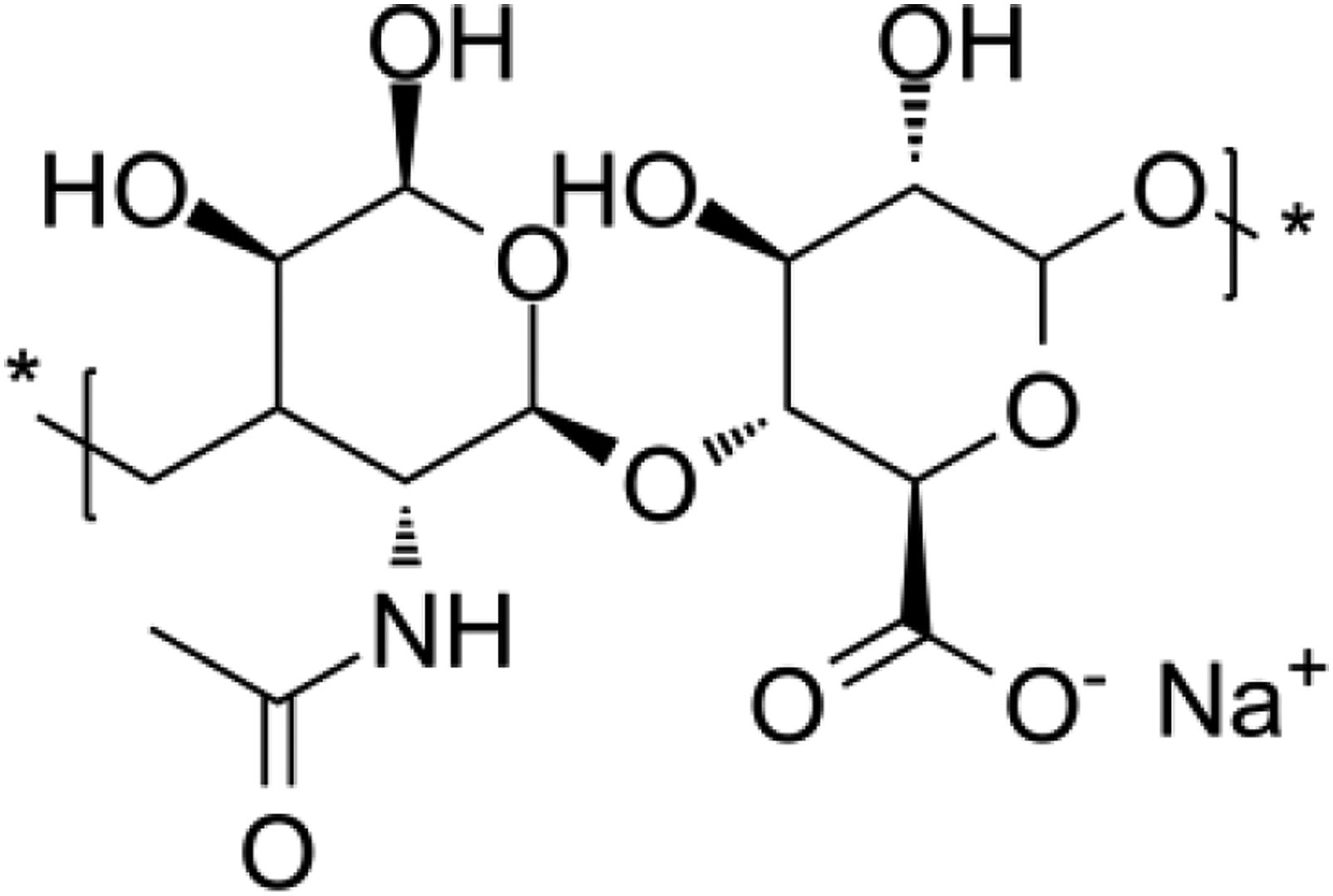
Silver and zinc salts have been incorporated into biomaterials to improve their antibacterial and healing properties as these biopolymers are very susceptible to bacterial agents.18-19 The use of zinc overcomes its deficiency in the human body and aids in wound healing, and synthesizes protein and collagen.20,21 Silver has been used to kill harmful bacteria by damaging the cell membrane and interrupting the enzyme’s activity of bacteria. Silver ions are non-toxic to the human body and generate a bacterial-free environment even at low concentrations.22-23
Researchers have developed alginate, chitosan, and hyaluronic acid composite fibers with improved antibacterial and absorption properties. However, the development process was not much environment friendly due to excessive use of hydrochloric acid in the hydrolysis of chitosan. Neibert et al. fabricated sodium alginate (SA) fibers loaded with silver nanoparticles using a wet-spinning technique to repair the wound area and reduce the bacterial burden. Such fibers were crosslinked using calcium chloride, which resulted in highly swollen fibers. These antibacterial fibers possess high mechanical strength and swelling properties due to the carboxyl groups. 24 Zare-Gachi et al. synthesized various ammonium salts using alginate and developed a fibrous wound dressing. Results reported that the tributyl-ammonium-based alginate dressing had improved antibacterial and anti-hemolytic properties. 25 The current study developed bio-composite antibacterial fibers containing silver and zinc ions. 26 Though many reports have prepared zinc and silver ion embedded fibers separately, no study has reported comparing these two salts on fiber-forming properties, gelling behavior, ion release, tenacity, and others.
Experimental
Materials
Sodium alginate (high guluronic acid contents) was supplied by FMC Biopolymer, Norway. Commercial grade hyaluronic acid (bovine source) was purchased from Pure Bulk, Inc. Roseburg. Reagent grade silver carbonate, acetone, zinc carbonate, sodium chloride, and calcium chloride were purchased from Merck.
Fiber Spinning Process
A dope was prepared for 0.05% (w/v) silver and zinc followed by adding 0.2% (w/v) of hyaluronic acid while stirring at 750 r/min for 2 h at 30°C. Sodium alginate (5% w/v) was added to the solution and stirred for 2 h at 30°C. The solution was degassed by leaving it overnight in a dope tank. Dope was ejected through a spinneret (200 μm) under the pressure of 2 bar in a coagulation bath (2% calcium chloride) to produce fibers. Fibers were then collected over collecting rollers and passed through washing bath, and drawn between first and second pairs of rollers. Collected fibers were thoroughly rinsed using deionized water and were dried using various acetone and deionized water ratios of 25:75, 50:50, 75:25, and 100% acetone. The digital images of the prepared samples are shown in Figure 3. The fibers loaded with silver salt and zinc salt were abbreviated as silver ions loaded composite fibers (SLCF) and zinc ions loaded composite fibers ZLCF, respectively. The digital images of (a) control sample (b) SLCF (c) ZLCF.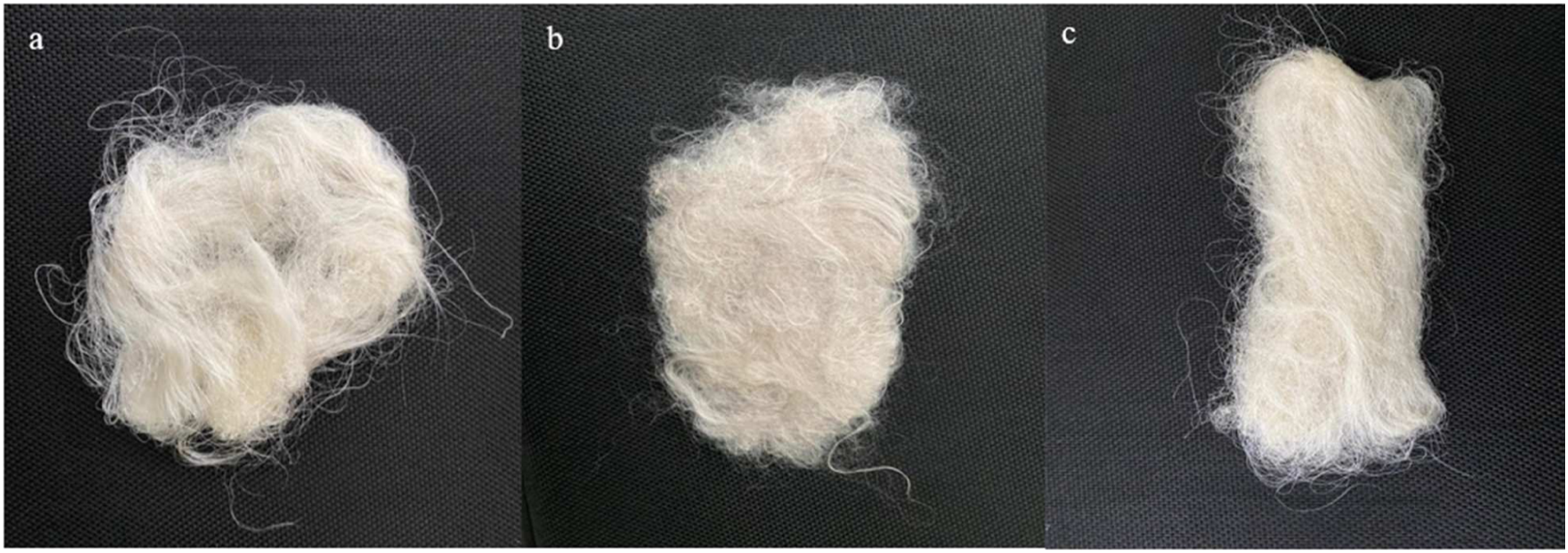
Testing and Characterization
Five specimens for each sample were analyzed, and mean values were plotted for graphs wherever possible. Fourier transform infrared (FTIR) spectroscopic analysis of developed samples were recorded using (Perkin Elmer-Spectrum two, USA) ZnSe-HATR Module, at a scanning range of 4000-600 cm−1. Fibers were placed in a desiccator to remove moisture before analysis. The de-moisturized samples were then placed on a ZnSe stone and scanned on a mode of attenuated total reflection. The surface morphologies and textural properties of developed fibers were analyzed using a scanning electron microscope (FEI Quanta 250, Czech Republic). Samples were mounted using a double-faced adhesive conductive tape on stub and coated with platinum for 30 s on an automatic sputter coater (Agar Scientific). The antimicrobial testing was performed by following the AATCC 147 standard. The strains of Escherichia coli (Gram-negative) and Staphylococcus aureus (Gram-positive) were incubated overnight to provide a suitable environment for their growth. Then, strains were inoculated on a sterilized agar plate after appropriate dilutions. Samples were placed at inoculated culture media. After incubation for 24 h at 37°C, plates were examined for interruption of microbial growth underneath the sample and inhibition zone along edges of the samples. The tenacity of developed fibers was measured by ASTM D 3822–07 using single fiber strength tester M250–2.5 CT TESTOMETRIC, Rochdale, England) with a gauge length of 20 mm, load cell of 10 N, and at 12 mm/min rate of extension.
Absorbency (g/g) of developed samples were analyzed using deionized water, saline solution, and solution A. Developed samples were first soaked in saline, solution A and deionized water for about 1 h and then weighed. Samples were dried in an oven at 105 oC for overnight and weighed. Absorbency (g/g) was calculated by using the equation 1
Swelling % was determined via a MICROS optical microscope (MC-50) and a digital camera. The change in diameter of the fibers after being soaked for about 15 min at 30°C was recorded at the 10 times magnification. Each sample with five repetitions was recorded in solution A, saline solution, and distilled water. Equation (2) was used to calculate swelling %
About 0.5 g of fibers were soaked in solution A. 1 mL of samples were taken after the interval of 0.5 h, 1, 6, 8, 24, 36, and 48 from solution A. Samples were diluted in double-distilled water. These samples were then analyzed using atomic absorption spectroscopy (AANALYST 400 Perkin Elmer, USA).
Results and Discussion
Liquid absorbency (g/g)
Liquid absorption from exudating wounds is a significant factor that influences wound healing. Figure 4 represents the results of alginate/hyaluronic acid (control sample), silver, and zinc-containing alginate/hyaluronic acid fibers liquid absorption properties. Silver and zinc-containing alginate/hyaluronic acid composite fibers have improved liquid absorption compared to pure alginate/hyaluronic acid fibers. Developed fibers have shown better absorption of saline solution due to the presence of excessive Na+ ions in the solution. Alginate is the significant component of the composite fiber and is known for its unique ion exchange properties; Superior saline solution absorption is credited to alginate ion exchangeability that has enhanced the excess of liquid molecules to hydroxyl and carboxylic groups of both alginate and hyaluronic acid. The fibers have shown less absorption of solution A. Solution A contains calcium ions that usually block alginate’s ion exchange process, suppressing the access of liquid molecules to carboxylic and hydroxyl groups of alginate and hyaluronic acid, resulting in less solution A absorption. Absorbency of test fibers (g/g) in solution A, Saline solution, and DD water.
Zinc-containing fibers have shown better liquid absorption properties compared to silver-containing fibers. Zinc helped the liquid molecules access liquid binding sites (i.e., hydroxyl and carboxylic groups) more quickly than silver-containing alginate/hyaluronic acid and control alginate/hyaluronic fibers.
27
Secondly, hyaluronic acid also has a positive contribution to the liquid absorption properties of the fibers due to its excellent hydrophilic and gelation properties.
28
The sodium and calcium ions exchange phenomenon of alginate has also been illustrated in Figure 5. Ion exchange of fibers with coagulant and body solution.
When sodium alginate fiber meets the calcium chloride solution, sodium ions exchange with calcium ions, converting the SA fibers into calcium alginate fibers (soluble to insoluble). 29 Human body liquid or wound exudate contains both sodium and calcium ions. When these fibers contact the wound surface, calcium alginate fibers form gel because calcium ions are usually replaced by sodium ions present in the body liquid or wound exudate (insoluble to soluble). 30
Swelling (gelation) properties
When the fibers come in contact with wound exudate, these fibers absorb the exudate and form a gel known as gelation. The higher the gelation capacity, the better the fibers are for the highly exudating wounds. Figure 6 represents the swelling properties of alginate/hyaluronic acid, zinc, and silver-containing alginate/hyaluronic acid fibers measured using three solutions, that is, solution A, saline, and double-distilled water. The developed fibers have exhibited less swelling properties in deionized water and solution A than saline solution. It was expected that the addition of hyaluronic acid would have improved the overall swelling properties of composite fiber. However, developed composite fibers have less swelled in solution A and double-distilled water. It has been earlier reported that calcium ions act as a barrier for ion exchange as calcium hyaluronate contributes to lowering hyaluronic acid’s solubility and gelation properties. Hyaluronic acid has carboxyl and hydroxyl groups in its structure, making it hydrophilic. The presence of these groups also contributes to the swelling of fibers. However, the presence of calcium ions may have suppressed these properties of hyaluronic acid present in the fiber resulting in minor swelling and liquid absorption.
28
Swelling % in solution A, Saline solution, and DD water.
Swelling % in saline solution is high as there is a maximum ion exchange of calcium ions with sodium ions. The fibers absorbed an excess amount of solution, resulting in gel formation.31,32 Hyaluronic acid enhances the swelling % in saline solution as it forms sodium hyaluronate, and sodium hyaluronate offers good ion exchange. 26 The results confirmed that zinc-containing fibers have higher swelling % than silver-containing alginate/hyaluronic acid. Zinc salt has better solubility than silver salt, which may have assisted in an ion exchange process, resulting in a better-swelling %.
Mechanical properties
Figure 7 represents the tenacity and elongation % of the control sample, zinc, and silver-containing alginate/hyaluronic acid fibers. The addition of silver and zinc has shown a profound effect on the tenacity of developed fibers, that is, 19.03 (cN/Tex) and 23.92 (cN/Tex), respectively, than alginate/hyaluronic acid fibers (11.59 cN/Tex). It is clear from the results that silver and zinc addition significantly improved the tenacity of alginate/hyaluronic acid fibers. The improvement in the tenacity of alginate/hyaluronic acid fibers due to the inclusion of zinc and silver is connected with its better crosslinking and orientation of macromolecules of the fiber-forming material and reduction in the amorphous region in fiber. Calcium, zinc, and silver ions generally react with the carboxyl group of alginate and hyaluronic acid and improve intermolecular forces between alginate and hyaluronic acid molecules resulting in improved tenacity.27,31 However, the elongation properties of the fibers were not much affected by the addition of zinc and silver to alginate/hyaluronic acid fiber. Mechanical properties of SLCF and ZLCF.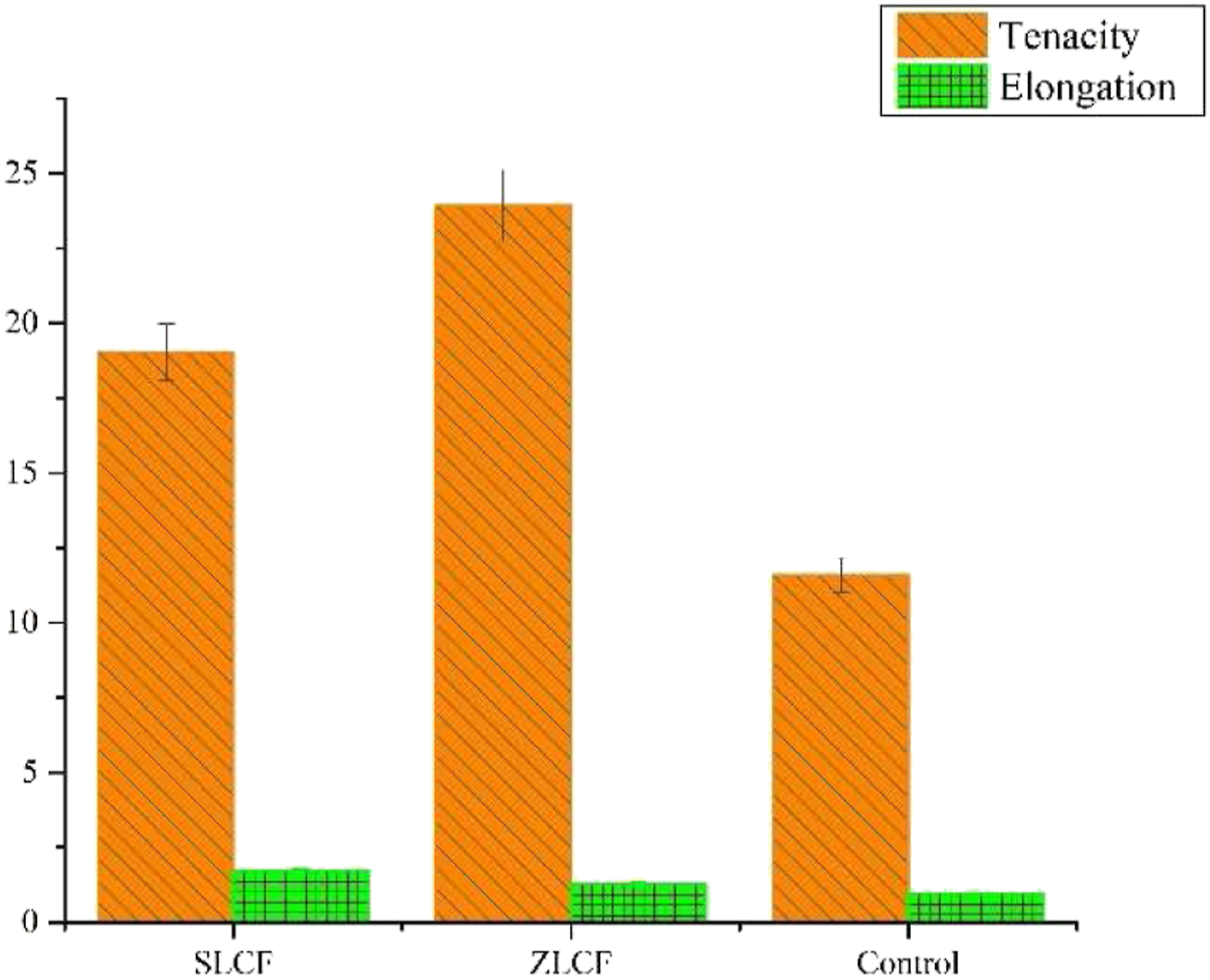
Fourier transform infrared Analysis
The FTIR spectrum of SA, HA, SLCF, and ZLCF are presented in Figure 8. Fourier transform infrared spectra of pure SA, HA, SLCF, and ZLCF in the mid-infrared region.
Spectra of Sodium alginate shows a band at 3182 cm−1 is ascribed to -OH of aliphatic chain, and the band 2876 cm−1 is attributed to asymmetric -CH stretching. Absorption bands at 1591 and 1407 cm−1 are assigned to the carboxyl group’s asymmetric and symmetric stretching vibration, respectively. Bands at 1089 and 1028 cm−1 are assigned to -CO stretching and -OH stretching vibrations.33–35 In IR spectra of Hyaluronic acid, the band at 3250 cm−1 is overlapped due to the combined stretching of -NH and -OH. The peak at 2889 cm−1 is attributed to asymmetric -CH stretching. The existence of a band at 1609 cm−1 confirms that the amide I. band at 1562 cm−1 corresponds to the presence of the amide II group. The bands at 1406 and 1148 cm−1 were assigned to aliphatic -CH stretching. A band at 1036 cm−1 showed the presence of the -OH group.36–39 In SLCF and ZLCF spectrum, peak at 713 and 716 cm−1 assigned to carbonate.40,41
Ion release
The release of ions from the developed fibers was checked using buffer solution at acidic pH. Figure 9 shows the relationship of ions release concerning their time dependence for both silver and zinc ions. Silver and zinc-containing alginate/hyaluronic acid fibers have shown controlled release up to 48 h. Zinc ions have shown quicker release than silver ions, generally attributed to their excellent solubility (pH-dependent), ion exchange properties, and lower density. Zinc salt’s solubility and ion exchange properties resulted in a better release from fiber’s complex composite structure than silver ions. However, this may result in reducing the prolonged effect of zinc-containing dressing in wound care applications. Drug release of SLCF and ZLCF.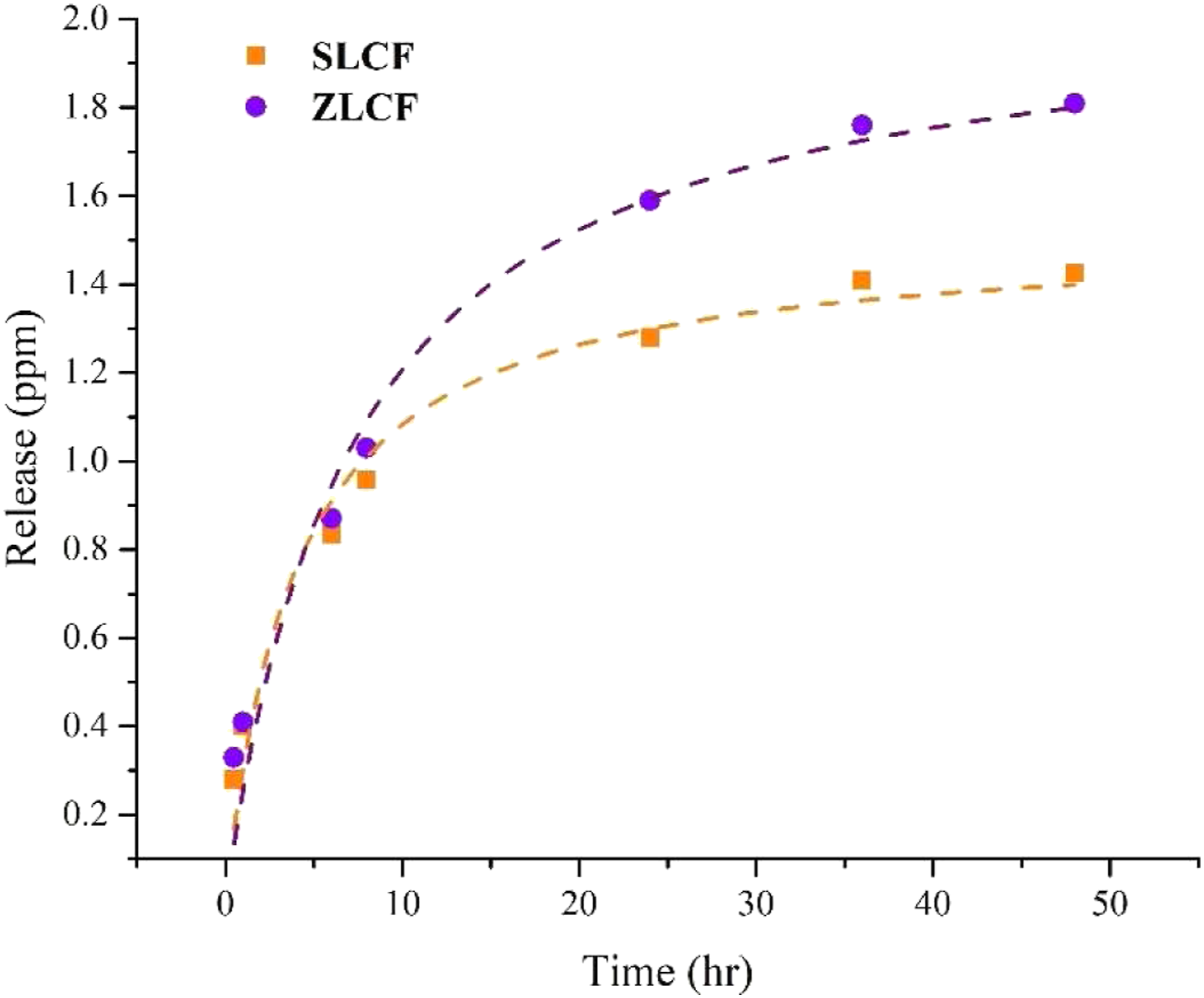
Antibacterial Activity
Figure 10 shows the antibacterial activity of zinc-containing alginate/hyaluronic acid fibers and silver-containing alginate/hyaluronic acid fibers. Both silver and zinc-containing fibers have shown antibacterial activity against Escherichia Coli and Staphylococcus Aureus by generating a clear zone of inhibition. Silver ions usually inhibit the respiratory chain and activity of DNA after reacting with the thiol group of bacteria.42,19,43 Silver ions also enter the bacterial structure and deposit negatively charged protein and nucleus, resulting in structural damage to bacteria’s cell wall and membrane.44,45 Antibacterial results (a) SLCF and ZLCF against E. coli (b) SLCF and ZLCF against S. aureus.
However, zinc ions encounter bacterial cell walls, causing cell integrity disturbance and generating reactive oxygen species that are bactericidal. 46 Zinc also penetrates bacteria’s cell walls and leads to death by zinc toxicity as an antimicrobial strategy. The bacterial cell wall has a negative charge and adheres with zinc through electrostatic forces, destroying bacterial cell walls and distorting cell activity.47,48
Scanning Electron Microscopy (SEM)
Scanning electron microscopy micrographs of alginate/hyaluronic acid fiber, silver, and zinc-containing alginate/hyaluronic have been shown in Figure 11. Scanning electron microscopy micrographs (
It is very clear from the micrographs that fibers have a rough surface with grooves of different depths and wrinkles resulting from excellent gelation properties of developed fibers, as marked in Figure 10. Excellent gelation properties of the SA polymer lead to the formation of grooves on the surface.49,50 It was observed that the fibers showed different morphology with the incorporation of zinc and silver ions.
Conclusion
Antibacterial composite biomaterials have been developed using environmentally friendly and non-toxic polysaccharides with excellent absorption (>25 g/g), gelling (>300%), tenacity (23.92 cN/Tex), and good antibacterial properties. The use of zinc and silver ions in the composite fiber is expected to be very useful in controlling the various wound infection. However, the release of zinc ions was pH-dependent. The use of hyaluronic acid will be capable of stimulating the granulating stage of wound healing. In addition to alginate fibers, zinc, silver, and hyaluronic acid could improve alginate fiber’s functionality in wound care applications. These composite biomaterials can help obtain a new generation of dressing materials for medical applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Higher Education Commission, Pakistan.