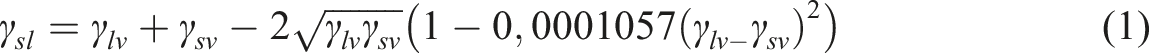Abstract
The study addresses changes in the surface energy of polycaprolactone nanofibre layers due to the different spinning electrodes. The value of surface energy and the associated wettability is an important aspect of polymeric biomaterials that influence the quality of interaction with biological material. The study involved the production of nanofibre layers by means of both needle and needleless electrospinning. The surface energy of the produced layers was determined via the contact angle measurement method. The experiment revealed that the spinning electrode exerts a significant effect on the resulting surface energy and, thus, on the wetting of the nanofibre layers. It was discovered during the measurement process, that the fibre layers produced via the needle electrospinning method evinced a low surface energy and are non-wettable/hydrophobic. In contrast, needleless electrospinning fibre layers evince a high surface energy and are wettable/hydrophilic.
Introduction
The surface energy and thus the wetting of nanofibre layers play an important role in tissue engineering, a multidisciplinary field that is concerned with the replacement, repair, restoration and regeneration of damaged tissue.1,2 The use of supporting materials, so-called scaffolds, comprises an important factor in all these processes via ensuring the adhesion and proliferation of cells throughout the material and, subsequently, the formation of new tissue. 3 The adhesion of cells to the material is affected by a wide range of factors including the wettability of the material. 4
Wetting comprises the ability of a liquid to spread over a solid surface. 5 It is affected by the surface tension of the liquid, the surface energy of the solid surface and the interfacial energy between the liquid and the solid. 6
In order to ensure the optimal degree of cell adhesion to the material, the material needs to be slightly hydrophilic. That is, a material upon which the applied liquid, for example, a nutrient medium, will form a contact angle of around 65°. 4 Obtaining a slightly hydrophilic surface requires the determination of a match between the surface tension of the liquid and the surface energy of the solid.
Surface tension is described as the effect of the surface of a liquid attempting to attain a state with the smallest possible area due to the surface tension comprising a force that acts upon the surface of the liquid. Intermolecular forces, such as Van der Waals, serve for the mutual attraction of individual atoms, that is, to the space of the liquid.7,8 A further factor that affects the wetting process comprises the surface energy of the solid, which is a similar case to the surface tension. Surface energy is a physical phenomenon caused by intermolecular interactions at the interface; numerically, it is equal to the surface tension, but is described in mJ/m2.
9
However, with respect to the surface energy, a higher value leads to a wettable surface, while a lower value leads to a non-wettable surface.
10
There are several ways to calculate the surface energy, one option is to use the Kwok-Neumann model (1),
11
which we used in this study
Currently, fibre scaffolds fabricated from polycaprolactone (PCL), whose chemical structure is shown in Figure 1, are used widely in the field of tissue engineering.
12
PCL is polyester with melting point 60°C and is prepared by ring-opening polymerization.
13
Chemical structure of PCL.
PCL, which is both a biocompatible and biodegradable material, is used, for example, for the regeneration of skin, bones, blood vessels and nerves.14,15 PCL scaffolds can be produced using several methods, including electrospinning, 16 forcespinning 17 and melt-blown technology. 18
This study focuses on electrostatic spinning using direct current voltage and divided into the needle and needleless electrospinning approaches. 19 A metal needle to which a high voltage is applied is used for needle electrospinning. 20 The needle electrospinning process involves the pull between capillary and electric forces. 21 Several types of electrodes are used in the needleless electrospinning process, for example, a smooth roller, a string roller, an inclined plate and even a string. 22 Needleless electrospinning results in the formation of Taylor cones via the self-organization of the solution. Nozzles are formed without the need for a capillary. 23 As demonstrated in this study, the electrode geometry in the electrospinning process exerts a major impact on the resulting surface energy of the nanofibre layer. Studies published to date suggest that polycaprolactone nanofibre layers are highly hydrophobic; thus, they have low surface energy properties.24–26 A further modification, for example, the use of plasma, is applied so as to modify the surface energy and change the degree of hydrophobicity. 27
The study demonstrated that the spinning process exerts a significant impact on the surface energy and, thus, on the wetting of the fibre layers. The study presented herein demonstrates that the surface energy is affected by the electrode used in the spinning process, thus establishing that it is possible to regulate the hydrophilicity/hydrophobicity of the resulting fibrous layers via the selection of the spinning technology and the spinning electrode.
Materials and methods
Materials: Polycaprolactone with a molecular weight of Mn 80,000 (Sigma-Aldrich, Germany) was used in the experiment with a solvent system consisting of acetic acid, formic acid and acetone (all Penta Chemicals, Czech Republic).
Preparation: The polymer solution was prepared at a concentration of 8wt% in the acetic acid, formic acid and acetone (ratio of 1:1:1) solvent system. The polymer solution was stirred on a magnetic stirrer at 300 r/min at room temperature. The solution was stirred for 24 h until the polymer was completely dissolved.
The fibre layers were prepared by means of both a needle and needleless spinner. A cut metal needle with a diameter of 1.2 mm was used as the needle spinner. The needle was attached to a 10 mL syringe, which was inserted into a dosing pump. A metal plate served as the collector. The NanospiderTM NS 1WS500U device with a string electrode was selected for the needleless electrospinning process. In both cases, spinning was performed on a Spunbond base fabric. In the case of the needle spinning approach, the dosage of the solution was set at 2 mL/hour. The electrical voltage applied to the needle tip was 15 kV, and the voltage applied to the collector−10 kV. The distance of the collector from the electrode was set at 15 cm. A NanospiderTM device with a string electrode was used for the needleless spinning method. A voltage of 50 kV was applied to the electrode and −10 kV to the collector and the distance of the collector from the electrode was set at 17 cm. In both cases, electrospinning was conducted at an ambient temperature of 22°C and a relative humidity of 45%. The electrospinning process was stable in both cases and exhibited no defects. Diagrams of the two devices are shown in Figure 2. Schemes of the electrospinning equipment. (a) Needle electrospinning, (b) needleless electrospinning.
Scanning electron microscopy and analysis: The nanofibre layers were gilded using Quorum Q150 R ES and the resulting gilded layers were scanned using a scanning electron microscope (SEM, Tescan Vega3) at an accelerated voltage of 20 kV. The images were evaluated by means of ImageJ software (NIH, Bethesda, USA).
Contact angle and surface energy of the fibre layers: The contact angle was measured using a See System E (Advex Instruments, Czech Republic) device. Glycerol with a surface tension of 64 mN/m was used as the measurement liquid. The Kwok-Neumann model was used to calculate the surface energy.
Fourier transform infrared spectroscopy: The Fourier transform infrared spectrometry (FTIR) was measured on the fibrous layers for the determination of their chemical composition using a Nicolet iZ10 instrument (Thermo Fisher, USA) and an ATR diamond crystal.
Differential scanning calorimetry: The layers were measured by means of differential scanning calorimetry so as to determine the differences in the crystallinity resulting from the two spinning approaches. The thermal properties of the polymer were determined using a Mettler Toledo DSC 3+. The testing temperature was set at 10°C per minute.
X-ray photoelectron spectroscopy: The chemical composition of the surfaces was determined by means of X-ray photoelectron spectroscopy (XPS) using an Omicron Nanotechnology ESCAProbe P Spectrometer (Omicron Nanotechnology GmbH, Germany). The detection angles of the secondary photoelectrons were set at 14° and 90° from the normal surface. The exposed and examined area had dimensions of 2 × 3 mm2. The spectra were measured applying a binding energy step of 0.05 eV. The evaluation of the spectra was performed in CasaXPS software.
Results and Discussion
Fibre morphology and analysis: Figure 3 provides a comparison of the resulting nanofibre layers. It is evident that both layers contain fibrous structures with bead defects. The fibre diameters, pore size and density of the resulting needle and needleless spun layers are shown in the Table 1. SEM images of the nanofibre layers. (a) Fibre layer produced via needle electrospinning, (b) fibre layer produced via needleless electrospinning. Scale 20 μm. Comparison of the fibre diameter, pore size and density of the samples prepared by needle and needleless electrospinning.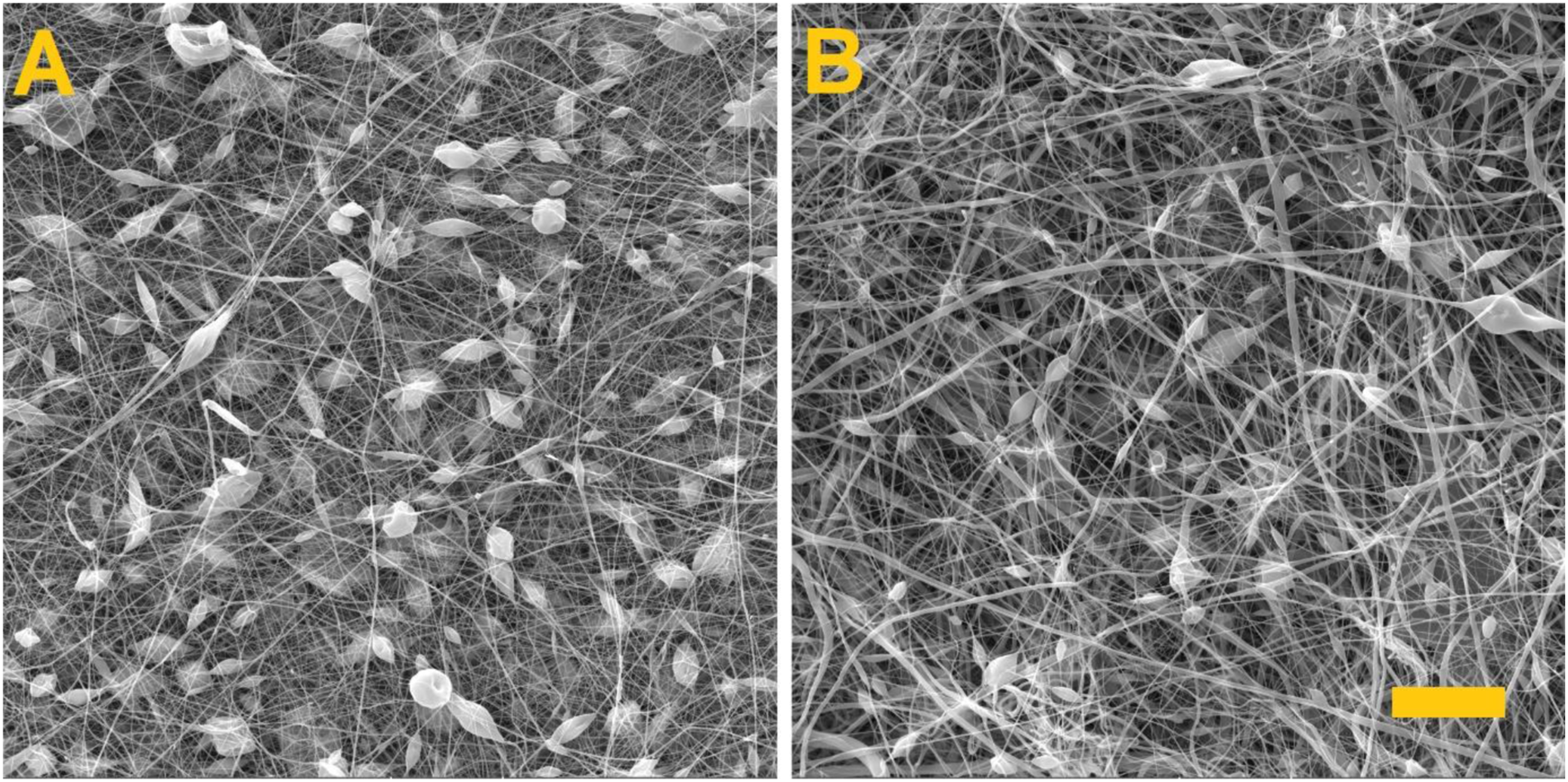

Contact angle and surface energy: The contact angle was measured so as to determine the surface energy of the resulting fibre layers. The graphs in Figure 4 show the values of the contact angles and surface energies of the produced layers. Measured values. (a) Contact angles, (b) surface energies of the produced fibre layers. 95% CI. **** p < .0001.
The results obtained illustrate that the fibre layer produced via the needle spinning method evinced a low surface energy value and was, therefore, non-wetting. Conversely, the needleless spun fibre layer evinced a high surface energy and was, therefore, wettable. As we have shown in our previous publications,28,29 wetting and surface energy values are not affected by the structure of the sample, that is, whether it is a purely fibrous material or a fibrous material with defects.
FTIR: The FTIR was measured in order to detect the differences in terms of the surface energy. Figure 5 shows the spectra of the fibre layers produced by the needle and needleless electrospinning approaches. Spectra of the fibre layers as indicated by the infrared spectrometry.
It is evident from the analysis of the spectra that the two layers had identical chemical compositions. Thus, clearly, one of the fibre layers was not modified during the spinning process.
DSC: The experiment also included a comparison of the crystallinity of the resulting layers. No significant changes in the crystallinity were determined due to the application of the differing spinning methods. Figure 6 illustrates the measurement curves. The fibre layer produced via the needle electrospinning method evinced a crystallinity of 58.43%, while the fibre layer from the needleless electrospinning method evinced a crystallinity of 61.54%. DSC measurement curves.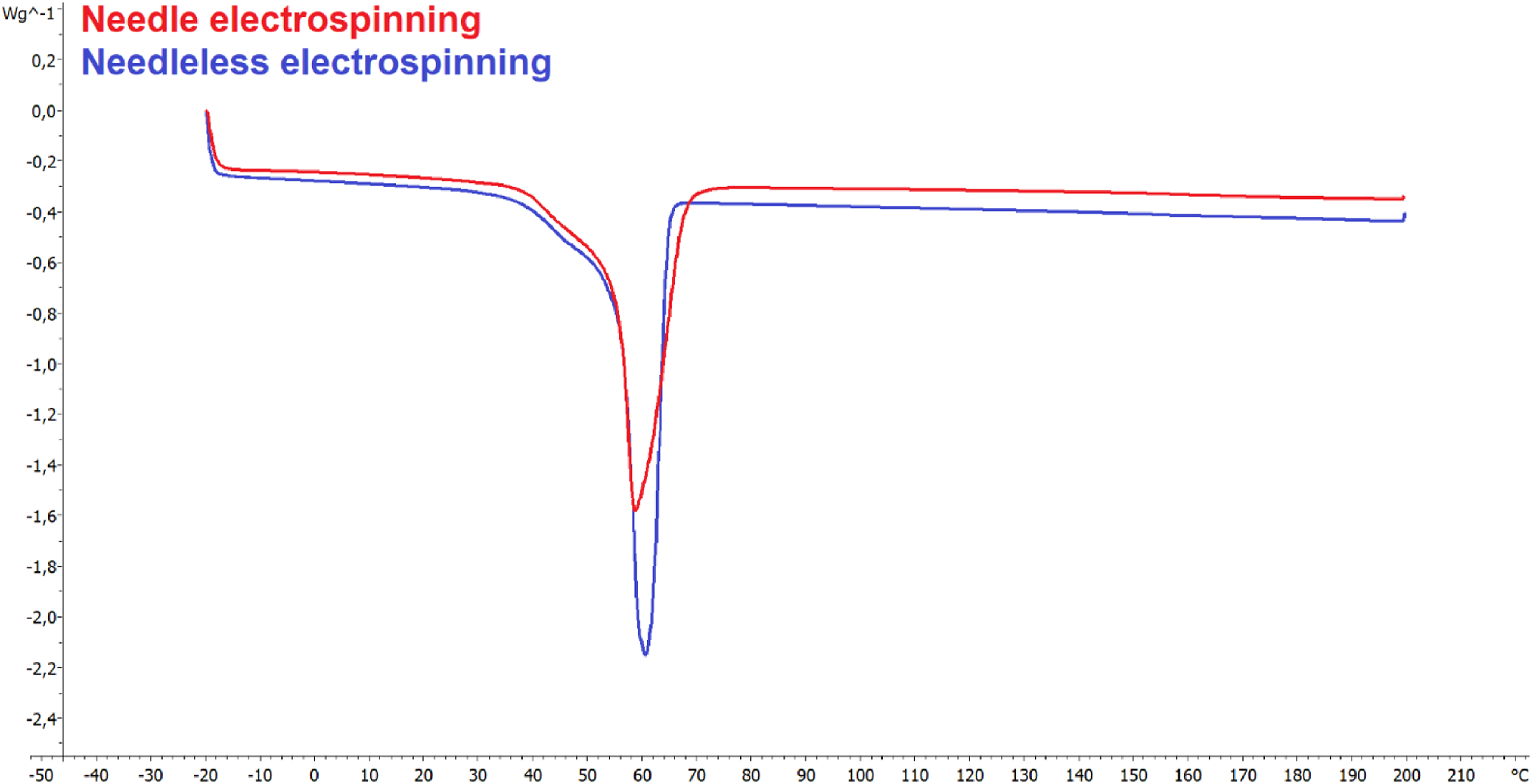
XPS: It is clear from the measured values that with respect to the use of the needle electrode, fibrous layers were produced with a low surface energy and were, therefore, non-wettable. XPS measurements were taken in order to determine whether fibrous layers with differing chemical compositions had formed on the surface of the fibres when using the needle electrode. Figure 7 shows the XPS measurement spectra at 90° and 14°. The XPS measurement spectra at 90°. (a) Needle electrospinning, (b) needleless electrospinning. And the XPS measurement spectra at 14°. (c) Needle electrospinning, (d) needleless electrospinning.
It is evident from the graphs that fibrous layers were produced with differing surface chemical compositions. The fibre layers fabricated via the needle electrode method contained more carbon and less oxygen on their surface than did the fibre layers fabricated via the string electrode approach. Hence, the results suggest that the electrode used in the electrospinning process affects the conformation of macromolecules in the resulting fibre layers.
The orientation of the polar groups of molecules and macromolecules in the electric field is a well-known phenomenon. For example, when measuring the electrical conductivity of polymers in a DC electric field, a time-dependent increase in the material’s electrical resistance occurs due to polarization. 30 The measurement of polymer solutions’ viscosity in an electric field shows the dependence of viscosity on the electric voltage caused by changes in intermolecular forces. 31 The process of orientation of the polar groups takes place over time, and the degree of orientation must therefore depend on the electrical voltage.
By these known contexts can be explained the surface energy and hydrophilic/hydrophobic behaviour of nanofibre layers prepared by electrospinning polymer solutions by needle or needle-free electrostatic spinning, respectively. The possible different orientation of macromolecules is shown in Figure 8. Possible orientation of macromolecules in the fibres from: (a) Needle electrospinning, (b) needleless electrospinning.
The surface energy of the fibres and thus the wettability is currently solved by modifying the surface of the fibres. 10 In the study, we showed that even by changing the orientation of macromolecules in polymers with a polar group, the surface energy of the fibres can be adjusted. If the polar group is ‘hidden’, the liquid will have access only to the non-polar part of the macromolecule and the fibres will be non-wetting. Conversely, if the polar group ‘protrudes’, the liquid will have access to it and the fibres will be wettable.
Conclusion
The experiment involved the fabrication of polycaprolactone nanofibre layers using two different types of electrodes. It was demonstrated that while the layers evinced similar fibre diameter and crystallinity values, the use of the two differing electrodes led to significant changes in terms of the surface energy. The needle electrode was found to produce fibrous layers with a low surface energy value of 7.60 mJ/m2, while, in contrast, the string electrode produced layers with a high surface energy value, that is, 40.71 mJ/m2. The change in the surface energy was due to the differing conformation of macromolecules, as demonstrated by the XPS measurements. The hydrophobicity of polycaprolactone is the result of its carbon chain, while the hydrophilicity was caused by the oxygen in the ester group. The XPS measurements demonstrated that the needle electrospinning process results in the formation of a structure that contains oriented polymer chains with an enhanced content of oxygen in the fibre volume. Conversely, the needleless electrospinning process results in a structure in which the oxygen atoms are oriented on the surface of the fibre. Hence, a hydrophilic fibre layer is produced via the needle electrospinning process and a hydrophilic fibre layer from the needleless electrospinning method. The results of this study led to the conclusion that it is possible to control the wettability of polycaprolactone nanofibre layers via the adjustment of the electrospinning parameters. These findings give us a potent tool for targeted surface modifications in tissue engineering.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Education, Youth and Sports of the Czech Republic, the European Union via European Structural and Investment Funds as part of the Research, Development and Education Operational Programme – Hybrid Materials for Hierarchical Structures project (HyHi, reg. no. CZ.02.1.01/0.0/0.0/16_019/0000843) and a Specific University Research Grant project (reg. no. SGS-2021-6041) awarded by the Ministry of Education, Youth and Sports of the Czech Republic in 2021.