Abstract
The filter paper of automobile engine air filter is easy to temper and causes combustion. Flame retardant is usually added to the reinforced resin styrene-acrylic emulsion (SAE) to improve the flame-retardant performance of the air filter paper. Ammonium polyphosphate (APP) was used as material in this research, and multi-layer coated APP was prepared by microcapsule coating. A layer of nano-scale SiO2 was coated and then introduced organosiloxane structure of the allyl polyoxypropylene ether side chain and boron. The chemical groups and structural composition were determined by Fourier transform infrared spectroscopy. TGA characterized the modified particles' high-temperature stability. By scanning electron microscopy and TEM, the morphologies of modified particles were analyzed to reveal the evolution of modified APP in SAE. Finally, nanoparticles with core-shell structures were formed. Applying the blended resin to the air filter paper, the flame-retardant performance was greatly improved. Total heat release (THR), heat release rate (HRR), peak heat release rate (pkHRR), mass loss rate (MLR), and effective heat of combustion (EHC) were reduced, respectively, by 30.8%, 30.7%, 42.0%, 16.7%, and 14.4%. The time to ignition (TTI) was doubled, so the fire spread could be effectively suppressed. Meanwhile, nano latex particles were attached to the interwoven fibers points and formed a “sea-island structure,” which significantly improved the paper’s mechanical properties, with bursting strength, tensile strength, stiffness, fracture work, and elongation increased by 18.60%, 5.72%, 10.64%, 17.00%, and 81.80%, respectively. It solved the problem that the inorganic flame-retardant particles often lead to the deterioration of the mechanical properties of paper.
Keywords
Introduction
Automobile air filter paper is the core component of automobile engine air filter.1–5 In order to achieve the “dual carbon” goal and planning, styrene-acrylic emulsion is always used as a reinforcing resin for air filter paper. Due to the backflow of hot engine air, high temperature of the cylinder, and other reasons, it is easy to cause the safety hazards of the burning of the filter paper, which poses a considerable threat to the safety of life and property. Improving the flame retardancy of the air filter paper and the flame-retardant modification of the styrene-acrylic emulsion are the key to ensure the safe use of automobiles.6–11 At present, companies such as Japan’s AWA, Germany’s Gessner, and South Korea’s Osloen have launched air filter papers with excellent flame-retardant properties and better mechanical properties.12–14 There are also a few domestic manufacturers that have produced corresponding products, but the overall performance of their filter paper is often not ideal. The scarcity of flame-retardant air filter paper with excellent performance in the domestic market has caused most filter companies to rely on imports to meet customer needs.15–17 To break the foreign monopoly in the field of flame-retardant air filter paper and improve the competitiveness of this industry in my country, the research and development of highly effective flame-retardant air filter paper with better mechanical properties have become an urgent issue to be solved in China.18–21
Ammonium polyphosphate (APP) is a high-efficiency flame retardant with P and N flame-retardant elements simultaneously, which has the advantages of low price, low smoke, non-toxic, halogen-free, and so on. It is widely used in flame-retardant paper, fire-retardant coatings, fire extinguishing agents,22–27 and other fields. However, APP also has some shortcomings, such as moisture absorption, poor polymer material compatibility, and particle segregation after long-term use. At the same time, improving the flame-retardant properties of flame-retardant paper will also degrade the mechanical properties of the paper.28–33
In this paper, the method of microcapsule coating was adopted.34–36 First, the surface of APP was coated with SiO2 NPs by the sol–gel method, and the surface of the particles was reacted in-situ with the addition of hydrosilylation, and organosiloxane structure with polyoxypropylene ether side chain was introduced. The structure, on the one hand, acts as a compatibilizer, improving the interface compatibility of inorganic particles and organic resins, and improving the disadvantage of poor compatibility with polymer materials. On the other hand, the organosiloxane structure has a certain degree of water resistance, which further improves the shortcomings of moisture absorption. On this basis, the lipophilic and hydrophilic properties of inorganic particles improved and adjusted through the complexation of B element to polyhydroxy compounds. Multilayer-coated modified APP was prepared to improve its flame-retardant properties. Finally, it was blended and compounded with styrene-acrylic emulsion. Through the interaction of the active groups between the modified particles and the organic resin, and the interpenetration, diffusion, and entanglement between the molecular chains, it evolved into the nanoparticles with core-shell structure, which was conducive to improving the mechanical properties, heat resistance, and flame retardancy of the air filter paper. At the same time, there will be no particle precipitation during use.
Materials and methods
Materials
Ammonium polyphosphate (APP) was purchased from Shanghai McLean Biochemical Technology Co., LTD. Ethyl orthosilicate, ethanol, ammonia, and toluene were purchased from Guangzhou chemical reagent factory. Polymethylhydrosiloxane (PHMS) (hydrogen content 1.0, viscosity 80–90 mm2/s) was purchased from Yangzhou Chenhua New Material Co., LTD. Chloroplatinic acid isopropyl alcohol solution with a solid content of 2% was self-made. Allyl polyoxypropylene ether (Mw= 650 g/mol) (PBO650) was purchased from Yangzhou Chenhua New Material Co., LTD. Boric acid was purchased from Tianjin Damao Chemical Reagent Factory. Styrene-acrylic emulsion with a solids content of 40% was self-made. Air filter paper (Basic weight 90 g/m2) was provided by Guangzhou Huachuang Chemical Material Technology Co., LTD.
Preparation of modified ammonium polyphosphate
30 g APP and 90 mL ethanol solution were added into the three-necked flask equipped with a mechanical stirrer, reflux condenser, and thermometer, and the temperature was raised to 50°C. Then 12 g tetraethyl orthosilicate, 120 mL ethanol, 9 mL ammonia, 90 mL ethanol, and 60 mL deionized water were added into the three-necked flask. After reacting for 2 h, removing the solvent to obtain 31 g white powder, which was recorded as APP-1.
10 g APP-1, 3 g PBO650, 20 g toluene, 3 g PHMS, and 1–2 drops of chloroplatinic acid isopropanol solution (catalyst) were added into the three-necked flask andnd reacted at 85°C for 1.5 h. Removing the solvent to obtain 10.4 g white powder, which was recorded as APP-2.
10 g APP-2 and 20 g toluene were added to the three-necked flask and increased the temperature to 80°C. Then 1 g boric acid was added to three-necked flask. After reacting for 1 h, removing the solvent to obtain 10.5 g white powder, which was recorded as APP-3. The preparation process and equation of particles are shown in Figure 1. The particle preparation process and hydrosilylation reaction equation.
Preparation of reinforced resin
The two particles of APP and APP-3 were blended with the styrene-acrylic emulsion at a mass ratio of 15:20 to prepare a blended composite reinforced resin, which was recorded as SAE/APP and SAE/APP-3, respectively.
Preparation of flame-retardant air filter paper
The base paper was immersed in the reinforced resin solution for 3 min and then taken out. The resin sizing amount was controlled to (30 ± 0.5) % (relative to the quality of the base paper). The resin-impregnated air filter paper was dried in an oven at 105°C for 30 min. And placed at standard temperature (23°C constant temperature and 50% RH constant humidity) for 24 h to prepare flame-retardant air filter paper. Corresponded to SAE/APP and SAE/APP-3, which were, respectively, recorded as flame-retardant paper FRP-1 and FRP-2. For comparison, the SAE impregnated air filter paper prepared by the above method was recorded as non–flame-retardant paper NO-FRP.
Measurements
The structure of a series of polymer systems was investigated by Fourier transform infrared spectroscopy (FTIR) (Bruker VERTEX 70). The measurements were conducted in the range of 400–4000 cm-1 with 4 cm-1 resolution. The particle size distribution of the four types of APP was measured by laser particle size analyzer (HORIBA, LA-960S, Japan). The contact angle measuring instrument (Dataphysics, OCA40 Micro, Germany) was used to test the surface contact angle through profile image analysis to characterize the hydrophobic strength of the sample. Differential scanning calorimetry analyzer (DSC 214 Polymer, Nietzsche, Germany) was used to characterize the crystallization behavior of the sample. The thermogravimetric analyses of the sample were characterized by a thermogravimetric analyzer (TGA) (TA Q750, America). The morphology of the samples was observed with scanning electron microscopy (SEM) (HITACHI, Japan) at 10 kV. All samples were sputtered with gold and compared the differences between the micromorphology of unmodified particles. On the other hand, the micromorphology of the samples after combustion was observed to explore its flame-retardant mechanism. The JEM-2010F transmission electron microscope (TEM, JEOL Co., Ltd.) was selected to study the relationship between material structure and performance. The prepared air filter paper was cut into five squares of 10 cm×10 cm, and conical calorimeter (FTT0007, Fire Testing Technology Company,UK) was used for conical calorimeter combustion test according to ISO related standards (ISO5660-1). Refer to the relevant ISO standards (ISO2758, ISO5635) to measure the bursting strength, tensile strength, bending stiffness, tearing, and elongation at break of air filter paper. The average pore size of air filter paper was tested using the capillary flow Aperture Tester (CFP-1100-A, PMI, USA). The initial filtration efficiency of the air filter paper was tested using the automatic filter material tester (8130, TSI, USA).
Results and discussion
Fourier transform infrared spectroscopy analysis
Figure 2 shows the FTIR spectra of APP, APP-1, APP-2, and APP-3. The characteristic infrared absorption of APP includes the characteristic absorption peak of -NH3+ at 3050.3 cm-1 and 2889.3 cm-1, and the N–H stretching vibration peak at 3185.3 cm-1, the P=O stretching vibration absorption peak at 1247.9 cm-1, and P–O symmetric and antisymmetric stretching vibration absorption peak at 1071.4 cm-1 and 877.6 cm-1. In addition to the characteristic absorption peak of APP, APP-1 also has Si–O–Si antisymmetric stretching vibration peak at 1087.8 cm-1, antisymmetric stretching vibration peak and bending vibration peak of surface-bound water -OH at 3220.1 cm-1 and 1709.8 cm-1, respectively. At the same time, the stretching vibration peak of Si–OH overlaps with the N–H stretching vibration peak of APP, which makes the deformation of the whole peak wide and dull, and the peak moves to a higher peak direction, indicating that SiO2 is successfully coated on APP. The infrared spectrum of APP-2 showed characteristic peaks of -CH3 and -CH2 at 2883.4 cm-1 and 2977.9 cm-1, characteristic peak of Si-C at 796.5 cm-1, and the absorption peak of Si-H bond near 2150 cm-1 is significantly weakened. The characteristic peak of the C=C bond near 1645 cm-1 disappears, indicating that the Si–H bond and C=C bond have undergone silohydrogen addition reaction, and the polyether side chain has been successfully grafted to the siloxane main chain. At the same time, Si-OH on the SiO2 surface interacts with -OH of the modified material or residual Si–H, which causes the characteristic peak of -OH at 3300.1 cm-1 to move to 3226.8 cm-1. On the other hand, the B element coated on the surface of APP-3 has a complexation with -OH, making the -OH peak narrow and moving to low wavenumber. Meanwhile, the coordination between B–O bond and -OH can form a ring, the infrared spectrum of APP-3 showed B-OH bending vibration peak at 1192.0 cm-1, B–O stretching vibration peak at 1407.5 cm-1 and O–B–O out-of-plane bending vibration peak at 554.5 cm-1, indicating the successful introduction of boron element on the surface. Infrared spectra of modified APP.
Particle size and its distribution
The median diameter of APP is 15.54 μm, and the median diameter of APP-1 coated with a layer of nano-SiO2 is 15.38 μm. The particle sizes of the two are relatively close. The median diameter of APP-2 increased to 17.55 μm. This is due to the hydrosilylation reaction between PHMS and PBO650, forming an organosiloxane structure coating with polyether side chains with a thickness of 1–2 μm on the surface of APP. With the addition of the B element, coordination and complexation between B and polyhydroxy groups occurred, resulting in the reduction of the median diameter of APP-3–17.41 μm. Further study found that the diameter, average particle size, and maximum frequency particle size of the modified APP particles coated by microcapsules are close. The distribution curve is close to the normal distribution. Indicating that the modified particles have good dispersion performance, which is beneficial to the compatibility of the particles with styrene-acrylic emulsion. The particle size distribution of various APP and reinforced resin is shown in Figure 3. Particle size distribution of various APP and reinforced resin.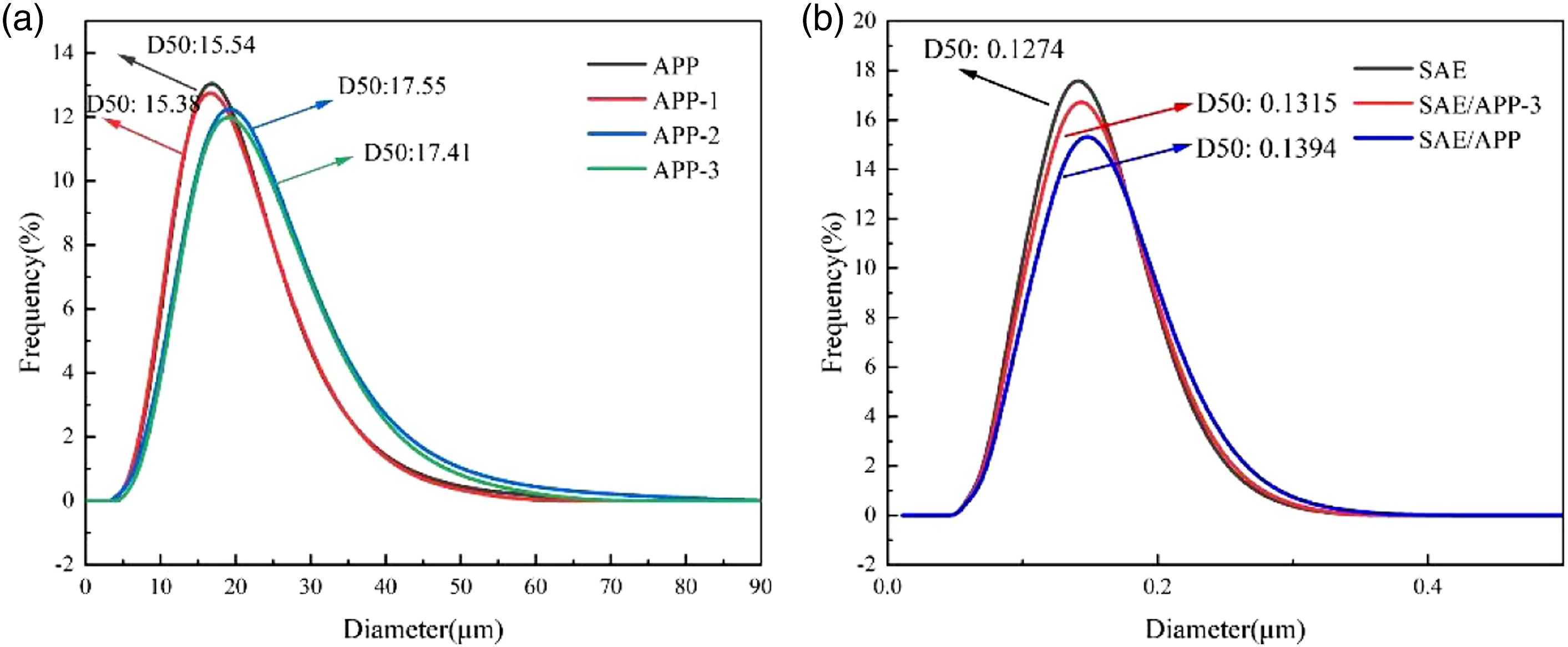
The median diameter of the latex particles of styrene-acrylic emulsion is 127.4 nm. When APP-3 is mixed with styrene-acrylic emulsion under the action of high-speed shearing, APP-3 particles will rearrange and distribute in the latex particles of styrene-acrylic emulsion, forming a particle size similar to that of styrene-acrylic emulsion latex particles, and the distribution curve is a normal distribution. Compared with APP-3, the particle size of the compound particles formed by diffusion into the emulsion is reduced to 131.5 nm, indicating that there is an interaction between APP-3 and the styrene-acrylic emulsion latex particles, and new latex particles are formed after blending. Meanwhile, we also found that APP and APP-3 have a better blending effect with styrene-acrylic emulsion. Compared with APP, APP-3 introduced organosilicon structure and flame-retardant elements, which could further improve its flame-retardant performance after blending with styrene-acrylic emulsion.
Differential scanning calorimetry analysis
Figure 4 shows the DSC curves of SAE and reinforced resin. From the figure, we find that the pure styrene-acrylic emulsion is amorphous, and at the same time, a glass transition occurs at 60°C–70°C. During the heating process, SAE/APP and SAE/APP-3 showed exothermic peaks near 178°C and 164°C, respectively, which means that APP and the molecular chain of styrene-acrylic emulsion are entangled, including physical and chemical effects, to form crystals. A lot of heat is released, and this interaction also makes the molecular chains denser and more orderly. This stable structure will greatly improve its mechanical properties. DSC curves of reinforced resin.
On the other hand, SAE/APP-3 has exothermic behavior at a higher temperature than SAE/APP, indicating that APP, after microencapsulation, is combined with styrene-acrylic emulsion to form a more compact and orderly crystal, which not only improves its mechanical properties, but also further improves its high temperature resistance.
TG analysis
The TGA curves of various APP are shown in Figure 5. All kinds of APP’s initial thermal decomposition temperature is between 320°C and 350°C, but the thermal degradation mode is different. APP and APP-1 are decomposed in two-stage. The first stage is the thermal decomposition of APP to produce NH3, H2O, and other non-combustible gases, forming crosslinked pyrophosphate. In the second stage, near 650°C, pyrophosphoric acid is decomposed to form phosphorus oxide. With the coating of nano-SiO2 and the organosiloxane with polyether side chain produced by the in-situ reaction, the thermal decomposition initiation temperature and maximum thermal weight loss gradually decrease, and the carbon residue rate at 800°C increases from 21.73% to 53.03%. Because the coating of SiO2 and silicone oil can increase the resistance of the chain end movement, resulting in a significant decrease in the thermal decomposition rate and improving the thermal stability of the material. After introducing the B element, the maximum decomposition rate of APP-3 is lower than 6%/min, and the maximum mass loss rate is reduced from 60.71% of APP to 23.19%. This is due to the formation of coordination bonds between P and B and the synergistic effect of B and Si, which makes the carbon layer formed during the combustion process more stable at high temperatures and dramatically improves its flame retardancy. TGA curves of various APP.
scanning electron microscopy analysis of particles
The SEM images of APP and APP-3 particles are shown in Figure 6. We can find that APP is a smooth and irregular cube with an average particle size of 16.85 μm. After the synergistic modification by B and Si, many mastoid particles are attached to the surface of APP-3, which constitute micro-nano structures and increase their surface roughness. (a) SEM images of raw material APP; (b) SEM images of APP-3.
Electron microscopic analysis of reinforced resin and air filter pater
We can see from the transmission electron microscope image of Figure 7 that (a) styrene-acrylic emulsion and TEM image (a) SAE; (b) SAE/APP; (c) SAE/APP-3.
After drying, blending resin in the process of water removal, the latex particles collapse each other and gradually form a continuous film. The scanning electron microscope (SEM) of the blended resin is shown in Figure 8. The organosiloxane with polyether side chain introduced on the surface of APP-3 acts as a compatibilizer and coupling agent. It is close to the styrene-acrylic emulsion during the drying process and entangles with the molecular chain of the styrene-acrylic emulsion. The result is finally embedded in the styrene-acrylic emulsion to form the “island” in the “sea-island structure.” According to the analysis of nano measurer software, the average diameter of “island” is about 300 nm. (a) Scanning electron micrograph of cross section of reinforced resin SAE/APP-3; (b) Nano Measurer software analysis diagram.
Figure 9 shows the SEM images of raw paper and flame-retardant paper FRP-2. The blended resin is used as a reinforcing resin for the paper, forming a “membrane” at the interweaving points between the fibers to improve the mechanical properties of the paper. On the one hand, the “sea-island structure” of the blended resin improves the interface compatibility and blending stability between the inorganic particles and the organic resin. On the other hand, it has excellent advantages in enhancing and toughening mechanical properties, which can significantly improve the mechanical properties of the blended resin. At the same time, we can see from Figure 9 that the resin only exists in the fiber and crosslinking between fibers, and does not block the gap formed between fibers, so it does not affect the average aperture and filtration efficiency of the filter paper to a certain extent. The evolution law and formation process of reinforced resin are shown in Figure 10. (a) SEM images of raw paper; (b) SEM images of flame-retardant paper FRP-2. Blending mechanism of reinforced resin SAE/APP-3.

Flame-retardant performance analysis of air filter paper
The cone calorimeter is an instrument designed based on the principle of oxygen consumption to measure the combustion performance of materials. Because its test environment is consistent with the real combustion environment of the fire material, the test results can genuinely evaluate the burning behavior of materials in the fire.
Combustion parameters of air filter paper.

The reason why the air filter paper has excellent flame retardancy is the addition of APP-3. APP particles are modified by Si and B synergistically. After combustion, a dense carbon layer structure is formed on the surface, and the volume expands, showing a loose and porous shape. Two flame-retardant mechanisms mainly cause the reason for this phenomenon. The first is the condensed phase flame-retardant. After introducing B and Si elements, B will catalyze the formation of a carbon layer during combustion. The dense carbon layer isolates the pyrolysis process. The heat generated prevents heat transfer, and Si is used to increase the stability and strength of the carbon layer to achieve the purpose of flame retardancy. The second is gas-phase flame retardant. APP rapidly releases incombustible gases such as NH3 and H2O through combustion, diluting oxygen concentration, removing heat, forming a fluffy layer, and reducing energy loading between the substrate and the heat source to achieve a synergistic flame-retardant effect. Scanning electron microscopy (SEM) of carbon residue after modification of APP-3 at 800°C is shown in Figure 11. SEM of carbon residue after modification of APP-3 at 800°C.
Analysis of mechanical properties and filtration efficiency of air filter paper
Paper is a network structure composed of fibers entangled and interwoven. The bonding force between fibers depends on the action of hydrogen bonds. However, the bonding force is weak. Therefore, the prepared air filter paper must be impregnated with reinforcing resin to meet the processing and use requirements. As the modified APP coated by nano-SiO2 and siloxane molecular chain with polyether structure improves the interface compatibility with the resin. The formation of the “sea-island structure” can produce a synergistic toughening and strengthening effect so that the impregnated and reinforced paper-based material has excellent mechanical properties.
Compared with air filter paper NO-FRP, the bursting strength of air filter paper FRP-1 increased by 8.36%, tensile strength increased by 2.99%, stiffness increased by 4.26%, breaking work increased by 3.96%, and elongation increased by 4.70%. The air filter paper FRP-2 prepared by modified APP-3 particles increased its bursting strength by 18.60%, tensile strength increased by 5.72%, stiffness increased by 10.64%, breaking work increased by 17.00%, and elongation increased by 81.80%.
Mechanical properties and filtration efficiency of air filter paper.

Conclusion
In this work, we adopted the way of microencapsulation coating. First, nano-SiO2 was coated on the surface of APP by the sol-gel method. Then organosiloxane structure with polyether side chain was introduced through in-situ polymerization and hydrosilylation reaction, which played the role of compatibilizer and made inorganic particles have good interface compatibility with styrene emulsion. The problem of poor blending stability between inorganic particles and organic resin was solved.
Through the coordination and complexation reaction between the B element and the polyhydroxy compound, the flame-retardant particles of B, Si, N, and P synergistically flame retardant were successfully prepared, which were used to improve the flame-retardant properties of resins and paper sheets. At the same time, the flame-retardant performance of the prepared air filter paper has been greatly improved, the total heat release (THR) has been reduced by 30.8%, and the heat release rate (HRR) and peak heat release rate (pkHRR) have been reduced by 30.7% and 42.0%, respectively. The loss rate (MLR) was decreased by 16.7%, the effective heat of combustion (EHC) was reduced by 14.4%, and the ignition time (TTI) was doubled, thus effectively suppressing the spread of fire.
The reinforced resin SAE/APP-3 was composed of nano latex particles with a core-shell structure, which was applied to the reinforcement of air filter paper. The latex particles were dried at the fiber weaving point to form a “sea-island structure,” which significantly improved the mechanical properties of the blended resin and air filter paper. Compared with NO-FRP, its bursting strength increased by 18.60%, tensile strength increased by 5.72%, stiffness increased by 10.64%, breaking work increased by 17.00%, and elongation increased by 81.80%.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Program of Marine Economy Development Special Fund (Six Marine Industries) under Department of Natural Resources of Guangdong Province, (Project No. GDNRC [2021]33).
