Abstract
A water-soluble epoxy resin emulsion was synthesized by diethanolamine-assisted succinimide modified epoxy resin (DSEP) and used to reinforce the strand integrity of polyimide filament (PI). FTIR, XPS, and 1H NMR provide an evidence for the succinimide (SI) and diethanolamine (DEA) bonded onto the epoxy resin (EP) structure in the form of C-N-C. The DSEP emulsion shows high storage and dilution stability, with its particle size distribution and PDI of 118∼232 nm and 0.106∼0.638, respectively. Compared with DEA modified EP, DSEP exhibits better strand integrity for PI filament. The breaking strength of PI filament infiltrated by DSEP can reach 2.59 GPa, which is increased by 47.04% than that of PI filament, and is close to that of commercially available water-soluble polyimide resin (2.63 GPa). In addition, the fracture microstructure of PI filament further confirms that DSEP significantly reinforces the aggregation of PI filament. Importantly, there is no wire splitting phenomenon of DSEP reinforced PI filament after more than 200 times of friction. These benefit from the similar material groups of imide ring and benzene ring between DSEP and PI filament structure, as well as the strong hydrogen bonding interaction between them, as further confirmed by FTIR and SEM analysis.
Introduction
High-performance fibers such as aramid fiber [1,2], carbon fiber [3,4], basalt fiber [5,6], glass fiber [7,8], and polyimide [9–15] have broad application prospects in the military, transportation, construction, electronic, aerospace, and electrical fields, due to their high strength, high modulus, and high-temperature resistance. Polyimide (PI) filament is a species of high-performance fiber with the imide ring structure in the molecular chain. Due to its excellent thermal stability, radiation resistance, and exceptional dielectric properties, PI filament is extensive used in the field of high-temperature resistance, fire protection, and composite materials [16–22].
However, due to the limitation of processing methods, PI filament usually shows poor strand integrity, which will produce a lot of static electricity and filament separation in the weaving process [23,24]. In recent years, researchers mainly use sizing agent to reinforce the strand integrity between mono-filaments. The structure of typical starch, polyvinyl alcohol, and polyacrylic acid adhesives in the textile size field is totally different from that of PI filament, so they cannot use as sizing agents to improve the binding ability of mono-filaments [25]. Although water-soluble polyimide resin (WPI) has appeared [26], its high price limits the extensive development as a sizing agent for PI filament. Therefore, it is urgent to develop a low-cost sizing agent to reinforce the bundling property of PI filament.
Epoxy resin (EP) is a kind of solvent dispersion with low cost, high yield, and easy availability. The structure of EP contains the same benzene ring structure as that of PI filament, which provides an advantage for enhancing the bundling property of PI filament. However, EP has high viscosity and insoluble in water, and it cannot be used as the PI filament sizing agent alone. Generally, the waterborne epoxy resin (WEP) was prepared by the mechanical, reverse rotation, emulsification, and chemical modification methods, which promoted the developments of WEP in the fields of high-performance fiber [27–30] and composites [31,32]. The chemical modification method can introduce the desired imide ring into the EP structure because the active group epoxy group in the EP structure provides the reaction site for chemical modification [33–37]. It provides an advantage for the preparation of WEP that can reinforce the bundling property of PI filament.
Diethanolamine (DEA) is a small molecule containing polar hydroxyl and has an excellent hydrophilic ability, and the waterborne epoxy resin emulsion can be realized by bonding the DEA onto the EP structure. We found that the strand integrity of single DEA modified EP dispersion on PI filament was not obvious. Based on the above finding, we tried to introduce succinimide (SI) molecule on EP structure, which has the same imide ring group with PI filament. It is expected to obtain a WEP emulsion that can significantly reinforce the strand integrity of PI filament through the synergistic modification of EP with DEA-assisted SI.
In this work, we will use DEA-assisted SI to modify epoxy resin to obtain a stable water-soluble epoxy resin (DSEP) emulsion and further reinforce the strand integrity of PI filament. The chemical structure, particle size, stability, and thermal property of DSEP were studied by FTIR, XPS, 1H-NMR, DLS, and DSC, respectively. The microstructure, mechanical properties, and their interaction of WEP reinforced PI filament were investigated by SEM, stress-strain and FTIR, respectively, as well as the comparative study of the strand integrity of PI filament reinforced by commercial WPI and DSEP was carried out.
Experiment
Materials
The polyimide (PI) filament was offered by Bangwei special fabric Co, Ltd, with length density and diameter of 55 D and 73.23 μm, respectively. Epoxy resin (EP, E-51, epoxy value: 0.48–0.54), with iviscosity of 10–18 Pa·s, was obtained from Nantong Xingchen Reagent Co, Ltd Succinimide (SI) was supplied by Shanghai Macklin Biochemical Co, Ltd Diethanolamine (DEA), acetic acid, and sodium hydroxide were purchased from Tianjin Fuyu Fine Chemical Co, Ltd The water-soluble polyimide resin (WPI, YE-1022) was acquired from Shanghai Yehe Trade And Industry Co, Ltd Its viscosity is 15–25 Pa·s and decomposition temperature is 295–303°C. Dialysis membrane (MWCO 1.0 KDa) was purchased from Xi’an Yobios BioTech. All the chemical reagents were analytical grade and used without further purification.
Synthesis process of DSEP emulsion
Synthesis and parameters of DSEP emulsion.
FTIR analysis
The samples were cut into powders and mixed with KBr, then pressed for the measurement. The chemical structures of the samples were characterized using the Fourier transform infrared spectrometer (PT-IR, UK) in the wavelength range of 4000∼650 cm−1.
XPS analysis
XPS spectra of the samples were measured using a X-ray photoelectron spectroscopy (Thermo Scientific K-Alpha+) with Mono Alka (hv = 1486.6 eV). Surface spectra were collected over a range of 0∼1350 eV and high-resolution spectra of C 1s, N 1s, and O 1s regions were also gathered.
1H-NMR analysis
1H NMR measurement of sample was conducted on a nuclear magnetic resonance spectrometer (Bruker 400M, Germany). The solvent is D2O and the internal standard is trimethylsilane (TMS).
Particle size analysis
The average particle size of the DSEP emulsion was tested by a Malvern Zetasizer Nano Instrument (Malvern, UK). The samples were diluted to 0.5 wt% with deionized water and then measured at 25°C.
DSC analysis
The thermal property of DSEP was analyzed by differential scanning calorimeter (Q500, USA). About 5 mg of powder sample is used to test under the protection of nitrogen. The test temperature is between 30 and 300°C and the heating rate is 10°C/min.
Morphologies analysis
The surface and fracture morphologies of the PI filament before and after infiltrating were observed by scanning electron microscope (SEM, Quanta-450-FEG, USA) and transmission and reflection metallographic microscope (LW300LJT, China), respectively.
Mechanical properties
The breaking strength of PI filament was determined using an electronic yarn tensile tester (YG020, China) with the stretching interval and speed of 500 mm and 500 mm/min, respectively. The average value was taken from the stress–strain curves of 30 examples.
The peeling strength between mono-filament and mono-filament was tested on an electronic fiber tensile tester (LLY-06EDC, China). Separate a mono-filament from the sample with a length of 10 cm, then use the upper clamp to fix the mono-filament, and the lower clamp to fix the remaining filament (the peeling point is placed in the middle of the upper and lower clamps). The distance is 20 mm and the tensile speed is 20 mm/min. When the clamp moves to more than 55% of the initial spacing, stop the test and record the maximum force in the stretching process, i.e. peeling strength. And the mean values of the peeling strength for mono-filaments were calculated by testing 10 samples.
Results and discussion
Synthesis of DSEP emulsion
DSEP was synthesized by one-pot method under the alkaline condition, the reaction principle is shown in Figure 1. Both the imide ring structure and the hydrophilic group -OH are introduced into the EP by the ring-opening reaction between the epoxy group and the imino group [38]. This caused that the EP not only contains the imide ring structure similar to the PI filament structure but has water solubility partly. Figure 2 (a1-a2) shows that the EP dissolved in ethanol appears white precipitation after being diluted by water, but the DSEP aqueous dispersion (50 wt%) is transparent (Figure 2 a3). When the concentration of the DSEP aqueous is diluted to 1%, the DSEP turns white but there is no precipitation (Figure 2 a4). The experimental phenomena show that the structure of EP modified by SI assisted by DEA has changed. Reaction principle of DSEP. Structural characterization of DSEP. (a) Appearance graphics. a1 is EP dissolved in absolute ethanol, a2 is the appearance of a1 after adding water, a3 is DSEP, and a4 is DSEP after dilution; (b) FTIR spectra of SI and DSEP; (c) FTIR spectra of different molar ratios SI reacting with EP; (d) XPS spectra of DEP and DSEP; (e), and (f) are C1s spectra of DEP and DSEP, respectively.

To confirm the group change of EP modified by DEA-assisted SI, the FTIR spectra of SI and DSEP were carried out, as shown in Figure 2(b) and (c). For the SI, the absorption bands at 1772 and 1703 cm−1 belonged to the carbonyl asymmetric and symmetric stretching vibration peaks, respectively [39] (Figure 2(b)). In the spectrum of DSEP, in addition to the benzene ring (1508 cm−1) and C-O-C (1292 cm−1) characteristic peaks of EP were occurred on the spectrum of DSEP, the asymmetric and symmetrical stretching vibration absorption bands of C=O, as well as C-N-C characteristic peaks were appeared at 1772, 1703, and 1359 cm−1 [40], respectively, which are attributed to the characteristic groups of SI and DEA. Meanwhile, the epoxy group at 913 cm−1 on the DSEP spectrum shows weak peak intensity, indicating that SI and DEA reacted with the epoxy group on EP structure. Figure 2(c) shows the FTIR spectra of the products with different molar ratios of SI and EP. The characteristic peak of the epoxy group at 913 cm−1 is weaker when the molar ratio is 2:1, which further indicates that the reaction between SI and EP takes place on the epoxy group.
To confirm the bonding form of DEA and SI on the EP structure, the XPS spectra of DEP and DSEP are measured as shown in Figure 2(d) to (f). The characteristic peaks near 280, 400, and 530 eV are related to the binding energies of C1s, N1s, and O1s, respectively (Figure 2(d)). From the C1s fitting spectrum of DEP (Figure 2(e)), there are three kinds of bonding forms of C element: C-C (284.8 eV), C-N (285.6 eV), and C-O (286.4 eV) [41]. The C-N peak is formed by the reaction of the imino group in the DEA structure with the epoxide group in EP. For the C1s spectrum of DSEP (Figure 2(f)), in addition to the C-C, C-N, and C-O peaks, the C=O peak appears at 288.7 eV [42], which indicates that the carbonyl group in the SI molecule exists in the structure of DSEP. FTIR and XPS results show that SI and DEA are mainly bonded to the epoxy group of EP in the form of C-N-C.
Figure 3 shows the 1H NMR spectra of DEP and DSEP. In the 1H NMR spectrum of DEP, the signal at δ = 6.78 and 7.21 ppm was assigned to the aromatic protons of bisphenol A, and the peak at δ = 1.58 ppm was attributed to the -CH3 proton of bisphenol A. The proton peak of -OH group produced by DEA occurred at δ = 3.48 ppm [43], which provided evidence for the successfully synthesized of DEP. Compared with the 1H NMR spectrum of DEP, a new signal peak appeared at δ = 2.74 ppm, which may be caused by the -CH2 proton peak on the amide ring in the SI structure. Combined the appearance of C=O bonding peak in XPS spectrum of DSEP, which further confirmed the successfully synthesis of DSEP. 1H NMR spectra of DEP and DSEP.
Particle size and DSC of DSEP
A series of DSEP was obtained by controlling the molar ratio of SI and DEA to EP, as shown in Table 1. It can been seen that with the increase of molar ratio of SI and DEA to EP, the particle size of DSEP emulsion decreases from 232 nm to 118 nm, which is attributed to the introduction of SI and DEA leads to the formation of a long carbon chain, and it is conducive to the formation of dense structure.
In order to confirm the stability of DSEP emulsions, the change of appearances of DSEP with the storage duration is measured and shown in Figure 4(a). DSEP exhibits good dispersibility in water when the molar ratio of SI and DEA to EP exceeds 0.5/0.5/1. This may be due to the increase of DEA content, which increases the contact probability between the imino group and the epoxy group of EP skeleton, leading to more hydrophilic hydroxyl groups that are introduced into the EP structure, it accelerates the salt formation reaction between the imino group and the carboxyl group, and improves the dispersion of DSEP in water. DSEP1 emulsion appeared stratification after 7 days, and the granules appeared at the bottom of the bottle for 1 month. While none of the other emulsions were stratified except DSEP1 and DSEP2 after 1 month. Furthermore, the DSEP3, DSEP4, and DSEP5 emulsions were observed by centrifuging treatment; it was found that DSEP3 produced precipitation when the rotating speed was 3000 r/min. The results indicate that the DSEP has high storage and centrifugal stability when the molar ratio of SI and DEA to EP exceeds 0.9/0.9/1. Performance characterization of DSEP. (a) The appearance of DSEP stored for different time. From top to bottom, the DSEP appearance graphics were placed for 0, 7, and 30 days; (b) Emulsion particle size distribution of DSEP4; and (c) DSC of DSEP.
Figure 4(b) shows the particle size distribution of DSEP4. It can be seen that DSEP4 has a narrow particle size distribution with average particle size and PDI of 126 nm and 0.106, respectively. The DSC curve of DSEP is shown in Figure 4(c). It can be seen that an endothermic peak appears at 215°C, indicating that DSEP exhibits excellent thermal stability and also meaning a new polymer was formed by the reaction of EP, SI, and DEA.
Strand integrity of PI filament
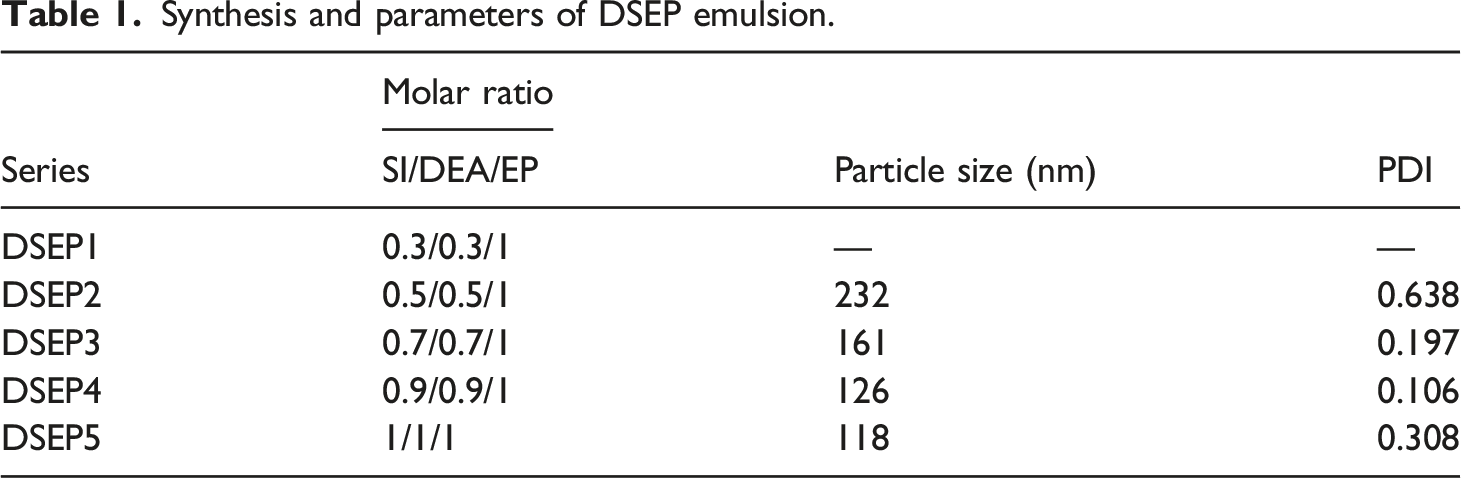
To prove that DSEP emulsion can reinforce the strand integrity of PI filament, we compared and analyzed the improvement effect of DEP, DSEP, and commercially available WPI on the strand integrity of PI filament, as shown in Figure 5(a). SEM images of PI filament infiltrated in DEP, DSEP, and WPI emulsions are shown in Figure 5 a2–a4. The mono-filament of PI filament is severely dispersed and has poor strand integrity (Figure 5 a1). Both DEP and DSEP emulsions can improve the strand integrity of PI filaments, but the DSEP has more significant strand integrity effect on PI filament (Figure 5 a2–a3), which is consistent with the microscopic morphology of the commercially available WPI reinforced PI filament (Figure 5 a4). Strand integrity of PI filament before and after reinforcement. (a) SEM images from left to right were PI filament (a1), DEP reinforced PI filament (a2), DSEP reinforced PI filament (a3), and WPI reinforced PI filament (a4), respectively; (b) stress–strain curves of PI filament before and after reinforcement; (c) peeling strength of samples; (d) fracture morphologies of the samples; and (e) morphologies after friction for different times.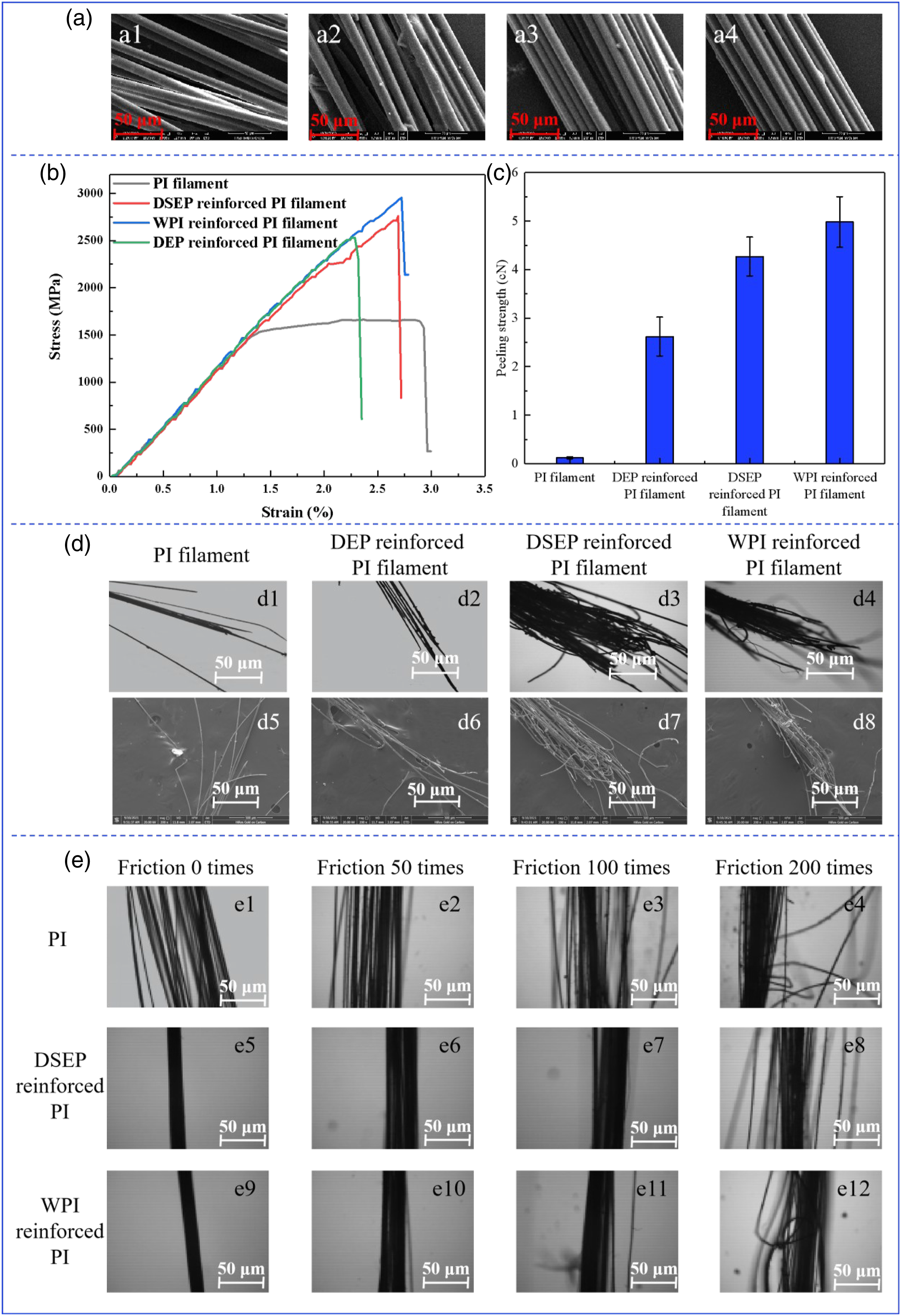
Figure 5(b) shows the stress–strain curves of the PI filament reinforced by DEP, DSEP, and commercially available WPI. It can be seen that there are a great difference on the tensile properties of PI filament before and after infiltrating. The stress–strain curve of PI filament includes two stress–strain phases. The first phase is the linear elastic region, and the second phase is the linear strain hardening region with large plastic deformation until the final fracture. However, the stress–strain curve of the infiltrating PI filament is mainly near the first elastic region [44]. Compared with PI filament, the breaking strength of the DSEP reinforced PI filament is increased by 47.04%, which is higher than the breaking strength of that of the DEP reinforced PI filament (40.16%), and close to that of the WPI reinforced PI filament (49.33%). The results show that the imide group bonded onto the DSEP structure can effectively reinforce the adhesion between PI filaments. This can be explained by the well-known fractal Hall–Petch effect [45], which is widely used in material design, that is, the nano DSEP emulsion particles coated on the inside and surface of PI filament can improve the mechanical property of the material. To further confirm the interaction between DSEP and PI filament, the peeling strength between mono-filament and mono-filament was tested, as shown in Figure 5(c). It can be seen that shows the DSEP reinforced PI filament shows higher peeling strength, indicating DSEP enhanced the bonding ability between mono-filaments.
Besides, the fracture of PI filaments was observed by transmission and reflection metallographic microscope and SEM, respectively. It was found that only a small amount of the dispersed PI filament was present at the fracture site (Figure 5 d1, d5). This is because there is no adhesion between mono-filament and mono-filament, and the mono-filament gradually fractured and the fracture was occurred at a small number of fibers during the tensile process [46]. A small number of filaments with clustering tendency can be observed at the fracture site of DEP reinforced PI filament (Figure 5 d2, d6). However, a large number of filaments with strong strand integrity can be observed at the fracture of DSEP reinforced PI filament (Figure 5 d3, d7), which is consistent with the effect of WPI emulsion reinforced PI filament fracture (Figure 5 d4, d8). The results showed that DSEP emulsion prepared in this work can reinforce the strand integrity of PI filament because DSEP structure contains imide ring groups similar to PI filament, which strengthens the bonding ability between mono-filament and mono-filament.
To confirm the enhancement effect of DSEP emulsion on PI filament, the surface friction treatment of DSEP and WPI resin reinforced PI filament was carried out, respectively, and their strand integrity was analyzed. As shown in Figure 5 e1, the strand integrity of PI filament was poor, both DSEP and WPI could improve the strand integrity of PI filament (Figure 5 e5, e9). After 50 times of friction, the PI filament reinforced by DSEP is still a whole (Figure 5 e6), while the mono-filament of PI filament dispersion is remarkable, which indicates that DSEP can promote the adhesion between the mono-filaments. When the friction reach 200 times, the mono-filament leaves from the filament matrix is occurred on the DSEP reinforced PI filament, and no mono-filament entanglement and fracture phenomenon are observed (Figure 5 e8), which is consistent with the effect of WPI reinforced PI filament (Figure 5 e12). However, the significant phenomenon of mono-filament disorder, slipping, and breakage has appeared on PI filament after 200 times of friction.
To further confirm the interaction between DSEP and PI filament, the FTIR of PI filament, DSEP, and DSEP reinforced PI filament was analyzed, as shown in Figure 6(a). Compared with the FTIR spectrum of PI filament, the intensity of C-N-C, asymmetric and symmetric C=O, and benzene ring characteristic peaks at 1359, 1773, 1703, and 1508 cm−1 in FTIR of DSEP reinforced PI filament were significantly enhanced, as well as there is a strong interaction between DSEP and PI filament. Figure 6(b) shows the mechanism of DSEP reinforced the strand integrity of PI filament. According to the principle of the dissolution in the similar material structure, there are the similar imide ring and benzene ring between DSEP and PI filament, which can improve the binding ability between PI mono-filaments. Also, the strong hydrogen bonding interaction will be formed between -OH in DSEP and C-N, C=O in PI filament, which can further improve the adhesion between PI mono-filaments. The above two synergistically effects strengthen the strand integrity of PI filaments. (a) FTIR spectra of DSEP, PI filament, and DSEP reinforced PI filament and (b) the mechanism of DSEP reinforced the strand integrity of PI filament.
Conclusions
In this work, a waterborne epoxy resin (DSEP) emulsion was obtained by introducing DEA and SI onto EP through the ring-opening reaction. When the molar ratio of SI and DEA to EP exceeds 0.9/0.9/1, DSEP has high storage and centrifugal stability, and its particle size distribution is 118∼126 nm. Benefiting from the similar structure and the strong hydrogen bonding interaction between DSEP and PI filament, the DSEP emulsion has a significant reinforcement effect on the strand integrity of PI filament, and the breaking strength of the PI filament is increased by 47.04%, which is close to that of the WPI reinforced PI filament. In addition, DSEP reinforced PI filament shows higher peeling strength, and there is no filament breakage in DSEP reinforced PI filament after more than 200 times of friction. This work provides a new way for developing water-soluble epoxy resin that can reinforce the strand integrity of PI filament.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Scientific Research Program of Shaanxi Provincial Education Department (21JK0664), Doctoral Research Program of Xi’an Polytechnic University (107020533), and the innovation team of ShaanXi Provincial Tri-Qin Scholars are greatly appreciated.
