Abstract
The recovery of impaired peripheral nerves is often not as expected, which makes the development of nerve conduits trendy nowadays. To enable the neural messages effectively being delivered as well as to prevent the secondary damage during the removal of nerve conduits, the conductivity and biodegradability are two essential requirements for ideal nerve conduits. In this study, electrospinning is used to produce polyvinyl alcohol (PVA)/carbon nanotubes (CNT) electrospun films, after which the morphology analysis, electrical property, water contact angle, and biological characteristics of the membranes are investigated, thereby determining the optimal nerve conduits based on the employment of electrospinning, PVA, and CNT. The test results indicate that with 0.25 wt% of PVA, the electrospun films exhibit comparatively lower resistance of 25.3 ohm, good fibrous morphology with a diameter being 1 μm. In addition, the electrospun films are cytotoxicity-free and facilitate the growth of cells. It is observed in the MMT assay that after co-cultured with cells for three days, PVA/CNT electrospinning fibrous membranes exhibit a cellular viability that is 18.5 times greater than that of the control group on Day 1. According to all property evaluations, PVA/CNT electrospinning fibrous membranes are a qualified candidate for the use of nervous conduits.
Keywords
Introduction
Tissue engineering is a knowledge combines multidisciplinary fields, which aims to rebuild or repair the organs and tissues with bioactive matters via in vitro culture or construction. Moreover, tissue engineering can target the impaired tissues in terms of the tissue regeneration, repair, and replacement, which means that complex damaged tissues also retain functions by means of the characteristics and structures of materials. Tissue engineering is considered providing the most possibilities regarding regeneration and function recovery as it realizes the remedy that combines both the cell transplant molecule delivery as well as biomaterial scaffolds. On the other hand, scaffolds are one of the three main categories of tissue engineering, providing the impaired sites with micro-environment for cell growth and attachment. In addition to good biological characteristics, good tissue engineering scaffolds demands sufficient mechanical strengths and stiffness in order to mechanically support the impaired tissues during the recovery and regeneration durations. If not as strong as the original healthy tissues, scaffolds should possess mechanical strengths and stiffness that at least support the impaired sites.
Nerve conduits are also one item that constitutes tissue engineering scaffolds, and they are deemed as stents that bridge the nerve stumps while guiding and supporting nerve regeneration [1,2]. In the meanwhile, they supply mechanical strengths and chemically stimulate the nerves, thereby facilitating the nerve growth and repair. Besides, peripheral nerves are commonly damaged due to the presence of trauma, compression, stretching, or ischemia. Despite good regeneration mechanism, nerves usually fail to regain the functions when being damaged and there are 50% of related patients who still need to receive a surgery as therapy [3]. As a result, the repair methods of nerve conduits become a trendy development currently. In early days, nerve conduits were primarily made of silicone tubes and were only able to mend 10-millimeter long impaired nerves. They also had disadvantages, e.g. lack of swelling ability and biodegradability as well as induction of inflammation and chronic foreign body reaction, which in turn oppressed the nerves and hampered the nerve regeneration [4]. In order to address the aforementioned problems, some scholars start producing biodegradable nerve conduits using natural and synthetic polymers [5,6]. On one hand, nerve conduits demands biocompatible materials, which means that they need to be cytotoxicity-free, non-carcinogenic, non-immunogenic, and non-mutagenic without causing partial or whole body stimulation and allergies [7,8]. On the other hand, nerve conduits require materials with a certain degradation rate and axon growth rate. Nerve conduits are supposed to be flexible and semi-permeable for they can adjust the regeneration process and the solute exchange between extra tunnel environments. Selective permeable tunnels allow the nerve regeneration, which appear to be more water impermeable or more water solute permeable tunnels comparing to normal static nerves [7,9]. Besides, many studies indicate that the presence of conductive materials improves the cell adhesion and transmission of neural message [10–13]. Carbon fibers, graphene, and other carbon-based conductive macromolecules are used in many biological applications, especially biosensors, tissue engineering, and nerve conduits, because they exhibit rather high conductivity, elasticity, and tensile properties [14–16].
Electrospinning is a production technique that employs static electricity to generate fine polymer fibers out of solutions [17]. Although the electrospinning technique is inclined to many factors, it still can produce nanofibers with a comparatively lower production cost, a greater specific surface area, and a higher porosity [10]. At the same time, combinations of different assembly components and solvents create fibers with different morphologies, such as hollow fibers, porous fibers, multichannel fibers, and shell-core fibers. A diversity of material selections and fiber structures further enable the electrospinning fibers for medicine applications, e.g., trauma protection materials, tissue engineering scaffolds, and artificial blood vessels. Furthermore, the biomedicine field has been increasingly paying attention to temporary implants and biodegradable materials with an attempt to remove the secondary surgery required by removing the non-degradable implants or long-term physically repulsive response toward the implant materials [18,19].
Polyvinyl alcohol (PVA) is toxicity-free polymer featuring goood physical properties, thermal stability, and flexibility, and therefore PVA has a widespread acceptance in different fields, involving livlihood, industry, food, and packagings [20–27]. In addition, PVA has biodegradability, biocompatibility, high hydrophilicity, and non-toxicity, due to which PVA has been commonly used in the medicine [28,29]. Asran et al. employed elelctrospinning to produce PVA/PHB electrospun films with an attempt to become skin substitute [30]. Thomas et al. proposed using citric acid to modify PVA electrospun films that were biotoxicity free and biocompatible with smooth muscle cells of blood vessels. The resulting products had a great potential to be used as vascular tissue engineering stents. Babaie et al. likewise produced PVA/PEDOT electrospinning stents and evaluated their applications to nevous conduits [31].
Following the advanced development of materials, the interest of using biodegradable material as temporary implants has come to the fore in the biomedical field. The purpose is to exclude and avoid second surgery required by non-degradable implants or many physical reactions when used an implant for a long term. In many studies, PVA used a biomedical material is comparatively safe and has ease of process. In the meanwhile, carbon materials are also found to have a positive effect on cell growth among various physical attributes. However, the combination of PVA and CNT via the electrospinning process is rarely seen in related literatures. Hence, in this study, electrospinning technique is employed to produce PVA electrospun films containing different CNT contents. The proposed PVA/CNT electrospun films harvest the biocompatibility from PVA, the conductivity of CNT, and micro-grooves that are suitable for cell attachment, making them a qualified candidate for nerve conduits. In addition, the morphology of electrospun films is observed in terms of the CNT content while the conductive property, water contact angle, degradation rate, and biocompatibility of PVA/CNT electrospun films are then evaluated as related to the CNT content.
Experimental
Materials
Polyvinyl alcohol (PVA) powders (Sigma-Aldrich, US) have a molecular weight of 89000-98000 Da. Carbon nanotubes (CNT, Advanced Nanopowder Inc., Taiwan) have a model number of CF182C and a diameter of 5-20 nm. LB culture medium, NB culture medium, and agar used in the biocompatibility test and MTT assay are purchased from Thermo Fisher Scientific, Taiwan. Phosphate-buffered saline (PBS) is purchased from Sigma-Aldrich, US. L929 cells are purchased from the Food Industry Research and Development Institute (FIRDI), Taiwan.
Preparation of PVA/CNT electrospun films
Deionized water at 120 °2 is used as the solvent to formulate 14 wt% PVA solution. Next, CNT (0, 0.1, 0.25, 0.5, 0.75, and 1 wt%) is added in order to form PVA/CNT conductive electrospinning solutions, after which the electrospinning solutions are processed with an ultrasonic vibrator (LEO-801, LEO, Taiwan) for 12 hours. During the process of formulating PVA/CNT electrospinning solutions, the containers with electrospinning solutions are sealed to refrain the evaporation. Therefore, the concentration of electrospinning solutions have comparable theoretical and empirical values.
In addition, the electrospinning solutions are tested for conductivity and viscosity with a pH tester (EC500, ExStike, US) and viscometer (Viscobasic+L, FUNGILAB S.A, Spain), respectively, because different CNT contents significantly change the properties of electrospinning solutions. The electrospinning assembly is composed of static electricity generator (Modol 200 Series, Kd Scientific, US) and solution syringe (280,220 Kd Scientific, US). For the starter, PVA/CNT conductive solutions are infused into the syringe and meanwhile, the solution flow velocity is set to be 0.06-0.08 ml/h with a spinning distance being 10 cm and a needle diameter being 18 mm. Next, the static electricity generator is turned on with a constant voltage between 19 and 21 kV that enables the electrospinning solution to overcome the surface extension and then form a jet. Afterwards, the resulting electrospun fibers are collected so as to form PVA/CNT electrospun films over the collection board [32–36]. The electrospun films are tested for fiber diameter analysis, surface observation, conductivity, hydrophilicity, and cell compatibility.
Measurements
Viscosity of electrospinning solutions
The viscosity of electrospinning solutions is measured as related to the CNT content being 0, 0.1, 0.25, 0.5, 0.75, and 1 wt% using a viscosity tester (VISCOBASIC+L, Fungilab, Barcelona, Europe). The tester is equipped with various detective probes and capable of detecting the difference in the viscosity of electrospinning solutions.
Conductivity of electrospinning solutions
The conductivity of PVA/CNT electrospinning solutions is measured as related to the CNT content being 0, 0.1, 0.25, 0.5, 0.75, and 1 wt% using a conductivity measuring instrument (EC500, ExStik©, EXTECH, USA), thereby examining the effects of CNT on the conductivity.
Diameter of electrospinning fibers
A sample is pasted and fixed to the platform of a scanning electron microscope (SEM, S-4800,HITACHI, Japan) and coated with a thin layer of gold. The fiber morphology of electrospun films is observed at an operation voltage of 15 kV afterwards. In the fiber diameter analysis, the average fiber diameter is examined from at least 200 sites of the electrospun films.
Scanning electron microscopy observation
The SEM images of PVA/CNT electrospun films are used to analyze the average fiber diameter using the image analysis software (Image-Pro Plus).
Conductivity of electrospun films
As specified in ASTM D4496-13 test standard, a four-point probe measuring instrument is used to measure the conductivity of electrospun films. It is required to use a high resistance meter when the conductivity is lower than 10-7 S/cm. In this test, the resistance (ρ) is measured and the conductivity is computed using the following equation.
Water contact angle measurement
A water contact angle tester (OCA-15 PLUS, SCA20, Germany) is used to measure the water contact angle of PVA/CNT electrospun films. The film is mounted over the platform of the tester, and a syringe is used to drip deionized water over the film. The water contact angle between the water drop and the film is observed and recorded with a microscope.
Degradation rate
Dried PVA/CNT films that are mixed with equal proportion are used for the degradation rate measurement. The sample is weighed in order to have the initial dried weight (W0), after which it is placed in a centrifugal tube containing PBS and then processed with shock slot at a constant temperature of 37 °7, during which the PBS is changed every 12 hours. After the designated degradation durations, samples are removed, dried, and weighed to have the weight after t time (Wt). The degradation rate is computed with the following equation.
Cell compatibility
As specified in ISO 10993-5 test standard, the electrospun films are co-cultured with fibroblasts. For the starter, fibroblasts are cultured in the substrate in a petri dish until they grow steadily. Next, the culture substrate is removed and PBS is used to rinse the petri dish in order to remove the deceased cells. Afterwards, 0.5 mL of trypsin is used to rinse to the petri dish, which in turn keeps the living cells afloat. Moreover, the petri dish is kept in an incubator at 37 °7 for five minutes and then 0.5 mL of PBS is added to dilute the trypsin. The cell suspension is withdrawn to a hemocytometer, thereby the cell counts are computed.
MTT assay
As specified in the ISO 10993-5, MTT assay is conducted to compute the cellular viability. Samples are sterilized beforehanded. For the starter, 10 μm of sample extract is infused into 96-well cultivate plate separately, after which L929 fibroblast suspension (90 μm; 104 cell/well) is added to the cultivate plate, too. The plate is placed in an incubation at 37 °7 for 24 hours and removed to a sterilized horizontal laminar flow table. A buster straw is used to remove the MEM and then add 70 μl MTT agent. The plate is kept in the dark for four hours, and MTT and medium are first removed, and 70 μl of dimethyl sulfoxide (DMSO) solvent is added. At last, an ELISA reader measures the optical density (OD) at 570 nm. OD is used to measure the cellular viability with the equation as follows
Results and discussion
Viscosity and conductivity of PVA/CNT electrospinning solutions
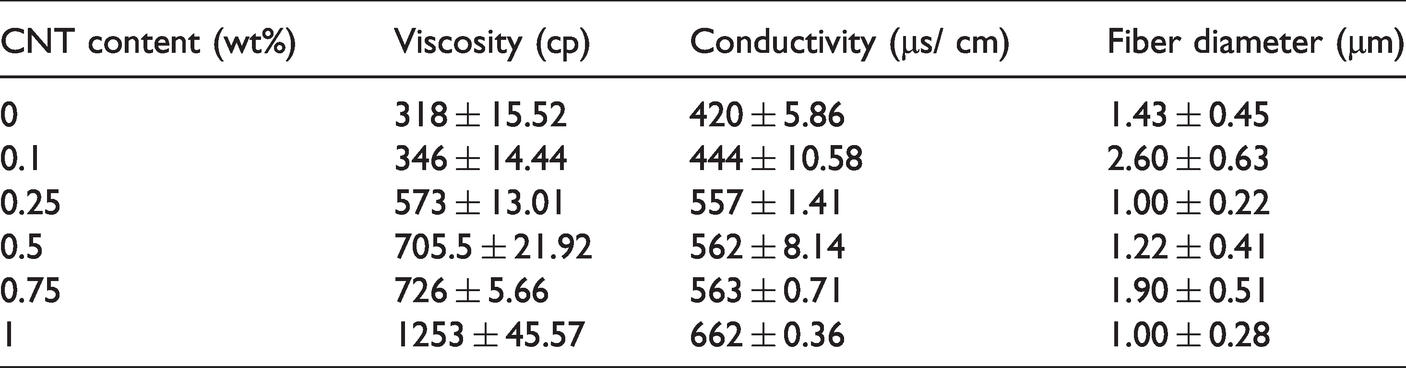
The electrospinning fibers have a morphology and size that are dependent on the parameters, involving the concentration, viscosity, electrical conductivity, and surface tension of electrospinning solutions [36]. Two main influential factors are the viscosity and conductivity at a suitable level, generating optimal fibrous morphology. When added to polymer materials, carbon nanotubes would form a network that restricts polymer chains and thus immobilizes the movement of molecular materials. Subsequently, the presence of carbon nanotubes increases the viscosity of electrospinning solutions [37–39]. Table 1 shows that a rise in the CNT content improves the conductivity and viscosity of PVA/CNT electrospinning solutions. A higher viscosity increases the surface tension of the electrospinning solution, which means that it demands a higher electrospinning voltage to overcome the surface tension and then forms an electric jet. The jet then reaches the collection board to form sleek fibers. Contrarily, with an excessively low viscosity, the viscoelasticity of electrospinning solution reflects a relaxation phenomenon, hampering the electrospinning fibers from electrostatic spraying. Therefore, the conductivity of electrospinning solution determines the fineness of electrospinning fibers, and a high conductivity provides the fibers with a finer diameter. The results are in conformity with the findings of Sebastian et al. [40].
Viscosity and conductivity of PVA/CNT electrospinning solutions as related to CNT content.
Morphology of PVA/CNT electrospun films
Figure 1 shows the SEM images and fiber diameters of PVA/CNT electrospun films as related to CNT content being 0, 0.1, 0.25, 0.5, 0.75, and 1 wt%.Meanwhile, the morphology of electrospinning fibers is also correlated with many factors, including the environment temperature, humidity, voltage, and spinning distance as well as the properties of electrospinning solutions. Specifically, the properties of electrospinning solutions (i.e. polymer molecular weight, conductivity, and concentration), the distance between the syringe and the collection board viscosity, and collection morphology of electrospinning fibers [41].

SEM images and corresponding insets (the fiber diameter distribution) of PVA/CNT electrospun films as related to CNT content being 0, 0.1, 0.25, 0.5, 0.75, and 1 wt%.
In this study, electrospinning is conducted with a voltage being 19-22 kV, the distance to the collection board being 10 cm, and a planar collection, the parameters of which are particularly designed to analyze the influence of concentration and conductivity of the electrospinning solutions on the diameter of fibers. Figure 1 shows that the electrospun films exhibit greater fiber diameters when containing 0.1 wt% of CNT than when containing 0 wt% of CNT. In the meanwhile, Table 1 shows that a rise in a CNT content increases both of the viscosity and conductivity of electrospinning solutions, which means that variation in the CNT content decreases the diameter of fibers. Meanwhile, the electrospinning voltage requires a fine adjustment when the conductivity of electrospinning solutions is changed. Namely, a greater conductivity demands a greater voltage, thereby surpassing the surface tension of the electrospinning solutions. Concurrently, a greater conductivity renders the electrospinning jets with higher static charges, higher extension, and thus smaller fiber diameters [42]. In addition to Figure 1(c) and (f) that show the smallest fiber diameter of PVA/CNT electrospun films, the corresponding insets also indicate a narrower fiber distribution. A narrow fibrous distribution suggests that all fibers are generated with an evener fineness. In particular, with a 0.25 or a 1 wt% of CNT content, PVA/CNT electrospun films acquire comparatively smaller diameter that is close to 1 μm.
FTIR of PVA/CNT electrospun films
Figure 2 shows the FTIR spectrum of PVA/CNT electrospun films as related to the content of carbon nanotubes (CNT). Regardless of the CNT content, the resulting electrospun films show comparable characteristic peak that belongs to PVA. The stretch vibration of –OH at 3315 cm-1 is associated with the stretch vibration of C = O, the result of which is ascribed to the acetate groups of PVA molecules [43]. Besides, the chemical structure of PVA/CNT electrospun films is not correlated with the CNT content distinctively. This result may be due to a small CNT content used in this study, which does not affect the FTIR measurement of PVA/CNT electrospun films to generate any new peak values. Namely, PVA does not change its quality or react with other chemical response because of the presence of CNT, which suggests that the PVA/CNT electrospun films retain the mechancial and physical attributes of PVA.

FTIR spectrum of PVA/CNT electrospun films as relate to CNT content being 0, 0.1, 0.25, 0.5, 0.75, and 1 wt%.
Water contact angle of PVA/CNT electrospun films
Figure 3 shows the water contact angle of PVA/CNT electrospun films as related to the CNT content. The water droplet along with the film’s surface comprises an angle; when the water contact angle is large, the materials have hydrophobicity, otherwise, the materials have hydrophicility. Materials are classified as super hydrophilicity for having a water contact angel lower than 10 degrees, hydrophilicity for 10-90 degrees, hydrophobicity for 90-150 degrees, and super hydrophobicity for over 150 degrees [44].

Water contact angle of PVA/CNT electrospun films as related to CNT content being 0, 0.25, 0.5, 0.75, and 1 wt%.
In the tissue engineering, biomedical materials are required to have good biocompatibility for the biomedical application. The interaction between materials and cells affects the cellular morphology, signal transduction, and gene expression, which means that the surface properties of biomedical materials are considered critical evaluations [45,46]. In addition to the materials’ structure, mechanical properties, and biochemical properties, especially cell binding sites, the hydrophilicity of biomedical materials is also an influential factor for it demonstrates a significant influence over the cell adhesion. The protein of cell membranes can adhere to the hydrophobic surface, demonstrated by a water contact angle of θ > 65f. For example, PEG has been commonly used as anti-adhesion layers due to good hydrophilicity, and thus the adjustment of the surface’s hydrophilicity is an effective measure to improve the cell adhesion [47–49].
In this study, PVA is hydrophilic but CNT is hydrophobic. Figure 3 shows that the control group (electrospun films w/o CNT) has a water contact angle of 32.6°, which is close to super hydrophilic. After incorporated with 0.25 - 1 wt% of CNT, the films exhibit a water angle that rises to 75°, which is then slightly increased with the increasing CNT content afterwards. Besides, the tissue engineering scaffolds exhibit cell attachment that is determined by whether the scaffolds’ surface have hydrophilicity and roughness. Despite the fact that the presence of CNT increases the water contact angle, the films have a water contact angle close to only 75°, suggesting that they are hydrophilic and meet the standard of cell adhesion.
Conductivity of PVA/CNT electrospun films
Figure 4 shows the conductivity of PVA/CNT electrospun films as related to the CNT content being 0, 0.1, 0.25, 0.5, 0.75, and 1 wt%, where the conductivity first increases and then decreases as a result of a rise in the CNT content. A conductive network is almost formed in the PVA/CNT electrospun films with the presence of 0.1 wt% of CNT, and therefore, the conductivity is not significantly improved. Next, a CNT content from 0.1 wt% to 0.25 wt% causes a distinctive rise in the conductivity and the optimal conductivity of electrospun films occurs when PVA/CNT electrospun films comprise 0.25 wt% of CNT. The resulting electrical resistivity is 25.3 ohm. Furthermore, the penetration overvalue percolation threshold is presented with a CNT content being 0.1-0.5 wt%. Nonetheless, when the CNT content exceeds 0.25 wt%, the conductivity shows a descending trend and eventually becomes mild with a CNT content being 0.75 wt%. This phenomenon occurs because a greater CNT content provides a complete conductive network in the electrospun films whereas an excessive amount of CNT contrarily causes agglomeration that adversely affects the conductivity [50–53].

Conductivity of PVA/CNT electrospun films as related to CNT content being 0, 0.25, 0.5, 0.75, and 1 wt%.
Degradation of PVA/CNT electrospun films
Figure 5 shows the degradation rate of PVA/CNT electrospun films as related to the CNT content being 0, 0.1, 0.25, 0.5, 0.75, and 1 wt%. In this study, dried PVA/CNT electrospun films are tested for degradation rate in order to evaluate how the CNT content, instead of electrospinning process (i.e. the fiber diameter), is correlated with the degradation. In addition, polymers, including PVA, show a degradation rate that primarily depends on the crystallinity, molecular weight, chemical structure, and mechanical properties. Figure 5 shows that with a test span being seven days, the degradation rate of PVA/CNT electrospun films does not fluctuate considerably and remains between 0.93% and 0.95%. In sum, the degradation rate of dried PVA/CNT electrospun films is dependent on the PVA matrices, rather than the CNT. A trivial amount of CNT (e.g. 0.1-1 wt%) does not change the crystallinity and mechanical properties of PVA matrices, which suggests that PVA films are not affected by a small amount of CNT [41,54].

Degradation of PVA/CNT electrospun films as related to CNT content being 0, 0.25, 0.5, 0.75, and 1 wt%.
Cell compatibility of PVA/CNT electrospun films
In addition to good mechanical properties and conductivity, CNT has been also used in the tissue engineering scaffolds because CNT has no biotoxicity but positive influences in terms of cell adhesion and proliferation as well as signal transmission [55]. In the meanwhile, nerve cells transmit messages to the receiving end via electricity, which means that the presence of CNT not only facilitates cell growth efficiency but also ensures effective transmission of nervous messages [56,57]. Figure 6 shows the cell compatibility of the control group (i.e. without PVA/CNT electrospun films) and the experimental group (i.e. with PVA/CNT electrospun films as dark substance in the images as related to the culture spans being 1 and 3 days. In Figure 6, due to the focus that is exclusively on cells, samples (i.e. the black part) in the image become blurred. Comparing Figure 6(a) and (b), both the control and experimental groups exhibit a comparable amount of cells. Namely, co-culture for one day proves that PVA/CNT electrospun films are toxicity free without significant cell proliferation on day 1. Furthermore, Figure 6(c) and (d) show that the experimental group outperforms the control group in terms of the cell amount, and the experimental group contributes to a far greater amount of cells. According to Figure 5, with a degradation span being 1-3 days, electrospun films gradually release toxicity-free CNT that has a positive influence on the cell growth afterwards. This finding is consistent with others in previous studies, and CNT is suitable for the production of nerve conduits.

Cell compatibility of control group on (a) Day 1 and (c) Day 3 as well as PVA/CNT electrospun films on (b) Day 1 and d) Day 3.
Figure 7 demonstrates the cell viability of PVA/CNT electrospun films, comparing the cell counts with that of the control group by ELISA reader in Figure 6. The control group shows 100% cell viability on day 1 and then 228.4% on day 3, indicating a 2.28 times greater cell growth. In other words, cells well adjust the culture environment. By contrast, with PVA/CNT electrospun films (i.e. experimental group) has a cell counts that is 18 times greater than that of the control group on day 1. The result indicates that PVA/CNT electrospun films are biotoxicity free and have biocompatibility that has a positive influence on the cell growth. On day 3, the experimental group exhibits 18.5 times higher cell growth than the control group whereas on day 1, it exhibits 1.03 times higher cell growth than the control group. To sum up, PVA/CNT electrospun films are helpful to the cell growth according to the outcome of the co-culture. In light of Figures 5 to 7, the biocompatibility of PVA/CNT electrospun films proves that they are a qualified candidate material in the tissue engineering field.

Cell viability of PVA/CNT electrospun films as related to CNT content being 0, 0.25, 0.5, 0.75, and 1 wt% (* (P < 0.05), ** (P<0.005)).
Ideal nervous conduits demand basic biological basic features, involving biocompatibility, noncytotoxic, and noncarcinogenic properties. At the same time, this material can simulate the extracellular matrix (ECM) structure while has electrical conductivity, porosity, biodegradation, multi-channels, and nervous growth factors [58,59]. The proposed PVA/CNT electrospun membranes possess aforementioned attributes and properties based on the results of physical and biological evaluations. As a result, it is substantiated that PVA/CNT electrospun membranes are a qualified material and can be considered a preliminary development.
Conclusion
PVA/CNT electrospun films are successfully developed and produced with the electrospinning technology in this study. The test results indicate that an even and finer diameter (approximate 1 μm) of fibers can be acquired when PVA/CNT electrospun films are made of 1 wt% or 0.25 wt% of CNT. Moreover, consisting of 0.25 wt% of CNT, PVA/CNT electrospun films exhibit lower resistivity of 25.3 ohm as well as a smaller water contact angle of 72.9°, indicating that they are highly conductive and hydrophilic materials. Besides, the biocompatibility test results show that co-cultured with fibroblasts for 3 days, PVA/CNT electrospun films generate a greater amount of cells than the control group, which substantiates that PVA/CNT electrospun films do not contain biotoxcitiy and have a beneficial effect on the cell growth. The aforementioned test results have proved that PVA/CNT electrospun films a qualified material for nerve conduits. Although nerve cell growth and animal evaluations are excluded in this study, the evaluations of a series of measurements, including conductivity, biodegradation, biocompatibility, MTT assay have proved that PVA/CNT electrospun films are feasible to be applied to nerve conduits. In our future studies, PVA/CNT electrospun films will be incorporated with other structures to form nerve conduits that are then evaluated for complete biological properties.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research project is financially supported by the Ministry of Science and Technology of Taiwan under the Contract MOST 109-2622-E-039-003- and CMU 108-S-02.


