Abstract
In the present study a double layer mucoadhesive buccal film containing nanocarriers encapsulated with neem extract was fabricated through electrospinning and solvent casting techniques for dental therapeutic applications. The morphological, physical and mucoadhesive properties of the resulting electrospun and solvent cast oral films were mutually compared, and their drug release behavior and antibacterial activity were further investigated. Chitosan/poly(vinylalcohol) (PVA) as a mucoadhesive component and phenylalanine amino acid nanotubes (PhNTs)-containing neem extract as a drug nanocarrier were used to fabricate oral films. A poly(caprolactone) (PCL) layer was used as an impermeable backing layer to protect the mucoadhesive component from tongue movement and drug loss. The results indicated an interconnected porous and fully filled solid structures for electrospun and solvent cast films, respectively. The physicomechanical parameters of the samples such as pH, weight, thickness, folding endurance and tensile strength were also evaluated. The crosslinked electrospun buccal film indicated better swelling and mucoadhesive properties compared to the solvent cast film. In addition, the drug loading capacity and encapsulation efficiency of the solvent cast film showed lower experimental values than those of electrospun oral film. On the other hand, the electrospun oral film had a well-controlled release of neem extract up to 82% at oral pH, which is best fitted to the Weibull model, and demonstrated the highest antibacterial properties against S. mutans bacteria with high biocompatibility on L929 fibroblast cells. Generally, the synthesized electrospun mucoadhesive film has a better potential for oral therapeutic applications than the solvent cast film.
Introduction
Buccal mucoadhesive drug delivery systems are introduced as a novel method and involve the administration of the drug through the buccal mucosa layer. These systems are considered as a functional biomaterial as opposed to structural biomaterials which are used to bear a load without being deformed [1]. Buccal drug delivery systems are used for the treatment of various diseases such as dental and oral problems [2], hypertension and atrial fibrillation treatment [3], migraine [4], pain management [5], nicotine replacement therapy [6] and for effective contraception [7]. Buccal mucoadhesion is the phenomenon of creating bonds between the mucoadhesive polymer and buccal mucosa in the first contact [8]. Buccal mucoadhesive systems include different types of drug dosage forms such as tablets, disks, wafers, films, semi-solid and liquid forms. Among them, thin and flexible films are promising dosage forms for the oral administration due to the high flexibility, desirable weight and thickness, longer drug-target residence time and good stability [8,9].
Generally, oral films have single or multilayered structure consisting of a mucoadhesive layer contained drugs or a and/or an impermeable backing layer which protects the mucoadhesive component from saliva, tongue movement and patients’ normal activities such as eating, drinking, or speaking [10]. For the fabrication of an ideal buccal film as a promising oral drug delivery system with controlled drug release and desirable characteristics, an appropriate mucoadhesive polymer and a suitable design are required. Chitosan is a biocompatible polymer obtained from deacetylation of chitin which is widely used in biomedical applications owning to its biocompatibility, non-toxicity, antioxidant characteristics as well as significant hemostatic and antibacterial activities [11–13]. In addition, chitosan is a good mucoadhesive polymer due to its cationic nature and electrostatic interaction with sialic acid-glycoprotein group in the mucus [14,15]. However, pure chitosan is not electrospinnable because of high solution surface tension [16,17]. Polyvinyl alcohol (PVA) is a biocompatible synthetic polymer which is frequently used in combination with chitosan for a successful electrospinning process. PVA has a high hydrophilicity, mucoadhesion and a high tensile strength and elasticity [18,19]. PVA/chitosan composite film is introduced as a suitable mucoadhesive film for buccal drug delivery due to their synergistic effect in the physical, chemical and mucoadhesive properties. In other words, PVA/chitosan composite films offer advantages such as improvements in stability, swelling, biocompatibility, and mechanical strength compared to the properties of the single components [18]. As well as the type of polymers, choosing a suitable design is necessary to obtain a film with desirable characteristics. In a matrix design, there is a bidirectional drug release to the mucosa and the mouth causing a partial absorption and lower drug bioavailability. In contrast, in a reservoir design, the drug is released to the absorption site and mucus layer without any drug loss and saliva penetration into the drug contained mucoadhesive layer [8]. Using drug nanocarriers in the mucoadhesive layer instead of direct loading of drug into the mucoadhesive component can enhance the bioavailability of the loaded drugs and provide a controlled drug release profile and a reduced side-effect. In addition, these nanocarriers protect the drug from enzymatic degradation [20,21]. Thus, the encapsulation of medicines such as herbal drug into the nanocarriers can prolong the drug residency time in the absorption site [22]. Nanotubes including peptides, aminoacids and carbon nanotubes are one of the most promising nanocarriers for a successful drug delivery applications. Among them, amino acid nanotubes possess high chemical diversity, good biocompatibility and ideal surface properties as well as stimuli-responsiveness to environmental factors such as pH. Phenylalanine and its derivatives are cyclic pH-responsive amino acids which can be synthesized via a self-assembly process. These types of biomaterials show reversible changes in their conformational characteristics as a function of pH and are considered to be a promising drug nanocarrier for an effective oral drug delivery [23]. Therefore, phenylalanine nanotube (PhNT) was chosen as a promising nanocarrier for a high efficient drug loading and delivery. Neem (Azadirachta indica) is a fast-growing tree with a high antibacterial, sedative and antiseptic properties. Furthermore, neem is an excellent antioxidant with a high biocompatibility to the human periodontal ligament fibroblasts [24,25]. Owing to its unique characteristics, neem extract has multiple potential uses in dentistry for oral hygiene and dental care. Many researchers claim that neem extract can successfully prevent or treat various oral-based problems such as periodontal disease, infections, tooth decay and bleeding. In other words, neem extract can successfully treat various oral-based problems such as infections, tooth decay and bleeding. Hence, using neem extract in a mucoadhesive buccal film can effectively control the dental and oral problems [26,27]. In addition, neem extract is an appealing material for reducing pain and inflammation due to the presence of a main bioactive compound known as “limonoid”. Limonoid is a furanolactone, known for its inhibitory properties and as a pain anesthetizer. In other words, Limonoid not only reduce inflammation but also work as a pain-relief agent owning to the activation of endogenous opioid pathway [28].
There are different techniques for the fabrication of mucoadhesive buccal films. Solvent casting is one of the most commonly used methods which fabricates multilayered buccal films for various applications. However, there are some limitations associated with solvent casting. Solvent cast films may contain the residual solvents which can be hazardous to patients and the environment. Moreover, there is the possibility of degradation of bioactive agents such as drugs and proteins in contact with organic solvents. Films developed by solvent casting are prone to brittleness and they have low surface-area-to-volume ratio and low drug loading capacity compared to electrospun films. The manufacturing of oral films with a suitable uniformity and quality is also problematic. It requires a huge investment to optimize the production conditions such as mixing rates, drying time and film thickness which may influence the commercial production [29]. Electrospinning is considered as a promising alternative to solvent casting method which produces amorphous fibrous films with high flexibility and plasticity, without using plasticizers, and higher surface area compared to solvent cast films [30].
In the current study, two types of mucoadhesive buccal films containing neem extract with the reservoir design were fabricated through solvent casting and electrospinning/electrospraying techniques for local treatment of oral and dental problems. The goal is to fabricate an appropriate buccal film with enhanced properties in terms of suitable mucoadhesion, high drug loading capacity, controlled drug delivery and antibacterial activity. To this aim, PVA/chitosan composite film with the ratio of 80:20 was used as ideal mucoadhesive polymers and genipin with optimum concentration was used as a crosslinking agent to control the swelling index, mechanical and mucoadhesive features. Subsequently, neem extract as an efficient herbal medicine for oral and dental problems with high antibacterial and pain relieving properties, was encapsulated into the PhNTs as a drug nanocarrier. Two fabrication methods including solvent casting and simultaneous electrospinning/electrospraying were used to prepare the final mucoadhesive oral films. The prepared buccal films were evaluated and compared in terms of physical, morphological, mechanical properties as well as swelling, mucoadhesion, drug loading capacity, in vitro drug release, cell cytotoxicity and antibacterial activity.
Materials and methods
Materials
Polyvinyl alcohol (PVA; Mw: 195000 kDa), poly(caprolactone) (PCL; Mw: 80 kDa) and phenylalanine amino acid were purchased from Sigma-Aldrich (U.K.). Chitosan (low molecular weight) was prepared from Aldrich (USA). Ethanol (96% purity), acetone and acetic acid were obtained from Merck (Germany). Genipin was purchased from Challenge Bioproducts Co., Ltd, Taiwan. Dried neem kernels were obtained from the local vegetable market, Iran. Na2HPO4.2H2O (99% purity), KH2PO4 and NaCl were purchased from Merck Millipore to prepare artificial saliva solution (pH = 6.75).
Preparation of neem extract
The dried neem seed kernels are ground into powder using a laboratory blender. Then, the dried kernel powder was added to ethanol solvent in the ratio of 1:5 (w/v) and kept for 24 hrs. Following the centrifugation process, a rotary evaporator was used to obtain neem extract. Then the remaining solvent was removed and the final extract was stored at 4 °C for further use [31].
Encapsulation of neem extract into nanocarriers
According to our recent research, PhNT was synthesized as a drug nanocarrier via a self-assembly technique with an approximate diameter of 22 nm. All the synthesis methods and results were reported in our previous study [23]. Neem extract as a sedative and antibacterial agent was loaded into the synthesized PhNTs. To this aim, 0.5 g of aqueous neem extract solution (1% w/v) was added to the nanotube solution with the ratio of (1:2 w/w) under the magnetic stirrer overnight at room temperature and the solution was exposed to ultrasonic radiations for 20 min via Ultrasonic Homogenizer (FAPAN 400 R, Iran). After 24 h, the solution was centrifuged at 6000 rpm for 20 min. The synthesized neem-encapsulated nanocarrier was used for further process.
Fabrication of solvent cast mucoadhesive buccal film
First of all, PCL film as an impermeable backing layer was prepared by solvent casting method. 3 g of PCL was dissolved in acetone/acetic acid (3:7 v/v) and a warm water bath (40 °C) was used for better dissolution of the granules. The solution (15% w/v) was stirred at 25 °C and then it was cast into a glass plate to achieve a film with a desirable thickness. The polymeric film was solidified by evaporating the solvent at room temperature overnight and then vacuum dried for 48 h. In the second stage, PVA/chitosan containing neem extract as a mucoadhesive layer was casted onto the PCL film to form a bilayer structure. First, chitosan was dissolved in acetic acid solution (2% v/v) at 45 °C to achieve a chitosan solution with the concentration of 3% w/v. PVA (8 g) was dissolved in water at 80˚C with stirring to obtain a 8% w/v solution. The solutions were mixed in the ratio of 80:20 PVA/chitosan with stirring at 60 °C for 10 min. Then, 500 mg of PhNTs containing neem extract was added into the prepared polymer solution with subsequent stirring and ultrasonication for 15 min. In order to study the effect of crosslinking agent on the final film properties, another sample containing genipin (0.05 g) as a crosslinking agent was prepared as mentioned. Finally, 0.50 g of mixture was transferred to Pyrex petri dishes and allowed to air-dry at ambient conditions for 48 h. In order to increase the adhesion of bioadhesive layer to PCL, a heat treatment at 70 °C for 20 min was applied in a dry oven. Figure 1 shows the schematic fabrication process of solvent cast mucoadhesive buccal films.

Schematic illustration of fabrication process of solvent cast mucoadhesive buccal films.
Fabrication of electrospun mucoadhesive buccal film
In order to protect the electrospun buccal film from saliva and to minimize the drug loss, a backing layer with a hydrophobicity behavior is required. To this aim, 3 g of PCL was added to a blend of acetic acid and acetone (acetone/acetic acid 3:7 v/v) to prepare a solution with the concentration of 15% w/v. Then, electrospinning was carried out with flow rate of 0.5 ml/hr at a collecting distance of 20 cm using 15 kv voltage applied to the needle tip. The prepared electrospun PCL was used as a backing layer. In the second stage, PVA/chitosan containing neem extract as a mucoadhesive layer was prepared. First, chitosan solution (3% w/v) was prepared by the addition of chitosan into an acetic acid solution (2% v/v) under the magnet stirrer at 45 °C and then it was added to a pre-heated PVA solution (8% w/v) to achieve a PVA/chitosan solution with the ratio of 80:20. Another electrospinning solution containing genipin (0.05 g) as a crosslinking agent was also prepared. To produce a bilayer buccal film, a combination of electrospinning and electrospraying methods was used. To this aim, successive electrospinning of PVA/chitosan (distance: 12 cm, voltage:15 kv and feed rate: 0.2 ml/hr) and electrospraying of neem extract encapsulated PhNTs (distance: 12 cm, voltage:15 kv and feed rate: 0.1 ml/hr) on the electrospun PCL as a backing layer was carried out. A subsequent heat treatment was performed to improve the adhesion between mucoadhesive and backing layers. Figure 2 shows the schematic fabrication process of electrospun mucoadhesive buccal films.

Schematic illustration of fabrication process of electrospun mucoadhesive buccal films.
Characterization
Physical characterization
First, to ensure the non-irritation effect of films on oral mucosa, the surface pH of the films was determined. The prepared films were immersed in 1 ml of simulated saliva solution (pH = 6.75) at room temperature and their surface pH was measured via a pH-meter (340i, WTW, Germany) by touching the film surface with the pH meter electrode. In addition, thickness of the films was measured at five distinct positions by a micrometer (INSIZE, SL-M) and the mean value was reported as the film thickness. Weight uniformity was also determined by weighing 5 films of each formulation by a calibrated digital balance and the average weight was stated.
Morphological characterization
Scanning electron microscopy (SEM) (FEI QuantaTM-200 ESEM) was used in order to study the morphology of mucoadhesive buccal films. The films were made electrically conductive by coating them with gold using the sputtering technique prior to imaging.
Determination of the swelling indices of films in simulated saliva solution
The prepared buccal films were weighted (W0) and allowed to swell in Petri dishes containing 10 ml of saliva solution at 37 °C for 30 min. Then, the samples were weighted (W1) after removing the excess water by a filter paper. The swelling index (%) was calculated using the following formula:
Contact angle
Since the most important purpose of using PCL is to protect the mucoadhesive layer containing the neem extract against saliva, it is important to study its hydrophobicity behavior. For this purpose, a drop of simulated saliva was poured on the PCL surface in both electrospun and solvent cast films. After 20 seconds, the contact angle of droplet with the surface was measured. To ensure the accuracy of the results, the experiment was repeated three times and the average contact angle was reported.
Folding endurance test
Folding endurance determines the ability of films to withstand repeated bending and folding without any breaking or cracking. Folding endurance is evaluated by repeated folding of the films at the same place [32].
Measurement of tensile strength
A tensile strength tester (Instron 5566) was used for determination of tensile strength and elongation at break. Tensile strength is the maximum stress applied to a point at which the film breaks. For the evaluation of tensile strength, the buccal films (200 mm2) were held between two fixed and movable clamps. The films were pulled by the movable clamp at a rate of 2 mm/s and then the force was measured when the films broke.
Mucoadhesive properties
The mucoadhesive properties of the films was evaluated by a texture analyzer (Farnel QTS25 model). A piece of goat buccal mucosa was moistened with a simulated saliva solution and fixed to the surface of a stainless-steel plate. Then, the prepared oral films were cut into 12 cm2 pieces and fixed to the probe. A preload of 20 g was placed over the probe for 2 minutes as a pressure for an initial attachment of the film with the mucosal membrane. The maximum required weight to detach the film from the mucosal surface presents the mucoadhesive strength [33]. Force of adhesion and bond strength were also calculated from mucoadhesive strength through equations 3 and 4, respectively.
Drug loading capacity and encapsulation efficiency of buccal films
In order to determine the neem extract loading capacity and its encapsulation efficiency in electrospun and solvent cast buccal films, the samples were added to 10 ml of distilled water. After centrifugation (6000 rpm for 15 min), the supernatant was filtered and collected for analysis by UV visible spectrophotometer at 210 nm which is attributed to the maximum absorption wavelength of neem extract. The encapsulation efficiency and loading capacity were calculated using the following equations [30]:
Release profile of neem extract
In vitro release of neem extract were evaluated in the simulated saliva solution (pH = 6.75) at 37 °C. The prepared samples were placed into the release medium with a speed of 50 rpm on the magnetic stirrer. 5 ml of samples were withdrawn at different time intervals and replenished with an equal volume of fresh saliva solution. The removed samples were analyzed via a UV-visible spectrophotometer at λmax=210 nm. According to the calibration curve, the release profile of neem extract from electrospun and solvent cast oral films were measured for 48 h.
To determine the release kinetics of neem extract from samples, different commonly used mathematical models including zero order, first order, Higuchi, Hixson-Crowell, Korsmeyer–Peppas, and Weibull were used. The values of the kinetic parameters were obtained by analyzing the data in the Excel software [23,34]. Table 1 shows the related equations for each drug release model.
Mathematical models for evaluating the release kinetics of drug.

Q: cumulative amount of drug release at time t, Qt: the amount of drug released at time t; Q0: the initial amount of drug in the formulation, Q∞: total amount of drug dissolved when the dosage form is exhausted; K: the release rate constants; n: the diffusion coefficient; m: accumulated fraction of the drug; α: scale parameter; β: shape parameter; Ti: location parameter.
Cell viability assays
Cytotoxicity and viability of L929 fibroblast cells were studied by methylthiazolydiphenyl-tetrazolium bromide (MTT) assay. L929 fibroblast cells were seeded in 96-well plates at 2 × 105/well in 300 μL culture medium and incubated at 37 °C in a 5% CO2 environment for 24 h. Then, the cells were treated with sample supernatants for two-time intervals (24 and 48 h). The supernatant was removed and 200 μL of MTT solution (5 mg/ml in the cellular culture medium) was added to the 96-well plates and incubated for 4 h at 37 °C to allow the formation of formazan crystal. The amount of formazan was directly proportional to the number of viable cells. The medium was then removed, and the blue formazan was eluted from cells using 150 μl of DMSO and the plates were shaken on an orbital shaker to solubilize the formazan crystals. After incubation (20 min, 37 °C), the optical density of the formazan solution was detected at 570 nm. Untreated cells were taken as control with 100% viability [35]. The cell viability of the samples was calculated from the average optical density (OD) values based on the following formula:
Antibacterial activity
In order to evaluate and compare the antibacterial activity of the electrospun and solvent cast films, a standard antibacterial test method of AATCC100 based on contact time was carried out. Samples were immersed in 10 ml liquid culture in a tube with a suspension of Streptococcus mutans (S. mutans) bacteria (1 × 106 CFU/ml) and incubated at 37 °C with for 12 h. Then, the bacteria suspension was diluted 100 times and 1 ml of the diluted suspension was inoculated on an agar plate at 37 °C for 24 h. In addition, an agar plate containing only bacterial solution was used as the control sample. The antibacterial efficacy (AE %) of the oral films was calculated by the following formula:
Statistical analysis
Statistical analysis of results was carried out by IBM SPSS statistics software. Results are reported as mean ± standard deviation. The statistical differences between mean values were assessed using ANOVA based on the Tukey test. A p-value of <0.05 was considered statistically significant.
Results and discussion
pH, weight uniformity and thickness measurement
Surface pH of the buccal films is one of the most important factors which should be taken into consideration to ensure that there is no potential risk of irritation or inflammation of the oral mucosa. According to Table 2, the surface pH for all samples is in the safe range showing no irritation to the buccal mucosa. The weight of non-crosslinked and crosslinked electrospun mucoadhesive films was 0.458 ± 0 and 0.470 ± 1 mg, respectively and their thickness was reported 0.45 ± 0.10 and 0.48 ± 0.11 mm which are lower than the values reported for the solvent cast films. Although both types of oral films have a weight and thickness in an ideal range [37], the electrospun films have more patient acceptance in terms of comfort compared to the solvent cast films.
Physical characteristics of the solvent cast and electrospun buccal films (µ± SD, n = 3).

Morphology of solvent cast and electrospun oral films
Figures 3 and 4 show the SEM images of the neem-containing electrospun and solvent cast oral films before and after crosslinking with genipin. Before crosslinking process, both electrospun and solvent cast films are flat. Non-crosslinked electrospun membrane has a smooth fibrous structure without beads with the average diameter of 220 nm and interconnected pores (Figure 3(a), (b), (c) and (e)), while solvent cast film is fully filled solid (Figure 4(a)). After using genipin as a crosslinking agent in the electrospun film, some bonding is observed due to the fiber fusion at crosslink sites while still showing an interconnected porous structure (Figure 3(d)). In addition, according to the diameter frequency distribution graph, the average diameter of fibers after crosslinking increased to 268 nm as shown in Figure 3(f). However, as shown in Figure 4(b), crosslinked solvent cast film has a filled solid and relatively slightly rougher structure due to the chemical crosslinking through genipin. These differences in morphology for electrospun and solvent cast films have a significant effect on their properties in terms of diffusion, mucoadhesion, dissolution and drug release capacity [38].

SEM micrographs of electrospun mucoadhesive buccal films (a, b, c: non-crosslinked films in three different magnifications, d: crosslinked film), Electrospun fiber diameter frequency distribution (e: non-crosslinked fibers, f: crosslinked fibers).

SEM micrographs of solvent cast mucoadhesive buccal films (a: non-crosslinked film, b: crosslinked film).
Swelling study
The neem-loaded electrospun and solvent cast buccal films were characterized to determine the swelling behavior through incubating samples in the simulated saliva solution (pH 6.75) at 37 °C for 24 h. According to the literature, when the oral film is placed in contact with the oral mucin, it starts to swell and various types of interactions including electrostatic attractions, Van der Waals forces, hydrogen bonding and hydrophobic interactions are formed between the polymer and oral mucosa. Therefore, swelling ratio is an important factor in mucoadhesion process and an optimum formulation is chosen based on its swelling behavior and mucoadhesive properties [39]. For an ideal buccal film, an optimum value of swelling index is needed to achieve a suitable mucoadhesion. The obtained results of swelling study are shown in Figure 5. The swelling index of non-crosslinked and crosslinked electrospun films were 279.38% and 193.66%, respectively demonstrating a significant hydrophilicity of the electrospun PVA/chitosan buccal films. Although using crosslinking agent decreases the swelling ratio, it can control the overhydration. Since, excessive swelling rate leads to a reduced mucoadhesion because of the formation of a mucilage without a complete adhesion. In other words, overhydration can decrease the adhesive strength, as a result of disentanglement at the polymer-tissue interface [40]. In comparison with the electrospun buccal film, solvent cast films have lower swelling index (101.93% for non-crosslinked and 74.11% for crosslinked solvent cast films). The reason is that electrospun oral films have large amount of water retained in the interconnected fibrous pores by the capillary action by virtue of the higher surface area [41]. Thus, solvent cast films have lower swelling ratio compared to electrospun oral films.

Swelling index (%) of different electrospun and solvent cast buccal films. Error bars represents mean ± standard deviation (n = 3).
Contact angle measurement
As previously described, the PCL film acts as a protective and impermeable backing layer in the oral films to prevent saliva from penetrating into the drug-containing layer. The contact angle test can provide useful information to express the hydrophilicity/hydrophobicity behavior of the protective layer. Therefore, the contact angle of saliva droplets on the PCL layer was measured for electrospun and solvent cast films. The values of contact angle for solvent cast and electrospun films were reported 76° and 105.64°, respectively. Although the results indicate the hydrophobic behavior of the PCL layer in both oral films, the hydrophobic properties of electrospun PCL is more than that of solvent cast film. In fact, fibrous topography of electrospun film significantly increases the contact angle of electrospun PCL compared to its solvent cast form. Since, as well as surface chemistry, topography has an important effect on the internal surface between the substrate and the liquid component. For a material that is chemically hydrophobic, the electrospinning web, due to its porous structure and trapping air molecules in the empty spaces between the fibers, prevents water droplets from penetrating into the structure and thus increases the hydrophobicity behavior [42].
Mechanical properties
The results of folding endurance and mechanical properties including tensile strength, Young’s Modulus and elongation at break for the solvent cast and electrospun films are presented in Table 3. According to the results of folding endurance, solvent cast buccal film was very brittle as it cannot be handled without cracking. However, repeated folding of electrospun film did not cause any cracks even after folding 315 times. It seems that for solvent cast film, there is an essential need to use plasticizer such as glycerol or polyethylene glycol in order to improve the flexibility and to reduce the brittleness. However, electrospun films have a substantial appropriate flexibility and plasticizer is not included in the preparation process [43].
Mechanical properties of electrospun and solvent cast buccal films (µ± SD, n = 3).
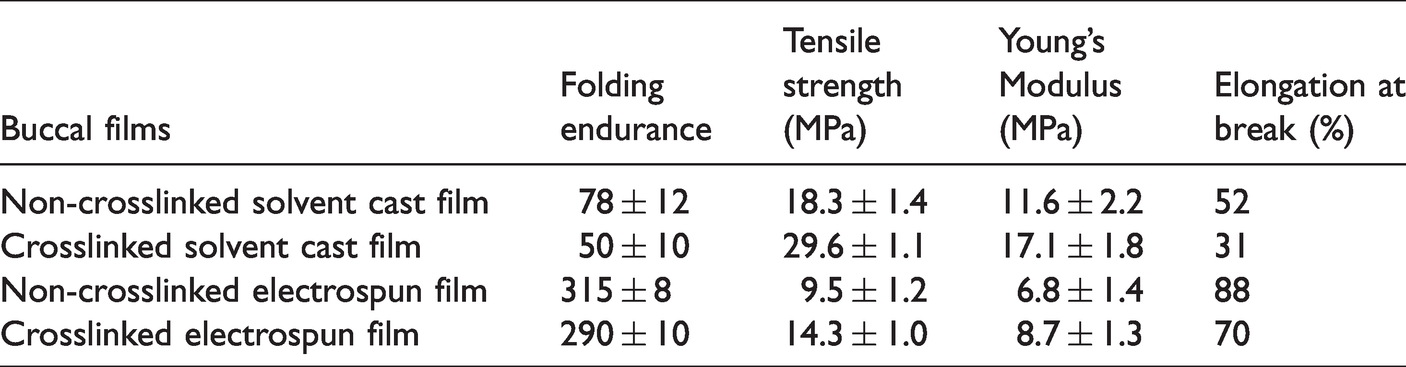
Generally a suitable buccal film should be flexible enough to follow the movement of the mouth and sustain a comfortable feel, as well as possessing sufficient mechanical strength to resist abrasion caused by tongue movement. Hence, high elongation (% elongation at break), high tensile strength and Young’s Modulus are the desirable mechanical properties of an ideal buccal film. Based on the results on Table 3, electrospun films exhibit high elongation accompanied by significant reduction in the specimen width. This high elongation due to small loads could cause significant changes in the film’s physical properties such as porosity and pore sizes which would then alter the mats expected functional performance. For optimum performance of an electrospun buccal film, a crosslinking agent can improve mechanical properties and minimize the high unrecoverable tensile elongations. The introduction of genipin as a crosslinking agent causes an increase in both tensile strength and Young’s Modulus which is attributed to the increased crosslinking density, improvement of the interaction among matrix polymers and restricted the movement of molecular chains. Therefore, the crosslinked electrospun film show a higher tensile strength (14.3 ± 1.0 Mpa) and Young’s Modulus (8.7 ± 1.3 MPa) in comparison with the non-crosslinked electrospun mat. In terms of elongation properties, the presence of genipin as a crosslinking agent leads to an increase in the network crosslinking density and a decrease in the film’s flexibility due to rigidity reasons. Thus, the elongation decreased from 88% to 70% for electrospun film in the presence of genipin. On the other hand, solvent cast films (both non-crosslinked and crosslinked films) show higher tensile strength and Young’s Modulus but low elongations at break compared to electrospun films. These differences are attributed to their different structures caused by different fabrication technologies. For solvent cast films, the material’s tensile properties are mainly influenced by the physical characteristics of the polymer and its molecular chain arrangement, whereas for randomly oriented nanofibrous mats, it is affected by fiber morphology as well as the fiber orientation and number of contact points between fibers in the mat [44]. Based on the results, it seems that crosslinked electrospun film provides a better mechanical properties in comparison with the other samples due to its sufficient tensile strength, stiffness and good flexibility.
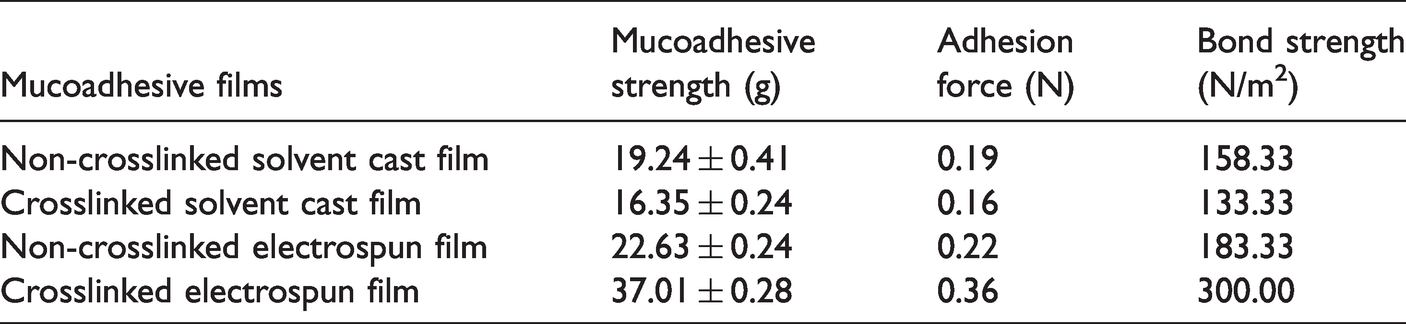
Mucoadhesive properties
Another essential factor for a buccal film is it’s mucoadhesive strength. Because, it is crucially important for the application of buccal mucosa to prolong the residence time of the drug in the absorption site [45]. The mucoadhesive properties including mucoadhesive strength, adhesion force and bond strength parameters are shown in Table 4. The results indicate that, the mucoadhesion strength of non-crosslinked electrospun film is 22.63 ± 0.24 g and it increased to 37.01 ± 0.28 g for crosslinked electrospun film. This increase is also observed for adhesion force and bond strength parameters. The reason is that in the non-crosslinked electrospun film, there is an excessive swelling making the film slippery and, as a result, prevents the mucoadhesive layer from an ideal bioadhesion to the oral mucosa. However, using crosslinking agent can control the overhydration and improve the mucoadhesive properties. Therefore, in the crosslinked electrospun film, owning to the presence of sufficient hydrophilic groups and a suitable swelling, a good mucoadhesion is achieved. In the solvent cast films, the mucoadhesive parameters are lower than the electrospun films. Since the number of functional groups for the formation of hydrogen bonds with oral mucin is insufficient. Generally, it seems that crosslinked electrospun film provides a better mucoadhesion strength, adhesion force and bond strength compared to the other samples. As mentioned earlier, electrospun oral films have higher surface-area-to-volume ratio and larger amount of water retention in the interconnected fibrous pores compared to the solvent cast films. As a result, fibers form more hydrogen bonds with oral mucin leading to a sufficient swelling and a suitable mucoadhesion.
Mucoadhesive parameters of electrospun and solvent cast buccal films (µ± SD, n = 3).
Neem extract loading capacity and encapsulation efficiency
The encapsulation efficiency and drug loading capacity values of the crosslinked electrospun and solvent cast oral films are presented in Table 5. Neem extract loading capacity in the prepared electrospun buccal film was found to be 17.62 ± 1.1%wt whereas solvent cast film showed neem extract loading of about 6.34 ± 0.51%wt. Moreover, the encapsulation efficiency of electrospun and solvent cast films were reported 95.11 ± 1.34%w/w and 73.50 ± 2.23%w/w, respectively. More encapsulation efficiency of neem extract in the electrospun film compared to the cast film is due to the higher porosity and larger surface-area-to-volume ratio. In other words, in electrospun film, the neem extract is loaded into the porous structure and gaps between nanofibers as well as loading to the PhNTs.
Data of neem extract loading capacity and encapsulation efficiency for electrospun and solvent cast films (µ± SD, n = 3).

In vitro neem extract release
The drug release behavior of the cross-linked electrospun and solvent cast buccal films are shown in Figure 6. As observed, a sustained release without serious burst of the neem extract was observed for up to 48 h. According to the literature, neem is considered as a good remedy for curing mouth ulcers and tooth decay and it acts as a pain reliever in toothache problems [46]. Therefore, fast releasing of neem extract in the first hours reduces the pain. The yield of release after 48 h from the electrospun and solvent cast mucoadhesive films was 82% and 61%, respectively. Large surface area of nanofibers membrane have some advantages over cast film in terms of drug release behavior. High porosity and large surface area of electrospun oral film cause the faster release of neem extract than that of cast oral film. Thus, the amount of neem extract release from the nanofibrous mat in the first 10 h was about 49% and it increased gradually within the rest of experimental time. This release trend shows a sustained delivery of neem from the fabricated electrospun oral film. However, in the case of solvent cast film, the drug release profile has some fluctuations and the amount of neem extract release in the first 10 h is recorded 31%.

The release profile of neem extract from electrospun and solvent cast buccal films. Error bars represents mean ± standard deviation (n = 3).
The mechanism of neem extract release from both types of buccal films can be investigated by fitting the drug release data to various mathematical models of drug release kinetics including zero-order, first order, Higuchi, Korsmeyer–Peppas, Weibull and Hixson-Crowell models [31,47,48]. These models were applied to the release profile of neem extract from solvent cast and electrospun films and their related kinetic parameters were reported in Table 6. According to the results, for both electrospun and solvent cast films, the neem extract release is best fitted with the Weibull model with an R2 of 0.9701 and 0.9597, respectively. The Weibull model describes different types of drug release as it is an empirical and generalized form of the exponential function, and has been shown previously to be the most applicable model for drug release from nanoparticles [49,50]. Weibull model is considered as a suitable model including parameters that are sensitive to ranges of drug release profile. In Weibull equation, β (shape parameter) characterises the shape of release curve and it can be linked to the physiological effects. β = 1 represents the exponential profile, while β > 1 and β < 1 demonstrate a sigmoidal form and a parabolic graph, respectively [51]. For both electrospun and solvent cast films, the shape parameter is lower than 1 (0.8696 and 0.8485), showing a parabolic drug release profile. On the other hand, the value of exponent β is an indicator of the mechanism of drug transport through the polymer matrix. In this classification, β ≤0.75 indicates Fickian diffusion in either fractal or Euclidian spaces while a combined mechanism (Fickian diffusion and Case II transport) is associated with β values in the range of 0.75 <β < 1. For β values higher than 1, the drug transport follows a complex release mechanism [52]. According to our result, the exponent β for both electrospun and solvent cast films is in the range of 0.75 <β < 1. Therefore, the drug release profile is a combination of Fickian diffusion and Case II transport. Weibull model seems to describe the release process with the major applicability compared to other mathematical models. This model is flexible enough to describe the effect of system properties on drug release process and be more applicable for identification of drug release from swellable polymeric system leading to the desired release profile in vivo [53].
Kinetic parameters of neem extract release from electrospun and solvent cast films.

Cell viability
The reduction activity of the methyl thiazolyl tetrazolium (MTT) was examined after 24 and 48 h of seeding L929 fibroblast cells on non-crosslinked/crosslinked electrospun and solvent cast buccal films containing neem extract. The results are shown in Figure 7. The MTT assays are evaluated from two diferent perspectives: First, the presence of crosslinking agent and second the fabrication method of buccal films. Although genipin is a green crosslinking agent, it has a slight minimum effect on cell viabity. Thus, it is expected to obtain lower cell viability percentage in crosslinked samples as compared to non-crosslinked ones. On the other hand, the fabrication method has considerable impact on the final structure of the buccal films. The electrospinning is giving rise to a porous structure consisting of entgalead nanofibers, whereas the solvent casting method leads to a non-porous bulk structure. The resulting porous structure of electrospun buccal films provides more surface area for cell interaction. Given the nature of synthetic materials used in the fabrication of the oral films (i.e. PVA, chitosan) as well as the remnant of crosslinking agent and neem extract within the structure, the porous structure of electrospun films exhibits slightly lower cell viability.

Cell viability of L929 fibroblast cells in the presence of different buccal films at two incubation time intervals (24 and 48 h). Error bars represents mean ± standard deviation (n = 3).
Generally, all samples have high cell viability in both time intervals and are classified in grade 1 cytotoxicity which means they are non-toxic. It means that the buccal films have excellent cell biocompatibility without any irritation to the buccal mucosa. Howeve, the presence of residual solvents in both fabrication mehtods as well as genipin and neem extract affect the cell viability (%) of L929 fibroblast cells.
Antibacterial properties
The antibacterial activity of crosslinked electrospun and solvent cast buccal films containing and without neem extract were tested against gram positive S. mutans bacterial pathogens and the results are shown in Figure 8. The reduction percentage of bacteria growth for the given samples is summarized in Table 7. According to the results, solvent cast and electrospun films without neem extract have a similar low antibacterial activity against S. mutans bacteria (41.12% and 39.74%, respectively). However, they show a slight antibacterial activity owning to the presence of amine groups of chitosan. These cationic groups electrostatically interact with negative charge of bacterial cell membrane and lead to the cell death [54]. In both electrospun and solvent cast buccal films containing neem extract, the antibacterial activity is significantly increased due to the presence of neem as a natural antibacterial agent. Neem extract has a strong antibacterial activity against pathogenic bacterial such as S. mutans owning to the presence of several active components like nimbidin, nimbin, nimbolide, gedunin, azadirachtin, mahmoodin, margolone and cyclic trisulphide [54]. In fact, neem extract penetrate into the organism and through an interaction with DNA, prevents the protein synthesis and causes cell death. The results also indicate that electrospun oral film has a greater antibacterial activity compared to solvent cast film. This phenomenon may be attributed to the more amount of the released neem extract in electrospun film as a result of its large surface area. Generally, it is concluded that neem extract and chitosan have a synergistic effect in antibacterial activity against S. mutans bacteria.

Antibacterial activity of the samples against S. mutans bacteria (a: control sample, b: solvent cast film without neem extract, c: solvent cast film containing neem extract, d: electrospun film without neem extract, e: electrospun film containing neem extract).
Antibacterial activity of samples.

Conclusion
In the current study, double layer mucoadhesive buccal films of PCL/PVA/chitosan containing neem extract-loaded PhNTs were successfully fabricated through electrospinning and solvent casting methods for use in oral and dental therapeutic applications. Chitosan and PVA have a significant effect in water absorption and swelling behavior of the oral films by virtue of their hydrophilic nature and the formation of hydrogen bonding with mucosa layer. Although both electrospun and solvent cast films had a proper surface pH, thickness and weight, electrospun samples indicated higher folding endurance and more hydrophobicity of backing layer compared to solvent cast films. The results of SEM showed that electrospun oral films had an interconnected porous structure, while solvent cast films were fully filled solid. Interconnected fibrous pores and large surface-area-to-volume ratio of electrospun film lead to higher swelling ratio and mucoadhesive properties. However, overhydration and excessive swelling cause a slippery mucilage and reduce mucoadhesion. Thus, using genipin as crosslinking agent can effectively control the swelling ratio and enhance the mucoadhesive features. As a result, crosslinked electrospun film allows more contact with the oral mucosa leading to a prolonged drug release. Neem extract as an efficient sedative and antibacterial herbal medicine for oral and dental problems was loaded into the synthesized PhNT and the drug loading capacity, encapsulation efficiency and release properties of neem extract from both electrospun and solvent cast films were determined. The neem extract loading capacity and encapsulation efficiency of electrospun film were 17.62% and 95.11%, respectively because of the high surface-area-to-volume ratio and porous structure of electrospun film were 17.62% and 95.11%, respectively. In addition, high porosity and large surface area of electrospun oral film cause the faster release of neem extract than that of cast oral film. The drug delivery profile of neem extract from electrospun oral film exhibited a relatively sustained release up to 82% at oral pH (6.75), well fitted to the Weibull model. While, the yield release for solvent cast film was reported 61% after 48 h. Furthermore, the electrospun film containing neem extract indicated an excellent antibacterial activity against S. mutans bacteria (94.67%) due to the presence of chitosan as well as sufficient amount of neem. All the samples showed excellent cell biocompatibility without any irritation to the buccal mucosa. In conclusion, the fabricated electrospun mucoadhesive film demonstrated higher mucoadhesive properties, drug loading efficiency, successful controlled drug release and antibacterial activity compared to solvent cast film. Therefore, these characteristics make electrospun oral film as a promising dosage form for clinical applications in oral medicine.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

