Abstract
In this study, the layered nanoclay was employed to impart ultraviolet (UV) protection functionality for electrospinning thermoplastic polyurethane (TPU) nanofibrous mats. This study is among the very rare studies which uses Montmorillonite (MMT) clay for UV protection. Due to its known UV protection property, titanium dioxide (TiO2) was used solely for comparison with MMT clay and as a blend with MMT for investigation of its synergetic effect with nanoclay. The morphology and chemical structure of virgin and nanoclay-loaded nanofibers were characterized via Scanning Electron Microscopy (SEM) and Fourier Transform Infared Spectroscopy (FTIR). Incorporation of TiO2 significantly improved UV protection performance of TPU electrospun nanofibers, as expected. UV protection factor (UPF) of electrospun mats including nanoclay was found as high as that of the ones including TiO2.Therefore, the study revealed that the clay has a considerable potential for producing nanofibrous layers with UV protection. Such a nanocomposite structure could be potentially employed as a layer in a multilayered technical textile such as tents, sun protective covers for automobiles, blind curtains, etc. This study proposed an eco-friendly and viable alternative to TiO2 which is a well-known material for its UV protection.
Introduction
Nanofibrous mats, having a fiber diameter less than 500 nm [1], not only offer a high surface area, high porosity, high pore volume [2] but also may be incorporated with various additives, i.e., nanoparticles, in order to impart fuctionalities such as sensing, inflammability, antibacterial protection, ultraviolet (UV) protection, etc. [3]. Although there are several methods for producing nanofibers, electrospinning is mostly preferred due to ease of use and low cost. In this process, a voltage difference between a nozzle (or a needle) and a metal collector leads to production of fibers. There are several factors affecting the morphological and the mechanical properties of the polymer and the polymer solution such as molecular weight, viscosity, and electrical conductivity, and the process parameters such as distance between needle-to-collector, feed rate of the polymer solution, and applied voltage [4].
UV radiation, having a lower wavelength and a higher frequency than visible light, covers the wavelength in a range of 100–400 nm, which is divided into three UV ray bands such as ultraviolet A (UVA, 315–400 nm), ultraviolet B (UVB, 280-315 nm), and ultraviolet C (UVC, 100–280 nm). The natural source of UV radiation is the sun, and all UVC radiation and almost 90% of UVB radiation are absorbed by ozone layer [5]. Therefore, UVA with a small portion of UVB radiation, which can still affect human health causing skin cancer, cataracts, immune system damage [6], can reach to the Earth’s surface [7]. Therefore, clothes and technical textiles with UV protection feature for various applications such as tents, sun protective covers for automobiles, blind curtains, etc. have gained importance in recent years.
Mostly, as a UV absorbing additive, TiO2, is incorporated into polymer solutions due to its photocatalytic performance, easy availability, low cost, long term stability and relatively high efficiency [8]. Lee and Lee [9] reported that the polyvinyl alcohol (PVA) nanofibers containing 2 wt.% TiO2 nanoparticles exhibited a UV protection factor (UPF) of greater than 50, indicating excellent UV protection. On the other hand, Kim et al. [10] produced very active photocatalytic graphene/carbon electrospun mats, by dipping them into a sol solution containing TiO2. In another study [11], it was reported that electrospun polyacrylonitrile (PAN) nanofibrous mats coated with TiO2 nanofilm by sol–gel process showed excellent UPF.
The studies revealed that ZnO is another powerful additive for UV protection. In the study of Fakoori and Karami [12], ZnO/polypropylene (PP) nanocomposite fibers were produced via melt spinning method, then as-prepared fibers were processed into the nonwoven fabrics by thermo-bonding method, which showed excellent protection in the wavelength of 250–300 nm. In the study of Abd Elhadyn [13], bleached cotton fabrics were padded into the suspension of chitosan/ZnO in water and the treated fabrics were found to exhibit higher UPF than that of the untreated cotton fabric. Moreover, UPF rating improved with the increase in the concentration of chitosan/ZnO nanoparticles from 0.5% to 4%. In another study [14], PAN/ZnO and PAN/MWNT composite electrospun mats produced with 5% and higher ZnO nanoparticles showed adequate UV protection. In the study of Lee [15], 100% PP nonwoven fabrics, covered with ZnO loaded polyurethane nanofibers via electrospinning process, showed UPF rating greater than 40.
Despite TiO2 and ZnO, novel additives have also been used for providing UV protection in the last few years. In the study of Qian et al. [16], rutin, a photostable chemical, was incorporated into electrospun pullulan (extracellular microbial polysaccharide)/PVA mats to obtain UV resistant properties and the UPF rating was obtained above 50 with a rutin content of 5.67% indicating comparable UV-resistance effect with traditional fabrics treated with ZnO or TiO2. In the study of Saricam et al. [17], 1,3,7-Trimethyl xanthine was used as a UV absorbant agent to develop UV protective electrospun polyamide 6.6 (PA6.6) mats and the UPF rating of nanofibers was found to be considerably enhanced to ‘‘excellent protection’’ level with 1,3,7-Trimethyl xanthine having concentrations over 15 w/w.% of the polymer.
Montmorillonite (MMT) clay has been attracting attention as a nanofiller in both polymer nanocomposites and nanofibers due to its excellent properties such as high aspect ratio, lightweight, low cost, and high stiffness [18], and its incorporation into polymeric matrix results improvement in thermal, mechanical, optical, barrier, and flame retardant properties of polymeric materials with only a small amount of loading (1–10 wt%) [19]. The final properties of clay loaded nanomaterials mostly depend on the dispersion level of the clay sheets within the polymer matrix, which requires some particular methods such as ball milling, soaking in an appropriate solvent, and ultrasonication. The known barrier property of clay make it a good candidate to be used as a protective agent against UV radiation.
There are very limited number of studies that employed nanoclay in the literature. Nanoclay was mainly used in wood polymer nanocomposites [20–23], and UV degradation test was applied in order to calculate weight loss of nanocomposites after UV exposure. In these studies, methods of compression moulding or impregnation were used for the fabrication of nanocomposite sheets, whereas our study employed electrospinning technique to produce porous nanofibrous nanocomposite mats. In addition, the developed nanocomposite mats are proposed to be used as a layer in textile-based layered systems such as tents, sun protective covers for automobiles, blind curtains, etc, while in aforementioned studies the produced nanocomposites were not proposed to be used in textile-based systems. Moreover, the previous studies focused on UV degradation behavior of polymer nanocomposites, whereas in our study we focused on UV protection behaviour of nanofibrous mats. On the other hand, to the best of our knowledge, the effect of MMT clay incorporation into PVA based electrospun mats for UV protection has only been reported by Elhami and Habibi [24]. In this study, it was revealed that the use of 3 wt. % MMT clay acted as a better barrier against UV radiation than virgin PVA nanofibers which was tested by exposing electrospun mats to UV radiation in a beaker filled with a methylene blue solution and then, measuring the absorption changes by spectrophotometer in the methylene blue solution.
The current study aimed to develop UV protective electrospun TPU layers incorporated with MMT nanoparticles for textile-based applications and to explore the use of MMT clay in UV protection that is evaluated based on UPF. Moreover, TiO2 and clay nanoparticles were used as a blend in order to investigate their synergetic effect on UV protection performance of TPU electrospun mats. Thus, MMT nanoclay was proposed as a viable and eco-friendly alternative to TiO2 for development of UV protective electrospun layers for the first time in this study.
Materials and methods
Materials
Thermoplastic polyurethane (Elastollan®C 59 D) in pellet form was purchased from BASF Chemicals. Titanium (IV) oxide (TiO2, anatase, molecular weight 79.90 g/mol, particle size 15 nm, 99% purity, Alfa Aesar) and the modified montmorillonite clay (particle size <20 µm, Nanoclay- Nanomer®, Sigma Aldrich) in powder form were used as additives for providing UV protection. N,N-Dimethylformamide (DMF, molecular weight: 73.09 g/mol, 99.5% purity, Merck Co.) and ethyl acetate (EA, molecular weight:88,10 g/mol, purity 99.5%, TEKKIM Kimya, Turkey) were used as solvents for the polymer. All materials were used as received without any purification.
Preparation of electrospinning solutions
Based on our preliminary studies with different weight ratios of TPU as 5, 10, and 15% in solution, the weight ratio of TPU was determined as 10%. The solution of TPU at a weight ratio of 5% was too dilute, whereas that of at a weight ratio of 15% was too concentrated, accordingly nanofibers could not be obtained with these polymer solutions, even if production parameters (voltage, feed rate, needle-collector distance) had been varied. On the other hand, since the boiling point of DMF is too high (153° C), it was not preferred to be used at a weight ratio of 100%, however used at a weight ratio of 80% instead. EA, having a boiling point of 77° C, had been added at weight ratio of 20%. So that the ratio of binary solvent system of DMF/EA was fixed as 80/20 wt.%. TPU was mixed in the solvent mixture of DMF/EA using a magnetic stirrer at approximately 70 °C for 6 hours. TiO2 was mixed in DMF for approximately 5 hours using a magnetic stirrer. Followingly, in order to obtain a better nanoparticle dispersion, it was ultrasonicated for 1 hour using Hielscher UP400S ultrasonicator. Meanwhile, ball milling process was performed on clay for 24 hours using MTI Corporation SFM-1 ball milling device. Subsequently, processed clay was kept in a beaker with DMF for 24 hours. Then, it was ultrasonicated for 1 hour to obtain a good dispersion. After the polymer solution and nanoparticle solution were combined, they were mixed using a magnetic stirrer for 30 minutes, ultrasonicated for 1 hour, and finally mixed using a mechanical stirrer (VBR 600, Yokes) for 15 minutes. The concentrations of TiO2 and clay additives were determined as 1, 1.5, and 2 w/w.% of polymer solution. The composition of the electrospun samples was given in Table 1.
The composition of the electrospun samples.

Electrospinning process
For the electrospinning process, an electrospinning device of Inovenso NE300 was used. The polymer solutions were fed into a 10-mL syringe. The applied voltage was in the range of 20–23 kV and tip-to-collector distance and feed rate were fixed at 15 cm and 1 ml/h, respectively. Deposition durations were determined as 1.5 and 4 hours to change the thickness of the electrospun layers. It was reported that thicker fabrics and nanofiber mats transmit less UV radiation [25–27]. Similarly, in this study, in order to eliminate the effect of thickness on UV protection of electrospun mats and demonstrate the ability of UV protection property of clay even in thin mats, the samples of 10% TPU (N1) and 10% TPU-1.5% Clay (N2) were produced at a deposition time of 1.5 hours.
Characterization of polymer solutions
Prior to electrospinning, the TPU polymer solutions were characterized by their viscosity and conductivity. Viscosity of the solutions was determined using a Brookfield DV-E digital viscometer with R2 spindle at 50 rpm. Electrical conductivity of the solutions was measured using a Windaus Winlab Data Line conductivity meter. Approximately 300 mL of samples were used for analysis at 22 ± 2°C.
Characterization and testing of electrospun mats
The thickness values of electrospun mats were measured by using a digital micrometer (Mitutoyo, 293-821).
In order to examine the morphology of nanofibers, a Tescan Vega3 scanning electron microscope (SEM), having a tungsten heated filament to create the electron beam, was used at 20.00 kV, with magnifications of 2 kx, 5 kx, 10 kx, and 25 kx. So that, for one sample four different SEM images were taken. Prior to imaging, samples were cut into a size of 1 cm x 1 cm and mounted on a stub with a double sided carbon tape and placed into a Quorum Sputter Coater to be coated with a thin layer of Au/Pd for 2 min. ImageJ software was used for measuring the diamater of the nanofibers.100 measurements were taken from each sample.
Perkin Elmer Spectrum 65 FTIR-ATR Spectrometer, equipped with an IR source, KBr beam splitter, and a LiTaO3 detector, was used in order to identify the bonds and functional groups of nanoparticles and nanofibers. For each sample, four FTIR-ATR spectra within the wavelength range of 500–4000 cm−1 were recorded with a resolution of 4 cm−1.
The spectrophotometer of Labsphere UV-2000F ultraviolet transmittance analyzer device was used in order to investigate the UV protection performance of the samples in UPF in accordance with the standard of AS/NZS 4399:1996 [28]. According to the standard, UPF is classified into three categories, i.e., ‘‘good’’ (15–24 UPF), ‘‘very good’’ (25–39 UPF), and ‘‘excellent’’ (40–50+ UPF).Textile products having a UPF value below 15 are not assessed as UV protective. Mean UVA and mean UVB transmissions, and UPF values were measured from four different locations of TPU electrospun mats including 0 wt.% and 1.5 wt.% UV absorbent particle.
Results and discussion
Morphological analysis
SEM images of the nanofibers were presented with fiber diameter distribution graphs in Figure 1(1–10). The viscosity and conductivity values of polymer solutions, and average fiber diameters produced from these solutions were given in Table 2. Nanoparticle addition can increase the viscosity of polymer solutions [29,30] as well as cause bead formation [31,32]. On the other hand, some nanoparticles can enhance electrical conductivity [29,30], which can reduce fiber diameter. In the current study, it was observed that addition of nanoparticles into TPU polymer solution decreased average fiber diameters. A decrease in fiber diameter was expected with addition of TiO2 nanoparticles due to the semiconductive nature of TiO2, which contributed to an increase in conductivity of the solutions (Table 2), and generated more stable electrospinning jet [33]. In addition, fibers (Figure 1(1–3)) were produced in continuous form with only a few circular beads with an average diameter of 368.8 ± 36 nm (shown with arrows) that may have formed due to nanoparticle agglomeration. On the other hand, the montmorillonite clay that was used in this study was a surface-modified clay with trimethyl stearyl ammonium chloride. It has been reported that the ammonium salt present as an organic surface modifier in the nanoclays improves the electrical conductivity (Table 2) of the polymer solution therefore, the diameter of fibers decreased [34]. On the other hand, some non-homogenous surface structures such as circular beads with an average diameter of 378.8 ± 15 nm (shown with arrows) in SEM images (Figure 1(4–6)) were most probably due to aggregation of clay. In addition, only in a few SEM images (Figure 1(6–8)) elongated beaded structures were observed.

SEM images of nanofibers (SEM photographs magnification: 10 kx). 1 (10% TPU/1% TiO2), 2 (10% TPU/1.5% TiO2), 3 (10% TPU/2% TiO2), 4 (10% TPU/1% Clay), 5 (10% TPU/1.5% Clay), 6 (10% TPU/2% Clay), 7 (10% TPU/1% TiO2-Clay), 8 (10% TPU/1.5% TiO2-Clay), 9 (10% TPU/2% TiO2-Clay), and 10 (10% TPU).
Polymer solution properties (viscosity and conductivity) and mean diameter of nanofibers produced by the electrospinning process.

As a conclusion, even if nanoparticle addition into TPU polymer solution decreased fiber diameters substantially due to enhanced conductivity, the TPU nanofibers including no nanoparticle were in a more continuous form (Figure 1(10)) compared to those of including TiO2 or nanoclay.
FT-IR spectrum analysis
The chemical structure of TPU is presented in Figure 2. The FT-IR spectra of TPU polymer, TiO2 and clay alone were given in Figure 3. The FT-IR spectrum of TPU has a peak at 3300 cm−1 that is attributed to the stretching vibration of N-H bond in urethane group. The characteristic bands of TPU were recorded at 1699, 1531, and 1309 cm−1 due to the C = O stretching vibration (amide I band), N–H bending vibration (amide II band) and C–N (amide III band) stretching vibration, respectively. The peaks at 2917 cm−1 and 2849 cm−1 in the FT-IR spectra can be attributed to the asymmetric and symmetric stretching vibrations of -CH bonds [35,36]. In the FT-IR spectra of TiO2, the broad band centered at 500–600 cm−1 was observed due to the bending vibration of Ti-O-Ti bonds in the TiO2 lattice [37]. In the FT-IR spectra of clay, the absorption peaks can be related to the fundamental stretching and bending vibrations of its basic functional groups of O-H and Si-O. The stretching and bending vibration peaks of O-H groups were recorded at 3700–3500 and 950–600 cm−1 regions, respectively. The peaks at 2924 cm−1 and 2848 cm−1 showed stretching of C-H aliphatic bonds [38]. In addition, Si-O stretching vibrations were recorded at 1000 cm−1 region, and the Si-O bending bands were recorded between 600–400 cm−1 [39].
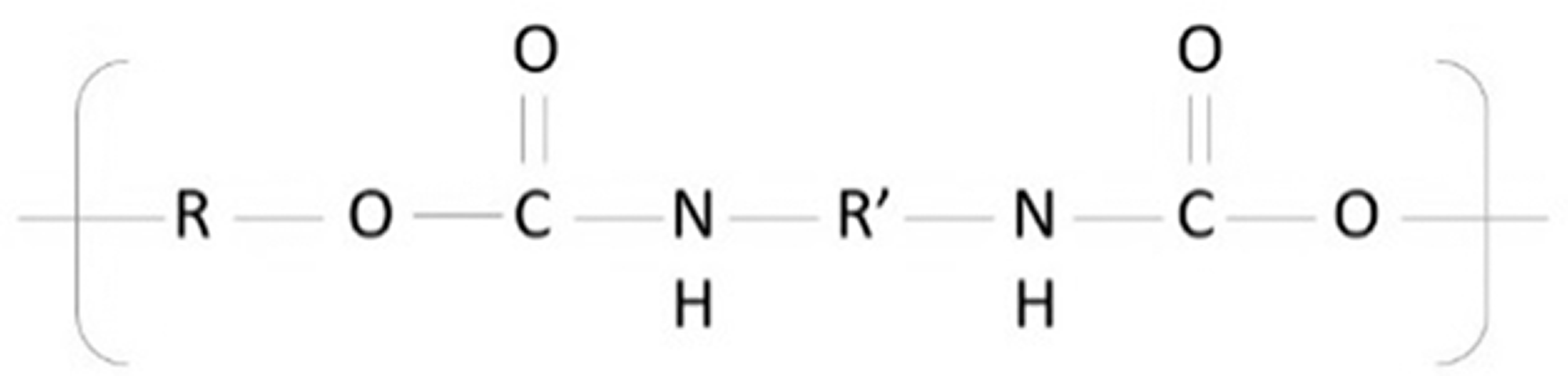
Chemical structure of TPU [40].

FT-IR spectra of TPU polymer, TiO2 and clay.
Figures 4, 5 and 6 show the FT-IR spectra of electrospun TPU nanofibers with TiO2, with clay, with both TiO2 and clay at a weight ratio of 1, 1.5, 2%, and without any additives recorded within the range of 450–4000 cm−1, respectively. In these spectra, even if specific peaks of TPU were dominant, specific peaks of nanoparticles were also observed and indicated on the FT-IR spectra.
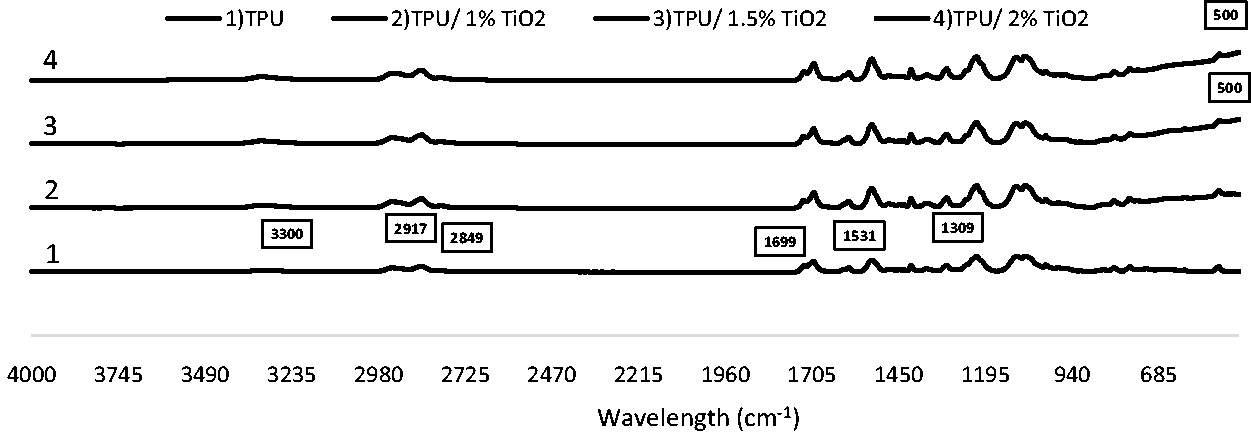
The FT-IR spectra of TPU and nanofibers including TiO2.

The FT-IR spectra of TPU and nanofibers including clay.

The FT-IR spectra of TPU and nanofibers including both TiO2 and clay.
In order to make a comparison between peaks of TPU nanofibers including TiO2 or clay, FT-IR spectra of TPU nanofibers with 2% TiO2, 2% clay, 2% TiO2-clay, and without any additives were given in Figure 7. Although small shifts can be observed at the TPU peaks due to the addition of clay or TiO2 into the TPU matrix, the characteristic peaks of these nanoparticles can also be recognized in the FT-IR spectrum at 1000–400 cm−1 region (shown with a rectangle in Figure 7) indicating that the clay or TiO2 were successfully incorporated into the nanofibers. The characteristic peaks of clay were observed in the spectrum of TPU/2% clay at 1000 cm−1 and at 400 cm−1, whereas that of TiO2 was observed in the spectrum of TPU/2% TiO2 at 500–600 cm−1.
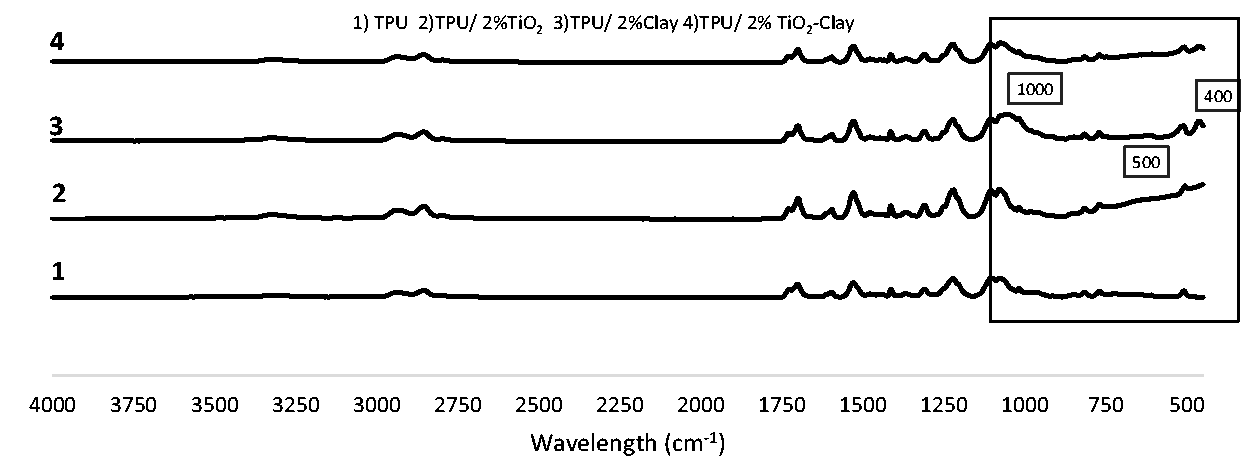
The FT-IR spectra of TPU and nanofibers including TiO2, clay and both TiO2 and clay at a weight ratio of 2%.
UV protection property
The mean UVA and UVB transmission and UPF results of the electrospun mats were given in Table 3.
UV protection performance of the electrospun mats.

In Table 3, it can be seen that addition of either TiO2 or clay into TPU polymer matrix at a concentration of 1.5 wt.% provided an increment in the UPF values compared to that of TPU nanofibers without any UV absorbent additives. The UPF value of electrospun TPU mats with 4 hours deposition time containing either TiO2 or clay was 2000 which corresponded to UPF rating of 50+, and defined as ‘‘excellent protection’’. It was expected to obtain a high UPF value for the nanofibers containing TiO2 due to its known UV protection property against UVA and UVB type radiations [9–11]. The conspicuous results of this study is that the electrospun mats containing clay showed the same UV protective performance as that of TiO2, indicating that clay has an obvious potential to be used in UV protection applications [20–24]. On the other hand, even if the UPF value of the electrospun mats containing both TiO2 and clay was higher than that of containing no UV absorbent additive, it was lower than that of containing only one type of additive (TiO2 or clay). It can be said that using TiO2 and clay in the same TPU polymer matrix had no synergetic effect.
Moreover, the UPF values of the thin samples (N1 and N2) that are produced at a duration of 1.5 hours were also given in Table 3. It can be seen that when the process duration decreased at a ratio of 65% (from 4 hours to 1.5 hours), UPF value of electrospun mat without any nanoparticle decreased at a ratio of 90% (from 1431.15 to 156.38), whereas UPF value of electrospun mat including 1.5 wt.% clay decreased at a ratio of 55%(from 2000 to 900.73).This result implies that considering the decrease rate in the UV protection performances, as the thickness of the electrospun mats continues to decrease [26,27], the one without clay would most probably lose its UV protection capability while the one with clay would still be classified as UV protective. Therefore, the use of clay in electrospun mats for UV protection brings the advantages in the electrospinning process i.e. a considerable reduction in duration and in the production of multilayered technical textiles where lightweight and breathability are critical. As a result, this study shows that MMT nanoclay is a promising additive material to develop UV protective electrospun layers.
Conclusion
In this study, nanoclay and TiO2 nanoparticle-loaded TPU nanofibers were successfully produced for developing UV protective electrospun mats. SEM analysis indicated that TPU fibers were obtained at nanoscale and nanoparticles were well dispersed with formation of some nanoscale aggregates. FTIR analysis indicated that the spectra are mainly dominated by the characteristic peaks of TPU. The main peaks of MMT clay and TiO2 were also observed in the spectra, which means that the nanoparticles were successfully loaded into the TPU electrospun mats.
UV protection analysis showed that TPU electrospun mats containing either TiO2 or nanoclay (1.5 wt.%) had a UPF value of 2000, which indicated that MMT nanoclay is a promising UV absorbent additive material as an alternative to TiO2. For lower deposition times, UPF value of TPU electrospun mat without any UV absorbent additive decreased at a ratio of 90%, whereas UPF value of TPU electrospun mat including 1.5 wt.% nanoclay decreased at a ratio of 55%. The use of clay was found to be advantageous in the fabrication of UV protective electrospun mats at low deposition durations leading to lightweight and breathable nanofibrous layers, which would find applications in multilayered technical textiles.
Footnotes
Acknowledgements
The support of undergraduate senior design students in the experimental studies is also acknowledged by the authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to acknowledge the financial support provided by Istanbul Technical University Scientific Research Project Office under the project number “BAP 36171”.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
