Abstract
Nano (or) microfiber structures made from biodegradable polymers offer many benefits for biomedical applications, including the tissue engineering system. In this study, a mixture of poly (lactic acid) (PLA)/gelatin (GEL) conjugated epidermal growth factor (EGF) electrospun nanofiber scaffolds, which may have potential applications for nursing care of diabetic wound healing. The amount of EGF conjugation on the PLA/GEL nanofibers was quantitated by X-ray photoelectron spectroscopy. A morphological examination of the electrospun nanofibers was performed using scanning electron microscopy (SEM) and transmission electron microscopy (TEM), which has bead-free morphology nanofibers. After characterizing, the PLA/GEL and PLA/GEL/EGF scaffolds were selected for further investigation based on their superior mechanical properties, including tensile strength, maximum elongation, and tensile modulus. We evaluated the antibacterial activity, cell proliferation and the efficiency of diabetic wound healing in vivo. The antibacterial effect of EGF present in the PLA/GEL nanofibers was assessed using the zone inhibition test against S. aureus and E. coli. In vitro cell proliferation activity was well maintained after the PLA/GEL conjugation and was compared to that of EGF with L929 fibroblast cells. The nanofiber PLA/GEL conjugated with EGF exerted greater curative activities in vivo compared to the control groups or EGF solutions. This study showed that the nanofiber PLA/GEL in combination with EGF could potentially be used as a novel material for nursing care of wound healing by antibacterial, increasing inflammatory cell, and re-epithelialization.
Introduction
Wound healing is one of the greatest challenges in medical and biomedical research because it is a complex process [1]. Diabetic foot problems are recognized as the most serious health problems in the world and complications, including peripheral neuropathy, eye disease, kidney disease, cardiovascular disease, and chronic wounds, become life-threatening [2,3]. Moreover, foot ulceration is the major complication of diabetes mellitus with a lifetime risk of 15% in all diabetic patients [4]. The exploration of therapeutic methods of wound healing for this population is very urgent and necessary. Our studies to solve this difficult clinical problem focus on the improvement of dressings that can improve wound healing. Many cells and factors influence the process of wound healing and slow or quick tissue manipulation. Important cell survival factors, such as stimulate cell migration, vascular endothelial growth factor, platelet-derived growth factor, and epidermal growth factor (EGF), angiogenesis and proliferation, which are crucial for wound healing and successful tissue engineering [5,6]. Consequently, several growth factors are used in the clinical analysis to treat wounds to quickly repair wounds and completely restore clean, scar-free skin.
Therefore, growth factors have been projected as therapeutic agents to enhance wound healing. Epidermal growth factor (EGF) is a unique 53 amino acid residue polypeptide chain and contains three intramolecular disulfide bonds required for biological activity [7]. One such potent factor for cell expansion is EGF, which activates signaling pathways to promote migration, proliferation, and survival without affecting the pluripotency of stem cells and also it enhances proliferation, and migration of fibroblast, keratinocyte, and endothelial cells at the wound spot. Initial clinical studies have shown that topical administration of EGF increases epithelialization and shortens healing time in venous ulcers, skin grafts, and diabetic foot ulcers [8,9]. The lack of data demonstrating a significant benefit from its application, as well as data indicating that EGF plays an important role in cancer development, has significantly hampered subsequent therapeutic use. Growth factors are also attractive composites for encapsulation in nanofiber dressings, as they are vital healing linkages for effective tissue repair [10]. Because the functions of EGF are known to be dependent on their spatial distribution, controlling the delivery of growth factors both spatially and temporally is crucial for their effective and safe use as regenerative medicines in clinical practice. Therefore, incorporation in electrospun nanofibers can overcome this restraint as these preparations protect EGF from the environment and thus improve its stability.
Electrospun nanofibers membranes are characterized by their mechanical strength, their high surface-to-volume ratio and their porous structures on a nanometric scale, which improves wound healing to various mechanisms [11]. High voltage transformations between the injection needles and the ground cause the charged polymer jets at the tip of the injection needle to be injected by electrostatic repulsion. Recently, nanofibers have been applied to medical applications such as drug delivery systems, tissue engineering scaffolds, vascular grafts, biological dressings and support for the human body [12–14]. As a bioactive polymer matrix, synthetic and natural polymers are widely prepared in nanofiber scaffolds for biomedical applications such as wounds [15–17]. Synthetic polymers used in electrospinning include extra-cellular matrix (ECM) components, such as polylactic-co-glycolic acid (PLGA), poly(vinyl alcohol), poly (lactic acid), polyglycerol, polyglycolide, polyurethane, etc. [18]. Synthetic materials are stronger and economical, have a distinct structure and are easier to electrospinning. Of the several biomaterials used in the synthesis of a biodegradable polymer scaffold, poly (lactic acid) (PLA) has currently acknowledged superior attention from researchers [19–22]. The improvement of mechanical properties and heat resistance of PLA-based materials is a matter of concern when they are used in biomedical and commodity applications [22]. PLA befits features including cytocompatibility, biodegradability and biocompatibility which makes it a great potential for implantation devices. The fiber diameter of nanofibers has been shown to regulate the spread, orientation and proliferation of osteoblastic cells as the main factor of temporary skin coverings or dressings. A major disadvantage of using PLA in the aqueous medium is the high solubility and degree of swelling [23]. Among the natural polymers used for electrospinning are chitosan, agarose, gelatin, pectin, silk, etc [24]. More importantly, the crosslinked PLA with gelatin (GEL) natural polymers improved the mechanical properties with a controllable degradation time [25–27]. The properties of these polymers (matrix cell interactions, non-toxicity, biocompatibility, and physicochemical properties) have made them suitable for wound healing [28–30].
In the current study, the aim was to develop a cross-linked PLA/GEL electrospun nanofibers loaded with EGF for the treatment of diabetic wound healing (Scheme 1). The PLA/GEL electrospun nanofibers were prepared by electrospinning and then subjected to mechanical, and biological in vivo evaluations. By improving the fabrication method of these two electrospun nanofibers, new strategies can be established to improve the transparency and mechanical properties of the nanofibers, making them more suitable for the endothelium of tissue engineering. GEL was used as the shell layer material because of excellent biocompatibility than PLA, which is beneficial for fabric engineering purposes. After conjugation of EGF on the surface of the PLA/GEL nanofibers, the wound healing effects of the PLA/GEL/EGF nanofibers were assessed for their potential applications in the healing of diabetic wounds.

The experimental approach of preparing PLA/GEL conjugated EGF nanofibers (PLA/GEL/EGF) with wound healing treatments for diabetic ulcers.
Experimental details
Materials
Polylactic acid (Mw = 186,000, Mw/Mn = 1.76) and Gelatin (type A, 240 bloom) was purchased from Jinan Daigang Biomaterial Co., Ltd. (China). Trifluoroacetic acid (TFA) was purchased from Sigma-Aldrich Chemical Co. The two polymers were dried before use and all materials were used without further purification. The highly purified EGF (99%) is supplied by Collaborative Research, our institute (China). It is extracted from the submaxillary salivary glands of mice according to the Savage & Cohen method. It was dissolved in 0–154 M NaCl at a final concentration of 50 µg/ml, fresh from the experiment every day. All general chemicals and buffer reagents (reagent grade or higher) were purchased from Sigma Aldrich company.
Electrospun nanofibers preparation
An emulsion containing EGF, PLA, and GEL were prepared a PLA/GEL and PLA/GEL/EGF electrospun nanofibers. Mixing solutions of PLA (1.0 g) and GEL (0.2 g) were prepared by dissolving them in 20 ml TFA and stirring at 50°C for 24 hours, after which a homoGELeous mixture solution PLA/GEL was produced. The amount of EGF (6 wt%; 60 mg/ml) was weighed and dispersed in the combination above prepared PLA/GEL solution to the mixture of PLA/GEL/EGF. The rate of addition two phases was set at 3 min. and the mixer speed was set at 300 rpm to form the emulsion (Vortex-genie 2, Scientific Industries Inc., USA). For electrospinning, the prepared polymer solutions PLA/GEL and PLA/GEL/EGF were transferred to a 5.0 ml syringe at a constant flow rate of 0.5 ml/h using an infusion pump (Veryark TCV-IV, China), with a needle-to-collector distance 12 cm and a voltage of 12 kV. Electrospinning is performed with the use of a horizontal instrument assembled and a piece of metal substrate (such as an aluminum foil) as the collector. The injected volume was normalized to obtain membranes of comparable thickness. The resulting PLA/GEL and PLA/GEL/EGF electrospun nanofibers were collected at room temperature and then dried under vacuum for at least 48 hours to complete the removal of any residual solvent. Finally, before the in vitro and in vivo studies the membranes were exposed to UV light (6 mV/cm2, 365 nm, 30 min).
Electrospun nanofibers characterization
The composition of the synthesized electrospun nanofibers materials was evaluated using the X-ray photoelectron spectroscopic technique (XPS ESCALab250, Thermo Scientific). The morphology of the electrospun nanofibers, i.e. membrane quality and fiber diameter, was analyzed using scanning electron microscopy images (SEM, Jeol JSM-6300). Transmission electron microscopy (TEM) images were obtained from electrospun PLA/GEL and PLA/GEL/EGF scaffolds and were imaged with Philips Tecnai G2 F30 ST at an acceleration voltage of 200 kV. The sample was prepared by air-drying with dilute dispersions of PLA/GEL and PLA/GEL/EGF on carbon films supported by copper grids.
Measurement of mechanical properties
For the analysis of the mechanical properties (tensile strength, elongation and tensile modulus) of the PLA, GEL, PLA/GEL and PLA/GEL/EGF electrospun nanofibers were measured with an Instron 5848 microtester, at a rate of deformation of 10 mm/min (Instron1, UK). All samples are cut into rectangles of 25 mm ± 4 mm. This system is based on the extension of the rate. The thicknesses of the samples were measured with a micrometer with an accuracy of 1 µm. The stress-strain was reported, and the maximum stress and strain were calculated (n = 3).
In vitro antibacterial test
The antimicrobial activity of the electrospun nanofibers PLA/GEL and PLA/GEL/EGF was examined by the zone inhibition method. Gram-positive Staphylococcus aureus (SA 29213) and Gram-negative Escherichia coli (E. coli 8739) were chosen as model microorganisms in this study. All bacterial cultures came from the American Type Culture Collection (ATCC, Manassas, VA, USA). Briefly, 1.0 ml of bacterial suspension containing approximately 108 CFU/ml for each bacterium was spread as stock onto nutrient agar plates. A volume of 100 µl PBS solution containing the two bacteria was added dropwise to the surface of each 1.5 x 1.5 cm fiber (PLA/GEL and PLA/GEL/EGF). The bacterial growth on the plate was immediately visualized, after 48 hours incubation of the plates at 37 °C, the diameter of the inhibitory zone was measured.
Cell viability and cell attachment
Cell viability was assessed using the MTT assay (3-[4, 5- dimethylthiazol-2-yl]-2, 5 diphenyltetrazolium bromide) 2 hours after cell seeding (fibroblast cells L929). The ultra-fine fiber scaffolds (0.1 cm2) in Dulbecco medium (DMEM) supplemented with penicillin (100 µg/ml), streptomycin (100 µg/ml) and fetal bovine serum (5% FBS). The samples were then incubated for 24 hours at 5% CO2 and 37 °C. After 24 hours, the medium was replaced with a fresh medium containing PLA/GEL and PLA/GEL/EGF at a concentration of 100, 200 and 300 μg/ml. After the incubation period, 100 μL medium with MTT (1 mg/ml) for 4 hours. The MTT solution was carefully replaced and 150 µL DMSO was added to each well. Cell viability was calculated from the following equation:
The cell attachment on the channel was studied using a fluorescent microscope with an optical filter of 330 and 450 nm excitation and emission wavelengths, respectively. The cells were seeded into a 96-well plate at a density of 6000 cells/well and grown overnight to allow attachment of the cells. The complete culture medium as a control. After 24 hours of cell seeding, the cells were stained with propidium iodide (PI; 10 µg/ml) and acridine orange (OA; 5 µg/ml). A PI/OA solution (concentration 1:500) was mixed to the cell channel constructions and incubated at ambient temperature for 5 minutes. To remove unbound PI/AO, the cell lines were washed three times with PBS. The growth condition of the chondrocytes was observed and the biological characteristics of the PLA/GEL and PLA/GEL/EGF electrospun nanofibers were assessed in vitro.
In-vivo wound healing and histopathological analysis
To access the anti-infective healing properties of the samples, we constructed the full thickness wound model with female Kunming mice (4∼7 weeks) on the back. All experimental analysis of in vivo animals was conducted under Home Office Project License No. 0736/2019 and IACUC Guidelines and in accordance with all the experiments was approved by ethical committee of Harbin Medical University, PR China. The mice were purchased from Harbin Medical University. The full-thickness excision wound model was generated in this study to evaluate the healing performance of PLA/GEL and PLA/GEL/EGF electrospun nanofibers. To create a wound in the animals, the mice's hair was completely shaved and hot round metal sticks (4 mm in diameter) were brought into contact with the hairless skin. The mice were randomly divided into 3 groups (n = 3). The excised wounds were covered with PLA (control) and an electrospun PLA/GEL and PLA/GEL/EGF membrane fixed with an elastic adhesive bandage and left untreated. According to the experimental conditions, the PLA was sown on fiber mats for 4 hours before fibrous tissue has adhered to the wound site. The photos of the injuries were taken with a digital camera to measure wound closure rate and wound size reduction was calculated using the equation as follows:
For histopathological analysis, animals were sacrificed under anesthesia and the wounds were dissected for histopathological examinations at the end of the 6th day after injury. The whole wound tissue with adjacent normal skin was obtained on days 3 and 6, immersed in 4% buffered paraformaldehyde and incorporated paraffin, sectioned and stained with hematoxylin and eosin. All rats were housed separately to avoid additional effects on the wound area.
Statistical analyzes
All experiments were repeated at least three times. The experimental data were expressed as mean ± standard deviation (SD). The statistical consequence was checked by running a Student t-test and data with a p-value <0.05 was considered significant.
Results and discussion
Characterizations of electrospun nanofibers
To determine the amount of EGF on the electrospun nanofiber PLA/GEL after the biotransformation reaction, XPS was used to quantify the amount of nitrogen in the electrospun nanofiber. As shown in Figure 1(a) and (b), XPS data showed that PLA/GEL and in the presence of EGF nanofibers are presented in the elements carbon (C), oxygen (O) and also nitrogen (N) showed C1s, O1s and N1s peaks [31]. Since the EGF molecule covers 73 N atoms, the concentration of EGF on the PLA/GEL nanofiber could be accurately calculated to the carbon atoms of PLA and GEL in the cross-linked polymer. The intensity of the N1s peak in the range of 399.5 eV was increased in the nanofiber PLA/GEL/EGF due to the interaction of EGF with the surface of PLA/GEL. XPS results showed that the elemental compositions of C, O, and N in the PLA/GEL/EGF nanofiber were 48.3%, 34.6%, and 17.1% respectively in the study of the survey scan performed for C1s, O1s and N1s [32]. In the case of untreated EGF, XPS survey spectra revealed atomic percentages of C, O, and N equal to 70.2%, 27.3%, and 2.5%, respectively (Figure 1). The N elemental composition was found to increase from 2.5% to 17.1% in their elemental composition, strongly suggesting EGF successful grafting with PLA/GEL, which was further supported by high-resolution XPS analyses. Figure 1(c) and (d) shows the narrow scan N 1 s spectrum for PLA/GEL, which were fitted with peaks at about 399.18 eV (N-C) and 401.78 eV (N-H) and PLA/GEL/EGF for 398.48 eV (-N-H), 399.18 eV (-C = N), and 399.48 eV (N-C), corresponding to N element, respectively. However, it should also be noted that many nitrogen atoms in the PLA/GEL electrospun nanofibers have been considered because their amount was very high compared to that of EGF.

X-ray photoelectron spectroscopy (XPS) of survey scan (a) PLA/GEL nanofiber without EGF, (b) EGF-conjugated PLA/GEL nanofiber (PLA/GEL/EGF), (c) N element oxidation states of PLA/GEL nanofiber and (d) N element oxidation states of PLA/GEL/EGF nanofiber.
The SEM images of PLA, GEL, PLA/GEL, and PLA/GEL/EGF showed that there was an important morphological change in the uniform and randomly oriented nanofiber scaffolds shown in Figure 2(a) to (f). According to the images, there is a well-formed nanofiber in the membranes produced by the core nanomaterials PLA/GEL and PLA/GEL/EGF due to axial electrospinning with a solution concentration of 10% (PLA/GEL) [33]. The thickness of the different membranes was comparable, with a value of 96.64 ± 0.52 µm and 63.12 ± 0.79 µm for the PLA/GEL and PLA/GEL/EGF nanofibers, respectively. The presence of EGF in PLA/GEL electrospun nanofibers enhanced the electrospinning of the polymer solution and appears to be clearer and uniform in bead-free fibers. However, the diameter of the nanofibers varies between the membranes, being 386.52 ± 83.61 nm in the PLA/GEL nanofibers and 259.35 ± 99.24 nm in the PLA/GEL/EGF nanofibers in Figure 2 [34]. The lower diameter of PLA/GEL/EGF have a greater surface area than PLA/GEL nanofibers produced by mainly result from its strong hydrogen bonding properties for EGF dissolution and the increase in applied voltage led to a decrease in fiber diameter. As the voltage was finally increased to 12 kV, the fiber diameter decreased, whereas a structure with bead-free was formed. It was also possible to produce nanofiber membranes with a lower concentration of PLA/GEL/EGF mixture solution. The above results indicate that the synthesized PLA/GEL and PLA/GEL/EGF electrospun nanofibers are smooth, continuous and suggest that the addition of EGF has a significant effect on the morphology of nanofibers. An additional study was conducted to highlight that small changes in molecular weight can strongly influence solution viscosity and resulting fiber morphology. First, the effect of solution viscosity on electrospun fiber morphology was investigated by mixing solutions of PLA/GEL in EGF at concentrations ranging from 10 to 30 wt%. As expected, increasing solution concentration resulted in increased viscosity. Representative images of fibers spun at low, intermediate, and high viscosity revealed beaded fibers at low viscosity, uniform fibers at an intermediate viscosity, and larger fibers at high viscosity.

SEM photographs of (a) PLA, (b) GEL, electrospun nanofibers of (c) PLA/GEL and (d) PLA/GEL/EGF. Diameter distribution of the nanofibers (e) PLA/GEL and (f) PLA/GEL/EGF nanofibers.
The development of the core structure in the electrospun axial nanofibers of PLA/GEL and PLA/GEL/EGF was confirmed by TEM as a piece of solid evidence is shown in Figure 3. Since a thin layer of nanofibers (∼200 nm) had to be placed on the copper grid to obtain a clear TEM image, the axial electrospinning was made on the grid plated on the collector for 60 s. The PLA/GEL morphology was spherical with a diameter of 198 nm [35]. Also, the morphology of the nanofibers and the presence of EGF on the PLA/GEL nanofibers were obtained in Figure 3(a) and (b). The diameter of the prepared nanofibers was about 172 nm. Therefore, TEM images confirmed the presence of EGF on the surface of the PLA/GEL nanofibers [36]. Uniform distribution of EGF is essential to improve the mechanical and biological properties (cell attachment and proliferation) of the membranes. It can be recognized in Figure 3 that the formation of nanofibers PLA/GEL and PLA/GEL/EGF has the selected core assembly with a clear boundary.

TEM photographs of (a) PLA/GEL and (b) PLA/GEL/EGF nanofibers.
Mechanical properties of electrospun nanofibers
To confirm the integrity of the membranes, the mechanical properties of electrospun nanofibers are significant for effective application in wound dressings. The tensile strength, maximum elongation and tensile modulus of the prepared nanofibers and pure materials are illustrated in Figure 4(a). It is noted that the addition of EGF to PLA/GEL electrospun nanofibers had a variable effect on tensile strength and total elongation [37]. It appears that the PLA/GEL and PLA/GEL/EGF nanofiber scaffolds showed tensile strengths of 3.4 ± 0.4 and 4.7 ± 0.2 MPa, respectively. (Figure 4(b)). Adding EGF to the PLA/GEL nanofibers made the scaffolding flexible and significantly increased their elongation at break and their average tensile strength. This was likely due to the combined effect of EGF content and PLA crosslinking of GEL, which both had a beneficial effect in improving the mechanical properties of the PLA/GEL/EGF nanofibers [38]. The core assembly membrane has a different structure than the non-crosslinked sample because the shell composed by GEL broke and detached from the PLA core due to the stiffness of GEL generated by the crosslinking reaction.
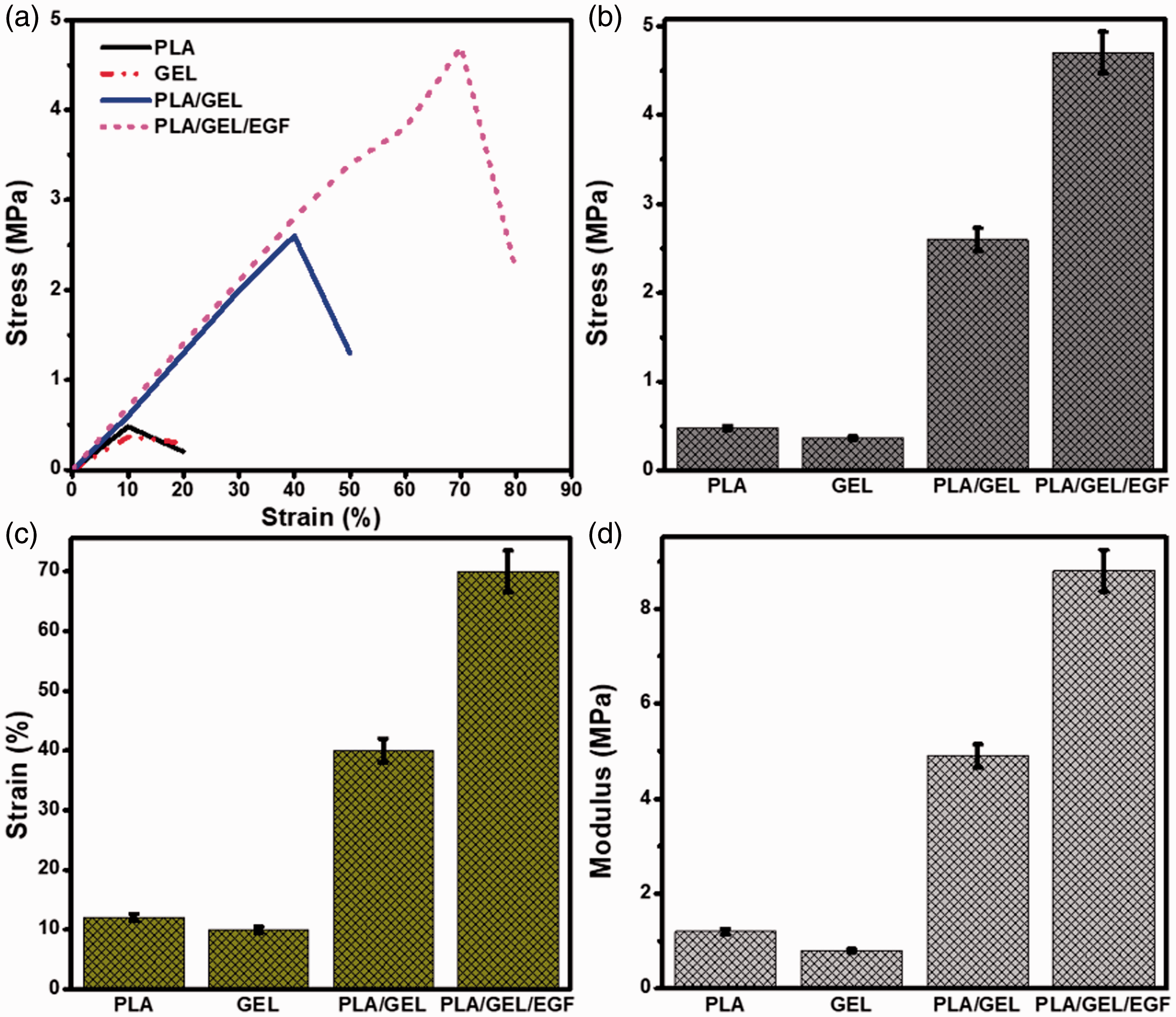
Mechanical properties comparison of (a) Stress-strain curve (b) Tensile strength (c) Elongation (d) Tensile modulus.
The elongation at break values represents the flexibility of electrospun nanofibers. The pure PLA, GEL and also prepared nanofiber scaffolds exhibited an elongation in the range from 10% to 70% at the breakpoints (Figure 4(c)) [39,40]. The result of the tensile modulus (Figure 4(d)) was consistent with the results of the stress test. The nanoscaled dimensions in the cross-sectional direction and the macroscopic length of the nanofiber axis. In addition, 3-D nanofiber network assemblies provide good mechanical properties and good handling characteristics. The Young’s modulus graph measured on the nanofiber cross sections are shown in Figure 4, where data clearly evidence the presence of a stiffer internal region nearby the nanofiber longitudinal axis, extending over about 40% of the cross-sectional area. This axial region exhibits a Young’s modulus up to 4.9 ± 1.2 and 8.6 ± 0.7 MPa. In general, adding the protein strong interfaces to the nanofibers led to increase the mechanical properties. The ultimate modulus and elongation rate of the PLA/GEL/EGF scaffolds was significantly higher than PLA/GEL, while their maximum strength was lower than that, and this is attributed to the different mechanical behavior of PLA/GEL and PLA/GEL/EGF. To achieve this goal, we conducted a detailed study of the effect of the EGF content on the mechanical properties and cellular responses of PLA/GEL electrospun scaffolds for a dressing application.
Antibacterial activity
The bactericidal activity of the PLA/GEL and PLA/GEL/EGF electrospun nanofibers was assessed to Gram-positive (S. aureus) and Gram-negative (E. coli) by measuring the inhibition of bacterial growth by disk diffusion method [41]. Photos of the bacterial growth after a 48 hours culture at 37 °C are shown in Figure 5. A significant increase in the number of viable bacteria was determined for the electrospun nanofibers PLA/GEL and PLA/GEL/EGF that was close to the growth of S. aureus and E. coli as control, PLA, GEL, EGF [34]. Besides, the antibacterial effect of PLA/GEL nanofiber scaffolds, PLA/GEL and EGF conjugates inhibited bacterial growth. It is well known that EGF is considered an antimicrobial agent and improves the epithelialization process. The diameter of the electrospun scaffold of the inhibitory zone on S. aureus was 14 ± 0.9 mm in PLA/GEL and 19 ± 1.2 mm in PLA/GEL/EGF (Figure 5(a)). Similarly, the diameter of the electrospun scaffold of the inhibitory zone on E. coli was 15 ± 1.3 mm in PLA/GEL and 22 ± 1.8 mm in PLA/GEL/EGF (Figure 5(b)). However, the nanofiber PLA/GEL/EGF scaffold exhibited better effects on both microorganisms [42]. The improved antibacterial activity of PLA/GEL/EGF nanofiber scaffolds can be explained by the mechanical properties of EGF with the high surface area of PLA/GEL nanofiber scaffolds. Because EGF was vastly incorporated in the PLA/GEL nanofiber matrix and exhibited better antibacterial activity. The antimicrobial activity associated with membrane permeability has been extensively studied in the literature [34,41–43]. These nanofibers are therefore promising materials for antibacterial applications. So, from the above data, the nanofibers PLA/GEL scaffolds loaded with EGF have the potential to enhance antibacterial dressings.
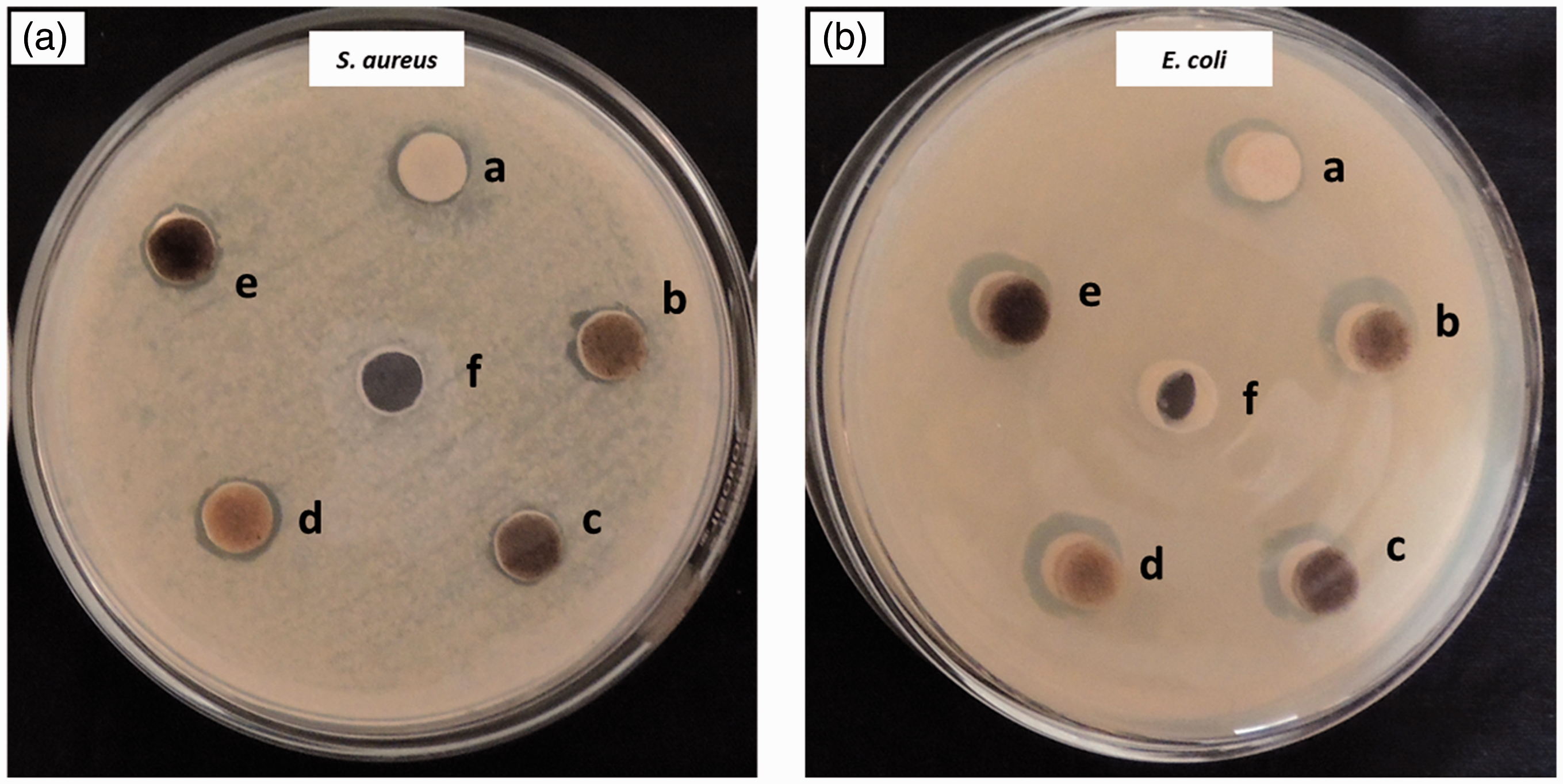
Inhibition zone of (a) control, (b) PLA, (c) GEL, (d) EGF, (e) PLA/GEL, and (f) PLA/GEL/EGF on (A) S. aureus and (B) E. coli culture.
In vitro bioactivity assay
In vitro cytotoxicity is an important technique for assessing biocompatibility with biomaterials for biomedical applications. The in vitro cytotoxicity of the PLA/GEL nanofiber scaffolds and EGF loaded with PLA/GEL was evaluated by the MTT assay in L929 fibroblast cells [44]. L929 fibroblasts were treated with different concentrations of PLA/GEL and PLA/GEL/EGF nanofiber scaffolds from 100 μg/ml to 300 μg/ml.
As shown in Figure 6(a), the cell viability of the PLA/GEL nanofiber scaffold was 84% after 24 hours; however, this value decreased to 72% and 63% with increasing concentration. Similar observations can also be found in EGF loaded with PLA/GEL nanofiber scaffolds. The proliferation of L929 fibroblast cells was considerably inhibited in a dose-dependent manner [44]. However, the nanofiber PLA/GEL and PLA/GEL/EGF scaffolds indicate concentration-dependent toxicity and the cell viability decreased from 86 to 32% with an increase in the concentration of EGF in PLA/GEL [45]. The cell viability of PLA/GEL/EGF at 100 μg/ml was slightly higher than that of PLA/GEL. This may be due to the change in the structure of the nanofiber scaffold due to the binding of EGF to the PLA/GEL surface. At the same time, cell viability decreased to 32%, depending on the higher concentration of PLA/GEL/EGF at 300 μg/ml. Fluorescence microscopy confirmed the binding of L929 fibroblasts to the control, PLA/GEL, and PLA/GEL/EGF scaffolds as shown in Figure 6(b) to (d). As illustrated, the number of cells attached to the PLA/GEL/EGF was higher than the PLA/GEL [46]. Based on the results suggest that the captured nanofiber scaffolds were biocompatible and have plausible for wound healing applications, which can, therefore, be used to advantage to avoid wound infections and promote cell proliferation [47].

(a) MTT cell viability profile of L929 fibroblast cells against different concentrations of prepared nanofibrous scaffolds (PLA/GEL and PLA/GEL/EGF) after 24 h. Representative photographs of the MTT cell viability assay for L929 fibroblast cells treated with (b) control and 300 µg/mL concentrations of prepared nanofibrous scaffolds after 24 h (c) PLA/GEL, and (d) PLA/GEL/EGF nanofibrous scaffolds.
In vivo wound healing assay
EGF conjugated PLA/GEL electrospun nanofibers based on PLA and GEL containing bioactive EGF, which has good mechanical, antibacterial and cell proliferation properties, are great therapeutic inputs for wound healing applications. EGF play an essential role in wound healing through stimulating epidermal and dermal regeneration. Medical nanofibers have been established to treat chronic ulcers such as diabetes in a short time. The developed PLA/GEL and PLA/GEL/EGF electrospun nanofibers have been successfully tested on normal mice and studied the wound healing process by calculating the percentage of wound closure measured at 9 and 18 days followed by a histopathological examination [32]. This nanofiber scaffold efficiency for wound healing applications was assessed using a full-thickness square model study with cotton gauze as a standard for positive control [34]. Figure 7(a) shows photos of wound imaging at different periods using a PLA/GEL/EGF nanofiber scaffold. The wounds in rats were observed once three days for healing progress. On the 18th day, the gauze stuck to the wound surface and its removal resulted in tissue loss. Wounds treated with the prepared nanofiber scaffold showed better wound healing compared to the control [48]. L929 fibroblast cells in vitro have demonstrated intensity enhancement of cell proliferation through the effect of nanofiber scaffolds over control, and it also shows a significant increase in new fibroblasts in a short response time.

In vivo evaluation of incised diabetic wounds treated with control, PLA/GEL and PLA/GEL/EGF electrospun nanofibers at different periods (days); (a) The analysis of wound closure percentages at 9 and 18 days post-treatment of the prepared electrospun nanofibers; (b) Macroscopic observations (Photo images) of incising wounds at different days (0, 9 & 18) with prepared electrospun nanofibers.
On the other hand, the decrease in wound surface overtime was much greater in the EGF loaded PLA/GEL electrospun nanofibers compared to the dressing groups [31]. Wound closure is quantitatively shown as a percentage of wound closure in Figure 7(b). The wound closure percentage graph showed that the nanofiber matrix device improved wound healing compared to the control group. The group of animals treated with a PLA/GEL nanofiber scaffold presented on the third day, the gauze control showed little growth in wound healing with a wound closure of 41 ± 1.63% and on day 6 showed a wound closure of 77 ± 2.32%. The PLA/GEL/EGF nanofiber scaffold showed significantly higher wound healing from day 9, showed a 54 ± 1.74% decrease in the wound area, and complete wound closure was observed on day 18 with a 99 ± 2.18% reduction [49]. In contrast, the wound closure rate for the PLA/GEL/EGF nanofiber scaffolds was higher than that of the wound covered with PLA/GEL scaffolds. The combination of EGF with wounds treated with PLA/GEL nanofiber scaffolds showed an important acceleration of wound healing and greater hair coverage than in the other groups. EGF has been observed to penetrate the damaged tissue into the cell membrane and proteins, indicating the therapeutic potential for the topical treatment of wound healing. According to our results, the presence of EGF had better wound healing properties compared to the control [49].
Histological evaluation
The formation of new blood vessels is a vital process in wound healing. The histological analysis focused on the healing ability of injured rat tissues due to the influence of hematoxylin and eosin (H&E) staining on PLA/GEL, PLA/GEL and control groups (Figure 8). The role of EGF has been extensively investigated in normal and pathological wound healing. On day 9, wound healing was initiated in the groups treated with EGF; however, the control and scaffold treated groups had ulcerated skin and no clear structure was noticed. On the 18th day, the treated group showed moderate inflammatory cells compared to the control [49]. Due to inflammation in the injured area, white blood cells are released into the injured tissue for protection around the injured area [49]. Also, at the end of the 6th-day experiment, the PLA/GEL/EGF electrospun nanofibers cover new epithelia covering the wound was observed, uniform and thicker than those treated with PLA/GEL and EGF. Interestingly, the wounds in rats treated with PLA/GEL/EGF electrospun scaffold healed much faster than the other groups (Figure 8(a)). While there was a tendency to an initial re-epithelialization percentage of PLA/GEL/EGF treated wound has better than PLA/GEL and control group (Figure 8(b)). In addition, the prepared PLA/GEL/EGF electrospun scaffold has reduced the activity of inflammatory cells to 63% and 92% at the time of days 9 and 18 (Figure 8(c)), which was superior activity than PLA/GEL and control group, indicating the effective control of inflammatory cells due to its combined anti-inflammatory effect EGF in the PLA/GEL/EGF electrospun scaffold. It should be noted that the EGF-conjugated PLA/GEL nanofiber mats showed increased cell proliferation of L929 fibroblast cells and epithelial migration on days 18, leading to earlier and more efficient collaGEL synthesis, probably due to the synergistic effect of EGF and PLA/GEL [34]. Likewise, the prepared EGF with an electrospun polymer membrane that has shown wound healing without scarring in the animal model [31,34]. Microscopy clearly visualized more efficient transdermal delivery of EGF conjugates PLA/GEL electrospun nanofibers to both normal skin and peripheral tissues around the wound area rather than that of EGF. In our effort to develop a wound dressing tissue engineering, we prepared a composite fiber material based on PLA/GEL conjugated with EGF, which was tested for the first time for wound healing application to the best of our knowledge. The results of this study illustrate the novelty and effectiveness of the PLA/GEL/EGF electrospun nanofibers for rapid healing applications of diabetic and infectious wounds.

(a) Characteristic H&E stained microscopic images displaying the regenerated structure of incised wounds on different days (9 and 18) treated with control, PLA/GEL and PLA/GEL/EGF electrospun nanofibers at 10x (scale bar 100 μm) and 40x magnifications, (b) re-epithelialization in percentage values and (c) Histogram of inflammatory cells.
Conclusion
In this study, we aimed to successfully prepare an EGF conjugated PLA/GEL electrospun nanofibers for the application of wound healing. The PLA/GEL and EGF mixture solution can be successfully electrospun into continuous, uniform, beadless nanofibers using an electrospinning process. The diameter of the prepared PLA/GEL/EGF electrospun nanofiber is 259.35 ± 99.24 nm as calculated from the SEM images. TEM images clearly showed that PLA/GEL had been distributed by the nanofiber matrix in the presence of EGF. The developed PLA/GEL/EGF has been well characterized and tested for its tensile strength, its elongation rate, its tensile modulus and it's in vivo wound healing abilities. The antimicrobial study showed that the EGF loaded PLA/GEL nanofiber scaffolds had a noticeable antibacterial effect against S. aureus, which was stronger than that against E. coli. The result of the MTT cell viability assay showed that the residues released from the electrospun PLA/GEL electrospun nanofibers improved cell proliferation of L929 fibroblasts after uptake of EGF. The in vivo healing study showed that wounds treated with PLA/GEL/EGF heal much faster than PLA/GEL treated wounds. The histology results revealed that the composite nanofibrous mat exhibited rapid healing process by promoting wound contraction and re-epithelialization. Hence, the development of novel PLA/GEL/EGF electrospun nanofibers used to new generation wound dressing material in the biomedical applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research investigation was supported by the Weigao Research Fund of Heilongjiang Provincial Nursing Association (NO. 201501132002), PR China.


