Abstract
Novel the bilayered electrospun biosheet with rapid cell mimiciking and proliferative efficacy will be suitable for wound healing application. The optimized concentration of gelatin (G) and sodium alginate (A) biosheet with nanofibrous Poly (3-hydroxybutyric acid) (P) as a bilayered elctrospun matrix through electrospinning. The engineered GAP bilayered biosheet involves tissue formation at extra cellular matrix (ECM) which further characterized its function in vitro and invivo. Here we fabricated GAP which exhibit better physiochemical properties, biological and mechanical properties with superior prosomes it enhance air passable at skin wounds. The Bilayered biosheet matrix possess better biocompatibility, cell adherence, fructuous and cell to cell interactions evaluated using cell lines. Furthermore, GAP bilayered matrix regulates growth factors to attain maximum wound closure efficiency during invivo. Thus, the fabricated GAP electrospun biosheet would be a possible wound dressing for skin wound applications.
Introduction
Biopolymers are the most promising biomaterials in the branch of tissue engineering and having a striking evidence to regenerate and heal physical skin damages. Wound healing is a complex process in majority of tissue related disorders and very challenging to a clinician. Current research on biomaterial scaffold development principally focused on people affected by skin wound, mainly caused by trauma, skin burn and diabetic wounds [1–3]. The fabrication of robust tissue remodeling scaffolds with biocompatible and biodegradable properties enhances wound healing. The elecrospun nanofiber provides good stability and oxygen permeability to impart cell proliferation to achieve accelerated wound healing [4,5]. In particular, degradable natural biomaterials such as collagen, chitosan, gelatin, silk fibroin, keratin and sodium alginate and synthetic biomaterials such as polylactide (PLA), polyglycolide (PGA), Poly (3-hydroxybutyric acid) (PHB), poly (vinyl alcohol) (PVA), poly(lactic-co-glycolic acid) (PLGA) and polycaprolactone (PCL) play a most significant role in drug delivery and tissue engineering [6–10]. However, an ideal nanofibrous scaffold requires a protein based biomimetic biological material. The researchers have developed many combinations using natural and synthetic biomaterials with merits and demerits [11,12]. Accordingly, the bilayer electrospun biosheet approach combines biopolymers with highly porous and well inter connected biomimetic materials to regenerate damaged tissues as a prominent wound dressing.
Gelatin (G), a denatured protein polymer derived from collagen. It has temperature sensitive properties, also conducts physical crosslinking structures [13–15]. Gelatin is widely found in nature, also contains high level of α-aminoacids such as glycine, proline and hydroproline [16]. Moreover, gelatin has an excellent feature which includes biocompatibility, biodegradability, optical property and swelling ability as a hydrophilic polymer. The Gelatin hydrogels structure can be formed easily with a melting temperature 35 to 45 °C and suitable in many medical industries [17,18].
On the other hand, sodium alginate (A) is an anionic linear polysaccharides comprised of 1, 4-linked β-D-mannuronate and 1, 4-linked α-L-guluronate residues in varying proportions [19,20]. They are extracted from brown sea algae cell walls. A is used in many biomedical applications such as drug delivery, wound dressing, bone regeneration, cartilage repair and 3 D bioprinting. Certain investigations reported that certain alginate dressings can enhance tissue regeneration by invigorating monocytes to produce prominent levels of tissue necrosis factor at wound site [21]. This activating features and its advantageous such as excellent biocompatibility, non-immunogenicity, low cytotoxicity helps to repair damaged soft tissues as wound dressing [22,23]. The Poly (3-hydroxybutyric acid) (P) is a natural hydrophobic thermoplastic material with good half-life. P is an eco-friendly non-toxic material with good mechanical properties and exhibit biodegradability [24]. During electrospun with other copolymer, nanoparticles and drug, P exhibits a proven functional support over fibroblast and keratinocytes [25,26].
The synergized nanofibrous wound dressing material has skin mimic function and proteins of native extra cellular matrix, which accelerate cell activities. Especially, the utilization of smart biomaterial such as natural and synthetic polymers as a blend will attain a good healing efficiency. In this study, the reinforcement of nanofibrous matrix with a protein biopolymers as bilayered biosheet makes cell adhesion, rapid healing process, granulation of tissues and promotes growth factors at wound site [27]. In addition, the simple and versatile method for the fabrication of polymeric fibers and single layered biosheet using simple casting method were combined to form a bilayered structure. Hence the durable bilayered structure is preferred for providing maximum efficacy in wound contraction and as a prominent skin wound healing substitute.
In this present study, we aimed to fabricate smart nanofibrous biosheet from poly (3-hydroxybutyric acid) (P) over the blend of Gelatin (G) and Sodium Alginate (A) for wound healing application. The microstructure of the biosheet was evaluated using Fourier Transform Infrared spectroscopy (FTIR), X-Ray diffraction (XRD), Scanning electron microscopy (SEM) and tensile strength. Then, the biosheet were further characterized for wound dressing application such as invitro biocompatibility, cell adhesion, cell proliferation using various cell lines and invivo studies using male Albino Wistar rats.
Materials and methods
Medium thickness alginic acid sodium salt from dark colored algae growth was gotten from Sigma Aldrich, CAS No: 9005 - 38 - 3. Type A Gelatin (G) was acquired from Sigma Aldrich, CAS No: 9000-70-8. Poly (3-hydroxybutyric corrosive) (P), 1,1,1,3,3,3 hexafluoro-2-propanol, PPC of normal atomic weight (Mn) ∼50,000, 2, 2, 2-trifluoroethanol, Dulbecco's changed falcon's medium (DMEM), fetal calf serum (FCS), keratinocytes development medium, and the beneficial anti-microbials for tissue culture were bought from Sigma Aldrich, Bangalore, India. Rest of the synthetics and culture products were likewise bought from Sigma Aldrich except if determined something else.
Fabrication of semi solid GA biosheet
The fabrication of completely air dried GA biosheet was readied. The 2% (w/v) of Gelatin (G) biosheet, 2% (w/v) Sodium Alginate (A) biosheet and 2% (w/v) of G arrangement were blended in with 2% (w/v) (A) in 1:1 proportion with 1.5 mL of ethylene glycol as a plasticizer to frame the GA blend. The readied G, A and GA blend was poured in polyethylene plate and completely air dried to acquire biopolymer platform. In any case, semi strong G, A and GA biosheet was manufactured by emptying the readied GA blend into the polyethylene plate (estimating 12 cm X 14 cm) and air dried at room temperature until to get a semi strong biosheets (80% dried). The readied semi strong biosheet with the polyethylene plate was taken for electrospinning to manufacture a bilayered nanofibrous double framework platform as given in Table 1 and marked as G, A, GA and GAP respectively.
The chemical source ratio of fabrication of bilayered nanofibrous matrix biosheet.

Electrospinning of bilayered nanofibrous matrix biosheet (GAP)
To prepare the GAP bilayered dual matrix, 4% (w/v) concentration of polymer solution were prepared by dissolving 0.4 g of Poly (3-hydroxybutyric acid) P in 10 mL of 1, 1, 1,3,3,3 hexafluoro-2-propanaol for 12 h constant stirring. The homogenous arrangement of P was permitted at steady mixing for 8 h for uniform arrangement. The homogenous arrangement P were electrospuned over the readied semi strong type of the GA biosheet put over the grounded square shape aluminum substrate a ways off of 13 cm opposite to the 24 G needle associated with positive terminal of the high voltage DC power supply (ZEONICS, Bangalore, India). The polymer arrangement was expelled with 1.5 mL/h utilizing a controlled syringe siphon exposed to an electric capability of 1.5 kV/cm. Therefore, after the evaporation of the solvent from the semisolid GAP scaffold, causing flatness P nanofiber over the solidified GA biosheet to form the bilayered nanofibrous matrix (Scheme 1). Further the prepared scaffolds were stored at room temperature until further use.

Schematic fabrication of GAP bilayered biosheet fabrication.
Physio-chemical characterization techniques
The functional group changes of the bilayered biosheet were researched utilizing Fourier Transform infrared (FTIR) estimations, to decide the practical gatherings present in the readied G, A, GA and GAP nanofibrous biosheet were examined. The spectra were estimated at a goals of 4 cm−1 in the recurrence scope of 4000-600 cm−1 utilizing ABB 3000 spectrometer with Grams as the working programming. The surface morphology of the frameworks were imagined utilizing FE-SEM, (Carl Zeiss AG, Jena, Germany) working at a quickening voltage of 5–20 kV in the wake of being covered with gold. The crystallinity of the bilayered biosheet were validated via X-ray diffractometer (X’Pert PROPANalytical). The XRD was performed over a range of 10°–90° with scan speed of 5°/min at step size of 0.02°.
Tensile strength measurement of the nanofibrous biosheet
Tensile strength properties of prepared G, A, GA and GAP biocomposite were cut into dumb bell shaped specimens of 5 mm wide and 12 mm length. Mechanical properties, for example, rigidity (MPa) and level of prolongation at break (%) were estimated utilizing an all-inclusive testing machine (INSTRON model 1405) at an augmentation pace of 5 mm/min.
In vitro biocompatibility test
The In vitro biocompatibility and cell reasonability was contemplated utilizing HeLa cell lines, MCF7 Cell lines. The cell lines were spread over bilayered biosheet and safeguarded in DMEM with fetal calf serum enhanced with 100 Units mL−1 Penicillin, 150 mg mL−1 Gentamycin, 60 mg mL−1 Streptomycin and 2 mg mL−1 Amphotericin B kept up at 37 °C. Cells without biosheet were refined in a clear well as control. To consider the biocompatibility of created biosheet which was kept up in a polystyrene culture plate which contains 96-well plate, it is segregated against different focuses, for example, 25 and 50 (lg/well). At that point, the fixation is hatched for 5 h at 37 °C with humidified climate of 5% CO2. At last, toward the finish of 24 h, the practical cell tally can be resolved utilizing MTT measure and the outcome is determined with p < 0.05.
In vivo biocompatibility test
For in vivo biocompatibility study, a male Albino Wister rats (220 to 275 g) were used. All the animal experiments were done under the ethical guidelines of ethical committee of Wenling First People's Hospital, Wenling, PR China. The rats were anesthetized by using thiopentone sodium as intra peritoneal injection at a dose of 50 mg/kg body weight. The rats were divided into three groups containing three animals in each group (Control, GA biosheet and GAP biosheet). Then rats were acclimatized for a week prior to the commencement of the experiments individually housed in 12 h light/dark cycle at 25 ± 1 °C and were monitored and provided standard feed regularly.
Under aseptic condition the dorsal surface of the rodent beneath the cervical locale was shaved on its back after the intraperitoneal infusion of standard sedation thiopentone sodium (portion 50 mg/kg body weight). A full thickness extraction wound estimating 2×2 cm2 were made. The GA biosheet and GAP biosheet wound dressings were used to evaluate its wound healing efficiency on excision wound models in rats. A square formed silicone brace was put, so the injury is focused inside the support. The support was fixed to the skin by suturing the corners to balance out the position. Control (Gathering 1), injuries were dressed with sterile cotton cloth. Gathering 1 creatures were dressed with control, 2 creatures were dressed with GA biosheet and bunch 3 creatures were dressed with Hole bilayered nanofibrous platform. In both GA biosheet and Hole bilayerd nanofibrous framework P nanofibrous side go about as the injury contact layer at the injury site. The dressings were changed intermittently at an interim of 5 days with the individual frameworks for control, GA and GAP treated gatherings. The injuries were cleaned with sterile refined water before dressing. Granulation tissues were gathered on every one of the five gatherings on fifth, tenth, and fifteenth day post injuring and were put away at - 80 °C until examination. The advancement of twisted recuperating in rodents was assessed by periodical checking of wound compression, biochemical and histological examinations.
Biochemical analysis of excision wounds
The biochemical analyses of the excision wounds on excised tissues were analyzed. First, the estimation of total amount of protein [28], estimation of hexosamine [29], estimation of total amount of collagen (hydroxylproline) content in defatted granulation tissue [30] and estimation of uronic acid [31] for both treated and untreated wound tissues.
Rate of wound contraction
The contraction of wound and its full re-epithelialization was calculated as the number of days required for the wounds to heal completely without a row wound left behind the rats. To estimate rate of wound contraction, wound biopsies were noted and surface area was measured by standard palimetric method, by tracing the wound on the transparent graph sheet. The results were measured and analyzed using the method of Thangaelu et.al. [32]
Statistical analysis
Results are presented as mean ± S.D. (n = 3). ANOVA (Analysis of variance) and student’s t-test were done to determine the significant differences among the groups. The observed differences were statistically significant when p < 0.05.
Results and discussion
FTIR spectroscopy
Figure 1 shows the FTIR profile of gelatin (G), sodium alginate (A) and gelatin-sodium alginate (G + A) biofilms. The peak occurs at 3350–3450 cm−1 corresponds to N-H stretching vibration. The FTIR spectra of functional range between 1700 and 1500 cm−1 is related to fibroin peptide spine of amide I (1700–1600 cm−1) and amide II (1600–1500 cm−1). The amide III band observes at 1350–1200 cm−1 [33]. The structure of unaltered “A” material band is concentrated in FTIR spectra. The band around 1610 cm−1, and 1408 cm−1 are credited to the unbalanced and symmetric extending of carboxylate – COO–, respectively. The peak around observes at 1034 cm−1 for O–H mode, due to material “A” Alginate [34]. Figure 1(d) shows the range of unadulterated GAP slim film show an expansive top at 2900-2800 cm−1 in all examples range are the blend of CH2, CH3 deviated and symmetric extending modes. The GAP shows a solid and sharp ingestion top at wave number 1734 cm−1 allot to C = O and however in their mixes indicated expansive pinnacle and reduction the worth [35,36]. The band C-H and C = O stretching of acetyl groups of GAP comprising peaks at 2924 cm−1 and 1652 cm−1, respectively [37,38]. The stabilized higher molecular weight polymers are merged with mupirocin, resulting in lower molecular weight compounds. This leads to significant interaction of the disulfide bond between protein polymers and the drug in a defined structure of the biopolymer film, and is beneficial for wound-healing applications.

FTIR profile of (a) Gelatin (G), (b) Sodium Alginate (A), (c) Gelatin-Sodium Alginate (GA) and (d) Gelatin-Sodium Alginate-Poly (3-hydroxy butyric acid) (GAP).
X-ray diffraction analysis
Figure 2(a) to (d) show the XRD patterns of the Gelatin (G), sodium Alginate (A), Gelatin-sodium-Alginate (GA) and GAP biopolymer films. The amorphous structure of the gelatin shows the broad hump at around 17–23° (G) in Figure 1(a) [39]. Figure 2(b) shows Sodium Alginate (A) typical halos were observed at 13.7° and 21.4°, corresponding to 6.45 and 4.42 Å, respectively [40]. All the mix frameworks displayed clear diffraction tops around 13.4° and 20°, which illustrated (A) segments unmistakably existed in the G/A mixing platforms, and the crystalline structure of GA was additionally for the most part the chitosan I structure dependent on the past investigations of scientists. Signs of low crystallinity of the readied platforms. With expanding of (G) and (A) content in the mix frameworks, the force of GAP trademark top around 20° was getting more vulnerable and more fragile, while the GA trademark top around 13.7° showed up unmistakably in Figure 2(c). These progressions were credited to expanding connection between G, A and GA atoms. Figure 2(d) the XRD case of the unadulterated GAP exhibited that it has a semi crystalline nature, which is in adequate simultaneousness with the results declared before [35].The XRD example of sodium alginate is appeared to have low crystallinity or nebulous nature [41,42]. The diffraction examples of physical blends demonstrated differed tops which is like that in unadulterated structure, showing that the crystallinity of GAP, Gelatin and Sodium Alginate was not changed [43–45]. The crystallinities decline from 73.6 to 50% as the GAP division was expanded [43]. The crystal structures were not affected by the addition of poly (3-hydroxybutyric acid) [46,47]. While by the addition of gelatin and sodium Alginate, the curves present a better resolution for peaks than that of poly (3-hydroxybutyric acid). Moreover, the intensities of the crystalline peaks of the blending obviously increased, thereby appear small peaks [35].

XRD pattern of (a) Gelatin (G), (b) Sodium Alginate (A), (c) Gelatin-Sodium Alginate (GA) and (d) poly (3-hydroxy butyric acid) (GAP).
SEM micrographs
Different ratios using blended Gelatin-sodium-Alginate (GA) and GAP biopolymer films are shown in Figure 3. The observed smooth surface for G and A with 1:1 than the other ratios and physical properties are made them to use for further analysis and poly (3-hydroxy butyric acid) blended biofilm preparation. Figure 3(a) and (b) show a photograph and SEM micrograph of the GA with 1:1 (further denoted as GA) and GAP biopolymers, respectively. The Gelatin: Sodium Alginate biofilm shows the smooth surface (Figure 3(a)). Figure 3(b) shows the GAP film, on GA film surface electro spun with poly (3-hydroxy butyric acid) shows a highly fibrous surface, owing to the disulfide bonds of keratin interacting with co-polymers as well as with the evenly distributed poly (3-hydroxy butyric acid [48,49].

SEM micrographs of (a) Gelatin-Sodium Alginate (GA) and (b) poly (3-hydroxy butyric acid) (GAP).
Tensile study
The tensile study of bilayered biosheet G, A, GA and GAP were evaluated as tensile strength and elongation break are shown in Figure 4. Figure 4(a) shows the GA and GAP matrix were flexible with tensile strength of 8.42 and 18.71 MPa respectively and percentage of elongation shows the GA and GAP 4.3 and 10.12% respectively (Figure 4(b)). Here the study reveals that bilayered electrospun matrix showed a remarkable enhanced tensile strength. Since G, A and GA biosheet were flexible and soft which exhibit moderate tensile strength. However, the GAP soft matrix was most prominent for the use during wound surface application (Table 2).
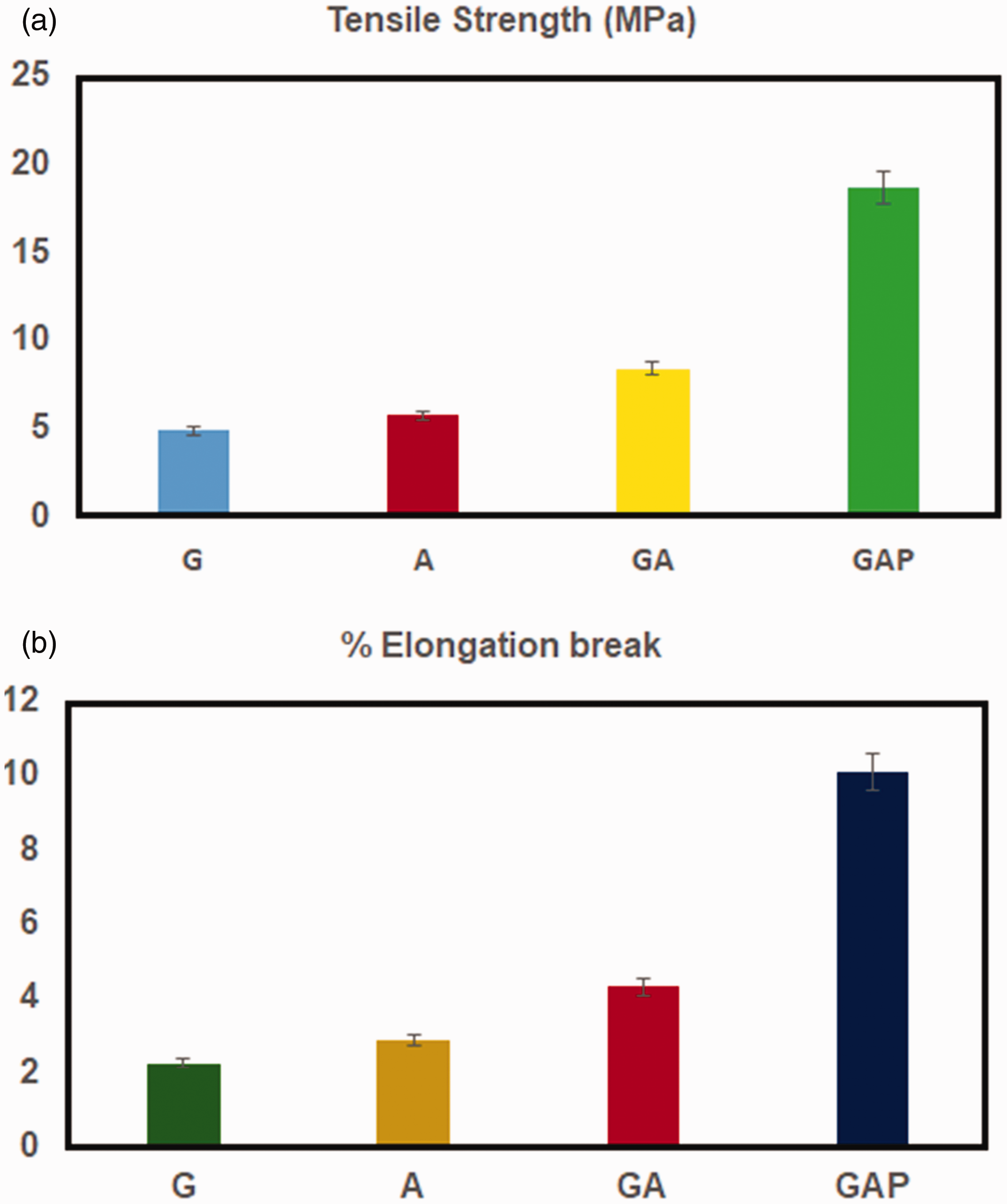
(a) Tensile strength properties of G, A, GA and GAP biopolymer biosheet, (b) Elongation break of G, A, GA and GAP biopolymer biosheet.
Tensile strength and elongation break properties of G, A, GA and GAP biopolymer biosheet.

In-vitro biocompatibility
Despite the fact that, geography and porosity of the biosheet assumes a critical job in cell connection and multiplication, biocompatibility of the readied bilayered biosheet right now assessed for their latent capacity use in wound dressing and skin tissue designing applications. HeLa Cells and MCF7 Cell lines were utilized as a kind of perspective cell lines. The rate cell reasonability was determined by MTT test (Figure 5(a)) which relies upon the mitochondrial movement of reasonable cells and speaks to parameters for their metabolic action. Contrasted and the control (nanofibrous surface) all the cells developed on the biosheet were seen as uniform with ordinary morphology. Broad MCF7 Cell lines and Vero cell development and connection were seen on the control polystyrene surface and no perceptible cell demise or restraint of development was seen in treated wells. More than 90% cell viability was seen in GA and GAP treated cells showed 88% of viability. But HeLa cell line growth was noticed as 92% cell viability in GA biosheet and 91% of viability on GAP treated cells (Figure 5(b)). As many researchers reported that biomaterial made of protein biopolymer attracts many cell types, which activates growth promoters in wound healing by collagen production at wound site.

(A) Fluorescence staining images of MCF7 (a-c) and HeLa cell (d-f) proliferation and adherence onto the GAP bilayered biosheet at 1 hour and 24-hour time interval. (B) In vitro cell % cell viability at different time intervals.
In-vivo biocompatibility
Photographic evaluation
The photographic evaluation and planimetric analysis studies on animal shown as single representative from each group (Figure 6). During this assessment, it was seen that GAP showed quicker recuperating than GA framework. Be that as it may, the GAP bilayered electrospun upheld cell development and expansion with consistent ingestion of exudates to the P side, which saves the injury liberated from contamination for fast mending. Along these lines the visual proof of in vivo wound recuperating was taken by utilizing NIKON DSLR camera DS5100 from an unaltered separation for every exploratory creature and the planimetric study were assessed occasionally. The conclusive outcomes additionally displayed to demonstrate quicker twisted withdrawal in the 5 gatherings with GAP when contrasted with different gatherings.

Photographic representation of contraction of rate showing the wound contraction area.
Biochemical analyzes, rate of contraction and wound re-epithelialization
The total protein, collagen, hexosamine, and uronic acid content in the granulation tissues of the control and the treated experimental groups of the rats on specified days of scarification after wound creation were shown in Figure 7(a) to (d). However the biochemical results reveals that GAP biosheet shows significant differences compared to control, G, A and GAP biosheet. Due to increase in collagen content on GAP treated wounds, the hexosamine, protein and uronic acid levels helps to heal the wound by mimic the function of extracellular matrix. The wound contraction assessment plays an important role in the prediction of tissue epithelium at excisional wounds, calculated using image J. Software program from the wound tracing sheet. Through ANOVA the statistical significance between the animal groups and reveals different data on the précised day. The rate of wound closure in all groups were shown in Figure 7(d). The complete closing of wounds was observed with slow rate of closure in control group which was treated with cotton gauze than the experimental groups at various intervals. The group treated with GA showed moderate wound closure when compared with the GAP group. The significant raise in wound closure was observed for all days. The bilayered electrospun biosheet possess faster wound closure over other treated groups. On the day 12 when compared to that of control, the GAP exhibits rapid wound closure. However, the G, A and GA shows similar amount of wound closure from day 10th onwards, whereas the GAP treated group exhibits enhanced wound closure from day 9th onwards. Thus, the complete healing with GAP treated groups was observed on day 18 and 19 and G, A and GA treated group on 20th day, whereas the control group has taken almost 22 days for complete healing. Overall the wound closure using planimetric results and photographic evaluation were well associated wound closure pattern.

Statistical significance between the animal groups and reveals different data on the précised day (a) Total Protein, (b) Hexosamine, (c) Hydroxyproline (d) Uronic acid and (e) Wound Contraction (%).
Conclusion
A novel source of bio-waste materials for the fabrication of GAP bilayered electrospun biosheet reported in this paper. The Gelatin and Sodium alginate were successfully blended with poly (3-hydroxybutyric acid) to form a GAP biopolymer film, whereas GA was combined with poly (3-hydroxybutyric acid) for organization of the GAP electrospun biosheet. The mechanical strength, surface properties of the GAP film were superior to those of the GA film. Moreover, compared with its GA counterpart, the GAP film exhibited superior cell adhesion and cell migration symptoms of Hela and MCF7 cells. The observed results suggest that the simple and biological waste generated by the prepared structure of GAP bilayered biofilm would be a fruitful wound-healing recipe that is suitable for in vivo studies. The final results also exhibited to indicate faster wound contraction in the 3 groups with GAP when compared to other groups. Overall the wound closure using planimetric results and photographic evaluation were well associated wound closure pattern.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
