Abstract
Nano-structures due to their unique properties can provide a biomimetic structure for cell attachment and proliferation in tissue engineering (TE) applications. But sometimes, their surface properties are not particularly suitable for directed tissue growth. In this regard, present study has focused on fabrication and hydrolysis of Poly (
Introduction
Electrospun nano-structures as tissue engineering (TE) scaffolds are attractive candidates in regenerative medicine [1–3]. Nano-structures due to their surface properties such as porosity and fibers with nano-scale diameter can hinder efficient cell proliferation, cellular ingrowth, and neotissue formation in TE applications [4]. The researchers have shown that the hydrophilic structures are crucial controlling parameter for cell activity in biomedical and biological applications in TE [3,4]. However, sometimes nano-structures can't support cell activity due to their hydrophobic surface. Therefore, their surface properties need to be modified and tuned for demonstrating better cell attachment and proliferation [5].
Plasma [6,7] and NaOH hydrolysis [8] have been suggested to hence modify the biocompatibility and hydrophobicity of nano-structures by increasing their water wettability. That NaOH hydrolysis as a cost effective, useful and simple surface modification method has attracted a lot of attention [9]. The results of other studies have indicated that NaOH hydrolysis increased the contact angle of nano-structures along with increasing the nanofiber’s roughness and generation of new active sites, hydroxyl groups on the nanostructures surface [10]. In other words the NaOH hydrolysis treatment, due to increase the surface energy induced capillary reaction and fully engaged of the water molecules with the nano-fiber structure were viewed significantly changes in contact angle and cell morphology of nano-structures [10,11].
The extracellular matrix (ECM) of native tissues is composite structure and consists of elastin and collagen that provide the flexibility and strength, respectively, at the same time in mentioned tissues [12,13]. In this regard, researchers have attempted to fabricate the TE scaffolds with mixture of two or more polymers in order to mimic and simulate the ECM and mechanical properties of native tissues [14–16].
Poly (
According to the author’s knowledge, there are studies about applying PCL/PU as designed hybrid nano-structures and characterizing their morphology, mechanical and biomechanical properties for biomedical applications [22,23]. PCL and PU in form of hybrid nano-structures can resembles the scale and structure of collagen and elastin fibers of the ECM in native tissue [24]. The hybrid PCL/PU nano-structures must be able to provide an optimum and biomimetic environment for cell attachment and proliferation for successful biomedical application in TE [21]. But their surface properties are not particularly suitable for directed tissue growth and their surface inertness is a drawback for PCL/PU matrices in most biomedical and biological applications. Therefore, the hydrolyzed hybrid nano-structures can be raised as unique approach for cover these specifications.
Clearly, further study is needed to investigate the influence of NaOH hydrolysis treatment on the morphology and mechanical properties of PCL/PU nano-structures for using in TE applications.
Above all, the main objective of present study was modification the surface of hybrid PCL/PU nano-structures using NaOH to improve the hydrophilicity of the nano-structures for TE applications. In addition, this study focused on determining the optimum NaOH concentration and submersion time in order to have structures with any undesirable changes on their morphology, mechanical and biomechanical properties. Therefore, the influence of the reaction parameters such as submersion time and concentration of NaOH on the surface of hydrolyzed neat and hybrid nano-structures was examined. All things considered, this article was split into investigation the influence the concentration of NaOH (0, 1, 2 and 3 M NaOH) at different submersion time (1, 2 and 3 h) in terms of evaluation the hydrophilicity and mechanical properties of PCL, PU and PCL/PU structures.
Materials and methods
Materials
PCL (MW = 80000) was purchased from sigma-Aldrich and PU segments (APILON 6505) were supplied by BDH and were used to fabricate the hybrid nano-structures by co-electrospinning method. Dimethylformamide (DMF), Tetrahydrofuran (THF), EtOH and Chloroform were purchased from Merck Company and were applied as solvent. All reactants were used without further purification
Fabrication and NaOH hydrolysis treatment of electrospun nano-structures
For co-electrospinning method, PCL (10 w/v %) and PU (15 wt %) were dissolved separately in (Chloroform/EtOH: 7/3 for 3 h) and (THF/DMF: 3/1 for 12 h), respectively.
Before electrospinning process, the polymer solutions were kept constant for 30 min to bubble removal. The custom designed electrospinning setup was used for fabrication the neat and hybrid nano-structures. The electrospinning setup typically consists of: High-voltage power supply (0–30 kV, LD Didactic GmbH521721) Syringe pumps (Model: wz-50c6 and pp-s14) 20 ml syringe fitted with needles of 18 G Cylindrical stainless steel mandrel collector (with 2000 mm/min and 800 rpm as linear and rotational speeds, respectivelt)
Also, the ambient temperature and relative humidity as the environmental factors were held at 28°C and 30%, respectively. Neat and hybrid nano-structures were fabricated with average thickness of 460 ± 28 µm (determined using a digital thickness meter, ASIMETO (±1 µm measurement accuracy, n = 7). For removing the nano-structures from the mandrel of collector, the Teflon spray was applied before starting the electrospinnig process. The fabricated nano-structures stored for 24 hours in a lab desiccator to remove any solvent from samples. Electrospun nano-structures were hydrolyzed by NaOH at three different concentrations (1, 2 and 3 M NaOH) and at 1, 2 and 3 h as submersion time. For hydrolysis treatment, the structures were immersed at the different concentrations of NaOH at room temperature. Then hydrolyzed structures were washed with distilled water to remove any NaOH on the surface of structures and dried for 24h under laminar hood at room temperature.
Surface characterization of electrospun nano-structures
SEM and FESEM
The surface morphology of the electrospun nano-structures was evaluated using a scanning electron microscopy (SEM) XL-30 ESEM (FEI, Co., Ltd., American) and Field emission scanning electron microscopy (FESEM) (TESCAN BRNO-Mira3 LMU). It is necessary to coat the nano-structures with gold-platinum for 2 min to protection against electron beams and provide a homogeneous surface for analysis and imaging. Therefore, the small size of surface of samples was selected and coated by thin layer of gold-platinum before taking SEM and FESEM images.
The average fiber diameter of the non-hydrolyzed nano-structures (neat and hybrid) was determined by selecting 100 fibers at random from 1000x and 10000x magnification of the SEM images and then they was analyzed by Image J software. Also, the hydrolyzed hybrid nano-structures were examined by FESEM to evaluate the influence of NaOH concentration and submersion time on morphology of all the structures.
Fourier transforms infrared spectroscopy
The non-hydrolyzed and hydrolyzed nano-structures were analyzed by Fourier transform infrared spectroscopy (FTIR, Nicolet Thermo Scientific Co.). All samples were scanned from 4000 to 400 cm−1 to investigate of chemical changes during the NaOH hydrolysis treatment.
Weight lost and shrinkage
The weight loss ratio and shrinkage of hydrolyzed nano-structures (n = 5) was determined after washing the neat and hybrid nano-structures by phosphate buffer saline (PBS) with pH 7.4 and water. After that, the samples dried in desiccator for 24 h, and the percentage of the weight loss and shrinkage of samples were calculated according to equations (1) and (2), respectively:
W1 = First Weight (non-hydrolyzed)
W2 = Second Weight (hydrolyzed)
S1 = First Area (non-hydrolyzed)
S2 = Second Area (hydrolyzed)
Contact angle
The static contact angle of the electrospun nano-structures (n = 5) was measured using the sessile drop method with a (5 µl) distilled water droplet at room temperature. The contact angle between the dried and hydrated structure and the pure water was determined using images were taken with a digital camera.
Mechanical properties of hybrid nano-structures
The tensile strengths, strain and Young's modulus as the most important mechanical properties of the non-hydrolyzed and hydrolyzed nano-structures (in form of mat: 2 cm length × 0.5 cm width, n = 5) were determined by mechanical testing machine (Universal Test Machine STM) by a 50 N load cell at a crosshead of 5 mm/min.
Results and discussion
Surface characterization
A schematic design of fabrication and NaOH hydrolysis treatment of the nano-structures was presented in Figure 1. In addition, the electrospinning conditions to achieve the nanofibers with optimum and perfect fiber diameter were obtained in Table 1. The effect of NaOH hydrolysis treatment on topography, fiber diameter and morphology of electrospun nano-structures were analyzed by SEM and FESEM images. The SEM images of the non-hydrolyzed nano-structures reveal random and non-beaded fiber morphology in all structures.

The schematic design of co-electrospinning method.
Co-electrospinning conditions.

The average fiber diameter of non-hydrolyzed PCL, PU, and PCL75%:PU25% and PCL25%:PU75% structures were reported 411 ± 78, 433 ± 80, 547 ± 89 and 437 ± 12 nm, respectively. According to another studies, the ECM of native tissues composed of three dimension structure with a fiber diameter of 50–500 nm [25,26]. As a result, using co-electrospining method in this study, let to the fabrication neat and hybrid nano-structures with fiber-scale similar to the ECM in body.
The obtained images (Figure 2(a) to (d)), indicated morphological changes on the fiber surface of structures by following their contact with different concentration of NaOH at different submersion time.

SEM and FESEM of non-hydrolyzed and hydrolyzed nano-structures: (a) PCL, (b) PU, (c) PCL25%:PU75%, and (d) PCL75%: PU25%.
Random and aligned networks were retained and fiber surfaces were observed with visible changed topographical features. Fiber diameter distributions remained comparable between test groups indicating no substantial change in fiber size. Generally, the reaserchers confirmed that NaOH hydrolysis treatment does not impact on fiber diameter [27].
As can be seen in Figure 2, at all concentrations at different submersion time (≥1 h) the fibers of hydrolyzed neat and hybrid nano-structures were degraded and many pore created on the surfaces of fiber due to NaOH hydrolysis treatment. The obtained images confirmed, that the degradation of the fiber and size amount of pores are directly related to the increase of submersion time and the NaOH concentration.
Fourier-transform infrared spectroscopy (FTIR)
The FTIR results of the non-hydrolyzed and hydrolyzed nano-structures were presented in Figure 3(a) and (b).

FTIR spectra of neat and hybrid nano-structures (a) before hydrolysis and (b) after hydrolysis.
For the PCL structure, the broad absorptions in the range 2850–3000 cm−1 and 1705–1725 and 1000–1300 cm−1 were attributed, respectively, –CH2 (-stretching), –C = O (carbonyl ketone – stretching) and C-O stretching bending vibrations. The FT-IR spectrum of PU nanofibers reflected the asymmetric stretching of C-O-C bending, the –CH2 (Stretching) by showing peak at 1100–1248 cm−1, 1543 cm−1, respectively. In addition, the peak at 1731 cm−1 range was attributed to the PU carbonyl (C = O) vibration. Also after hydrolysis (Figure 4(b)

The shrinkage results of neat and hybrid nano-structures.
PCL has similar functional group to PU [23]. The bands in the range of 2948 cm−1 and 2868 cm−1 in hydrolyzed structure attributed to the carbonyl stretching of a carboxylic ester.
Therefore, this change could be due to the ester bond cleavage yielding a carboxylic and an alcohol group as a consequence of the nano-structure degradation [28]. After more passed submersion times, were not observed any additional changes in hydrolyzed structures [28].
Weight loss and shrinkage
The weight loss and shrinkage of the hydrolyzed PCL, PU, and PCL75%:PU25% and PCL25%:PU75% structures at 1, 2and 3 M NaOH at different submersion times were reported in Figure 4 and Table 2, respectively. The results confirmed that the maximum weight loss (14.1% at 1 M, 21.55% at 2 M, and 23.88% at 3 M NaOH) and shrinkage (6.23% at 1 M, 9.75% at 2 M, and 12.51% at 3 M NaOH) between all structures it belongs to the neat PU structure after 3 h.
Weight loss of neat and hybrid-nano structure.

PCL introduce as semi-crystalline thermoplastic polyester and composed of five methylenes [(CH2)5] and an ester functional group (CO2, stretching vibrations) as the repeating unit [29,30]. The polyurethane is consisting of hard (isocyanate) and soft (polyol) segment in its structure [31,32]. This polymer is produced using reacting an isocyanate with a polyol containing on average two or more hydroxyl in the presence of a catalyst [33]. The carbonyl (C = O) groups of the PCL and of the PU is very reactive zone that are favorable for the hydroxide ions attack of the NaOH solutions.
That is to say, these groups are sensitive to hydrogen bonding in hydrolyzed structure [33]. Also, the ester group (-O-C = O-) of the repeating-unit of the PCL polymer tends to be hydrolyzed. Conversion of carbonyl groups (C = O) to -ol (-OH/, alcohol) and carboxylic acid groups (COOH) and removing the ester group can cause short chains in hydrolyzed PCL nano-structures [27,34]. In NaOH hydrolysis treatment of PU, the OH- attack mainly is on the urethane group (-O-CO-NH-) and leads to amine and ol (OH, alcohol) end groups, and also to lose CO2 gas [33]. After NaOH hydrolysis treatment of PCL and PU, the polymeric chains tend to fragmented into short polymeric chains with low molecular weight [34]. In other words, during NaOH hydrolysis treatment of PCL and PU, the molecular chains of polymers break, and the main polymeric network are lost.
As a result, low molecular segments of the chains is removed by work-up (washing by water), and weight loss and shrinkage (the area decrease of nanostructures) of the nano-structure happens after NaOH hydrolysis treatment. Furthermore, many hydrogen bondings are produced between -NH2, -OH/-O- and –COOH/-COO- end groups generated by NaOH hydrolysis treatment of PCL and PU.
This reduces the distance between the two polymer chains, which can explain the contraction (shrinkage) of the nanostructure (the area decrease of nanostructures) after NaOH hydrolysis.
In hybrid nano-structures with more composite ratio of PU, the degree of weight loss was found to be much greater than other nano-structures due to lose CO2 gas after NaOH hydrolysis. Because of the generation of -NH2 and -OH/-O- end groups on the PU nanofibers, stronger hydrogen-bonded occur, and therefore, it can be seen more shrinkage in PU compare to other structures. Also, by increasing NaOH concentration and time submersion, increasing weight loss and shrinkage are clearly observed. As observed in the FT-IR spectra of all samples hydrolyzed, they show clear peaks of NH (-NH2) and OH (-OH and –COOH) end groups at about 3300–3500 cm−1 after NaOH hydrolysis.
The PCL25%:PU75% structure showed 13.37%, 17.03%, and 20.02% percentage of weight loss at 1, 2 and 3 M NaOH, respectively, at 3 h of submersion time. The results indicated by increasing the composite ratio of PU from 25 to 75 in the hybrid nano-structures, the shrinkage of the structure increased. In addition,
Contact angle
The significant differences were observed in water contact angle between neat and hybrid nano-structures. As can be seen in Table 3 at all concentrations of NaOH and at different submersion time (1 hour or more) compared to non-hydrolyzed nano-structures the hydrophilicity of the surface was improved. The obtained results indicated that after hydrolysis NaOH treatment at 3M and after 3h, the contact angle of all hydrolyzed nano-structures was determined 0° for all structures.
The contact angle of non-hydrolyzed and hydrolyzed neat and hybrid nano-structures at 1 M, 2 M, and 3 M NaOH and 1, 2 and 3 h as time submersion.

For electrospun nano-structures at 1 M concentration of NaOH, the contact angle of PCL75%:PU25% reduced from 92° ± 0.5° to 27° ± 1.6° (at 1 M NaOH after 2 h), from 92° ± 0.5° to 17° ± 0.9° (at 2 M NaOH after 2 h), and from 92° ± 0.5° to 10° ± 0.4° (at 3 M NaOH after 2 h). Also, the results of PCL25%:PU75%s contact angle have confirmed the 66°, 76° and 86° decrease at 1, 2, and 3 M NaOH after 2 h. In neat electrospun nano-structures were observed 55°, 68° and 82° of reduction of contact angle for PCL and 72°, 82° and 91° for PU at 1, 2 and 3 M NaOH, respectively after 2 h.
Mechanical properties
The obtained results of the mechanical properties (stress, strain and Young's modulus) of the non-hydrolyzed and hydrolyzed nano-structures were presented in Table 4(a) to (c).
Characterization the mechanical properties of neat and hybrid nano-structures before and after NaOH hydrolysis treatment: (a) Stress, (b) strain, and (c) Young's modulus.
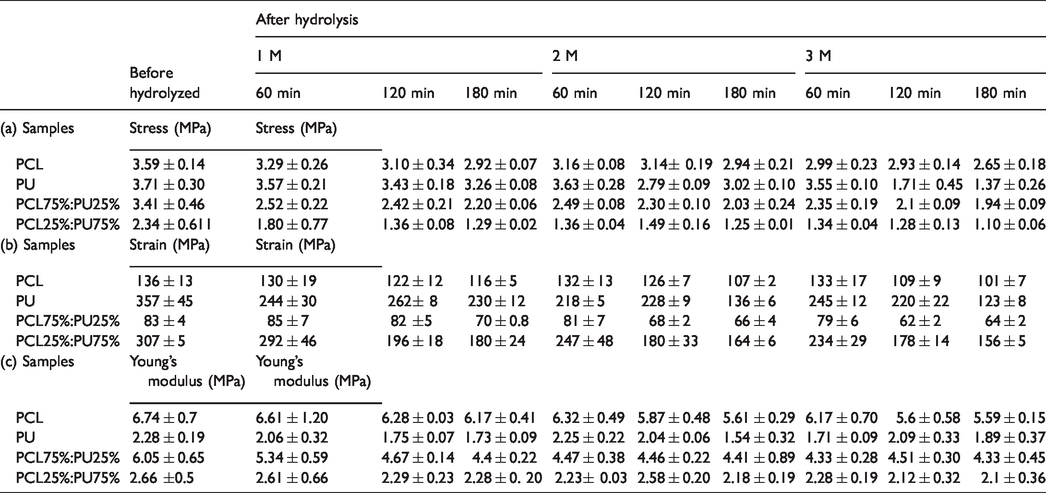
The stress, strain and Young's modulus of hybrid nano-structures with more composite ratio of PCL (75% of PCL), has been reported 3.41 ± 0.46 MPa, 83 ± 4% and 6.05 ± 0.65 MPa, respectively) Table 4(b)). Also, in structure with 25% of PCL, 2.34 ± 0.611 MPa, 307 ± 5% MPa and 2.66 ± 0.5 MPa were obtained for stress, strain and Young's modulus.
PCL is a semi-crystallization polymer and as an ideal reinforce was applied with another approved FDA polymer in fabrication suitable scaffolds for TE applications. The obtained results (Table 4(b) and (c)) have confirmed by increasing the composite ratio of PCL as a strength polymer in non-hydrolyzed hybrid nano-structures, the Young's modulus increased and the strain decreased.
Also, in non-hydrolyzed hybrid nano-structures with higher composite ratio of PU as elastomer polymer (0.75% of PU) the Young's modulus decreased and the strain increased compare to PCL75%:PU25% structure.
In the present study, the co-electrospinning process was used in order to achieve the hybrid nano-structures of independent and separated form of the PU and PCL polymer fibers (Figure 1). The results have demonstrated that in fabrication polymer scaffold by co-electrospinning method, polymers retain their unique properties in structures. In this process, two polymers were electrospun in a separate manner but they jointed with together on the surface of collector. Considering to the our obtained results [21], and other research [10], we hypothesize that in co-electrospinning process due to use different solvent with different evaporation rate, the residue solvent on the collector could stick polymer fibers to itself and to other fibers at junction point.
In addition, according to the obtained results in another study [15], in the PCL/PU as a hybrid nano-structures there are point-bonded between PCL as strength polymer and PU as a thermoplastic and elastomer polymer. Therefore more composite ratio of PCL’s fiber as strength polymer in hybrid nano-structures plays an important role in increasing the strength and decreasing the strain of the structure. The mechanical properties after hydrolysis treatment showed, the modulus Young's of PCL75%:PU25% nano-structures decrease from 6.05 ± 0.65 to 4.4 ± 0.22, 4.41 ± 0.89 and 4.33 ± 0.45 MPa with increasing NaOH concentration from 1 to 3 M NaOH after 3 h. Investigation the effect of NaOH hydrolysis treatment on the mechanical properties showed that the stress, strain and Young's modulus of of the hydrolyzed nano-structures decreased by increasing the NaOH concentration. That it seemed to be related to the degradation the fiber during the NaOH hydrolysis treatment and create the more porosity on the fiber in structures and therefore decreased the strength of the fiber in hydrolyzed nano-structures.
Considering to the mechanical properties of native soft tissue such as graft [19,21] and skin [35,36], the obtained mechanical properties of hydrolyzed PCL75%:PU25% nano-structures is within acceptable range for soft TE applications.
Also, in the higher concentration of NaOH and at the longer submersion time in NaOH hydrolysis treatment, the more erosion of fibers has happen. Therefore, in the case of NaOH hydrolysis treatment, according to the type of structures there are limitations in terms of NaOH concentration and submersion time.
The results demonstrated the greatest reduction in stress at 3 M NaOH 3 h (1.1 ± 0.06 MPa with 63% decreases), strain at 3M 3h (123 ± 8% with 65% decreases), and Young's Modulus at 2 M 3 h (1.54 ± 32 MPa with 32% decreases) were observed for PU structure. As a results of following treatment in neat structures, the PCL25%:PU75% structure showed the greatest reduction in stress and strain at 3 M 3 h (1.1 ± 0.06 MPa with 52% decreases) and (156 ± 5% with 49% decreases), respectively. Also the structure with 75% of PCL polymers indicated 28% reduction in Young's Modulus (4.33 ± 0.45 MPa) at 3 M NaOH after 2 h.
Conclusion
Persent study, PCL, PU and PCL/PU as electrospun neat and hybrid nano-structures with different composite ratio were fabricated by co-electrospinning method and characterized. The surfaces of neat and hybrid nano-structures were modified by a novel NaOH for water wettability significant improvement. The effect of the NaOH concentration and submersion time on the neat and hybrid nano-structures were systematically evaluated by examination of the surface morphology and mechanical properties. The obtained results, confirmed the optimum hydrolysis treatment conditions were found at 3 M NaOH 2 h. Considering to the FESEM examination, longer submersion time leads to degrade the surface fiber of all structures. In addition, the contact angle of all structures decreased by increasing NaOH concentration after 3 h. according to the obtained mechanical properties and structural features, the hydrolyzed PCL75%:PU25% hybrid nano-structure is promising candidate for TE applications.
Footnotes
Acknowledgements
The authors would like to express deep gratitude to Professor Ali Reza Modarresi-Alam for his valuable and constructive help in this research and for comments that greatly improved the quality of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by University of Sistan and Baluchestan (Grant No. 932/2/1006).
