Abstract
This work reports our recent efforts on improving the fire safety of nonwoven polypropylene fabric by increasing surface polarity followed by a conventional finishing. The fabric was firstly surface treated in a surfactant solution containing cetyltrimethylammonium chloride under ultrasonic to increase the hydrophilicity, and then was treated by a pad-dry process with a water solution of ammonium polyphosphate and pentaerythritol. The contact angle of polypropylene fabric treated with 0.2% cetyltrimethylammonium chloride was decreased to 71° from 130° of the control sample. The flammability was evaluated by limiting oxygen index, vertical flammability test, and cone calorimetry test. The limiting oxygen index value was increased to 23.5% from 18.1%, the peak heat release rate was decreased to 159.1 from 292.5 kW/m2, and the melt dripping was completely eliminated by the finishing treatment. Thermogravimetric analysis showed that treated polypropylene fabric samples had higher thermal stability and formed more char residue in high temperature range (above 450 °C) than the control polypropylene sample. The char morphology observation by scanning electron microscopy demonstrated that the treated fabric sample formed continuous char structure without voids, which effectively isolated underlying polypropylene fabric materials from fire and oxygen. It is proposed that decomposition compounds from both cetyltrimethylammonium chloride and ammonium polyphosphate catch free radicals in the gas phase, and the presence of ammonium polyphosphate and pentaerythritol promote char formation in the condensed phase during the burning process of polypropylene fabric.
Introduction
Rich sources and excellent processing properties make polypropylene fabric (PP fabric) widely used in indoor decoration materials, geotextiles, automotive textiles and so on [1]. However, the inherent flammability and burning with dripping of PP may cause fire hazards. Therefore, the demand for flame retardant PP fabric has been steadily increased in the last two decades.
The flame retardancy of textiles can be improved by finishing, chemical grafting [2,3] or introducing flame retardant during spinning process [4,5]. Polypropylene was mixed with melamine cyanurate and a mixture of phosphinate together and then melt spinning method was used to prepare flame retardant PP fabric. The LOI could reach up to 24.9% when 6% flame retardant was added [6]. Since the introduction of flame retardants during melt spinning could cause serious deterioration in mechanical and aging properties [7], the finishing method is widely applied. Microencapsulated diammonium phosphate has been blended with PU and coated on PP fabric. It is suggested that modified PP fabric shows better flame resistance and more char residues after combustion than pure PP fabric [8]. The LOI value of PP fibers coated with polysulfonamide (PSA) could increase to 25% [9]. Traditionally, halogenated flame retardants and antimony trioxide formulation are effective to endow flame retardancy to PP fabric; however, the release of corrosive smokes and toxic gases during combustion will cause negative effects on both human health and environment. Environmental friendly flame retardant in textiles have attracted more and more attention [10–12]. The inorganic flame retardants, including red phosphorus, magnesium hydroxide and aluminium hydroxide, also play important roles, but the high loading usually destroys the mechanical properties of PP [13]. Intumescent flame retardants (IFR) have been widely used because of their low cost, low toxicity, low smoke and dripping resistance properties [14]. It is demonstrated that IFR system could provide efficient fire proofing properties and keep other properties, such as mechanical properties, at an acceptable level [15,16]. Moreover, the adding of synergistic agents such as kaolinite could effectively catalyze the formation of protective char layer [17]. Numerous articles have been reported the use of IFR system on improving the flame retardancy of PP composites [18,19]. However, the direct application of IFR solution on the finishing of PP fabric has not been reported so far because of the inherent low hydrophilicity of PP fiber. Surfactant contains amphiphilic group which could improve hydrophilicity for PP fabric in a simple and effective way without affecting its bulk properties [20]. It was reported that little dosage of surfactant (0.0018 g/L and 0.002 g/L for hexadecyltrimethylammonium bromide and octadecyltrimethylammonium bromide, respectively) can significantly affect the hydrophilicity of PP fabric [21,22].
In this work, cetyltrimethylammonium chloride (CTAC) was selected to improve the hydrophilicity of PP fabric, and the IFR solution of ammonium polyphosphate (APP) and pentaerythritol (PER) was then applied to improve the fire resistance of PP fabric through a pad-dry process. The surface hydrophilicity of PP fabric was evaluated by contact angle tests. The flame retardancy, thermal stability of treated PP fabric were investigated, and the combustion mechanism was also discussed. The flame retardant PP fabric produced by this technology is expected to be applied to interior decoration (such as curtains, carpets, wallpaper, and bedding) and special clothing (such as disposable medical gowns and party costumes) [23].
It is expected that this work provides a possible method to introduce intumescent flame retardants on the PP fabric with minimal damage to the mechanical properties through facile conventional finishing.
Experimental
Materials
Polypropylene non-woven fabric with a density of 80 g/m2 was purchased from Dongguan Xinmei Textile Co., China. Water-soluble APP (degree of polymerization: 10∼20; solubility in water at 20 °C: > 400 g/L) was provided by Shandong Shi’an Chemical Co., China. Ammonium polyphosphate (APP), ethanol and pentaerythritol (PER) were provided by Beijing Chemical Company, Beijing, China. Cetyltrimethylammonium chloride (CTAC) was provided by Tianjin Guangfu Fine Chemical Research Institute, China.
Fabric preparation and treatment
The polypropylene nonwoven fabrics (PP fabrics) were firstly immersed in ethanol for 24 h and dried at 70 °C to constant weight in order to remove impurities, and then immersed in cetyltrimethylammonium chloride (CTAC) solution (the concentration was set as 0.1%, 0.2% and 0.3%) under ultrasonic for 5 min before being dried at 70 °C. The IFR solution with different concentration and mass ratio of APP/PER (Table 1 and Supporting Information Table S1) were prepared respectively. The above samples were finally treated with IFR solution through two dips and two nips with a laboratory padder. The pressure and the rotate speed of the padder are 0.3 MPa and 2 m·min−1, respectively. After that, the treated PP fabrics were dried at 70 °C for 2 h. The preparation of flame retardant PP nonwoven fabric is shown in Figure 1.
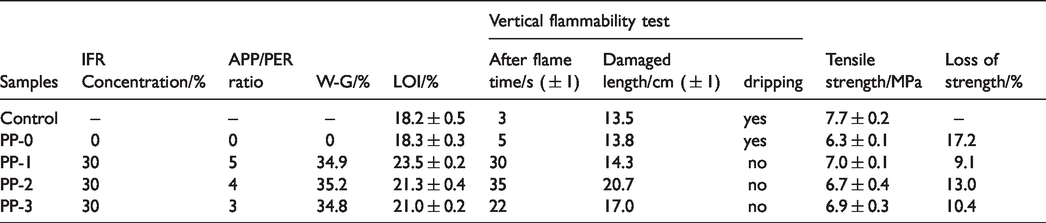
Flammability and mechanical property of PP fabric samples.

The preparation process of flame retardant PP nonwoven fabric through finishing.
Characterization
The contact angle of water on PP fabrics was analyzed through a SL200A contact angle meter from Solon Tech. (Shanghai) Inc., Ltd. Each sample was tested in five different areas, and the final results were averaged.
The limiting oxygen index (LOI) was obtained according to ASTM D2863-2000 by using a LOI instrument (JF-3, Jiangning, China). The dimensions of samples are 15 × 6 cm2.
The vertical burning test was carried out according to ASTM D6413-08 by using a CZF-3 instrument, Jiangning, China with a sample dimension of 30 × 8 cm2. Five specimens of each sample were measured and an average value was recorded.
The thermogravimetric (TG) tests were analyzed using a TGA Q500 TG analyzer with a sample mass of around 4 mg under a flow of nitrogen (60 mL/min) from ambient temperature to 800 °C. The heating rate is 10 °C/min.
The calculated char residue value is obtained according to the following procedure:
Calculated char residue = the percentage of FR × residue of FR + the percentage of fabric ×residue of fabric
For example, the calculated char value of PP-1 is obtained as follows:
The percentage of flame retardant is calculated according to the weight gain of treated PP-1 sample (34.9%):
The percentage of fabric: 100%−25.9% = 74.1%
Therefore, the theoretical char value should be:
Calculated char residue (700 °C) of PP-1 = 25.9%×residue of FR + 74.1%×residue of fabric = 25.9% × 18.4% + 74.1% × 7.1% = 10.0%
The cone calorimeter test (CCT) was carried out according to ISO5660 standard using a FTT007 Cone Calorimeter (Fire Testing Technology Ltd., UK). The external heat flux is 50 kW/m2. Five parallel samples were examined for each formulation in the cone test, and each sample contained 4 layers fabric with a size of 10 × 10 cm2. The mass of PP nonwoven sample is 4.04 ± 0.07g.
The surface morphologies of PP fabric samples and char residue samples obtained from the cone calorimeter test were observed by a scanning electron microscope (HITACHI S4700).
The possible flame retardant mechanism was discussed according to the data obtained from Fourier transform infrared (FTIR) spectrometer (Thermo Nicolet Nexus 670, USA) with a variable-angle attenuated total reflection (ATR) accessory (PIKE ATR Max II).
The mechanical property of PP fabric samples was measured according to GB/T 3923.1-2013 by using a universal testing machine (LR30K PLUS, Lloyd, United Kingdom) with a drawing speed of 10 cm/min. The result was averaged over 5 measurements per each sample.
Results and discussion
Hydrophilicity of PP fabric
The contact angles of PP fabrics treated with different concentrations of CTAC are shown in Figure 2. The contact angle of control PP fabric is 130°, because of the inherent hydrophobic structure of PP, indicating the water droplets cannot penetrate into the fabric. The contact angle of PP fabric decreases significantly after treatment with a CTAC solution, and reaches its minimal value of 69° after treated with 0.3% CTAC solution. The contact angle of the sample after treatment with 0.2% CTAC solution decreases to 71°, which is only 2° less than that of the sample after treatment with 0.3% CTAC solution; therefore, 0.2% CTAC solution has been selected in the process of preparing flame retardant fabric samples for further study. It is suggested that the lipophilic groups of CTAC are adsorbed onto the surface of PP fabric by van der Waals force, and the hydrophilic groups extend toward the air to form an adsorption layer. The layer with high surface energy effectively changes the surface wetting property of the original PP fabric.

Contact angle of: (a) control PP fabric; (b) PP fabric treated with 0.1% CTAC; (c) PP fabric treated with 0.2% CTAC; (d) PP fabric treated with 0.3% CTAC.
Flammability
LOI and vertical burning test analysis. Table 1 and Table S1 presents the flammability of PP fabrics. The LOI value of control PP fabric is 18.2%. The LOI values of all treated samples have been improved. One can see from Table 1 that the LOI of PP nonwoven fabric slightly increases with the APP/PER ratio. The LOI reaches 23.5% with a concentration of 30% at an APP/PER ratio of 5. The flammability of the fabric samples treated with different IFR concentration solution is listed in Table S1. The IFR solution concentration has minimal effect on LOI value. Vertical test results are also listed in Table 1 and Table S1, and digital images of control and treated PP fabric samples after vertical flammability tests are presented in Figure 3. The control PP fabric sample produces lots of drippings during burning, whilst the treated PP fabrics generate intumescent char layer without any dripping. It indicates that the finishing treatment actually increases the after flame time and damaged length due to the wick effect; however, the dripping has completely been eliminated by finishing. Subsequently, the APP/PER ratio of 5 and the concentration of 30% were selected according to the overall performance to treat fabric sample for the following investigation.

Digital photos of samples after vertical flammability test of (a) control; (b) PP-0; (c) PP-1; (d) PP-2; (e) PP-3.
The curves from cone tests are displayed in Figure 4, and some key data are listed in Table 2. We can see from Figure 4(a) that the peak heat release rate (pHRR) of control PP fabric is 292.5 kW/m2, while the pHRR of treated PP fabric is decreased to 159.1 kW/m2. The total heat release rate (THR) (Figure 4(b)), and peak smoke production rate (pSPR) (Table 2) are also declined after finishing. The CO production is increased (Figure 4(c)), however, the CO2 production is decreased (Figure 4(d)). It is suggested that free radical scavengers produced from CTAC and APP in the gas phase promote the CO formation, and reduce the generation of CO2. In summary, PP fabric samples treated with IFR have much better flame retardancy than the control fabric sample.

HRR (a), THR (b), COP (c), CO2P (d) curves of control PP fabric and treated fabric samples PP-1.
Cone calorimeter data of PP fabric samples.

Mechanical properties
The mechanical properties of PP fabrics are listed in Table 1 and Table S1. It is obvious that the tensile strength of samples decreases after finishing. The tensile strength of the PP fabric sample treated with 30% IFR solution with an APP/PER ratio of 5, drops from 7.7 to 7.0 MPa, however, the reduction of 9.1% is acceptable in many commercial cases. It is worth noting that the tensile strength of the fabric sample (PP-0) decreases to 6.3 from 7.7 MPa after padding without any solution. It is suggested the padding process with a pressure of 0.3 MPa causes physical damage to the nonwoven fabric, resulting in a decrease of the tensile strength.
The comparison of mechanical properties between this work and literature work was shown in Figure 5 [24–27]. It can be seen that the strength loss of finishing is much less than that of melt spinning.

The mechanical property comparison.
Thermal behavior analysis
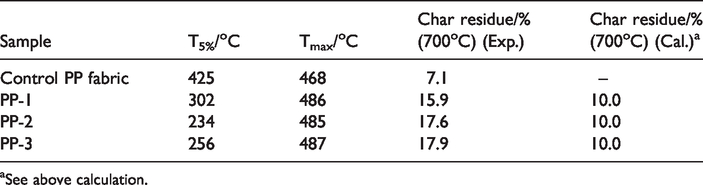
The thermogravimetric (TG) curves are shown in Figure 6 and Figure S1, and some related key data are summarized in Table 3 and Table S2. The treated PP fabrics have lower initial decomposition temperature (230-310°C) than pure PP fabric (425 °C), which is due to the early decomposition of APP and PER. However, Tmax (the temperature at the maximal thermal decomposition rate) of treated samples is about 20 °C higher than that of pure PP fabric, which is associated with the decomposition of PP backbone and the char formation process. It is suggested that polyphosphoric acids and polymetaphosphates produced from the degradation of APP promote the char formation which can protect PP backbone from further degradation [28]. It is apparent that the char residue of the treated PP fabric is much higher than that of the control sample, and it reaches 15.9% when treated with 30% IFR (5APP/PER) solution (PP-1).

TGA and DTG curves of PP fabric samples.
TGA data of PP fabric samples.
aSee above calculation.
In order to investigate the char forming effect of IFR formulation on PP fabric, theoretical char residue of treated PP fabric was calculated based on TG curves of control PP fabric and IFR in Figure 6. The experimental char residue at 700 °C is not less than 15.9%, which is much higher than the calculated value of around 10%, indicating the finishing significantly promotes char formation of PP fabric.
Surface morphology
Figure 7 are the surface morphologies of control PP fabric and PP-1 samples before and after cone test. The surface of control PP fabric is smooth (a), while PP-1 is rough with lots of accumulated flame retardant particles (b). Figure 7(c) shows that the outer surface of char sample is continuous without voids. This structure effectively isolates PP substrate from fire and oxygen, and protects the underlying fabric materials. In contrast, the internal char residue in Figure 7(d) exhibits an intumescent structure with many small closed cells and some open voids. The gas released from blowing agent form bubbles in viscous carbonaceous liquid during the combustion, and continuously enlarge bubbles to form intumescent char structures.

SEM images of the control PP fabric (a), the treated fabric sample PP-1 (b), char SEM images of sample PP-1 (surface layer (c), and inner layer (d)).
Characterization of char forming process
PP fabric treated with 30% IFR solution (PP-1) was calcined in a muffle furnace at 300, 400, 500, 600, 700 °C, respectively for 5 minutes, and the chemical structures of char residue have been investigated by FT-IR. The digital photos and the FTIR spectra of condensed products are shown in Figure 8.

FTIR spectra (a) and digital photos (b, c, d, e and f) of the treated fabric sample PP-1 calcined at different temperatures.
It can be seen from Figure 8(b), (c), (d), (e), (f) and (g) that PP fabric shrinks and melts with no obvious char structure at 300 °C. The black char layer starts to form and expands at 400 °C. The char volume reduces at 700 °C. It is suggested that the bubbles should be broken to release gas and cause the contraction of char structure at high temperature, meanwhile, the degradation of char at high temperature can also reduce the char volume.
In FTIR spectra, the peaks in 3500-3000cm−1 at 25 °C correspond to NH4+ in APP [18], and they disappear at 300 °C. Other peaks at 2950-2830cm−1 and 1450-1370cm−1 are characteristic peaks of -CH3, -CH2- of PP [29,30], and these peaks almost disappear at 500 °C. The peaks at which refer to P = O, P-O and P-O-P respectively appear at 500 °C [31,32]. The peak at 1643 cm−1 is ascribed to the absorptions of C = C bond [33,34]. It is suggested that APP decompose to release NH3 under 300 °C, and form P = O, P-O and P-O-P, and catalyze the PP chain and PER to form intumescent char layer.
Mechanism analysis
Based on the above test results, the possible flame-retardant mechanism of PP fabrics treated with solution of APP and PER via finishing is proposed and presented in Figure 9. In the gas phase, APP, PER in association with hexadecyltrimethylammonium chloride (CTAC) release inert gases (such as NH3, CO2, H2O and HCl) which can dilute the combustible fuels. Moreover, the presence of HCl and PO2·can catch active small free radicals like H· and OH·, which can effectively inhibit the oxidative radical reactions during the burning process [35]. In the condensed phase, the phosphoric acids produced from APP catalyze both PER and PP substrate to form more char residue (supported by TGA result) [28], resulting the formation of protecting char layers which can defer the transfer of heat, oxygen and combustible gases. The combination of both gas and condensed phase actions has resulted the significant improvement of fire performance for treated PP fabric, however, the effect in the condensed phase is the main contribution in terms of the small amount of HCl and PO2 free radicals.

The schematic diagram of flame-retardant mechanism of PP fabrics.
Conclusion
The surface treatment with a solution of cetyltrimethylammonium chloride (CTAC) effectively improved the hydrophilicity of PP fabric, which enabled the introduction of water soluble flame retardants onto the nonwoven fabric through conventional finishing. The flame retardancy of PP nonwoven fabric was significantly enhanced by the presence of APP/PER: The LOI was increased from 18.2% to 23.5%, and the char residue was increased to 17.9% from 7.1%. The pHRR, THR and pSPR values were decreased remarkably. The melt dripping of PP fabric was eliminated completely. The finishing process used in this work caused much less mechanical property loss to PP nonwoven fabric than the melt spinning process. It is proposed that APP/PER and CTAC release inert gases which can dilute the combustible fuels, produce free radicals scavengers which can effectively inhibit the oxidative radical reactions during the burning process. It is also suggested the continuous and compact intumescent char layer structure formed during the combustion is the main contribution to the improved flame retardancy of PP fabric.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The National Key R&D Program of China (Grant No. 2018YFD1100403) and the National Natural Science Foundation of China (Grant No. 21674008, 21875015, 51803007) financially supported this work.
Supplemental Material
Supplemental material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
