Abstract
In this study, we examined the notable textile materials in the literature, which have been produced to prevent the electromagnetic waves emitted by the electronic devices that are a significant part of our lives. Nowadays, these devices and technologies that bring us many advantages in our daily life cause health problems on human beings, which leaded this type of studies to deepen. In this study, non-woven and woven products were examined. Produced tissue types found in the literature were excluded from the research. This literature review is mainly focused on the textile materials treated with pyrrole and having electromagnetic shielding property by producing via different methods either by doping metal in the fabric or yarn structure or by weaving with metal. Because it is known that the most successful studies on electromagnetic shielding were made by polypyrrole coating. In the last part of the study, suggestions have been submitted about the type of studies that should be done to provide electromagnetic shielding feature to the textile materials. We investigated 56 articles about electromagnetic shielding in the literature. 48 of 56 articles were evaluated. It seems that the researchers used to polypyrrole for electromagnetic shielding on textile surface, however they don’t investigate to advantage and disadvantage of polypyrrole for using. We discussed this issue and which material was more effective for electromagnetic shielding on the textile surface in the discussion section.
Introduction
The wavelength of the electromagnetic spectrum increases from gamma rays to radio waves. In particular, sources of radiation in the X and gamma regions emit ionizing electromagnetic radiation, which is very dangerous. This is because, in this region of the spectrum the wavelength of light is very low. At this point, the beam acts as an energy cluster rather than a wave. These energy clusters are called quantum or photons. Photons in this region carry high intensity energy. In the other regions of the electromagnetic spectrum, light sources emit radiation in different amounts of energy depending on their wavelength and their energy levels are lower. There are many studies concerning radio frequencies, microwaves, infrared, visible and UV rays and the electromagnetic field emitted by the devices that use or transmit these rays and their negative effects on the biological systems and human health [1–4] (Stress, Sleeping disorder, heart health … etc.).
As we know for each region on the electromagnetic spectrum, a very different electronic device has been developed to work on these regions. Although these devices simplify our lives, the typical or leak waves that they release around the world have begun to affect human life negatively. For this reason, we have begun to investigate the ways of protection as we benefit from these devices.
The way to get rid of these effects is again using metals [5]. However, they are inflexible, expensive and heavy, they are open to oxidation and corrosion, which create a limitation for using these metals directly. Researchers have begun to use textile surfaces obtained from conductive polymers and textile-based composite materials instead of metals [6–9]. These materials, which are used instead of metals, have become more preferred recently due to their features such as being light, flexible and low cost [10].
In fact, these conductive textile materials are used not only for electromagnetic shielding but also in other fields such as sensors [12–14], antistatic applications [15,16], electrocardiograms [17], capacitors [18], wearable health monitoring systems [19]. The methods such as coating the textile surface with a conductive polymer [16–17], and placing metal particles into the textile layer [11,22,23] are used to provide conductivity in the use of textile materials.
Researchers have produced a wide range of electromagnetic shielding textiles with different compositions, via different techniques, and have measured the performance of these products. Some researchers have even considered the mesh geometry of these products and examined its effect on the measurements. They concluded that when a textile product does not contain metal in its structure, it is not efficient in electromagnetic shielding [24].
Polypyrrole is most used material for electromagnetic shielding on textile material surface. Polypyrole is a conductive polymer, so it is very useful material for using of electromagnetic shielding. The conjugated π electrons of polypyrrole conduct to electrical conductivity. The electromagnetic waves are absorbed by π electrons of polypyrrole. The Textile industry search to polypyrrole with this property for electromagnetic shielding.
Therefore, we tried to compile the studies that researchers have performed on electromagnetic shielding rather than the type of knitting. Regarding the works, we examined, we took into account the works on the pyrrole used textile materials prepared via coating. Thus, we examined how pyrrole is used in this area and how effective it is and what kind of problems it poses. We have tried to limit and classify our work in this way because there are too much data and resources related to products produced for electromagnetic shielding.
In this study, we divided the paper as 5 sections. In the next section was described Electromagnetic Shielding and Measurement Techniques. In the Textile materials with electromagnetic shielding section was investigated literature about: polypyrrole using and coating methods on textile material surface, metal doped or other compounds combining with polypyrolle and its’ electromagnetic shielding properties. In the Discussion section was described advantage and disadvantage of using polypyrrole for coating textile materials. End of the paper (in the conclusion), we made some suggestions for researchers in the future studies.
Electromagnetic shielding and measurement techniques
In this section, electromagnetic shielding and the measurement methods are discussed. In fact, the terms used in electromagnetic shielding, although slightly modified, have similarities with the optical phenomenon that the light experiences when passing through a material (Figure 1).

Exposure of a sample to the beam [25].
Regarding the electromagnetic wave coming to sample with intensity Eo, a part of it is absorbed by the sample while some part is reflected (R) and some of it continues its way by passing across the sample (I). SE (shielding effectiveness) or SE (shielding efficiency) is a parameter that shows how effective the shielding is and it is expressed in decibel (dB). SE value is calculated by the following formula.

It is difficult to apply standard tests for such measurements. Because there are many parameters such as the direction of the measurement, the polarization, the distance of the receiver and transmitter devices to the screen. There are two techniques in the literature, namely insertion loss technique and enclosure measurement [26–27]. “Enclosure measurement” technique is considered to be a better test to measure at higher frequencies compared to the “insertion loss technique” [28]. Figure 2 illustrates a measurement design made by enclosure measurement technique.

Enclosure measurement [28].
It is known that there are various standard test methods, new measurement methods, and measurement device recommendations to measure the shielding efficiency of the materials used in the shielding of electromagnetic waves. Chen et al. [28] used ASTM D4935, ASTM ES7 standards and a special measuring device developed by themselves. The study by Foster et al. [29] introduced a test method used for electromagnetic shielding control in small electronic devices that provide wireless communication. The study gave information about ASTM D4935 and Mil Std 285 standards that are used to test electromagnetic shielding efficiency and the suitability of relevant standards for electromagnetic shielding detection tests in small size wireless electronic device applications [30]. In the study by Ueng and Cheng [31], measurements were made according to EN EMC directive 89/336/ECC and EN-50082-1: 1992, EN-50081-:1992 test methods using D-2000 microwave leak detector to determine the electromagnetic shielding efficiency of the samples. In addition, a specially designed test device was used for the surface and volume resistance tests of the samples. Surface and volume resistance tests were performed according to JIS K 7194 standards. For EMSE (Electromagnetic Shielding) tests of the woven fabric samples, the customized coaxial transmission line holder method defined in ASTM ES 7-83 standard was used. The study by Wieckowski and Janukiewicz [31] provided information about the standard test methods (Mil Std 285, ASTM D4935- Figure 3) used for the measurement of electromagnetic shielding efficiency in thin planar materials and a special measuring device was designed. The measuring device consists of network analysis unit (circuit analyzer), coaxial transmission line measuring unit, power amplifier, antennas and transmission lines [32].

Measurement device of Modified Principle Mil Std 285, ASTM D4935 standard [31].
Textile materials with electromagnetic shielding
Pyrrole used textile materials produced by coating via chemical method
It is a technique used mostly by chemists. This method worked very well in the textile sector. An example of the polymerization with vapor phase test setup is shown in Figure 4. In the Figure 4, pyrrole solution in the N2 atmosphere sprayed on the cotton surface. At the same time, the cotton is passed through the medium with the pyrrole solution at a constant speed.
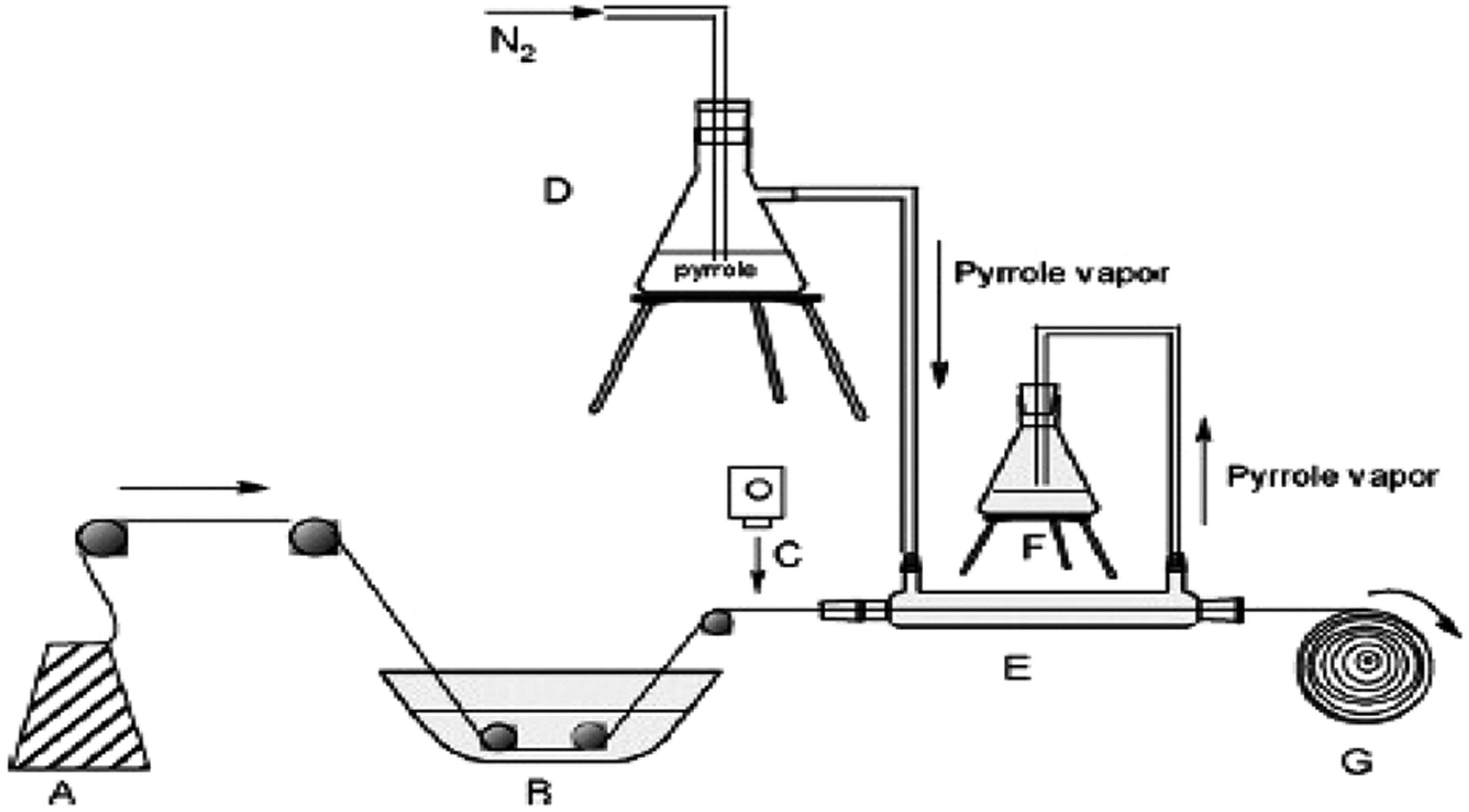
Polymerization with vapor phase test setup [33].
In the presence of iron (III) chloride starter solution, it is possible to coat cotton and polyester with polypyrrole by using polymerization with vapor phase technique. It is a very good method for yarn coating. Polypyrrole-coated yarns have different tensile strengths, electrical and morphological properties at different concentrations and electromagnetic shielding properties of the fabrics weaved with these cotton yarns vary as well.
Figures 5 and 6 illustrate EMS variations when different concentrations of iron (III) chloride are used. The highest EMS was observed when 0.6 M iron (III) chloride was used [33,34].

EMS values of cotton fabrics containing different concentrations of FeCl3 [33].

EMS values of polyester fabrics containing different concentrations of FeCl3 [34].
The ferroelectric properties and the high dielectric constant of Barium titanium oxide (BaTiO3) [5,35–38], which is used as filler in applications such as optics, sensors, IR detectors, make it useful in these areas. It also has electromagnetic shielding properties, because of electronic structure [38–40]. It is possible to coat the fabrics coated with Barium titanate poly (acrylonitrile-co-methyl acrylate) nanocomposite with polypyrrole through the polymerization process (Figure 7). Figure 8 shows EMS measurements of the fabrics prepared with different pyrrole concentrations. In the Figure 8(a) is shown FeCl3, PPy and BaTiO3 mixings and in the Figure 8(b) is shown different PPy concentrations with FeCl3 and BaTiO3.

Process scheme of polypyrrole deposition on nanocomposite coated fabrics [5].

EMS values of Barium titanate poly (acrylonitrile-co-methyl acrylate) nanocomposite coated fabrics [5].
The fabrics coated with BaTiO3 have a better EMS value than those coated with PPy-FeCl3. EMS values of all samples, except PPy-BaTiO3-P (AN-co-MA) coated fabrics, are increased in the frequency ranges of 1800-2100 MHz, 2150-2400 MHz and decreased over 2700 MHz. In the 2700-3000 MHz range, only the EMS values of the coated fabrics increased. The highest EMS value was obtained at 0.3 mol/L pyrrole concentration. It should be noted that pyrrole concentration played an important role again [39]. It is also possible to carry out mixed polymerization processes with pyrrole and polyurethane, which is prepared by adding very small amounts polyurethane compared to pyrrole. As can be seen from Figure 9, they have very good EMS values for coating carbon-based fabrics and yarns [40].
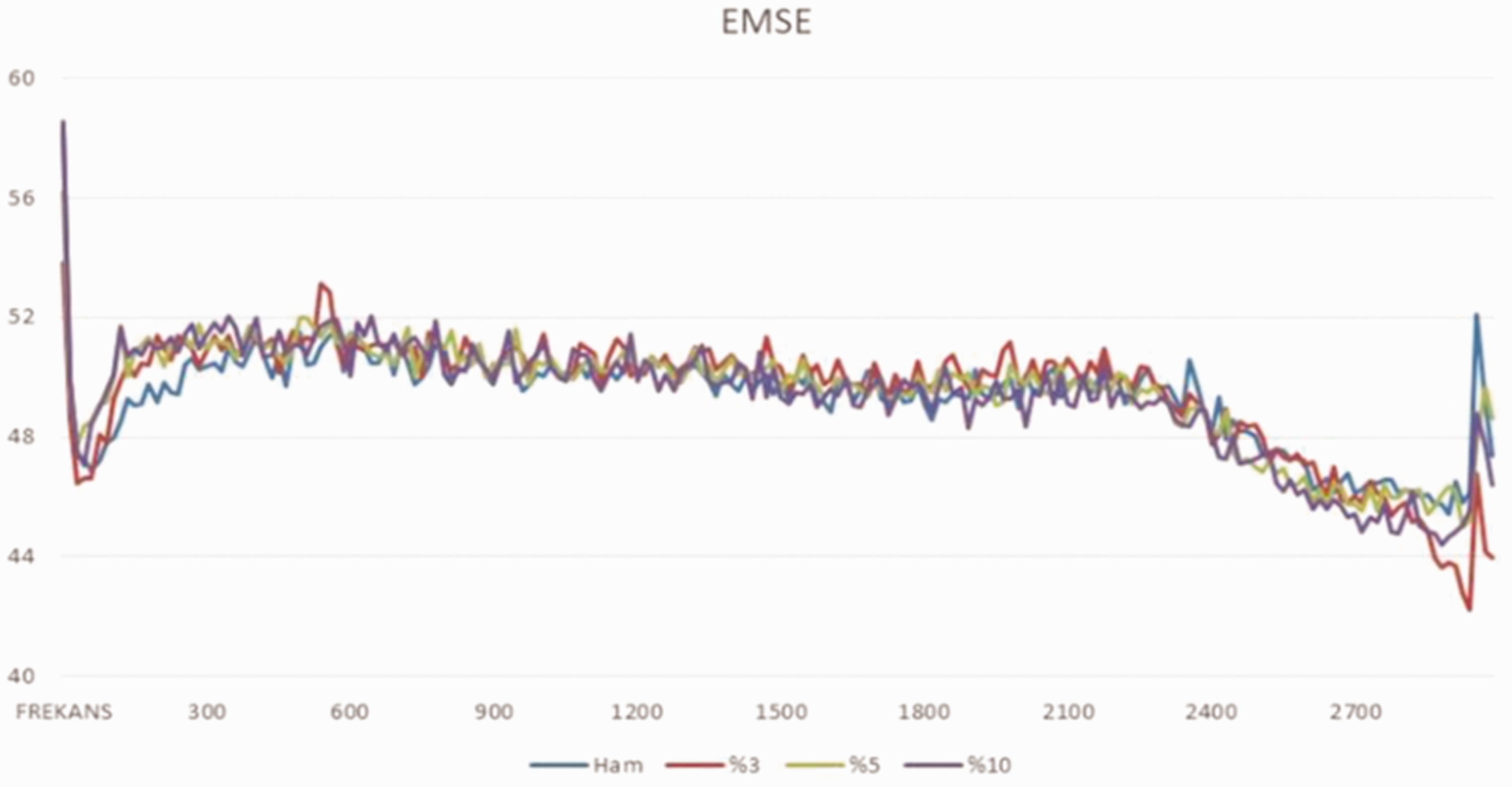
EMS values of pyrrole and polyurethane coated carbon fabrics [40] (X axis: EMSE, Y axis: Hz).
Pyrrole used textile surfaces produced by electrochemical coating
Regarding the pyrrole used coating made via polymerization with vapor phase, iron III chloride is not the only material used as starter. Some studies are convincing that the reaction can be accelerated by reagents and electrochemical methods that will allow more rapid polymerization of pyrrole. At the same time, it is seen that the EMS properties of textile materilas produced by electrochemical coating are better than the products produced by chemical polymerization technique. For this process; pyrrole dissolved in a polyvinyl alcohol or non-alcoholic aqueous solution, which is used as the surfactant, first it is sprayed on a PET fabric, and then it is oxidized by spraying an aqueous solution of the oxidant (Ammonium peroxydisulfate, APS and iron (III) p-toluene sulfonate, FTS) and the additive (1-naphthalene sulfonic acid, NSA). The polymerization time can be shortened up to 100 min. to determine the polymerization time of PPy on the fabric. While APS and FTS are compared, it was found that the surface resistance was lower in synthesis via FTS (Figure 10).

Comparison of APS’ and FTS’ oxidant effect [41].
Subsequent electrochemical polymerization is carried out in an aqueous electrolyte solution, by applying a constant current density at room temperature to PPy coated (chemically) fabric and to the stainless-steel plate as working and counter electrode. An aqueous solution containing Anthraquinone-2-sulfonic acid sodium salt (AQSA-Na) (0.01, 0.03, 0.05 or 0.07 M) and 0.3 mol/L pyrrole is used as the supporting electrolyte solution.
Regarding Figure 11(a) and (b), it can be seen that FTS is a better oxidant than APS, providing better PPy coating. Figure 11 is the SEM images of the synthesis of the polypyrrole with FTS and APS. As we can see that synthesis of the polypyrrole with FTS have more suitable surface than the synthesis of the polypyrrole with APS.

(a) FTS (b) APS [41].
It was found that the conductivity of the composite increased with electrochemical polymerization. It is seen that electrochemically synthesized PPy has much higher electrical conductivity compared to chemically synthesized PPy.
EMS SE, reflection (Re), absorbance (Ab) and permeability (Tr) data of PET fabric/PPy composites with various volume resistances produced by this method are given in Table 1.
EMS SE, Ab, Re and Tr values of PET fabric/PPy composites with various specific volume resistivity [37].

As shown in Table 1, there is only a small change in SE, Re, Ab or Tr in the frequency range of 50 MHz–1.5 GHz, thus researchers provided the average values of SE, Re and Ab as a function of the specific volume resistivity in Figure 12.
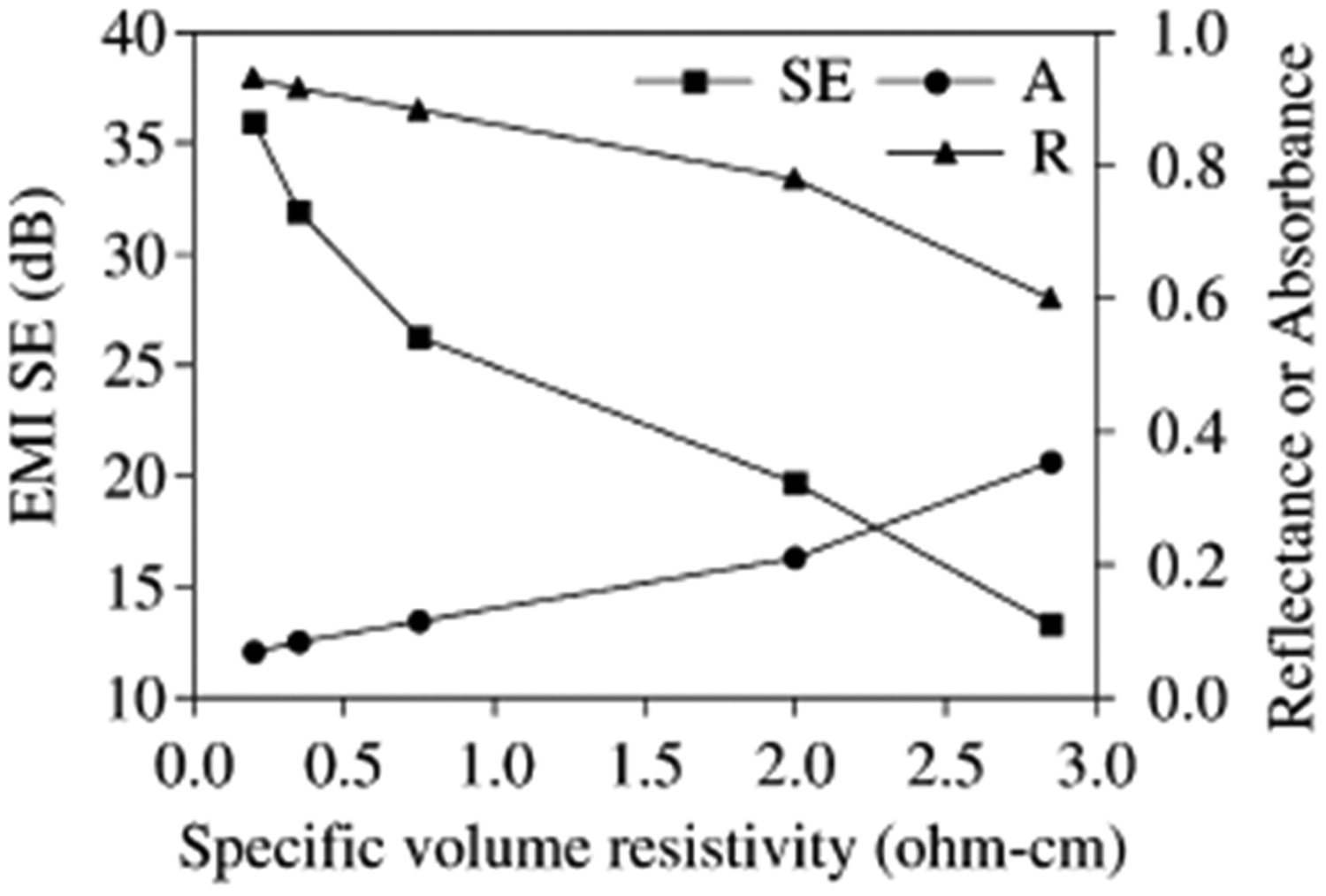
SE, R and Ab of PET fabric/PPy composites with specific volume resistivity [41] (A: Absorbance, R: Reflectance).
The review of the results indicated that the value of EMS SE was found to increase from 13 to 26 dB by decreasing the specific volume resistivity from 2,85 to 0,75 ohm-cm and then increase to 36 dB at 0,75 ohm-cm by increasing the conductivity rapidly. The study suggests that the shielding efficiency can be easily controlled through electrical conductivity [41].
Pyrrole used textile materials produced by chemical and electrochemical coating methods (multiple layers)
Researchers showed that, when a single layer of coating is not sufficient, they may not be able to coat, especially Cu and Ni layers, by electrochemical deposition method. It was found that layered linen fabric/polypyrrole/nickel (LF/PPy/Ni) composites can be produced by combining a chemical in situ polymerization approach with a chemical coating method. LF/PPy/Ni composites are synthesized in 2 steps. (I) The PPy layer is built on LF substrate by self-polymerization of the pyrrole monomer. Pyrrole is used as monomer, ferric chloride (FeCl3) as oxidant and sodium p-toluene sulfonate (STS) as dopant. (II) Nickel film was placed on 3-aminopropyltrimethoxysilane (APTMS) self-assembled single layer modified LF/PPy by electrolyzed nickel coating. APTMS used here serves as a molecular bridge between organic (PPy film) and inorganic (Ni layer) [38]. The synthesis route of LF/PPy/Ni composites is shown in Figure 13.

Synthesis route of LF/PPy/Ni [43].
In this study, the researchers evaluated the quality of the PPy layer in terms of both EMS SE and Rs (surface resistance). They knew that the quality of the PPy layer depends on the amount of the starter. To obtain LF/PPy composites, FeCl3 concentrations were tested in the range of 0.2 to 0.8 mol/ml. The graph showing the EMS values is given in Figure 14(a). Regarding the results, they found that the ideal SE and the lowest Rs value for LF/PPy composites were obtained at 0.4 mol/ml concentration. They investigated the effect of polymerization time on EMS SE in the range of 5-20 min and they determined that EMS SE values of composites increased in the range of 5-10 min. Therefore, they set the ideal polymerization time as 10 min. The graph showing the effect of polymerization time on EMS SE is given in Figure 14(b). Coating thickness was also effective for EMS shielding products. In this study, synthesis cycles in the range 1-4 were screened. Due to the difficulty of coating the homogeneous PPy film, high Rs and low EMS SE values were obtained when N = 1 and 2 (N: cycle number). They observed a decrease in Rs and an increase in EMS SE when N = 3. There was no significant increase in EMS SE when N = 4, but thick PPy binding occurred. Therefore, the ideal coating thickness was determined to be n = 3. The graph showing the effect of synthesis cycles on EMS SE is shown in Figure 14(c).

The effects of a) concentration b) polymerization time c) synthesis cycles on EMS SS SE [43].
The EMS SE graph of LF/PPy, LF/Ni and LF/PPy/Ni is given in Figure 15. The review of the results revealed that the composite products containing both PPy and Ni had higher SE value (20.22–43.51 dB) than the products containing PPy or Ni. In other words, it is suggested that LF/PPy/Ni composites can be used as EMS shield in electronic products such as radio receivers, television sets, video recorders, DVD players, digital cameras, cameras, personal computers, video game consoles, telephones and mobile phones.

Shielding effects of a) LF/PPy, (b) LF/Ni and (c) LF/PPy/Ni [43].
The shielding mechanism of LF/PPy/Ni composites is given in Figure 16. It is suggested that in case of double layer coating, the electromagnetic waves passing through the first layer will be further absorbed by the other layer [43].

Shielding mechanism of LF/PPy/Ni composites [43].
In the same manner, copper was coated as the second layer using electrochemical method. Multilayered cumprammonium fabric/polypyrrole/copper (CF/PPy/Cu) composite was produced. PPy layer can be coated onto the CF substrate by in-situ polymerization. By using STS (sodium p-toluene sulfonate) as dopant and FeCl3 (iron (III) chloride as an oxidant), PPy can be coated on CF via two-step bath process. The Cu film can then be built on PPy coated CF (PPy/CF) using electrolytic copper coating method. The production scheme of the produced CF/PPy/Cu composites is given in Figure 17.

The production scheme of CF/PPy/Cu composites [44].
The mechanism used to examine the electromagnetic shielding properties of this composite is shown in Figure 18. The graph showing EMS SE values for CF/Cu and CF/PPy/Cu is given in Figure 19. When the results were compared, it was found that the CF/PPy/Cu composite has a quite high SE value of 30.3–50.4 dB, in 30–1000 MHz range. As a result, it was found that CF/PPy/Cu composite has a higher SE value than the other reported conductive textiles and the proposed CF-based composite is suitable for EMS shielding [44].

Shielding mechanism of CF/PPy/Cu composite [44].

Shielding efficiency of (a) CF/Cu and (b) CF/PPy/Cu [44].
Pyrrole and polyaniline used textile materials produced by ink-jet coating method (multiple layers)
Polyaniline (PANI) is one of the best conductive polymers because of its environmental stability [45] and partial solubility in various solvents. Its high conductivity, controllability, thermal and chemical stability, and low specific gravity can be named among the reasons of being a versatile polymer [46]. Therefore, as polypyrrole, polyaniline is also suitable for electromagnetic shielding in the textile industry.
It is also possible to observe method trials for coating polyaniline (PANI) and polypyrrole (PPy) on different textile fabrics via reactive ink-jet printing. PANI and PPy coated fabrics can be obtained by chemical oxidation of aniline hydrochloride and pyrrole with ammonium persulfate on polyacrylonitrile, cotton, polyethylene terephthalate (PET), cotton/PET, wool and cotton/wool fabrics. The method of obtaining PANI or PPy via oxidative ink jet of aniline hydrochloride or pyrrole is shown in Figure 20. The ink jet printing technique is given in Figure 21.

PANI and PPy formation scheme [47].

PANI and PPy deposition scheme in fabrics via ink jet printing method [47].
The mechanism for coating PANI and PPy layers on the textile surfaces is given in Figure 22.

PANI (a) and PPy (b) formation mechanism on PAN fabric [47].
Electromagnetic shields are obtained with highly promising results of conductive textile materials produced with PANI and PPy. The values showing the electromagnetic shielding efficiency of PANI/PAN and PPy/PAN composites are given in Figure 23. The average EMS SE value for PANI is 11.1 dB, whereas EMS SE value for PPy is 2.8 dB. These results are in line with the results published in the literature [5,13,14,20,21,33,39,42,43,47]. Regarding the polymers without additives, EMS SE was found as 6–13 dB for PANI/fabric composites, and 1–10 dB for PPy/fabric composites in the frequency range of 30–1500 MHz. Constantly increasing values were observed. The highest value was 25 dB for PANI, whereas the highest value of PPy was 9.3 dB. The review of the results for both conductive polymers showed that PANI had higher EMS SE than PPY [47].

EMS shielding efficiency of a) PANI/PAN and b) PPy/PAN composites according to frequency and conductive polymer layer thicknesses [47].
Textile materials produced by polymerless coating methods
Researchers showed that metal coating can be performed on polymer yarn surfaces without using pyrrole. The best example is the coating of metals such as aluminum, copper, titanium and silver onto the surface of polyester (PET) substrates using a vacuum coating system.
As these metals have different evaporation rates, the coating thicknesses are different from each other. The properties of the coating materials are given in Table 2.
Properties of the coating materials [48].

Regarding fabrics weaved with metal coated PET filaments, it is also important to see how different metal thicknesses affected EMS values in 2250-2650 MHz frequency range. EMS graphs of the materials coated at various thicknesses are given in Ag (in Figure 24(a)), Cu (in Figure 24(b)), Al (in Figure 24(c)) and Ti (in Figure 24(d)).

(a) EMS effect (efficiency) of sample A at different thicknesses [48]. ✦ 31.80 µm; ∇, 27.04 µm; ▲, 22.28 µm; ★, 16.58 µm; ■, 12.44 µm; ✧, 0 µm. (b) EMS (efficiency) of sample B at different thicknesses [48]. ✦, 34.66 µm; ∇, 27.67 µm; ▲, 23.28 µm; ★, 17.58 µm; ■, 14.69 µm; ✧, 0 µm. (c) EMS (efficiency) of sample C at different thicknesses [48]. ✦, 33.80 µm; ∇, 27.04 µm; ▲, 24.34 µm; ★, 15.52 µm; ■, 11.04 µm; ✧, 0 µm. (d) EMS (efficiency) of sample D at different thicknesses [48]. ✦, 35.16 µm; ∇, 28.49 µm; ▲, 25.33 µm; ★, 16.23 µm; ■, 12.39 µm; ✧, 0 µm.
EMS relations of the coating materials at 2450 MHz frequency are shown in Figure 25. According to these results, it was found that sample A had the highest EMS value whereas sample D had the lowest EMS value. It was found that EMS values of the coated fibers varied with coating thickness. It is known that this is due to electrical conductivity of Ag (in Figure 24(a)), Cu (in Figure 24(b)), Al (in Figure 24(c)) and Ti (in Figure 24(d)) have, decreasing respectively [48].

The effect of film thicknesses on EMSE [48]. ■, sample A; Δ, sample B; ✧, sample C; ★, sample D.
And also, we found new materials for coating co-polymers with polypyrrole in the literature. However, we are not evaluating these papers, because they arenot investigated electromagnetic shielding [48].
The other methods: Coating nanoparticles
Moazzenchi and Montazer [49] coated nickel nanoparticles on polyester fabric by chemical bath methods in Figure 26.

Coated nickel nanoparticles on polyester fabric [49].
They synthesized nickel nanoparticles were on polyester fabric. They used Nickel acetate (1–2.5%) and hydrazine hydrate (2–4%) for synthesized nickel nanoparticles. They found that nickel treated fabric has higher EMSE values than the untreated fabric in the Figure 27.

EMI Shielding untreated and nickel treated fabric.
Wang et al. [50] investigated EMI Shielding properties of CoFe2O4/CNTs nanocomposites. They synthesized CoFe2O4/CNTs nanocomposites by hydrothermal methods at 6-10 nm. CoFe2O4/CNTs nanocomposites exhibited excellent electromagnetic shielding performance with values 22–25 dB in the Ku frequency range with a thickness of 2 mm in Figure 28.
EMI Shielding of CoFe2O4/CNTs nanocomposites.
Karbownik et al. [51] studied an important issue about EMI shielding of nanoparticles in Figure 29. Silver and gold nanoparticles were coated on the structure of polyacrylonitrile (PAN) fibers as a result of a synthesis. They synthesized nanoparticles in the dimethylformamide (DMF) solution, was a typical solvent for polyacrylonitrile.
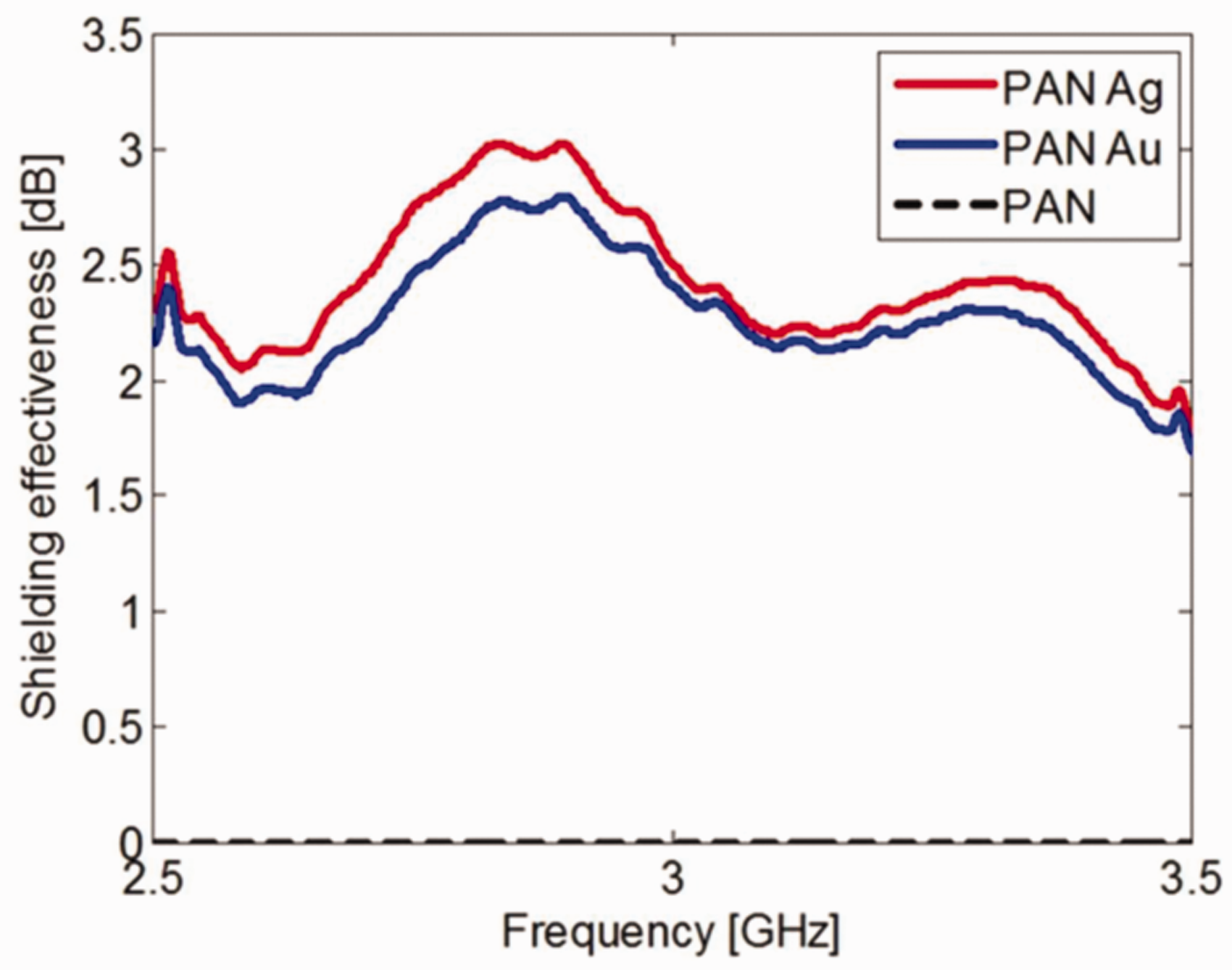
EMI Shielding of Ag/Au nanocomposites.
The average values of shielding effectiveness were found a frequency range of 2.5 GHz to 3.5 GHz for the samples are as follows: SEPANAg = 2.42 dB and SEPANAu = 2.27 dB.
Discussion
In this study, polypyrrole coating methods, which are frequently used in textile materials having electromagnetic shielding properties, were discussed and these methods were compared. Moreover, we tried to understand how EMS values have varied in the studies we have found in the literature and without using polypyrrole. It is important that which materials and methods have more effective EMS than the others with using polypyrrole. In Table 3, the highest SE values are investigated in the literature. The values that the authors could not specify exactly were read form the graphs and interpreted. These studies are the ones selected from the literature. In addition to these studies, it is possible to encounter a few studies but other studies that are overlooked will not obstruct the generalization that we will make.
Comparison of the notable studies in the literature.
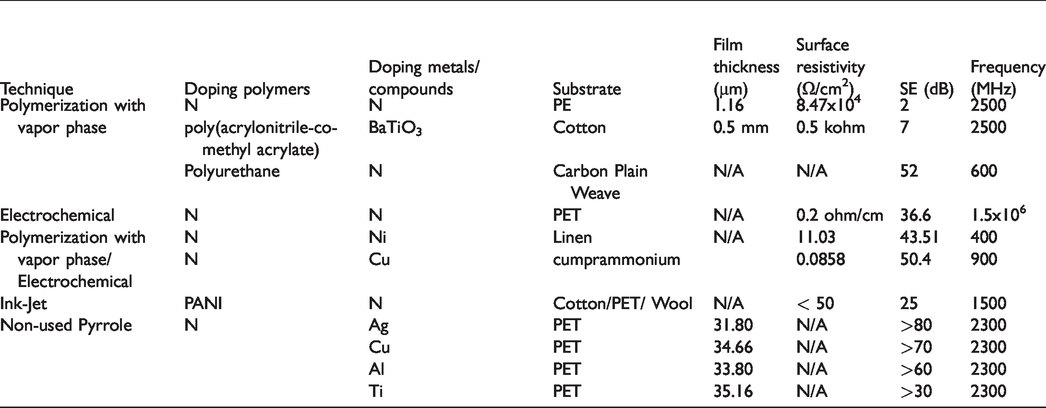
According to Table 3, pyrrole used textile coating produced by polymerization with vapor phase method are usually effective in electromagnetic shielding at high frequencies. However, it can be seen that electrochemical methods and multi-layer coatings were much more effective. Especially, the value of SE was found to be 43 dB through doping with metals having ferromagnetic properties, which is a pretty good result. However, a better SE value was observed when doped with copper instead of nickel, which indicates the importance of conductivity in electromagnetic shielding. Regarding Ink-Jet method, although it is used very rarely, it exhibited better electromagnetic shielding (25 dB) than chemical polymerization. The ink-jet method is more suitable for industrial applications than the other methods. Polymerization with vapor phase method have some risks for human health. The electrochemical method coating is more expensive cost than the other methods, however it shows quality surface coating. Fortunately, the ink-jet method is low cost and fast producing than the other methods.
On the other hand, metal coating under vacuum showed us that we can produce textiles products with very good electromagnetic shielding without using pyrrole. Ag surpassed other metals with a value of 80 dB and thanks to the superiority of its conductivity.
At same time, Ag have more electromagnetic shielding properties than the other metals. In the polymers, the polypyrrole is used more than the other polymers. However, what are the advantages and disadvantages of pyrrole?
It brings certain advantages, including: Chemical resistance, 2.Electrical conductivity, Low density.
At the same time, in case of doping metal to the surface, pyrrole can encapsulate or bind the metal ion to the textile surface during polymerization.
Regarding the disadvantages, the problem starts at this point. So far in the literature, only 1-2 articles have investigated the washing resistance of textile materials with EMS properties and they were not coated products. It is an inevitable that textile materials will ultimately face with water and washing. However, they will be exposed to many physical and chemical wear such as knitting and drawing. It is also possible to find some studies on this subject. Above all, the toxic properties of these products have not been examined by anyone. Pyrrole has very toxic properties. Despite these disadvantages, textile materials produced by coating with polypyrrole are seen to have superior EMS properties at low frequencies compared to others.
Conclusion
In this study, we have tried to point the missing points in the existing studies by reviewing notable works that are mostly read, in other words that are a kind of leader in EMS. We have tried to limit and categorize the samples as much as possible because there are a lot of papers in this field and too much data in these papers. Accordingly, the biggest deficiency in the studies is assessment of the durability of the products. In particular, the lack of toxicity analysis in the studies concerning the use of pyrrole and polymers with similar type toxic effect is a serious shortcoming. Since they have a serious impact on human health, researchers should make these tests on the samples of textile materials that they produce.
The researchers don’t want to selected polypyrrole in the future studies, because of toxic effect of the polypyrrole. They must study on toxic effect of the polypyrrole textile material. We have no evidence very toxic effect of the polypyrrole such as pyrrole.
In the future studies, the researchers will study on polyaniline and silver metal coating or silver doped polyaniline textile materials. The toxic effect of polyaniline is lower than the polypyrrole. The silver coatings are not given quality coatings on textile surfaces. However, the silver doped polyaniline coatings can be a good solution for these problems.
Footnotes
Acknowledgement
This study was carried out by R & D team of Comfytex company. We thank the employees of the company and Hasan KÜÇÜK, Hüseyin MUSLU and Selim ŞAHİN for their valuable contributions to the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
