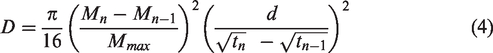Abstract
Natural fibers are increasingly being used as composite reinforcement for both thermoplastic and thermoset resin, mainly for automotive application. Due to their hydrophilic nature, natural fibers have certain limitations during composite manufacture especially owing to their poor resin wettability, weak fiber–polymer interface, high moisture absorption, and being affected by high temperature in case of thermoplastic resin. This work investigates the impact of sisal fiber modification techniques on moisture absorption, thermal, and mechanical properties of the fiber. Four sisal fiber samples were prepared; untreated, alkaline treated, acetylated, and a combined alkaline-treated/acetylation samples. The samples were evaluated for their hygroscopic nature, thermal stability, and tensile properties. It is found that acetylation resulted in a reduction of moisture absorption of sisal fiber as the acetylated and alkaline-treated/acetylated samples recorded a decrease of 42% and 28%, respectively. Alkaline treatment increased the absorbency owing to the removal of hemicellulose and lignin. The thermogravimetric result revealed that alkaline treatment improved the thermal stability as the alkali-treated and alkali-treated/acetylated samples showed improvement in thermal properties. The acetylated sample resulted in a significant reduction in tensile strength. But, the results from tensile tests of the alkaline-treated samples showed an insignificant decrease in tensile strength and improvement in the modulus for all treated samples. Fourier-transform infrared and scanning electron microscopic analysis were included in the study to supplement the results with structural and microstructural changes. The effect of those treatments on the sisal–PET composite properties was studied and will be submitted in part 2 of the study.
Keywords
Introduction
The concept of utilizing green materials has emerged as mainstream for industries, material scientists, and researchers. The issues of raw material sustainability, new directives on waste management, and considerable awareness on preventing environmental pollution has led to the utilization of renewable, bio-degradable, and environmentally friendly materials [1–6]. Although excellent mechanical properties characterize glass fiber composites, their end-of-life disposal and recycling is complicated and inappropriate disposal may result in environmental pollution, soil degradation and machine wear, and respiratory problems [5,6]. In this respect, cellulosic fibers are getting appealing as they possess advantages over conventional reinforcements such as low density, guarantee environmental safety, being renewable, ease of processing, and bio-degradable than traditional reinforcements of glass fibers with acceptable specific mechanical properties [2–7]. When ecological aspects in the material selection are considered, replacement of synthetic and inorganic fibers by natural ones is the only solution [3,7]. There is a wide variety of natural fibers that can be applied as a reinforcement with polymers. The most broadly utilized natural fibers are jute, flax, hemp, and sisal [2,3,7] due to their property and easy availability. Sisal fibers are bast fibers extracted from Agave sisalana Perrine leaves [2]. Naveen et al. [4] explained that sisal fiber has comparable specific strength and modulus with glass fiber [4–6]. Ramesh et al. [5] described the benefit of natural fibers in terms of ease of processing and environmental advantages [5,6]. They clarified that minimizing the content of glass fiber by hybridizing sisal with glass fiber as reinforcement can reduce the tool wear while processing and respiratory irritation problems. As detailed by numerous specialists, sisal fiber-based polymer composites can be applied effectively in automobiles, such as their internal engine covers, door panels, package trays, seat backs, and exterior or under-floor paneling [4,7–9]. Hulle et al. [1] explained that in contrast to other leaf fibers, sisal fibers are characterized by low density, high tenacity, and high moisture absorbency. The moisture absorption of sisal is profoundly subject to the chemical constituents of the fibrils [2,3,7,10]. Rohit et al. [2] listed the major chemical components of some of the natural fibers. Accordingly, sisal fiber is mainly composed of 65% cellulose, 12% hemicellulose, 9.9% lignin, and 2% waxes [2]. As far as its utilization as a reinforcement in thermoplastic matrices is concerned, its high moisture absorption is an undesirable property. Water molecules create polar differences and thus poor wettability with the thermoplastic matrices [11–13]. As concentrated in their writing by Farsi [7] and Shrivastava [10], the main drawback of natural vegetable fibers as reinforcement in thermoplastic matrices is their higher water absorption or desorption behavior and the resulting incompatibility between the hydrophilic sisal and hydrophobic matrix during mixing and composite forming [2–5,7,10,14–16]. As a result, strength and other mechanical properties of the subsequent composites are adversely affected when they are exposed to light, heat, and water [7,10]. Farsi [7] and Gürdağ and Sarmad [17] revealed that cellulose and hemicellulose are the primary components responsible for high moisture absorption [7,8,10,17,18]. Cellulose is a linear polysaccharide with long chains containing primary hydroxyl in a methyl group (–CH2OH) and secondary hydroxyl groups (–OH) both of which are hydrophilic and are capable of forming hydrogen bonding with other cellulose units and water molecules [17]. Besides, due to the presence of organic and inorganic substances on the fiber surface that prevents molecular contact between the fiber and matrix, the wetting ability between them is shallow [8,18–20]. Besides, the processing temperature of cellulosic fibers when they are processed with thermoplastics is limited due to possible degradation at higher processing temperatures [7,11,21,22]. Furthermore, reinforcing with thermoplastics involves processes such as mixing, extrusion, and injection molding that requires a base temperature equal to the melting temperature of the concerned thermoplastics. Therefore, it is difficult to form a composite of natural fibers with thermoplastic polymers that have a melting point above 200°C. Kumar et al. [20] reviewed sisal behavior as a reinforcement in composites and stated that sisal fiber starts degrading above 200°C, below which there are no significant changes in the chemical structure of the fibrils [11,20].
It is, therefore, necessary to alleviate these problems for sustainable utilization of natural fibers as a reinforcement in polymers. Fiber surface modification techniques constitute the most studied and applied remedies for enhancement of processing and performance properties [2,3,7–11,14–16,19–23]. There are several chemical and physical modification techniques to enhance physical, mechanical, and process performance properties, and have been reported by researchers [3,8,15,16,18,19]. Selective removal of noncellulosic compounds constitutes the main objective of all fiber chemical treatment methods [8,11,18,21]. Among modifications is alkaline treatment. Alkali treatment is the treatment of natural cellulosic fibers with hydroxides such as NaOH or KOH to improve surface roughness, strength, and wettability. It is capable of making the hydroxyl groups of cellulose more reactive by removing impurities and making hydroxyl groups more accessible [17]. Equation (1) shows the reaction mechanism during the alkaline treatment [3,23]
Bisanda [19] reported that alkaline treatment improves the fiber surface adhesive characteristics by removing the natural and artificial impurities from the surface of natural fibers. It is reported by Reddy et al. [24] and Ramesh et al. [25] that alkali treatment with NaOH involves the decomposition of natural fibers into several individual fibers called fibrils because of the removal of the cementing compounds by delignification thereby increasing the active surface area of contact with polymeric matrices [7,24,25]. They also, therefore, added that alkaline processing directly influences the cellulosic fibril, the degree of polymerization and the extraction of lignin and hemicellulosic compounds. Mokaloba and Batane [8] explained that as a result of the removal of impurities, the fiber surface becomes more uniform and thus stress transfer capacity between the ultimate cells improves. In addition to this, it reduces the fiber diameter and thereby increases aspect ratio, which results in better fiber interfacial adhesion. The surface tension, hence wetting ability, of alkaline treated fibers will be higher [8,19]. Kumar et al. [20] reported the effects of different alkali concentration on sisal surface roughness and observed that 10% concentration of NaOH results in a higher fiber roughness [20,21]. Acetylation is another fundamental surface modification technique. It involves surface treatment of natural fibers with acetic anhydride, which assumes the substitution of the hydroxyl group of cellulose with acetyl groups and makes the fibers more hydrophobic [3,8,18,21]. Bledzki et al. [16] stated that during acetylation, full esterification of all the hydroxyl groups takes place, which results in the substitution of hydroxyl groups of cellulose by acetyl ones, removal of noncrystalline constituents and alteration of surface topology all of which contribute to an improved stress transfer at the fiber–matrix interface. The plasticization effect of cellulosic fibers due to acetylation was reported by Li et al. [3]. Acetylation of sisal is also reported to be beneficial in improving the fiber–matrix adhesion [3,8,21]. The combined alkaline treatment with acetylation for better results was also reported [3]. Equation (2) shows the reaction of acetic anhydride with the hydroxyl group of the fiber [3,8]
The effect of acetylation on water absorption characteristics of sisal fiber has been studied by researchers [3,16,18,21] and review papers [2,3,11,20]. Mohebby and Hadjihassani [18] also concluded that acetylation increases the moisture repellency of natural fibers by replacing the hydrophilic hydroxyl groups by hydrophobic acetyl ones. The review by Kumar et al. [20] explained that acetylation is beneficial in reducing moisture absorption of natural fibers with a reduction of about 50% for acetylated jute fibers and of up to 65% of acetylated pine fibers. As stated by Célino et al. [12], natural fibers follow Fickian diffusion in the case when they are exposed to vapor during relative humidity aging. Martins and Joekes [21] also investigated the effect of alkaline and acetylation treatments on the thermal stability of sisal fiber. Accordingly, thermograms show an improvement in the thermal stability of the modified fibers in relation to the untreated fibers. An investigation by Martin et al. [22] showed that thermal degradation of cellulose occurs at higher temperatures compared to hemicellulose. Oladele et al. [15] investigated the effect of alkaline treatment and other treatments on the tensile properties of sisal fiber and concluded that alkaline treatment increases the tensile strength while Young’s modulus and extension at break slightly reduced. Martins and Joekes [21] also studied the effect of alkaline treatment and acetylation of sisal on its mechanical properties and found a slight reduction in tensile properties. Several researches have been published on sisal fiber utilization as a reinforcing fiber for thermosets and thermoplastics [2,3,7–11,14–23]. Most of these researches focus on improving some of the limitations that affect its full utilization as a reinforcement. This research work is a preliminary study for utilizing sisal fiber as a reinforcement with PET polymer. As the mixing and injection molding temperatures are governed by the melting temperature of PET during composite processing, and the moisture absorption, interfacial and mechanical properties of the resulting composite are highly dependent on the fiber–matrix compatibility, studying the mechanical, thermal, and water absorption properties of the fiber and finding solutions on improving these properties is an essential step to come up with better processing behaviors and conditions. This work addresses the different approaches of enhancing hydrophobicity, thermal and mechanical properties of sisal fiber for potential application as a reinforcement in high melting point thermoplastic polymer, PET. The study aims at minimizing the hygroscopic nature of sisal fiber for better compatibility with the hydrophobic thermoplastic matrix, and minimizing the thermal effect during composite forming. The results of the raw and treated samples were compared and reported. Incorporating these modified fibers in the PET matrix and investigating the impact on the fiber–matrix adhesion, water absorption, thermal, and tensile properties will be described in part 2 of this study.
Materials and methods
Materials
Commercial grade sisal fiber was purchased as a stretch band from Seilerei SAMMT, Elsterberg, Germany. Acetic acid (glacial), sodium hydroxide, acetic anhydride, and sulfuric acid are commercially available and were of analytical grade and used without any alteration.
Employed fiber modification methods
Alkali treatment
Fifteen grams of raw sisal, abbreviated as RS hereafter, was chopped into a 3–5 cm length and treated with 10% (0.2 M or 0.038 moles) NaOH (≥98% conc.) at 50°C for 2 h at a material liquor ratio of 1:5. It was then washed with distilled water diluted with droplets of acetic acid to neutralize the pH and finally dried at 80°C overnight [3,9,19,20]. Hereafter, MS represents the alkali-treated fiber.
Acetylation
Fifteen grams of sisal fiber was first immersed in glacial (acetic) acid (99.7% conc.) for 1 h. It was then transferred to a solution containing 75 g of acetic anhydride and 0.1 wt% of sulfuric acid (96% conc.) used as a catalyst for 5 min [3,16,18,20]. Final washing with distilled water and drying at 80°C were performed. Hereafter, the sample is abbreviated as AS.
Alkaline treatment followed by acetylation
The alkaline-treated sample was acetylated as per the above steps [3]. Hereafter, the sample is abbreviated as MAS.
Fiber characterization methods
Fiber diameter
The diameter of fibers used for the determination of the diffusion coefficient and tensile strength of the treated and untreated sisal was determined with KEYENCE Digital Microscope VHX-500F equipped with a real-time observation, recording, and measurement of diameter on the monitor screen. A bundle of 30 fibers were mounted with a mounting instrument facing the cross-section outwards, placed into a cup and filled with a mixture of thickener and epoxy, as shown in Figure 1.

Determination of fiber diameter under the digital microscope (A) after polishing, (B) under the microscope of (a) RS, (b) MS, (c) AS, and (d) MAS samples (C) on the screen measurement.
The epoxy-thickener filled cups were allowed to solidify in the laboratory at room temperature for two days to make them suitable for polishing, polished with a Struers Tagramin-25 polishing machine and finally observed under the digital microscope for measurement. Twenty measurements were taken from each sample.
Scanning electron microscopy
The morphology of treated and untreated fibers was investigated using a scanning electron microscope (SEM), CAM SCAN 44, Electron Optics. Sisal fibers were fixed on a metal surface with an adhesive and sputter-coated with gold for 20 min before the investigation and observed with a magnification of 250×.
Fourier-transform infrared spectroscopy
Fourier-transform infrared (FTIR) was conducted with Bruker-Alpha-FTIR and sample preparation follows the attenuated total reflectance technique. The test was performed with a spectral resolution of 2 cm−1 in the mid-infrared region of the 400–4000 cm−1 spectral range.
Moisture absorption kinetics
A 1.5 grams of sisal from each sample was taken and dried to a constant mass in an oven at 80°C for 24 h. The dried fiber was then allowed to absorb moisture in a conditioning cabinet with a controlled relative humidity of 65% and temperature of 20°C for 240 min. In the meantime, the mass was measured in grams, with an electronic balance having an accuracy of ±0.01 g, and recorded at consecutive and increasing time intervals. As indicated in ASTM D 5229 [26], the amount of moisture in a material is taken as the ratio of the mass of the moisture in the fiber to the weight of dried fiber expressed as a percentage (Mt%) at each time interval t and was determined as per equation (3)
Thermogravimetric analysis
The thermal property and decomposition pattern of the fiber before and after treatment was studied with TGA, with model TGA-5000, supplied by TA Instruments.
A 6.5 mg sisal was taken for analysis from the raw and treated ones. The samples were heated up; steadily at a rate of 20°C/min from 30°C to 800°C in nitrogen as a purge gas.
Tensile strength
Sample preparation was performed according to ASTM C 1557, which is a standard test method for tensile strength and Young’s modulus of fibers. A thick paper of 90 g/m2 is used to hold the fiber firmly. The sample preparation step is performed in step a to c, as shown in Figure 2. The tests were carried out with a universal tensile testing machine of Zwick Roell, TestXpert®II, with test speed of 2 mm/min, load cell capacity of 200 N, and a gage length of 25 mm.

Sample preparation for tensile testing of natural vegetable fibers [27].
Twenty fibers from each sample were tested. The standard establishes glueing the fibers in frames and recommends that the total fixed fiber length should be 1.5 times greater than the median free gap of the frame [28]. Since the results of the tensile test were in maximum force, elastic modulus, and elongation at break, the tensile strength and modulus of the untreated and treated samples were determined based on equations (5a) and (5b) as per ASTM C1557-20 [28].
where σ is the tensile strength or stress; Fmax is the maximum tensile force recorded; A is the cross-sectional area of the fiber; d is the fiber diameter; E is the modulus; ϵ is the strain; ΔL is the elongation at break, mm; L is the length of the test specimen.
Results and discussion
FTIR analysis
Figure 3 presents the spectra of treated and untreated sisal fiber, revealing the differences in occurring peaks. As depicted in Figure 3, the broader band in the range of 3350–3200cm−1 appears in all the treated and untreated sisal in which its intensity varies from treatment to treatment. This band represents the O–H stretching of hydrogen-bonded hydroxyl groups of cellulose [29]. The depth of the band is higher for the RS and more intensified in the case of MS due to O–H stretching vibration indicating that some alkali-sensitive materials are wiped off during alkaline treatment, resulting in the more accessibility of hydroxyl groups. The intensity of this peak reduced after acetylation and thus was weaker at the AS and MAS samples. Therefore, as a result of the acetylation process, the substitution of the hydroxyl groups of cellulose by the acetyl functional groups takes place [18,23,29]. The peak in the range of 1740–1720 cm−1, responsible for C = O stretch [29] in the hemicellulose of the RS, appears weaker after alkaline treatment indicating the incomplete removal of hemicellulose. Besides, the peak that appears at around 1596 cm−1 in the RS shows the C = C stretch [29] that is present in aromatic compounds, in this case in lignin and pectin, disappears after alkaline treatment indicating the removal of lignin and pectin.

FTIR spectra of (a) RS, (b) MS, (c) AS, and (d) MAS samples.
The spectral bands ranging from 1765 to 1735cm−1 are resulted due to the C = O stretching of the ester group. These bands showed an augmentation at the AS and MAS samples, which resulted from the addition of the acetyl groups attached to the acetylated sisal fiber [29]. Likewise, the band 1300–1000 cm−1 is also a characteristic of the C–O stretching of the esters of acetyl components (appearing around 1234 cm−1). This band is intensified after acetylation, as shown in Figure 3 for the AS and MAS [14,23]. The intensity of the peaks in the wave number ranging 2990–2850 cm−1 that indicate the methyl groups in cellulose decreases for the AS and MAS samples, which is an indicator for cellulose degradation by the action of sulfuric acid during acetylation [29].
SEM surface analysis
Figure 4 shows the characteristic SEM of raw and treated sisal fibers. On the adjusted image (B), the whiter surfaces are characterized by particles and surface attachments while the dark parts show pores and cleaned surfaces. On the other hand, Figure 4(C) represents the particle distribution profile in which case the darker spotted patterns indicate particles. The fiber surface in Figure 4(A) shows the original SEM image of the samples. The RS (Figure 4(Aa)) is marked by several white spots and scales, which indicates the attachment of waxy cuticles over the fiber cells [14].

(A) Original SEM images, (B) adjusted (by thresholding) images and (C) particle distribution outline of the (a) RS, (b) MS, (c) AS, and (d) MAS samples.
The flattened shape of the RS becomes convoluted and more circular after alkali treatment, as shown in Figure 4(Ab) and (Ad) as a result of swelling of the cell wall [8,19]. An eroded and cleaned surface marks the alkali treated sample (Figure 4(Ab)) along its length, which is an indication for the removal of the impurities and waxy cuticle layers on the fiber surface and defibrillation of the raw fiber due to the extraction of hemicellulose and the cementing components, such as lignin. As shown on the adjusted images in Figure 4(B) and (C), the surface of AS (Bc and Cc) and MAS (Bd and Cd) fibers become rougher than the RS (Ba and Ca) and MS (Bb and Cb) ones, which can be an evidence for the formation of cellulose acetate in the fibrils [14,16]. The difference in the surface particle of the samples was analyzed with ImageJ. Table 1 shows the result of counted particles, including holes or voids, on the given area of each sample surface in which their outline is represented in Figure 4(C). Nearly the same area for each sample was taken for the analysis.
Particle analysis of treated and untreated sisal samples with ImageJ.
Experimental results of maximum moisture absorption.

The observed difference in the number of surface particles is a proof for the consolidation (in case of a higher count of particles) and elimination (in case of a lower count of particles) of compounds. As can be observed from Table 1, the surface of the MAS sample has the highest number of particles compared to the RS, MS and AS samples. This is attributed to the attachment of alkoxides and void formation during alkali treatment and the addition of acetyl groups on the surface during acetylation. On the other hand, the surface of the MS sample contains a lower count of particles per a given area relative to the RS. The result is an evidence for the removal of waxy cuticles, hemicellulose, lignin, and other surface impurities due to alkaline treatment. The reason for the lower particle count of the AS compared to the MAS is during acetylation of the mercerized fiber, the degradation effect of cellulose due to sulfuric acid was minimized as NaOH neutralizes the acid, while more degradation is possible during acetylating of the raw fiber. The particle distribution outline presented in Figure 4(C) also indicates higher particle attachment on the AS and MAS samples than the RS and MS samples.
Fiber diameter
Fiber diameter and aspect ratio (length-to-diameter ratio) are essential parameters that affect the tensile strength and other mechanical properties of the fiber and its products. The variation in sisal fiber diameter due to the surface treatment are plotted in Figure 5. From the vertical plot chart, it can be observed that a relatively significant difference within measurements was observed for the MS and RS samples (standard deviation of 28.90 and 21.98 µm), while the AS and MAS samples recorded lower average diameter. The distribution of individual test specimens of the AS and MAS shows that most of them recorded more precise values relative to their average diameter. The average diameter of the treated samples is reduced compared to RS. The result is expected in the case of MS and MAS samples as impurities such as lignin, hemicellulose, and waxes are removed during alkaline treatment [8]. Besides, a single sisal fiber is composed of a multiple of individual more delicate tissues bound together by lignin [7,17,19], which depends on the efficiency of the fiber extraction process.

Column scatter plot for fiber diameter of treated and untreated sisal.
The reduction in the diameter of the AS and MAS samples demonstrated the degradation of cellulose because of the presence of sulfuric acid in the acetylating solution, which is minimized during acetylation of the alkali-treated one. Since alkaline treatment swells the cell wall of cellulose [3,7,8], which depends on the NaOH concentration and treatment conditions, an increase in diameter is expected [8,19,21]. As it is shown from the column scatter plot, some of the test coupons of the MS sample recorded a higher diameter.
Moisture absorption
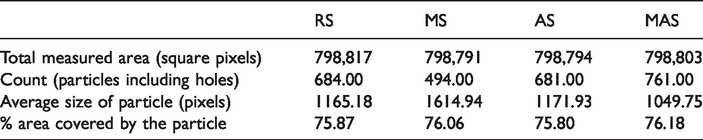
Figure 6 represents the absorption kinetics of the four samples while their corresponding maximum absorption values are presented in Table 2. The curves, especially on the acetylated and alkaline-treated/acetylated samples, have a sigmoidal shape. This particular shape is as a result of minimum or slow absorption behavior and the consequent delay time in the establishment of water concentration equilibrium at the fiber surface [9,14].
Moisture absorption of the untreated and treated samples.
As depicted from Table 2, the acetylated sisal shows minimum moisture absorption as a result of the acetylation process. As compared to the acetylated sisal sample, alkaline-treated/acetylated sisal sample recorded higher moisture absorption. This is attributed to the low efficiency of the acetylation process in which sulfuric acid may get neutralized by the alkaline effect of the alkaline-treated fiber. Relative to the RS, MS shows an increase in moisture absorption by 11.0%, which is attributed to the removal of hydrophobic groups like lignin and pectin [23]. The AS had a decreased moisture absorption by about 42%, and MAS by 28%, in comparison to RS. This confirms the enhancement of hydrophobicity of sisal fiber due to the replacement of hydroxyl groups of the cellulose by hydrophobic acetyl components as a result of acetylation.
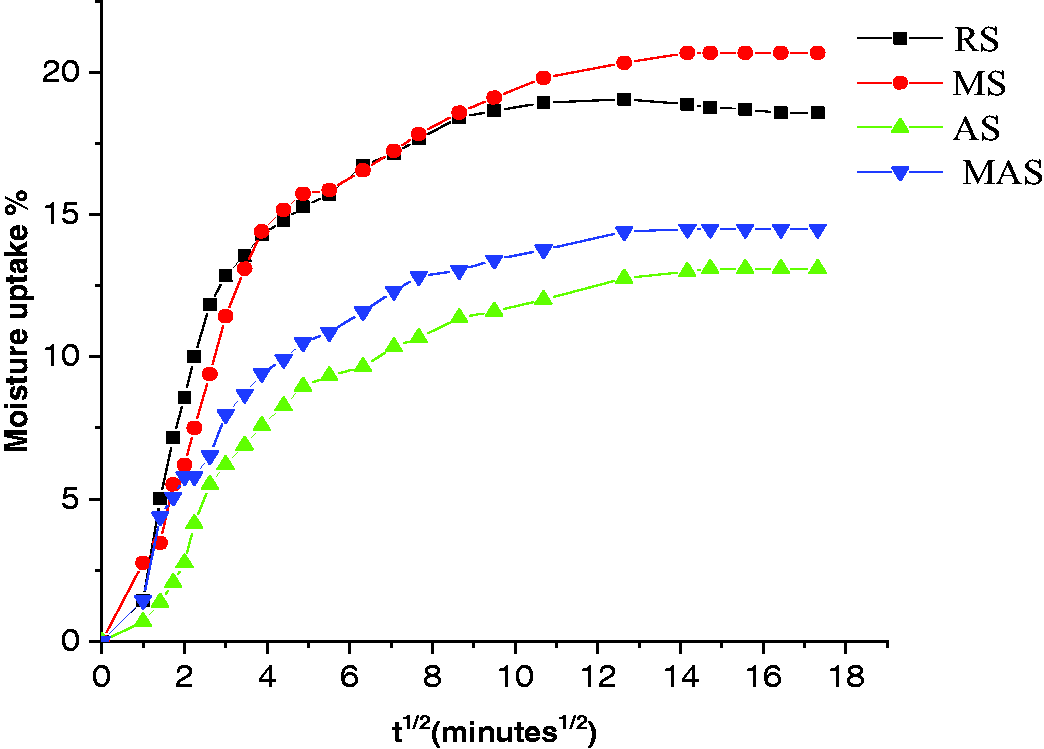
The high absorption of RS and MS samples can be illustrated by the diffusion coefficient calculated from equation (4).
The calculated results are shown in Figure 7. It is depicted that the MS and RS samples yielded a higher diffusion coefficient than the AS and MAS. The higher diffusivity of the MS and RS samples are the characteristics of a higher rate of diffusion of water or moisture in the through-thickness of the fiber. However, the AS and MAS fibers yielded a lower diffusion coefficient due to an increased hydrophobicity of sisal fibers.
Diffusion coefficient of untreated and treated samples.
Thermal analysis
Figure 8 shows the DTG and TGA profiles of RS, MS, AS, and MAS samples being overlaid. The overlaid TGA/DTG curve is divided into water loss, onset, maximum peak, carbonization, and residual temperature regions. DTG curves show a small initial peak between 30°C and 150°C, which corresponds to water loss in all the samples with different quantities [21–23]. The onset temperature, the temperature at which the degradation of the primary polymer, in this case, cellulose, is starting, is higher for MS and MAS samples while the AS shows the least value. Similarly, the highest maximum peak temperature is recorded for MAS and the least is the AS one. The reason behind the low thermal stability of AS sample is probably due to the degradation of some cellulose during the acetylation process as the acetylating solution contained sulfuric acid as a catalyst. It can be further confirmed by comparing it with the result of MAS.

Overlaid TGA/DTG curve for untreated and treated samples.
The onset and maximum decomposition temperature of the AS are 169.5°C and 340°C, while for MAS are 258°C and 353.9°C, as highlighted in Table 3. The reason is due to the neutralization effect of sodium hydroxide on sulfuric acid during the acetylation of the MS fiber. The damage of cellulose is more considerable during acetylation of the raw fiber as cellulose is more susceptible to strong acids like sulfuric acid.
Primary decomposition regions of the samples.

In comparison to the RS, the modified fibers have resulted in higher oxidative carbonization, end set temperature values and higher residue percentage, as indicated in Table 3. This is due to the addition of covalent bonded C = O functional groups as a result of acetylation and the metallic alkoxide, –O−Na+, due to alkaline treatment [21,22]. Besides, smaller peaks labeled (a) and (b) on the DTG curve appear only on the RS and AS samples respectively, while they are negligibly little (or disappear) at the MS and MAS samples. These temperature regions are the regions for hemicellulose degradation. These peaks do not exist for MS and MAS samples as hemicellulose was removed during alkaline treatment. Table 4 illustrates the thermal decomposition pattern of each sample during thermal analysis with TGA. The decomposition up to 150 °C involves water loss. The decomposition pattern from 150°C to 250°C for RS, MS, AS, and MAS are 1.92%, 1.66%, 9.36%, and 3% and from 150°C to 300°C (processing temperature of most thermoplastics), it is 13.39%, 9.49%, 26.25%, and 11.5%, respectively.
Decomposition pattern (%) of raw and modified sisal fibers at different temperature regions.

Thus, according to the decomposition study up to 300°C, the MS and MAS samples show a minimum weight loss (14.7% and 16.8%) than the RS sample (22%), while the AS recorded the highest weight loss (30.7%). In other words, compared to the RS, the thermal stability of the MS and MAS samples was enhanced by 49.6% and 30.9% respectively, while the AS samples resulted in a drop by 39.5%. Therefore, the alkaline treated samples, MS and MAS, have improved thermal stability. This can be attributed to ionization of the hydroxyl groups by alkoxide (fiber–O-Na+) [7,21,22].
Tensile strength
The stress–strain curves of the four samples are displayed in Figure 9. The curves show an initial linear region characterized by initial modulus. Stress variability between individual test specimens within a sample as well as between samples was observed. The reason is the nonuniformity of natural fibers and the presence of weak points and microstructural differences throughout the fiber length [30]. A relative comparison shows that the MS and MAS showed a linear variation between the stress–strain curve. This is due to the surface cleanness and the resulting uniformity of microstructure of cell wall [8]. On the contrary, the RS show high strain with given stress as compared to the MS, AS, and MAS. This was a result of the presence of lignified fiber bundles which start to delaminate as the load is applied; as the load increases, these bundles begin to separate before breakage which resulted in higher strain [8,9,30]. Because of the weak cell wall created due to cellulose degradation during the acetylation process, the stress resistance of the AS samples was smaller than for the RS and MS samples. The deterioration was minimized in the MAS samples [8,23]. The average values of the tensile test are presented in Table 5. As illustrated from the table, a variation in tensile strength and modulus of the samples was observed. Extreme outliers were recorded during measurement, especially for the tensile strength and modulus.

Stress–strain curve of untreated and treated sisal samples.
Average values for tensile properties of untreated and treated samples.

This variability is mainly due to the difference in tensile properties of natural fibers throughout their length [30]. Less variability can be achieved by reducing the gage length during the tensile test [23,27,30]. Acetylation was shown to reduce the tensile strength significantly while the others show a slight reduction [15,18,20]. But, the modulus of all the treated samples showed improvement as a result of the removal of amorphous compounds during mercerization and the plasticization effect of acetylation process and the consequent stiffness [8,18,21].
As compared to the raw sisal, the modified fibers, especially the AS and MAS samples, breaks at a lower strain rate due to the plasticization effect of acetylation process, which induces rigidity to the fibers. The AS sample recorded the least average tensile strength and strain values. The reason being the damage of cellulose during acetylation due to the effect of sulfuric acid in the acetylation solution and the resulting decrease in secondary covalent bonds. The reduction in tensile strength is due to the removal of lignin, hemicellulose, and other cementing compounds, that impart rigidity and functions as a binder between the cells generating a composite structure with outstanding strength and elasticity [7], during alkaline treatment and to the damage of cellulose by sulfuric acid during acetylation as cellulose is highly sensitive to strong acids [7,17].
Conclusion
The current investigation focuses on finding out the changes in the moisture, thermal, and mechanical properties of sisal fiber as a result of fiber surface treatments. Therefore, based on the investigated range, the following conclusions can be drawn: Acetylation and the combined alkali treatment/acetylation treatments resulted in a very considerable improvement of hydrophobicity of sisal fiber. Relative to the RS, 42% and 28% reduction in moisture absorption was achieved for the AS and MAS samples respectively. On the other hand, alkali treatment increased the moisture absorption property owing to the removal of hydrophobic components such as lignin and waxes. An increase of 11% was observed for the MS one. Thermal stability of sisal fiber was improved after alkali treatment, especially 250–300°C, which is the processing temperature region of PET. Accordingly, up to 300°C, the thermal decomposition minimized by 49.6% and 30.9% for the MS and MAS specimens than the RS equivalent. In comparison to the RS, the MS and MAS samples showed a slight decrease in tensile strength, while the modulus of the fiber was improved after treatment for all the samples. The average sisal fiber diameter was decreased after treatment owing to the removal of impurities and cementing compounds during alkali treatment and degradation of cellulose during acetylation. A significant drop of tensile strength and thermal stability was observed on the AS sample. This is because of cellulose degradation during acetylation (noted on the FTIR spectra of the AS sample) due to the presence of sulfuric acid in the acetylating solution as a catalyst, in which cellulosic fibers are highly sensitive to strong acids. Therefore, the use of strong acids as a catalyst is not recommended during acetylation of natural fibers and the process shall be optimized in terms of catalyst selection. Generally, for improved hydrophobicity, thermal stability and modulus, with a slight change in tensile strength, acetylation shall be performed after alkali treatment of the raw sisal.
Footnotes
Acknowledgements
This work is a part of the Ethio-German Homegrown PhD program. Thus, the authors are thankful to the German Academic Exchange Service, DAAD and Ethiopian Ministry of Education (MoE).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: There is no specific funding for this work except that it is a scholarship-based PhD research.