Abstract
In the present work, nanocomposite cellulose fabrics with in situ generated silver nanoparticles were prepared by bioreduction method employing aqueous dispersion of low-cost natural turmeric powder as a reducing agent and different concentrated aqueous AgNO3 as source solutions. The prepared nanocomposite cellulose fabrics were characterized by scanning electron microscopy, Fourier transform infrared spectroscopy, X-ray diffraction, thermogravimetric analysis, and antibacterial tests. The nanocomposite cellulose fabrics had roughly spherical silver nanoparticles in the size range of 41–130 nm with an overall average of 78 nm. The X-ray analysis indicated the generation of both silver nanoparticles and Ag2O nanoparticles in the nanocomposite cellulose fabrics. The nanocomposite cellulose fabrics retained the generated AgNPs even after repeated detergent washings. The prepared nanocomposite cellulose fabrics exhibited excellent antibacterial activity against both the Gram-negative and Gram-positive bacteria and hence can be considered as antibacterial hospital-bed materials, apparels, etc.
Keywords
Introduction
Due to their unique properties such as large surface area to volume ratio, large surface energies, Plasmon excitation, quantum confinement, etc. [1], metal nanoparticles (MNPs) find immense applications in the fields of catalysis, medicine, sensors, optoelectronics, etc. The properties of the MNPs often depend on their shape, size, and distribution [2]. There are various methods of synthesizing MNPs such as plasma spraying, laser pyrolysis, chemical vapor deposition, mechanical milling, sputtering, laser ablation, electro explosion, etc. [3]. However, these methods are complex and expensive. On the other hand, biosynthesis is an eco-friendly method in which bacteria, yeast, fungi, plant extracts in which the inherent biochemicals present act as reducing and stabilizing agents [4]. Of the MNPs, silver nanoparticles (AgNPs) exhibit outstanding antibacterial activity and hence, find immense applications in medical field [5–7]. Some researchers synthesized AgNPs using leaf extracts such as Corandrum sativum [8], Musa balbisiana [9], banana peel [10], Ocimum sanctum [11], etc. Instead of using the generated AgNPs directly, for many applications, it is advisable to introduce them in suitable polymer matrices to prepare the nanocomposites. However, dispersion of the already prepared AgNPs in polymer matrices often leads to agglomeration. In order to overcome this problem, some researchers in situ generated AgNPs in polymer matrices. For instance, Sadanand et al. [12] made cellulose/AgNP films by in situ generation of AgNPs using O. sanctum leaf extract as a reducing agent. Sivaranjana et al. [13] used Cassia alata leaf extract as a reducing agent and prepared cellulose/AgNPs films with in situ generated AgNPs. Similarly, Muthulakshmi et al. [14] prepared cellulose/AgNPs films using Terminalia catappa leaf extract as a reducing agent. Though these nanocomposite films exhibited excellent antibacterial activity, for medical applications such as wound dressing, bed materials, apparels, etc., antibacterial fabrics are necessary. In this direction, recently, our group prepared cellulose/AgNPs fabrics with in situ generated AgNPs by hydrothermal method [15] and also using red sanders powder extract [16] and C. alata leaf extract [17] as reducing agents. However, these nanocomposite fabrics required higher concentrated source solutions to achieve significant antibacterial activity. Hence, it is necessary to use an inherently antibacterial natural reducing agent to achieve better antibacterial activity even for low concentrations of the source solutions. In this direction, in the present work, the authors prepared nanocomposite cellulose fabrics (NCFs) with in situ generated AgNPs using the aqueous dispersion of the traditional turmeric powder as a reducing agent and various concentrated aq. AgNO3 as source solutions. Turmeric powder is widely used in many Asian countries as a home remedy, food additive, and also as a natural dye [18].
The turmeric powder is composed of 60–70% of carbohydrates, 6–13% water, 6–8% protein, 5–10% fat, 3–7% dietary minerals, 2–7% dietary fiber, 3–7% essential oils, and 1–6% curcumin [19]. The curcumin having hydroxyl groups can reduce the metal salts into their respective MNPs. Taking into the medicinal properties and the presence of reducing agents [18,19], the authors prepared NCFs with in situ generated AgNPs using aq. turmeric powder dispersion as the reducing agent. The prepared NCFs were characterized by scanning electron microscopy (SEM), Fourier transform infrared (FTIR) spectroscopy, X-ray diffraction (XRD), thermogravimetric analysis (TGA), and antibacterial tests. Besides, the retention of the AgNPs in NCFs even after repeated detergent washings was established by SEM analysis.
Materials and methods
Materials
In the present work, white cellulose (cotton) fabrics were procured from the local market. Initially, these fabrics were washed thoroughly with mild detergent to remove the sizing material if any and then rinsed thoroughly with distilled water before they were dried. The turmeric roots obtained from the local market were dried and made into fine powder and sieved. The yellow colored turmeric powder which passed through 25 µm sieve was used in this work. Silver nitrate (Aldrich fine chemicals) was used as received. Deionized water was used in all the preparation methods.
Preparation of the matrix
Aqueous dispersion of turmeric powder (10 wt.%) was prepared and kept in standard flasks. The cleaned and dried cellulose fabrics were cut into the size of 200 × 150 mm, which were subsequently dipped in the standard flask having aq. turmeric powder dispersion under constant stirring on magnetic stirrer maintained at a speed of 300 r/min at ambient conditions for 24 h. Subsequently, these fabrics were removed from the standard flasks, which were subsequently thoroughly rinsed in water and finally air-dried. These lemon yellow fabrics treated with aqueous dispersion of turmeric powder constituted the matrix materials.
Preparation of NCFs with in situ generated AgNPs
For the preparation of NCFs, in the first step, aqueous AgNO3 solutions of concentrations 1 mM, 2 mM, 3 mM, 4 mM, and 5 mM were prepared in separate standard flasks using deionized water. In each standard flask, two pieces of the matrix fabrics were dipped and completely wetted. These standard flasks were then kept on magnetic stirrers with constant stirring (300 r/min) at room temperature for 24 h. During this period, the color of the fabrics gradually changed from lemon yellow to brown indicating the in situ generation of AgNPs in them. The wet NCFs were thoroughly washed with deionized water and dried in shade.
Characterization
Microscopic observation
The SEM images of the NCFs were recorded using Zeiss EVO 18 SEM at an operating voltage of 10 kV. The specimens were sputter coated with gold prior to SEM observation. Using the built in SmartTiff software of the microscope, the size of the in situ generated AgNPs was measured. The energy-dispersive X-ray (EDX) spectra of the NCFs were also recorded using the accessory of the same instrument.
FTIR spectra
The smart iTR ATR Nicolet is 10 FTIR spectrophotometer was employed to record the FTIR spectra of the cellulose fabric, matrix and the NCFs in the wavenumber region of 4000 to 500 cm−1 for 32 scans at a resolution of 4 cm−1.
X-ray diffraction
The XRD patterns of cellulose fabric, matrix and the NCFs were recorded using Bruker Eco D8 XRD diffractometer operated at a voltage of 40 kV and a current of 25 mA in the 2θ = 10–80° range.
Thermogravimetric analysis
The primary and derivative thermograms of the matrix and NCFs were recorded using Perkin Elmer Model STA 6000 thermobalance in the temperature range of 30–700 °C at a heating rate of 10°C min−1 in nitrogen atmosphere.
Antibacterial test
The antibacterial test of the cellulose fabric, matrix, and the NCFs against both the Gram-negative (Escherichia coli and Pseudomonas) and Gram positive (Staphylococcus aureus and Bacillus subtilis) bacteria was carried by the disc method as described elsewhere [20]. Briefly, in this method in the first step, the nutrient agar gel medium was prepared by mixing appropriate amounts of peptone, beef extract, and sodium chloride in water by adjusting the pH at 7. This agar medium was then transferred to the presterilized Petri dishes which were kept in the laminar airflow chamber. In the second step, the respective bacterial cultures were spread separately on the agar media. After inoculation, the circular discs of the matrix and NCFs were placed over the media and incubated for two days at 37°C. The clear zones observed were photographed and the corresponding zone diameters in each case were measured. In each case, three sets were tested and the average diameters of the clear zones are reported.
Tensile test
The tensile test of the cellulose fabric, matrix, and the NCFs was conducted using Instron 3369 universal testing machine. The specimens with dimensions of 100 mm × 10 mm were used in the test. The test was conducted at an extension rate of 5 mm min−1 with a gauge length of 50 mm. For each test five specimens were used and the average values along with the standard deviation are reported.
Results and discussion
In order to visualize the appearance of the matrix and the NCFs, their digital images are presented in Figure 1. From Figure 1, it is can be visualized that the color of the matrix is lemon yellow due to the interaction of the cellulose fabric with curcumin, the coloring component of turmeric powder. The color of the matrix remained unchanged despite several washings with distilled water indicating interactions between it and curcumin. In the case of the NCFs, the color changed to brownish yellow, which deepened with the concentration of the source solutions (1–5 mM). The color change preliminarily indicates the in situ generation of the AgNPs. The color of the NCFs remained unchanged despite washings with distilled water several times.

Digital images of matrix (a) and NCFs with in situ generated AgNPs using 1 mM (b), 2 mM (c), 3 mM (d), 4 mM (e), and 5 mM (f) aqueous AgNO3 source solutions and aqueous dispersion of turmeric powder as a reducing agent.
In order to prove the in situ generation of AgNPs in the NCFs, their SEM images were recorded along with their EDX spectra. Using the SEM images, the particle size of the AgNPs in each case was measured using SmartTiff program. For clarity, the SEM images of the NCFs prepared using 1 mM and 5 mM source solutions are presented in Figure 2(a) and 2(b), respectively. The EDX spectra of the NCFs prepared using 1 mM and 5 mM source solutions are presented in Figure 2(c) and 2(d), respectively. Using the SEM images, the particle sizes were measured and as an example, the histograms of particle size distribution for the NCFs prepared using 1 mM and 5 mM source solutions are presented in Figure 2(e) and 2(f), respectively. The presence of roughly spherical AgNPs on the surface of NCF can be observed in Figure 2(a) and 2(b). The presence of silver element in the NCF was established by the presence of its peak in the EDX spectra (Figure 2(c) and 2(d)). The histograms representing the particle size distribution (Figure 2(e) and (f)) indicate that the size range of in situ generated AgNPs in the NCFs in both varied between 41 nm and 100 nm. The average diameter of the AgNPs generated in NCFs made using 1 mM and 5 mM source solutions was found to be 51 nm and 66 nm, respectively. In the case of NCFs made using 2 mM, 3 mM, and 4 mM source solutions (not shown in Figure 2), the average particle size was found to be 69 nm, 76 nm, and 79 nm, respectively. Thus, the SEM analysis confirmed the in situ generation of AgNPs in the NCFs due to the effective reduction of silver nitrate into AgNPs by the aqueous dispersion of turmeric powder as a reducing agent.

SEM images of the NCFs prepared using 1 mM (a) and 5 mM (b) source solutions. EDX spectra of the NCFs prepared using 1 mM (c) and 5 mM (d) source solutions. Particle size distribution of the generated AgNPs in the NCFs prepared using 1 mM (e) and 5 mM (f) source solutions.
In order to examine the retention of the in situ generated AgNPs after repeated detergent washings, the images of the prepared NCFs after 20 detergent washings were photographed and are presented in Figure 3. From Figure 3, it is evident that the intensity of the color of the NCFs decreased after 20 washings with detergent.

Digital images of the NCFs with in situ generated AgNPs using 1 mM (a), 2 mM (b), 3 mM (c), 4 mm (d), and 5 mM (e) aqueous AgNO3 source solutions and aqueous dispersion of turmeric powder as a reducing agent.
In order to probe whether these NCFs even after 20 washings still have AgNPs or not, their SEM analysis was carried. For clarity, the SEM images of NCFs prepared using 1 mM and 5 mM source solutions are presented in Figure 4(a) and 4(b), respectively. The EDX spectra of the NCFs prepared using 1 mM and 5 mM source solutions are presented in Figure 4(c) and 4(d), respectively. The histograms of the NCFs made using 1 mM and 5 mM source solutions are presented in Figure 4(e) and 4(f), respectively. From Figure 4(a) and 4(b), it is evident that the NCFs retained most of the AgNPs even after 20 detergent washings. Hence, these NCFs can be repeatedly used even after washings. The EDX spectra (Figure 4(c) and 4(d) indicate that the in situ generated nanoparticles in the NCFs belong to Ag element. From Figure 4(e) and 4(f), it can be observed that the size of the AgNPs in the NCFs varied between 51 nm and 130 nm. The average sizes of the AgNPs in the NCFs prepared using 1 mM and 5 mM source solution were found to be 92 nm and 88 nm, respectively. In the case of the NCFs prepared using 2 mM, 3 mM, and 4 mM source solutions (not shown in Figure 4), the average particle sizes after the washings were found to be 86 nm, 80 nm, and 73 nm, respectively. In order to examine the possible chemical interactions between in situ generated AgNPs and the matrix in the NCFs, the FTIR analysis was carried.

SEM images (a) and (b), EDX spectra (c) and (d) and histograms of particle size distribution (e) and (f) of NCFs prepared using 1 mM and 5 mM source solutions after 20 washings.
FTIR analysis
To probe the interactions between the functional groups of cellulose fabric and curcumin present in the turmeric powder, the FTIR spectra of both the cellulose fabric and matrix were recorded which are presented in Figure 5(a). From Figure 5, it is evident that the common bands appearing at 3338 cm−1, 2897 cm−1 were assigned to –OH group and CH stretching vibrations, respectively, the other peak at 1741 cm−1 arose due to C=O group while the peak at 1639 cm−1 was assigned to predominantly mixed C=C and C=O bonds. The peak at 1427 cm−1 was assigned to CH2 scissoring vibration while the other low-intensity peaks at 1365 and 1337 cm−1 were attributed to the CH2 bending vibrations. The peaks appeared at 1200–1260 cm−1 were attributed to wagging of CH2 [21]. The bands observed at 1149 cm−1and 1018 cm−1 were due to C–O–C st of pyranose ring and that at 878 cm−1 was attributed to β-glucosidic linkage [15]. These observations indicate the cellulose structure in both the matrix and the pristine fabric. This is understandable as the turmeric powder has carbohydrates as major components. Further, the intensity of the major peaks of the matrix was higher than for cellulose fabric indicating the addition of hydroxyl groups due to the interaction with turmeric powder. The noise (corresponding to CO2) in all the spectra in the wavenumber range of 2300 to 1800 cm−1 was deleted.

FTIR spectra of cellulose fabric and matrix (a) and matrix and NCFs made using 1–5 mM aqueous AgNO3 source solutions (b).
In order to examine the possible chemical interactions between in situ generated AgNPs and the matrix in the NCFs, the FTIR spectra of the matrix and the prepared NCFs are presented in Figure 5(b). From Figure 5(b), it is evident that almost all peak positions were same in the matrix and composites indicating that no structural change took place. Similar observation was made by Sadanand et al. [15] in the case of NCFs with in situ generated AgNPs by hydrothermal method. As we have used only milli molar (maximum 5 mM) source solutions, no structural change in the NCFs was initiated by the in situ generated AgNPs. However, the intensity of the major peaks of the NCFs was found to be lower than that of the matrix indicating the role of OH groups in the reduction and stabilizing reactions.
In order to examine the effect of curcumin of turmeric powder on the crystallinity of matrix and the generated AgNPs on the crystallinity of NCFs, the X-ray analysis was carried out. The diffractograms of the cellulose fabric and the matrix are presented in Figure 6(a). From Figure 6(a), it can be observed that the intensity of the diffractogram of matrix is higher than that of the cellulose fabric. The matrix fabrics are lemon yellow in color (Figure 1) indicating the binding of curcumin, the natural dye present in turmeric powder to the white fabric. The diffractograms of matrix and the NCFs are presented in Figure 6(b). From Figure 6(b), one can observe that the intensity of the peaks of NCFs prepared using 1–5 mM aqueous AgNO3 source solutions is higher than that of the matrix. It clearly indicates that the in situ generated AgNPs increased the crystallinity of the NCFs. For more clarity, the diffractograms of the matrix and the NCF using 5 mM aqueous AgNO3 source solution are presented in Figure 6(c). From Figure 6(c), it can be seen that the crystallinity of the NCFs was increased by the in situ generated AgNPs. A close observation of Figure 6(c) indicates the presence of additional low-intensity peaks in the diffractogram of the NCF which was obscured by the high-intensity peaks of cellulose fabric. In order to observe these additional low-intensity peaks, the diffractogram of the NCF prepared using 5 mM source solution was expanded in the 2θ = 35°–80° range and is presented in Figure 6(d). From Figure 6(d), it can be observed that there are many low-intensity peaks in the expanded diffractogram of which, the prominent ones at 2θ= 38.9°, 45.7°, 47.2°, 55.4°, 63.7°, 66.6°, and 77.5° belong to AgNPs and Ag2O nanoparticles (Ag2ONPs) [22,23]. Of these, the peaks observed at 2θ= 38.9°, 45.7°, 63.7°, and 77.5° belonged to the reflections from (111), (200), and (311) planes of AgNPs, respectively [22]. The other prominent peaks observed at 2θ= 47.2°, 55.4°, and 66.6° resulted from the reflections from (211), (220), and (222) planes of Ag2ONPs of the NCFs, respectively [22,23]. Thus, the NCFs had both in situ generated AgNPs and Ag2ONPs.

X-ray diffractograms of cellulose fabric and matrix (a); matrix and NCFs prepared using 1–5 mM aqueous AgNO3 source solutions (b); matrix and NCF prepared using 5 mM aqueous AgNO3 source solution (c) and the expanded diffractogram of NCF made using 5 mM aqueous AgNO3 source solution in the 2θ = 35–80° range (d).
The possible mechanism for the reduction of the AgNO3 to AgNPs in the NCFs is presented in Scheme 1. The main components of Curcuma longa (CL) include Curcumin-I (71.5%), Curcumin-II (19.4%), and Curcumin-III (9.1%). The Curcumin-I mainly contains poly phenols (3–15%) as principal components [24]. The hydroxyl functional groups in phenol of Curcumin-I can play a role in the bioreduction of Ag+ ions into metallic silver. In this bioreduction two Ag+ were reduced by two hydroxyl functional groups of the diphenols. The involvement of two hydroxyl functional groups for simultaneous reduction into two silver metal atoms was reported by earlier researchers [23,25]. The mechanism of generation of Ag2ONPs is presented in Scheme 2.

Diagrammatic representation of mechanism of the generation of AgNPs.
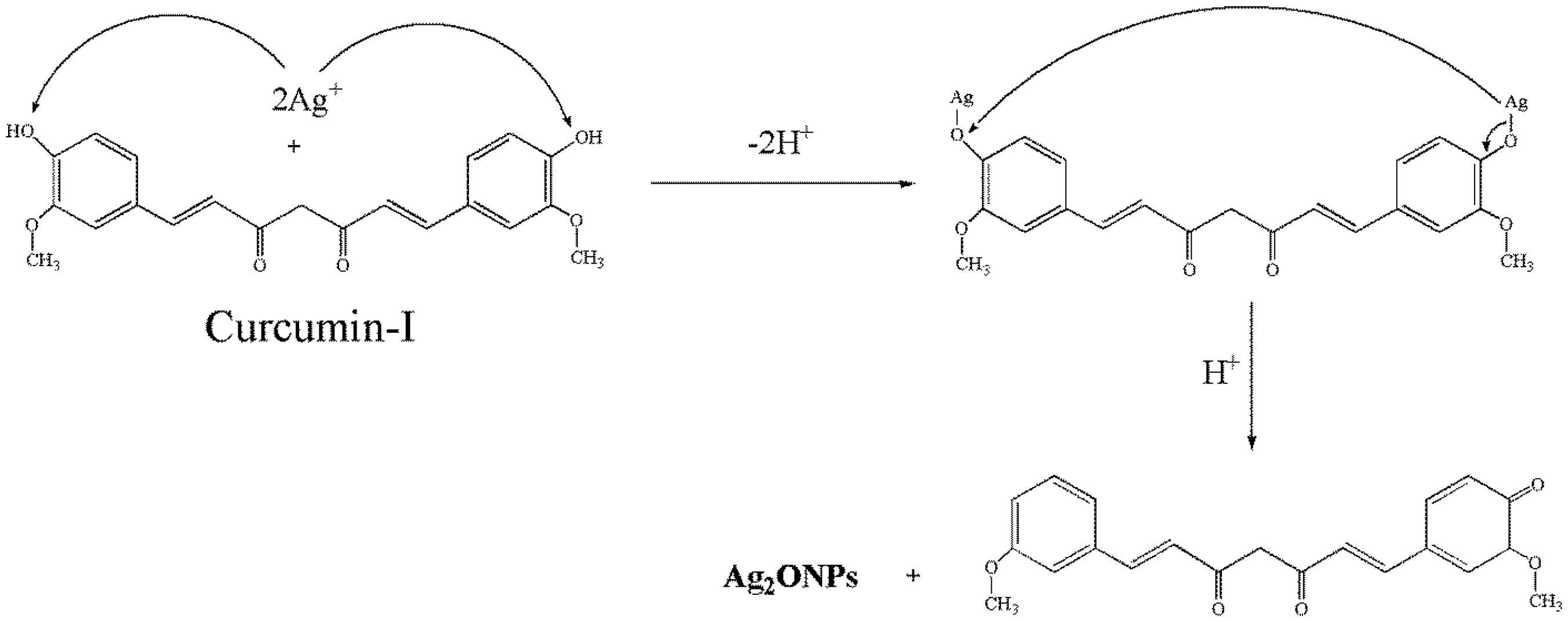
Diagrammatic representation of the mechanism for the generation of Ag2ONPs in the NCFs.
The utilization of hydroxyl functional groups of phenols in curcumin-I in the reduction reaction was also confirmed by the lowering of the intensity of the peaks at 3338 cm−1 in the FTIR spectra of NCFs (Figure 5).
Due to high catalytic activity of silver atoms, some silver atoms might have reacted with oxygen atom present in form of phenome group from enol form of curcumin-I to generate Ag2ONPs (Scheme 2) along with AgNPs. These results indicate that some hydroxyl groups in phenols of curcumin were involved in bio reduction of Ag+ ions into AgNPs and Ag2ONPs.
Thermogravimetric analysis (TGA)
In order to examine the effect of generated AgNPs on the thermal stability of the NCFs, the TGA was carried out. The primary and derivative thermograms of the cellulose fabric and the matrix are presented in Figure 7(a) and 7(b), respectively. The primary and derivative thermograms of the matrix and the NCFs with in situ generated AgNPs using 1–5 mM aqueous AgNO3 source solutions and aqueous dispersion of turmeric powder as the reducing agent are presented in Figure 7(c) and 7(d), respectively. From the derivative thermograms (Figure 7(b) and 7(d)), it can be observed that there exists a minor weight loss in the temperature range of 45–107°C which arose due to the removal of the volatiles and water from the fabrics. From Figure 7(a) and 7(b), it can also be observed that the inflection temperature (the temperature at which the degradation rate is maximum) of the matrix is slightly higher than that of the cellulose fabric. This may be due to the presence of curcumin, a diphenyl compound (in matrix). However, no major change in the inflection temperatures between the matrix and the NCFs was detected. Further, both the matrix and the NCFs were found to be thermally stable up to 300°C and complete thermal degradation occurred only after 400°C. Thus, the NCFs can be used up to a minimum temperature of 300°C.

Primary (a) and derivative (b) thermograms of cellulose fabric and matrix; primary (c) and derivative (d) thermograms of matrix and NCFs.
Antibacterial activity
The antibacterial test of the NCFs prepared was conducted against both Gram-negative and Gram-positive bacteria by standard disc method [20]. The zones of clearance observed were photographed and the corresponding images are presented in Figure 8. From Figure 8, it can be noticed that no antibacterial activity was observed for the cellulose fabric and the matrix. However, all the NCFs prepared using 1–5 mM source solutions formed the zones of clearance against both the Gram-negative (E. coli and Pseudomonas aeruginosa) and Gram-positive (S. aureus and Bacillus sutilis) bacteria. The diameters of the clear zones formed which indicate the effectiveness of the antibacterial activity [15–17] were measured and are presented in Table 1. From Table 1, it is evident that the diameters of clear zones for the NCFs varied between 15 mm and 18 mm. Thus, the NCFs prepared possess significant antibacterial activity and hence can be considered for applications as antibacterial apparels and hospital bed materials.

Digital images of the clear zones formed by the NCFs made using 1 mM (3), 2 mM (4), 3 mM (5), 4 mM (6), and 5 mM (7) source solutions against E. coli (a); Pseudomonas aeruginosa (b); Staphylococcus aureus (c), and Bacillus subtilis (d) bacteria. For comparison, the antibacterial activity test was conducted for the cellulose fabric (1) and matrix (2).
Clear zone diameters of NCFs made using different concentrated aq.AgNO3 source solutions against Gram negative (E. coli and P. aeruginosa) Gram–positive (S. aureus and B. subtilis) bacteria.

The antibacterial activity of the system under study was compared with that of the NCFs prepared by hydrothermal method [15] and the NCFs made using the aqueous extraction of red sanders [16] and C. alata leaves [17]. The diameters of the zones of clearance (indicating the quantitative antibacterial activity) of these along with the present system against the bacteria E. coli, P. aeruginosa, S. aureus, and B. subtilis with concentrations of the source solutions are presented in Figure 9(a), 9(b), 9(c) and 9(d), respectively. From Figure 9, it is evident that the antibacterial activity of NCFs prepared using aqueous dispersion of turmeric powder as a reducing agent was higher than the rest. Further, from Figure 9, it is also evident that the NCFs in the present study exhibited excellent antibacterial activity even when lower concentrated source solutions were used. Hence, this comparative study indicates that aqueous dispersion of turmeric powder is more effective in bioreduction than the rest [15–17].

Antibacterial activity of NCFs prepared by hydrothermal method and by using the aqueous extracts of red sanders powder, Casia alata leaves and dispersion of turmeric powder against E. coli (a), Pseudomonas aeruginosa (b), Staphylococcus aureus (c), and Bacillus subtilis (d) bacteria.
Tensile properties
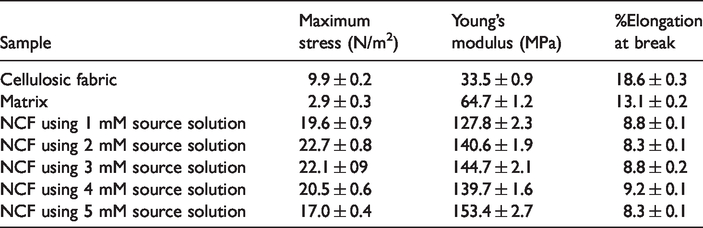
In order to study the effect of the generated AgNPs on the mechanical properties of the NCFs, the tensile test was conducted as per ASTM D638 specifications. The maximum stress, Young’s modulus, and %Elongation at break of the cellulose fabric, matrix and the NCFs prepared using 1–5 mM source solutions are presented in Table 2. From Table 2, it can be observed that maximum stress and Young’s modulus of the matrix were higher than those of the cellulose fabric. This may be due to the interaction between rigid curcumin component of the turmeric powder and the cellulose fabric. However, the %Elongation at break of the matrix was found to be lower than that of the cellulose fabric. From Table 2, it can also be seen that, maximum stress and Young’s modulus of the NCFs were found to be higher than those of both the cellulose fabric and the matrix. The higher crystallinity of the NCFs due to the generation of silver based nanoparticles (as proved in X-ray analysis) may be responsible for the increase in the maximum stress and the Young’s modulus. However, no regular trend was observed in the tensile parameters of NCFs. The %Elongation at break of the NCFs was found to be lower than that of the cellulose fabric and the matrix.
Maximum stress, Young’s modulus, and %Elongation at break of cellulose fabric, matrix, and the NCFs prepared using 1–5 mM aqueous AgNO3 source solutions.
Conclusions
The NCFs with in situ generated AgNPs were prepared using various concentrations of aq. AgNO3 as source solutions and aqueous dispersion of low-cost herbal turmeric powder as the reducing agent. The generated AgNPs were retained by the NCFs even after repeated detergent washings. The NCFs before and after detergent washings had roughly spherical nanoparticles in the size range of 41–130 nm with an overall average of 78 nm. The crystallinity of the matrix was found to be higher than that of the cellulose fabric due to the interaction between the functional groups of aqueous dispersion of turmeric powder. Further, the generated AgNPs enhanced the crystallinity of the NCFs. The X-ray analysis also indicated the generation of both AgNPs and Ag2ONPs in the NCFs. The NCFs were found to be thermally stable even at 300°C. The NCFs prepared in this study had higher tensile strength and the modulus than the matrix and the cellulose fabric. The NCFs exhibited good antibacterial activity against both Gram-negative and Gram-positive bacteria even when lower millimole concentrated source solutions were used and hence can be considered as antibacterial apparels and hospital bed materials.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Natural Composite Research Group (NCR), Department of Mechanical and Process Engineering(MEPE), TGGS, King Mongkut's University of Technology North Bangkok (KMUTNB), Thailand (Grant No. KMUTNB-64-KNOW-001). Thailand Patent filed with Registration Number: 1803001275.
