Abstract
Aquaculture is usually carried out in cage nets made of textile materials. Fouling organisms settle on the cage nets over time, close the mesh openings, and cause unwanted weight gain. In order to prevent this problem, fishing nets or cages are treated with antifouling paints. Therefore, there is a great need for developing eco-friendly antifouling biocides since negative ecological effects have been proven for the current antifouling biocides and paints. In this study, it was aimed to take advantage of the slow and sustained release of microcapsule technology. For this purpose, econea active agent (an eco-friendly antifouling chemical) was encapsulated with spray-dryer method, carried out capsule optimization studies, and impregnated to the ultra-high-molecular-weight polyethylene fishing nets with two different binders as polyurethane and acrylic. Fishing nets, which transferred microcapsules, were compared with a commercial copper oxide-based dye. Microcapsule-treated and untreated net samples were immersed in a marine ecosystem next to an aquaculture zone for six months in the Aegean Sea. Fouling organisms on nets were observed by taking underwater photographs at periodic intervals. Physical tests (tensile strength, variation in mass) were performed on the nets before and after the treatment with microcapsules. Solidity of the nets and analyses of fouling organisms were examined after the field study. These results show that copper-free eco-friendly antifouling microcapsules are effective against the fouling mechanism. The novel ecofriendly formulation has promising results, and slightly dissolving versions of studied ecofriendly microcapsules could be an alternative for copper-based antifouling biocides.
Introduction
Biofouling is the attachment of organisms to the surface in contact with water for a certain period of time. Biofouling is one of the most common problems in the marine sector. Marine aquaculture was developed in a rapid way with the improvement of human living standards and the demand for seafood. At present, the aquaculture industry is mainly offshore, and the proportion of aquaculture is increasing year by year. With the development of the industry, new problems arise which is the pollution problem of fishing nets. In aquaculture, the intensity of the biofouling event varies depending on the season, geographic region, and environmental conditions like temperature, pH, and salinity, and these are major problems for aquatic systems. The adhesion of marine organisms, such as seaweed, shellfish, and other invertebrates, to the ship hulls, fishnets, and some other marine facilities has been one of the most serious environmental problems in the world with the marine pollution becoming more and more serious. Fishnet is one of very practical tools in the fisheries industry, and most of fishnets are made of nylon-6, PE, ultra-high-molecular-weight polyethylene (UHMWPE), etc. which can be easily adhered by the seawater microbes, especially seaweed. The adhesion of the seaweed can do much harm to the mariculture and bring about much trouble to the fishers [1–7].
Antifouling is the ability of specifically designed materials and coatings to remove or prevent biofouling by any number of organisms on wetted surfaces. Since biofouling can occur almost anywhere water is present, biofouling poses risks to a wide variety of objects such as medical devices and membranes, as well as to entire industries, such as paper manufacturing, food processing, underwater construction, and desalination plants [4].
According to different antifouling mechanisms, the antifouling technology of fishing net can be divided into chemical, physical, and biological antifouling methods, of which the most efficient is to use antifouling coatings, one of the chemical methods. Tributyltin (TBT) is the main active ingredient in antifouling (AF) dyes used to prevent bioaccumulation in ships’ submarines. However, the production and use of TBT-based antifouling paints were banned in 2003 and 2008, respectively, due to their genotoxic effects on non-target marine organisms. In modern times, antifouling paints are formulated with cuprous oxide (or other copper compounds) and/or other biocides—special chemicals which impede growth of barnacles, algae, and marine organisms. However, it is clear that dyes containing copper oxide have a good antifouling effect but also harmful for the environment in ecological sense. Although copper(I) oxide (main biocide) and commercially used booster biocides have satisfactory antifouling effects, antifouling paints prepared from these biocides have low service times, and they exhibit high eco-toxicities against non-target marine organisms [8–30]. Thus, various efforts have been made in order to create new antifouling methods as potential alternatives. A relatively recent method, which involves the microencapsulation of biocidal agents, gives us eco-friendly solution for this purpose [21].
In this study, econea capsules, which were environmentally ecological active substance, were obtained by using spray-dryer method. For this purpose, optimization of the production of microcapsules was done, and optimum formula was obtained. With microcapsulation technology, it was aimed to achieve controlled release by using less active substance (ECONEA®) than other methods. Thus, it was aimed to achieve long-lasting effect with less chemical usage. It was expected to have a high export potential for the aquaculture sector, which has quite high investment costs, and long-term antifouling effect was expected. Econea, which is known to be environmentally friendly compound, was encapsulated and impregnated to the UHMWPE fishing nets with two different binders as polyurethane and acrylic. Fishing nets were not dyed or coated by copper oxide or with any substance as distinct from unlike conventional methods. On the other hand, microcapsule-transferred fishing nets were compared with a commercial copper oxide-based dye. In this study, the same type of fishing nets was used to examine the effectiveness of microcapsules. Subsequently, field work was made for capsules transferred fishing nets for six months in a fish farm in the Aegean Sea in Turkey (from June to December). Fouling organisms on nets were observed by taking underwater photographs at periodic intervals for six months by AEE Lyfe Titan Action Camera which have 16 megapixels resolution. Strength, weight, pore size, and fouling organism analysis of fishing nets was performed before and after field work. As a result, it was determined that microcapsulation technology gives antifouling properties to fishing nets according to the analysis, and it is thought that it may be an alternative to copper-containing paints for antifouling properties because of their ecological property.
Material and methods
Material
In this research, 100% UHMWPE fishing net, which have 1/2″ mesh size and 2.0 mm twine diameter, was used. In order to evaluate microcapsule efficacy, the type of fishing net used was kept constant, and only UHMWPE fishing net was used. Marine antifouling agent econea was purchased from Janssen PMP. Econea (tralopyril–IUPAC: 4-bromo-2-(4-chlorophenyl)-5-(trifluoromethyl)-1H-pyrrole-3-carbonitrile)) is an arylpyrrole compound (Figure 1).

Chemical structure of econea.
Econea is metal-free and represents the latest generation of active ingredients for antifouling paints for ships and other marine structures. Econea has a broad spectrum of activity against hard-shelled and soft-bodied invertebrate animal fouling organisms including barnacles, hydroids, mussels, oysters, polychaete tube worms, ascidians, bryozoans, and sponges.
Econea is a nonmetallic organic biocide based on chlorfenapyr. It is considered as a promising antifouling compound since it can replace copper due to its wide range of antifouling activity including hard fouling. Econea is a non-persistent compound susceptible to hydrolysis (half-lifetime of 3 h in sea water) [31]. However, this feature implies premature degradation and loss of antifouling activity; therefore, encapsulation could enhance its stability in antifouling process, especially in water-based formulations. Because of mechanical movements in the sea (impact of fish, sea creatures, stream, etc.), the active substance econea can release into the environment.
Ethyl cellulose premium 4 (4 cP viscosity) and ethyl acetate were purchased from Sigma Aldrich Co. (USA) for microencapsulation. All other materials (acrylic and polyurethane binders) were analytical grade. Tween 20 auxiliary agent was used as a dispersing agent and purchased by Merck Co. (USA).
Preparation of the microcapsules
In order to obtain the microcapsules, the solvent was prepared like coacervation method. In this process, the interactions of water-insoluble polymers with water were utilized to form the microcapsules. Ethyl cellulose and econea were dissolved homogenously in organic solvent in a specific ratio. Polymer-rich organic phase was added to polymer-free aqueous phase. Active ingredients were mixed with Silverson high shear mixer. Afterward, a spray dryer (SD-Basic LabPlant, Huddersfield, UK) was used to collect the microcapsules. The compositions were fed to the spray dryer at the following conditions for each batch size: feed flow rate of microencapsulating composition 10 mL/min; inlet air temperature 135°C; and outlet air temperature 90°C. Microcapsules were collected from the product vessel using a soft brush in the fume hood and transferred to glass containers for storage. The studies were done for producing microcapsules with active substance (econea). For this purpose, 2:1, 4:1, and 8:1 “polymer (ethyl cellulose):econea” ratios were used. Chemical quantities and test conditions for spray drying are given in Table 1.
Chemical quantities of capsules and test conditions for spray drying.

Mass yield of microcapsule
The total powder obtained after spray drying was weighed, and the process yield was calculated as a percentage of the amount of solids added during the preparation process according to equation (1)
Active substance determination
In order to determine the amount of active ingredient in the capsules containing econea, analyses were carried out with Perkin Elmer UV–vis spectrophotometer. The econea wavelength was read against the blank solution, and it was found that the λmax corresponds to the absorbance value of 296 nm. The solutions were diluted in predetermined proportions to form auxiliary solutions. The absorbance values were recorded at the 296 nm wavelength of the solutions transferred to the quartz cuvettes for samples. In some prescriptions, only the binder was transferred for control purposes. In order to see the effect of encapsulation, some of the prescriptions are also made without capsulation for econea.
In order to find the econea ratio in the capsule, the 50 mg absolute weight capsule was weighed in a 100 mL volume on a precision scale, 50 mL of ethyl acetate was added, followed by stirring in a shaking water bath at 700 r/min for 12 h at room temperature. Samples were mixed in an ultrasonic bath twice for 20 min with 20 min intervals and treated in centrifuge for 10 min. The resulting filtrate was analyzed using the UV–vis spectrophotometer at the predetermined absorbance value.
Particle size of microcapsules
Particle size and distribution analysis was performed on the Haribo Partica LA-950 V2 model by laser diffraction measurements.
Particle morphology of microcapsules
The morphologic properties of the capsules were evaluated using scanning electron microscopy (SEM-Quanta 250 FEG). Samples were gold-coated (15 mA, 2 min) to assure electrical conductivity. The measurements were taken at 2 kV accelerating voltage.
Application of the microcapsules to the UHMWPE fishing nets
The application of the selected optimum formulations to the fishing nets was carried out according to Table 2 which was given below.
Capsule transfer prescription.

On the other hand, water-based acrylic and polyurethane binders for fixing were selected, and their effects were investigated. For this purpose, 10 different transfer prescriptions were coded and implemented with different capsule ratios and binders as shown in Table 3.
Fish nets coding system.

The microcapsules were dispersed in a commercially available water-based binders. Afterward, microcapsules were transferred to the fishing nets with dip-coating method [31]. Thus, a surface was created on the fishing nets as in antifouling paint applications. As a result of mechanical movements in the sea (impact of fish, sea creatures, stream, etc.), the active substance Econea was released into the environment.
Field work
The fishing nets were placed on the submarine in the fish farm (Guven Balık) in Karaburun, Aegean Sea, Turkey (38°27′31.7′′N 26°36′58.1′′E) at a depth of 4–5 m (Figure 2) for six months by stretching to 30 × 30 cm specially prepared frames, and periodically, images were taken with AEE Lyfe Titan Action Camera which have 16 megapixels resolution.

Images from fish farm in Aegean Sea.
The average seawater temperature over this time (from June to December) was 21.6 ± 0.01°C (range 18.3–24.0°C), and salinity was almost 36% for sea in the same period.
Solidity of nets
The net solidity (Sn) was expressed as the ratio of the solid fraction of the net to the total net outline area, and it is measured from images according to equation (2)
To quantitatively study the porosity of the net with/without biofouling, the net solidity Sn is defined as the ratio between the projection area and the outline area of the plane net. Both the clean and biofouled nets were photographed from the front view, and solidity was calculated by photoshop. On the other hand, weight of the biofouled fishing nets was measured.
Strength and weight analysis
Upon termination of submarine work, fish nets were cleaned with water spray gun. After the cleaning of the fishing nets whose field works were terminated, the weights before and after the treatment were measured. On the other hand, tensile strength and elongation values were measured by DYNA 300 DP Dynamometer for fishing nets. The strength and elongation analyses were performed from 30 different mesh points of fish nets, and standard deviation and confidence intervals of each sample were calculated.
Results and discussion
In this study, capsules were successfully prepared by spray-drying method. This method is a simple, viable method to obtain microcapsules, suitable to prevent drug biological activity loss, avoiding exposure to elevated heating and to organic solvents.
Mass yield of microcapsule
The yield of MPs, produced by laboratory-scaled spray dryer, may not be high due to loss of light-weighted particles by vacuum suction and adherent to the inside wall of the spray-dryer apparatus. The mass yields ranged between 15.0 and 65.3% (w/w) as given in Table 4.
Mass yield of microcapsules.

The yield values of the capsule experiments with econea and three different molar ratios were calculated, and it was concluded that the capsule of the highest yield was a 2: 1 molar ratio. As the ratios of the shell material (ethyl cellulose) were increased, the microencapsulation yield tended to decrease because of the lack of sufficient quantity to the active agent (Econea).
Active substance determination
The method used to find the amount of active ingredient contained in the capsules was based on the spectroscopic measurement of the active substance released by complete dissolution of the shell material. For this purpose, the UV spectrum and λmax of econea should be determined. The econea in the ethyl acetate solvent of the determined concentration was read against the blank solution, and the λmax was found to correspond to the absorbance value of 296 nm. By taking this value, the standard calibration line was drawn as shown in Figure 3.

Standard accurate graph of the econea substance in the solvent.
The concentration of active substance in the capsules was calculated by means of the calibration curve. The amounts of econea in ethyl cellulose capsules are given in Table 5.
The amounts of econea in ethyl cellulose capsules.
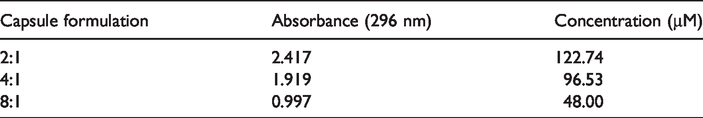
As a result of the data obtained, it was determined that the active ingredient was successfully encapsulated, and the active substance taken into the capsule decreased linearly with the decrease in the active substance rate added to the organic phase during the capsule formation phase.
Particle size of microcapsules
The mean particle size of MPs was determined by laser diffraction method and changes in MP size due to changing polymer: active agent ratios were observed. Particle size distribution graphs of microcapsules were indicated in Figures 4 to 6.

Particle size distribution of the 2:1 capsules.

Particle size distribution of the 4:1 capsules.

Particle size distribution of the 8:1 capsules.
When the particle size analysis of the capsules produced at different ratios was evaluated, the 4:1 capsules produced had a particle size of 375.3 μm and a high homogeneity. The 2:1 and 8:1 capsules produced had a particle size of 658.2 and 1152.6 μm, respectively. Econea-based capsules with 2:1 and 8:1 ratios were homogeneously distributed and had a close value with 4:1 ratio capsules but standard deviations were high (48.6% for 2:1 and 8:1 for 110%).
Particle morphology of microcapsules
Spray-dried microcapsules were usually characterized by spherical shape and narrow particle size distribution. Typical photomicrographs that were obtained by SEM of the microcapsules show that the spray-dried product is composed mainly of spherical-shaped particles (Figure 7).

SEM images of microcapsules.
According to SEM analysis, microcapsules filled with active substance (econea) were obtained. However, when the micrographs of 8:1 ratio microcapsule were examined, it was observed that not all particles appeared morphologically spherical. Some of the microcapsules’ center was collapsed due to sudden solvent evaporation when the polymer solution introduced into hot air chamber and also capsule distribution was not seen homogeneously. On the other hand, the biggest cause of econea-induced collapse in capsules is the active substance that cannot be encapsulated is thought to accumulate on the shell material. According to the SEM images of microcapsules, the optimum econea content to get homogenous spherical microspheres was seen in 2:1 ratio microcapsules.
After characterization studies, 2:1 and 4:1 ratio microcapsules were chosen for field work and made application to the fishing nets. Although the microcapsule with the 2: 1 ratio was optimum content according to the SEM images, submarine studies of the 4: 1 ratio capsules were performed because of high homogeneity of their particle size distribution.
Figure 8 represents the SEM images of microcapsules after application to fishing nets.

SEM images of microcapsules after application to fishing nets.
Field work
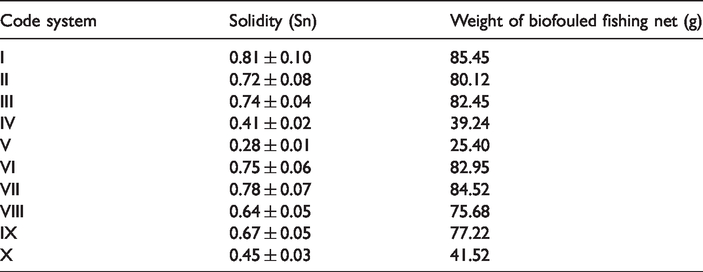
The effectiveness of microencapsulation is related to the nature of the active agent (chemistry and size), to the composition of the biopolymer, to the method of production, and to the features of the obtained microparticles (particle’s size, etc.). As a result of the studies, the time-dependent effectiveness of econea capsules was evaluated visually at the field work, and then the solidity properties were calculated as shown in Table 6. Solidity of nets was evaluated from different meshes of fishing nets, and the number of experiments was 30.
Solidity and weight of biofouled fishing nets.
After characterization studies like SEM, particle size and active substance determination 2:1 and 4:1 ratio microcapsules were chosen for field work and made application to the fishing nets as Table 3. Micro and macro-organisms that settled on the fishing nets were identified after six months of seawater immersion. The images of the fishing nets, which have been studied on the underwater camera, are shown in Figure 9.

Images of fishing nets from field study.
After the field work, when the images are examined, it was observed that the pores of the fish nets, in which were produced with the coded as “V” which was 2 : 1 (shell material : active substance) and transferred with the polyurethane binder, were more open and did not show viable proliferation on these nets. This shows that microcapsules were provided long-lasting effects due to their slow release properties. In terms of its antifouling properties, fishing nets, which were produced with the coded as “IV” which was 2 : 1 (shell material : active substance) and transferred with the acrylic binder, gave the second best result as shown in solidity values of fishing nets (Table 6). Polyurethane durability contributes significantly to the long lifetimes of many products. The extensions of product life cycle and resource conservation are important environmental considerations that often favor the selection of polyurethanes [32]. Due to these properties of polyurethane binders, it was thought that better antifouling properties were obtained when compared to acrylic binder.
When the images obtained from the field study were examined, it was determined that the non-encapsulated econea which was impregnated by binders did not show the desired antifouling effect for both polyurethane and acrylic binder. In addition, it was found that the binder without any other auxiliaries and active agents could not provide an antifouling effect too.
When pore size of fishing nets was compared in terms of different microcapsule ratios, it was observed that 4:1 capsules did not provide the antifouling effect which was obtained in 2:1 capsules transferred fishing nets. The reason for this : capsules that were produced in a 2 : 1 ratio are thought to provide a long-lasting effect as there was higher amounts of the active ingredient than the 4:1 capsules.
Econea, which is the low soluble in water, presented the greatest encapsulation efficiency followed. As a consequence, antifouling biocide, which is generally hydrophobic and with low water solubility, was expected to be encapsulated successfully with the coacervation–spray-dryer technique. In addition, after field studies, it was observed that capsule-transferred nets were cleaned more easily because less biofouling organisms were attached to fish nets.
As a result of field studies, it has been observed that capsule-transferred fishing nets gave better results when compared to commercially used copper oxide containing dyes. It was contemplated that capsules having slow and prolonged release may serve as an alternative to commercially used dyes.
Macro-organisms analysis
After removal of the nets from the marine environment, samples of fouling organisms were collected, preserved in formaldehyde, and photographed by digital camera. Macro marine organisms such as the diatoms, Tunicate sp., Bivalvia sp., and Bryozoa sp. were identified on the nets and are shown in Figure 10.

Marine organisms attached to the surfaces of fishing nets.
These macro-fouling organisms have important biological roles in the Mediterranean ecosystem. It is of great importance for sustainable aquaculture to develop biocidal agents that do not have a lethal effect upon these species.
Solidity of nets
The solidity and weight of biofouled fishing nets after six months are presented in Table 6.
According to the present study, solidity increased without microcapsule, and it was shown that solidity reached the lowest degree in the presence of 2:1 microcapsule-transferred fishing nets with polyurethane binder (V). Biofouling weight was increased without microcapsule as solidity also. As both solidity and growth amount were continuously increased, it was considered that the level of biofouling on the nets increases without microcapsule treatment.
Strength and weight analysis
Table 7 shows the weighting results of the fishing nets before and after field work.
Weighting results of fishing nets before and after the field study.

Considering the pre-test results, it was observed that the transfers made by the impregnation process increased the weight of the fishing nets compared to the untreated fishing net. After six months of experimentation, a decrease of 2% was observed in the weights of the fishing nets which were impregnated. It has been observed that the untreated fish nets show less change in weight than the other nets. It was thought that this situation may be caused by the release of chemicals on fish nets, when compared to untreated fish net.
The strength results of the fishing nets performed in field studies are given in Table 8. Strength and elongation values of nets were evaluated from different meshes of fishing nets, and the number of experiments was 30.
Strength and elongation values of fishing nets.

a+ values shows standard deviation.
When the results of tensile strength and elongation (mm) required to break the fishing nets were evaluated, no significant changes were observed before and after the field study.
Conclusion
In this study, econea capsule, which is ecological and an environmentally friendly active substance, was obtained by using spray-dryer method as an alternative for copper-oxide dyes to gain antifouling properties to fishing nets. As an active agent, econea was successfully encapsulated. 2:1 and 4:1 capsule ratios were selected with optimization studies and impregnated to the 100% UHMWPE fishing nets with water-borne polyurethane and acrylic binder. The antifouling activities of encapsulated econea also were evaluated, and they were found to be mechanically durable for six months in the fish farm environment. Especially, the nets transferred 2:1 ratio microcapsules with the polyurethane binder exhibit better antifouling properties than other chemicals. It was observed that the pores of the fishing nets, which were produced with, were more open and did not show viable proliferation on these nets. On the other hand, microcapsulation process has no considerable effects on the strength and elongation properties of fishing nets after the field study. With microcapsulation technology, it was provided to achieve controlled release by using less active substance (ECONEA®) than other methods. Thus, long-lasting effect was achieved with less chemical usage. It is expected to have a high export potential for the aquaculture sector, which has quite high investment costs, and long-term antifouling effect is expected.
Footnotes
Acknowledgements
We especially thank Güven Balık Üretim ve Ticaret, LTD.ŞTİ for supporting our underwater studies. We would also like to thank Prof Dr Levent Çavaş and Assoc Prof Dr Halis Erdoğan for their advice in keeping our progress on schedule.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article : TÜBİTAK-BİDEB 2211.
