Abstract
Silver ions (Ag+) and silver nanoparticles (AgNPs) are effective antimicrobial agents that act against a broad spectrum of bacteria. The releasing quantitation of free Ag+ is exclusively responsible for the biological toxicity, while limiting the free Ag+ in AgNPs or in polymers would largely ease the conditions. In this study, the different concentrations of silver nitrate (AgNO3) in poly(vinyl alcohol) (PVA) and an optional preheat treatment on the spinning solution before electrospinning to form the Ag+/AgNPs/PVA fibrous membranes through electrospinning technology were investigated. The morphologies, AgNPs dispersity within the PVA matrix, and the sterilizations (UV irradiation and autoclave) for Ag+/AgNPs/PVA fibrous membranes were characterized. The antibacterial activities of Ag/PVA composite membranes combined with the in-house prepared light-cured resin were also investigated. Results showed that the AgNPs/PVA fibrous membranes with antibacterial capability can be produced with the addition of at least 5 wt.% of AgNO3 to PVA. The antibacterial activities of Ag content in the PVA matrix were increased in the high presence of Ag. Noteworthy, the antibacterial enhancing effect was observed for the spinning solution groups after preheating treatment at 100°C for 1 h. The result suggests that Ag/PVA fibrous membranes sterilized by autoclave hindered antibacterial effectiveness due to the significant particle size changes in the generation of large-sized AgNPs. Nevertheless, the direct application of UV light irradiation to Ag/PVA fibrous membranes preserves their active antibacterial profile against Staphylococcus aureus and Escherichia coli. We also demonstrated that these designed AgNPs/PVA composite membranes can equip the resin with an active antibacterial capability, could benefit from the prevented bacteria breeding in microleakages and thus further reduce the possibility for secondary caries.
Keywords
Introduction
Silver especially when they are presented as Ag+ or been synthesized into nanoparticles are considered to be broad spectrum antibacterial agents due to their large specific surface to volume ratio appearance [1]. Since silver is effective against Streptococci in the oral cavity and periodontal pathogens and prevents adhesion of bacteria to surfaces and formation of biofilms, it can be used as a useful antibacterial additive to dental materials [2–4]. Composite resins have been widely used in dentistry for restoration purposes and have grown popular over the last 50 decades because of their capability to adjust the color for aesthetic specifications, which makes it easy to obtain shades similar to enamel. However, microleakages between the composite resin and tooth tissue due to polymerization shrinkage during operations, thermal expansion or shrinkage after oral thermal fatigue aging, and changes in dental tissues after acid etching caused by oral bacteria remain the foci of clinical attention in dentistry [5].
A very commonly used monomer for the polymeric matrix is 2,2-bis [4(2-hydroxy-3-methacryloxypropoxy)-phenyl]propane, also known as bisphenol A glycidyl methacrylate (Bis-GMA). Bis-GMA would composite with diluent monomers, and among them, triethylene glycol dimethacryate (TEGDMA) is the most extensively used in current resin restorations. The majority of dental composites undergo solidification via free radical chain polymerization which enables the formation of a cross-linked network within a very short time curing [6]. In the past few decades, the strategies for improving the composite resin properties by changing the coupling agent and adding nanoparticles (NPs) as reinforced particles have significantly improved the properties of the resin composite and effectively reduced the probability of resin collapse, resulting in marginal discoloration, secondary caries, and pulpitis [7]. However, secondary caries caused by microleakages is often unavoidable [8–10]. Therefore, antibacterial agents incorporated into dental composite resins to suppress bacterial growth and acid production and thus reduce the possibility of secondary caries have been extensively studied [11].
The antibacterial effect of the composite resins can be achieved by incorporating fillers that contain organic or inorganic bacteriostatic agents. Clinical studies have reported that the inclusion of antimicrobial monomers in adhesive systems is an effective dental treatment strategy [12]. Organic antibacterial agents provide a rapid and powerful bacteriostatic action. However, problems regarding the limited application of organic antibacterial agents exist, such as easy migration, poor heat resistance, and biotoxicity [9]. Inorganic antibacterial agents possess good compatibility, durability, and antibacterial properties. Silver nanoparticles (AgNPs) are commonly used inorganic antibacterial agents, but AgNP agglomerations often occur, which results in insufficient bacteriostatic effect [1]. Many AgNP preparation strategies are available, including the use of reducing agents, such as sodium borohydride, sodium citrate [13,14], N, N-dimethylformamide [15], or hydrazine hydrate [16]. However, these compounds are toxic, and using such agents may evoke issues on environmental toxicity and induce biological hazards. Therefore, nontoxic chemicals and renewable materials are preferably used to develop a green synthesis for AgNPs in reducing environmental risk.
Novel studies and electrospinning technologies are devoted to preparing an antibacterial hybrid membrane with AgNPs incorporated inside the fibers, thereby preventing AgNPs from aggregation by dispersing the particles through the fibrous structures [17–20]. The common environmentally friendly reducing agents used in solving environmental hazards are glucose, polyethylene glycol, poly(vinyl) alcohol (PVA), and polyvinylpyrrolidone (PVP), all of which are popular ways to reduce Ag+ into AgNPs. Among them, the aqueous solutions of PVA and PVP can simultaneously function as stabilizers to create small and stable AgNPs, ion reductants, and fiber main constituents. Our aim in this present contribution was to prepare the antibacterial PVA fibrous membrane using silver nitrate (AgNO3) as a precursor through optional preheat treatment to investigate the synergistic effect of Ag+ combined with AgNPs against gram-positive and gram-negative bacteria. An investigation into the efficacy of the Ag+/AgNPs/PVA membranes through the sterilized behavior and then incorporated within the resin to evaluate the antibacterial capability systematically.
Materials and methods
Materials
The PVA (Mw: 146,000–186,000 g/mol) and chemicals used for the in-house synthesized resin compositions with bisphenol-A glycerolate dimethacrylate (Bis-GMA), triethylene glycol dimethacrylate (TEGDMA), camphorquinone (CQ), butylated hydroxyl toluene (BHT), and N, N-dimethylaminoethyl methacrylate (DMAEMA) were purchased from Sigma-Aldrich. The AgNO3 precursor was purchased from Riedel-de Haën, whereas the bacteria culture in tryptic soy broth (TSB) (Soybean-Casein Digest Medium, BD Bacto) and the agar for the bacteriological culture media were obtained from Acumedia.
Ultraviolet-visible analysis
The absorption spectra of the suspended solutions before electrospinning from each preheat treatment procedure for Ag+ reduction observation were determined using a ultraviolet-visible (UV–vis) spectrophotometer (UV-1800, SHIMADZU, Japan) within the range of 250–800 nm.
Electrospinning process to fabricate the fibrous membranes
As suggested by the previous experimental result [21], the electrospinning in this study was applied on the 7.5 wt.% PVA solution in which the 0.75 g PVA powder was added to 10 mL of d.d. water and was stirred at 80°C for 16 h. The mixture was then cooled to room temperature to create the 7.5 wt.% PVA spinning solution. Afterward, different amounts (0.0075 to 0.1125 g) of AgNO3 powders were added into the above PVA aqueous solution which were referred to varying AgNO3/PVA weight ratio levels (1 wt.% to 15 wt.%) and then the solutions were stirred for 30 min. The sample bottle was wrapped with aluminum foil to protect from light degradation. Table 1 shows the detailed processes of the spinning solution before the electrospinning and designated groups with different AgNO3. The preheat treatment involved heating the spinning solution in hot water baths at 60°C for 24 h or at 100°C for 1 h to facilitate the AgNP reduction [22]. The prepared Ag/PVA solutions were immediately subjected to room temperature for electrospinning. The nozzle’s inner (Di) and outer diameters (D0) and length are 0.686 mm, 1.067 mm, and 2.6 cm, respectively. A syringe pump (KDS 100, KD Scientific) was used to deliver the solutions to the nozzle at a controlled flow rate of 0.4 mL/h. An electrical voltage of 15 kV (high-voltage power supply, Matsusada Precision Inc.) was applied in all AgNO3/PVA groups to the spinneret, whereas 11 kV was applied to the PVA group only. To construct a needle-to-plate electrode configuration, an aluminum board (30 × 30 cm2) was used as the collector for the electrospinning fibers at a fixed tip-to-collector distance of 20 cm. Accordingly, Figure 1 shows the detailed experimental designs, parameters, and electrospinning processes, where the optional preheat treatment was applied to the spinning solution.
Experimental conditions: the effects of solution concentration and heat treatment methods applied on the solution before electrospinning.

AgNO3: silver nitrate; PVA: poly(vinyl alcohol).

Schematic of the experimental design for antibacterial activity of electrospinning membranes.
Morphology and characterization of fibers
The morphology of the fibers was observed using a scanning electron microscope (SEM, S-4800, HITACHI Co., Japan). A transmission electron microscope (TEM, JEOL-2100F; JEOL Co., Japan) was used to determine the locations of the AgNPs in the PVA-matrix fibers. The fiber diameters in the SEM images and the particle sizes of the reduced AgNPs in the TEM images are calculated using ImageJ software to determine the mean and median diameters.
Combination of the membranes with dental resin
Supplementary Table S1 lists the detailed compositions of the in-house fabricated resin. The curing procedures were processed after attaching the composite membranes on both sides of the samples to validate the bacteria contact measurement. The test resin was light-cured using a light intensity between 580 mW/cm2 and 600 mW/cm2 for 40 s for two cycles for both sides of the samples. After electrospinning, all samples and the as-prepared combination of the membranes with resin were stored in the dark until the subsequent measurements.
Methods applied for membrane sterilization and antibacterial characterization
Sterilization methods
Two sterilization methods were used in this study: autoclave and in-situ UV irradiation. For the autoclave condition (TM-328, Tomin), the as-prepared AgNO3/PVA fibrous membrane groups were processed at 121°C under 1.2 kg/cm2 steam pressure to sterilize for 15 min. The UV irradiation was performed using a UV lamp at 365 nm for 8 h at an intensity of 700 mW/cm2.
Inhibition zone–agar diffusion test
Staphylococcus aureus (ATCC number: 25923) and Escherichia coli (ATCC number: 10798) were used in this study. S. aureus and E. coli cells were cultured in Luria–Bertini broth. The bacterial suspensions were diluted to achieve an OD595 of 0.2 to 1 × 107 cells/mL using an ELISA reader. Then, 300 µL of the bacterial suspension was evenly spread using a sterilized glass spreader on the surface of the solidified TSB agar. Samples of the as-prepared AgNO3/PVA groups were cut into circular sheets with a 6 mm diameter. Each circular sheet was pressed tightly on the 10 cm plates in contact with the bacteria. The pure PVA fibrous membrane without AgNPs was used as the control group. After 24 h at 37°C incubation, the inhibition zones were recorded and photographed.
Biocidal measurement
The bacterial suspensions of S. aureus and E. coli were diluted to achieve an OD595 of 0.2. Then, 1 mL of each suspension was placed in a sterilized tube containing 20 µL of as-prepared AgNO3/PVA individual groups. After vortexing for 10 s, all mixtures were incubated with gentle shaking at 37°C. Then, 100 µL of the bacterial suspension was withdrawn and processed to achieve a 1000× dilution in the TSB broth. Finally, 300 µL of the diluted bacterial suspension was withdrawn and evenly spread on the prepared agar plates by using a sterilized glass spreader. Colony counting occurred after culturing at 35°C for 24 h.
Results and discussion
UV–vis analysis
To standardize the AgNP synthesis route and evaluate the combined effect of the spinning solution through preheat treatment and the electrospinning technique on AgNP reduction and AgNP/PVA fibrous membrane formation, two preheat procedures of AgNO3/PVA spinning solutions were tested using the results of the UV–vis spectrophotometers: 60°C for 24 h and 100°C for 1 h. The reduction of Ag+ in the spinning solution under the latter condition was confirmed and compared with the result of the former condition. The spectrum ranged from 430 nm to 450 nm of the absorption, and the strong absorption band was revealed at 436 nm (Figure 2). The resonance peak at 436 nm indicates the formation of AgNPs. Therefore, the condition for Ag/PVA at 100°C for 1 h was used for further analysis (Table 1).

Surface plasmon resonance of 5 wt.% AgNO3/PVA solution under 100°C for 1 h (blue line, C5 group) and solution under 60°C for 24 h (red line) preheat treatment before electrospinning.
In vitro antibacterial capability of varied Ag-doped AgNPs/PVA fibrous membrane concentrations: Inhibition of zone–agar diffusion test
Figure 3 shows the systemic survey of the qualitative antibacterial activity against E. coli and S. aureus in the groups of PVA, A1, A5, A10, and A15 with AgNO3/PVA. The spinning solutions were not subjected to preheat treatment, the B1 spinning solution was pretreated at 60°C for 24 h, and the C5, C10, and C15 groups with AgNO3/PVA spinning solution were pretreated at 100°C for 1 h before electrospinning. The results showed no antibacterial effect in control groups (i.e. A1 and B1). Therefore, the incorporation of Ag+ concentration in PVA fibrous membranes via the electrospinning method plays a critical role in antibacterial capability. The results indicated that when the Ag content in PVA was raised to 5 wt.% in the A5 and C5 groups, the inhibition zone of the fibrous membranes against E. coli and S. aureus appeared. The largest inhibition zone against S. aureus was achieved by the increased Ag+ addition in the A15 group. Moreover, only the A5, A10, and A15 groups demonstrated increased antibacterial efficiency against S. aureus when the AgNO3 concentration in PVA was increased; the inhibition zones consequently increased from 2.39 mm to 3.74 mm. Although the A5, A10, A15, C5, C10, and C15 groups exhibited a better antibacterial capability against S. aureus than against E. coli, the inhibition zone diameters against both bacteria showed insignificant relationship when the AgNO3 concentration in fibrous membranes was increased.
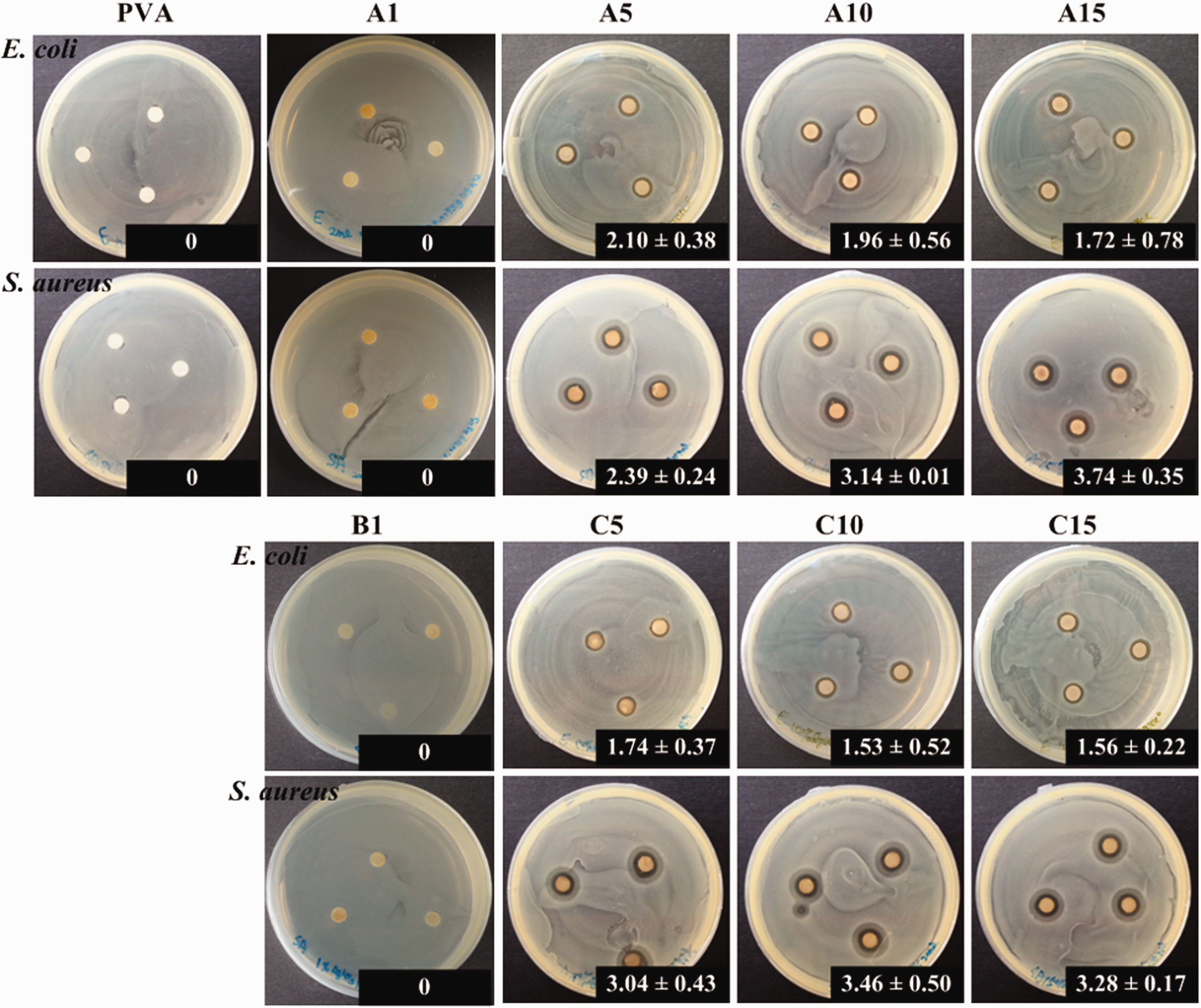
Inhibition zones (mm) against E.coli and S. aureus of PVA-only and Ag+/AgNPs/PVA without (A groups) and with optional spinning solution preheat treatment (B and C groups).
In vitro antibacterial capability of varied Ag-doped AgNPs/PVA fibrous membrane concentrations: Biocidal observation
To clarify the hybrid AgNO3/PVA fibrous membrane effect of AgNO3 concentration in the A5, A10, and A15 groups and the effect of preheat treatment in the C5, C10 and C15 groups on the antimicrobial capability against E. coli and S. aureus, the colonies of both bacteria after being cultured with the as-prepared AgNO3/PVA suspension solution at 35°C for 24 h was investigated (Figure 4). The antibacterial test in the control group showed that the live bacteria demonstrated colony spread. However, in the group with increased AgNO3 (i.e. A5, A10, and A15), the number of colonies decreased. The colonies of E. coli and S. aureus in the A15 group (with AgNO3/PVA) achieved the highest rate of 15 wt.%; the results showed aseptic growth on dishes. Hence, the reduced AgNPs seriously ruptured the part of the cell wall, leading to the leaching out of nutrients and a nucleic material swollen cell structure, consequently causing the death of the bacterium [22]. The results in Figures 3 and 4 indicate that the spinning solutions preheat treatment at 100°C for 1 h before electrospinning is beneficial to the antibacterial capability against E. coli and S. aureus. The study indicated precursor compounds such as AgNO3 can be blended in the solution to be electrospun and conversion to their active form. This approach is effective in providing a sustained release of the active Ag+/AgNPs agents, regulated by its contact and diffusion through polymer matrix by the degradation of the matrix, or by the combination thereof [23]. Notably, the size-controlled reduction of AgNPs over the different particle size range and the Ag+ distribution on the fiber surfaces showed that the results are extremely crucial for their antibacterial efficacy [24]. As the PVA molecule consists of many active OH groups on its side chains, it can serve as a chemical reducer and a template agent for electrospinning simultaneously [25]. Such a reduction process could be accelerated by heating even though the preheating treatment has little effect on the diameters of AgNPs than posttreatment (autoclave sterilization and UV light irradiation). The antibacterial activities of Ag amount in PVA matrix were increased in the highest amount of 15 wt.% Ag (A15 and C15), the enhancing effect was observed for the spinning solution groups after preheating treatment at 100°C for 1 h. The result showed that the combinations with AgNPs in PVA matrix have better antibacterial effects and the optimal group for almost totally antibacterial ability was in the group of C15.

(a) Antimicrobial tests for as-prepared Ag/PVA solutions without preheating treatment before electrospinning against E. coli and S. aureus (24 h incubation time). (b) Antimicrobial tests for as-prepared Ag/PVA solutions preheat treated at 100°C for 1 h before electrospinning against E. coli and S. aureus (24 h incubation time).
Morphology and characterization of fibers during and after sterilization
The formation of stabilized AgNPs was confirmed by the UV/vis and the appearance of the strongest absorption at 436 nm. The AgNP size was concentrated in narrow diameters (mean = 9.36 ± 3.84 and median = 8.36 nm) (Figure 5(a)).
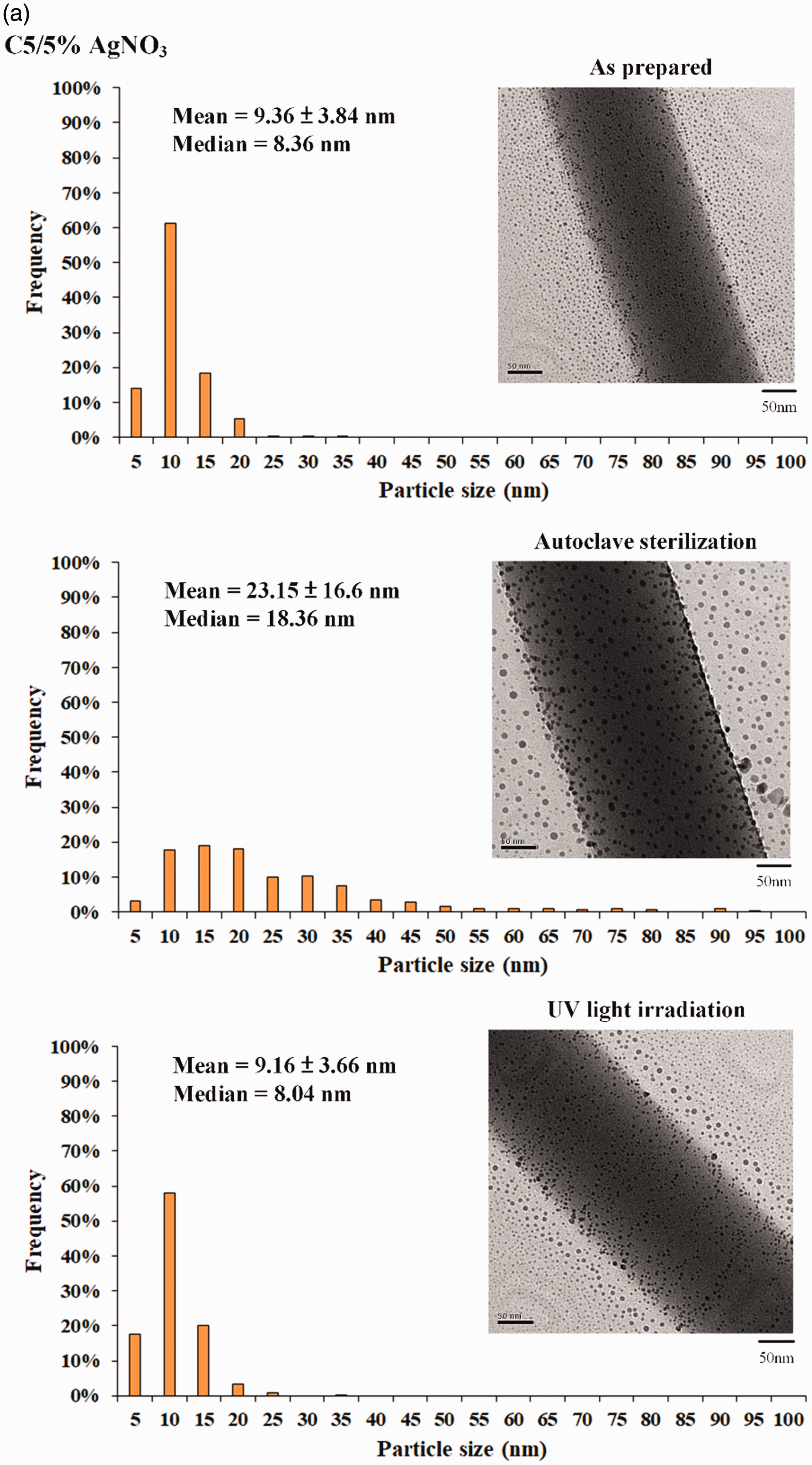
(a) Different sterilizations on the particle size distribution of AgNPs, bright-field TEM images (right); histograms of Ag particle size distribution (left) at different frequencies. (b) Noninhibition zones of group C5 against E.coli and S. aureus after autoclave sterilization.
Few traces of AgNP clusters may contribute to particle size variation due to further AgNO3 reduction, or AgNP crystals moving to fuse the grain at 121°C during autoclave sterilization. The middle part of Figure 5(b) shows the morphology. The AgNPs generated via electrospinning form aligned arrays along the orientation of the PVA fibers due to autoclave sterilization. The size distribution of AgNP contents exhibited insignificant changes under UV irradiation (lowest image in Figure 5(a)). The intensity of the cross-linked PVA’s color change from light white to yellow-brown can be attributed to the coarsening of AgNPs after steam autoclave sterilization (Figure 5(b)). This result showed that UV irradiation is a promising sterilization strategy for fabricated AgNO3/PVA fibrous membranes without changing the membrane’s antibacterial capability.
Practical applications show that AgNPs with sizes ranging from 3 nm to 10 nm demonstrate the optimal property combination due to its exposure surface (i.e. the contact area is considered) [22]. The decrease in the sizes of AgNPs can be controlled by performing Ag+ concentration, be stabilized from oxidative of active hydroxyl ion (OH-) in the PVA molecules, and applying thermal reduction to the entire system [22,25]. Therefore, aqueous PVA solutions are suitable for the electrospinning of fabricated antibacterial fibrous membranes to carry the metal antibacterial agent and oxide in advance as a reducing agent. For example, Abdelgawad et al. reduced AgNO3 in situ with a mixture of chitosan (reducing agent) and PVA to obtain a composite fiber [17]. Tijing et al. used polyoxyethylene [18] and Tian et al. used PVP [19] as an ion-reducing agent and fiber stabilizer for Ag+, respectively, to produce composite fibers with excellent antibacterial capabilities. In this study, the AgNPs were initially reduced by spinning the AgNO3/PVA solution at 100°C for 1 h before electrospinning and then stabilized using PVA during high-voltage electrospinning.
The electrospinning processes of the sub-microfibrous membranes of as-prepared PVA only and 15 wt.% AgNO3/PVA were observed using TEM (Figure 6). The well-dispersed and approximately spherical AgNPs are greatly reduced within the fibers. The PVA molecule consists of OH- on its side chains, and the efficiency of OH- exposure on the fiber surfaces enhances the side-chain alignment through electrospinning [22, 26]. The PVA also simultaneously serves as a chemical reducer, template agent, and fiber stabilizer for Ag+ during electrospinning. As well as the high voltage (15 kV) applied to the AgNO3/PVA, the electron transfer of the oxidation-reduction reaction between the hydroxyl radical and Ag+ to produce the final AgNPs [25,27]. When a high voltage is applied to the AgNO3/PVA solution in the syringe, the pendent droplet at the nozzle of the spinneret becomes highly electrified. In this study, the reduction of AgNPs in the membrane of the C15 group was composed by sub-microfibrous components with narrower fiber diameters and a more concentrated distribution than the controlled PVA only group (Figure 6).
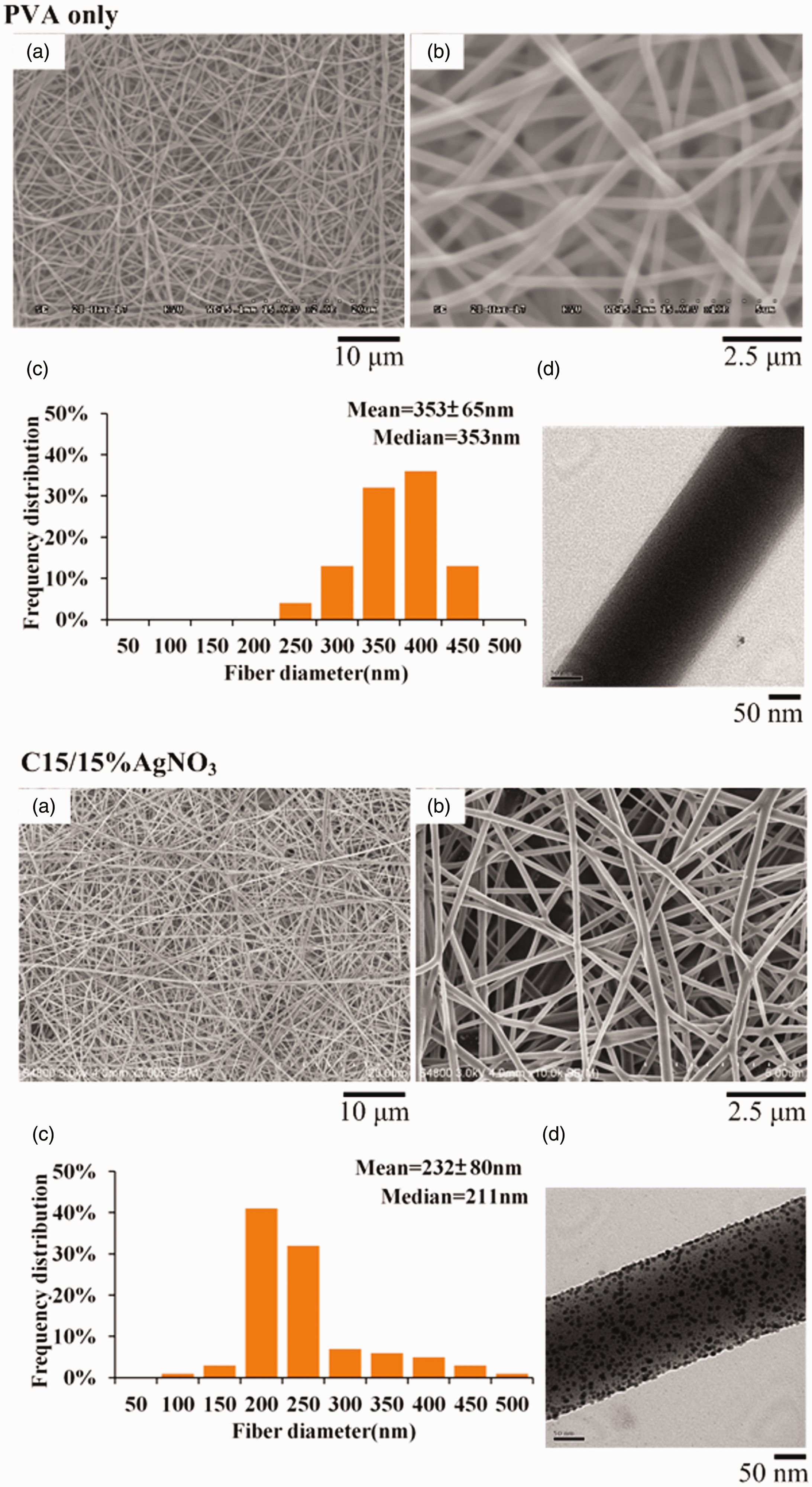
SEM and TEM images of PVA only and C15 group: (a) low-magnitude SEM image; (b) high-magnitude SEM image; (c) histograms of the fiber diameters; (d) bright-field TEM image.
Several studies have directly added antimicrobial NPs into dental resin to produce the bacteriostatic properties of the composite resins [28]. Researchers have introduced AgNPs and AgNP-decorated hydroxyapatite to improve the antibacterial properties of the resin [29–31]. However, the AgNP fillers with high specific surface area lead to poor AgNP dispersibility, thereby worsening the resin color stability and declining the resin’s mechanical strengths. Given the adverse effect of AgNPs on resin properties, Asmussen et al. added AgNO3 in the resin by using resin inhibitors to reduce AgNO3 into AgNPs, in which Bis-GMA or Bis-EMA will act as a protective agent to disperse AgNPs [32]. However, when the AgNP agglomeration occurred as well as the AgNO3 content increases, the polymerization of the resin would be inhibited and the degree of polymerization will greatly decline.
The concept of this study is to utilize the antibacterial fibrous membrane prepared by electrospinning as the interface membrane that will be potential placed/attached as a protective membrane in pre-filled cavities and then filled with the dental resin (Figure 7 and Supplementary data S3). The membrane in which well-dispersed AgNPs contain attached to the dental resin. If microleakage occurs between the resin boundary and tooth, then the interfacial membrane with AgNPs can play the role to effectively inhibit bacteria from breeding in the microleakage, and thus prevent the operation of secondary caries.

Schematic of the experimental design of the AgNP doped within the fibers, which can produce electrospun membranes with antibacterial activity via dental restorative materials.
Conclusions
The proposed electrospinning fabrication could efficiently produce PVA-matrix membranes doped with AgNPs by utilizing AgNO3 as a precursor. The adequate preheat treatment on the spinning solution was proven by the observed strengthening antibacterial effect. Moreover, the reduced sizes of AgNPs and the amount of doped AgNO3 exerted a large effect on the antibacterial capability. We validated the potential hygienic application of this functionalized membrane by evaluating their antibacterial effect when the Ag precursor (AgNO3) concentration in the PVA fiber reached 5 wt.%. The results of the present study showed the composite membranes containing AgNPs can significantly inhibit the growth of two typical bacteria: gram-positive bacteria of S. aureus and gram-negative bacteria of E. coli. The sterilization strategy of the antibacterial membranes with AgNPs emphasized in situ UV irradiation did not affect the antibacterial capability. An opposite result was observed when the in situ autoclaving was performed would invalidate the AgNPs’ effect on the antibacterial capability. The results showed a possible application that the dental resin combine with antibacterial membranes in oral health care is feasible.
Supplemental Material
sj-pdf-1-jit-10.1177_1528083720913345 - Supplemental material for Preparation of electrospun silver/poly(vinyl alcohol) fibrous membranes and characterization of the effect of sterilization processes on the antibacterial activity
Supplemental material, sj-pdf-1-jit-10.1177_1528083720913345 for Preparation of electrospun silver/poly(vinyl alcohol) fibrous membranes and characterization of the effect of sterilization processes on the antibacterial activity by Wen-Cheng Chen, Chia-Ying Ko, Kai-Chi Chang, Chih-Hua Chen and Dan-Jae Lin in Journal of Industrial Textiles
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Ministry of Science and Technology, Taiwan (Grant Nos. MOST 107-2314-B-037-033 and 106-2622-E-035-006-CC2).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
