Abstract
In the present project sodium alginate-based polyurethane dispersions were synthesized by two shot processes, using isophorone diisocyanate, polyethylene glycol (Mn-300), dimethylol propionic acid as internal emulsifier along with other reagents including triethylamine and dibutyltindilaurate catalyst. Molecular characterization was performed by Fourier transform infrared spectroscopy and proton nuclear magnetic resonance spectroscopy. Physical properties were observed and samples were found to be translucent yellow with a stability of more than one year. Biological properties such as blood hemolytic and antibacterial action were also noted in order to check if the samples can be used inside human body for bandage coatings. Synthesized dispersions were found to have considerable blood hemolytic activity and good antibacterial activity. After the complete characterization, dispersions were applied on polycotton blend fabric (50/50). After the treatment, fabric was analyzed for its tear strength, tensile strength pilling resistance and morphological properties by scanning electron microscopy. Fabric treated with polyurethane dispersions has decreased tear strength, enhanced tensile strength, improved pilling resistance and more intact appearance as compared to the untreated fabric.
Keywords
Introduction
Polyurethanes are interesting class of compounds with their tailor-made properties [1]. Urethane linkage is the main feature of these polymers, which is formed by the reaction of an isocyanate and polyol. Polyaddition reaction of polyols (containing –OH group) or diamines (containing active –H group) with –NCO group containing isocyanates leads to the formation of a urethane linkage
Natural material-based dispersions are in demand nowadays due to their non-toxic nature and environment friendly way of synthesis, giving rise to a wide variety of materials with desirable and biocompatible properties via combination of natural and synthetic raw materials. In the same way, blend of natural materials/polymers into the polyurethane backbone makes them more biocompatible, non-toxic, biodegradable and ideal for many biomedical applications. Vegetable oils are most commonly used for the production of such polymers at lab, pilot and industrial scale, after undergoing trans-esterification and other modifications occasionally [10]. Natural polymers such as hemicellulose, hyaluronic acid, alginate, cellulose and guar gums can be obtained from plants, whereas chondroitin chitin and chitosan are of marine origin that may also be used as raw materials for polyurethane synthesis, which are aimed to be used in biomedical field [11]. Alginates are also a class of biological polymers which are being used for biomedical applications [12,13]. Their natural source is brown algae and bacteria. Alginates are being investigated for their interesting and important applications in textile and food industries as stabilizers, thickeners, film formers and gel formers etc. As they are abundant in waterbodies, a variety of alginates are present naturally with excellent chelating ability, low cost, biodegradability and biocompatibility [14].
Antibacterial finishes for textile material are being investigated, as most of the textile materials used in hospitals may cause infection due to the pathogenic bacteria adhered to its surface. Therefore, textile hygiene is utmost important issue to be dealt with [15]. Researchers are investigating certain materials to meet the requirement of antibacterial finishes to be applied on fabric. The material to be applied should adhere firmly to the fiber so that it may not wash out on laundering [16]. PUDs are successful candidates for this application which on combining with natural materials get enhanced biocompatibility and antibacterial action. They are being synthesized using chitosan, cyclodextrin and collagen as chain extender with different compositions. Efforts are being made to enhance the antibacterial properties of these PUDs by incorporation of biocompatible materials. Therefore, in the present study, aliphatic isocyanate-based PUDs using sodium alginate (a natural antibacterial polymer) were synthesized. Dimethylol propionic acid (DMPA) was used as internal emulsifier which gave stability to the dispersion in aqueous media. These alginate-based dispersions were suggested to be good applicants for antibacterial finishing of textile.
Experimental
Materials
All the chemicals used were of analytical grade and the materials such as polyethylene glycol (PEG Mn = 300 g/mol), Isophorone diisocyanate (IPDI), sodium alginate (SA Mn = 216.12), dimethylol propionic acid (DMPA), dimethyl sulfoxide (DMSO), methyl ethyl ketone (MEK), triethyl amine (TEA), dibutyltin dilaurate (DBTDL) catalyst (95%) were purchased from Sigma Aldrich Co., USA, and used without further purification. DMPA was dried at 40℃ in oven for 2 h to remove entrapped moisture that may react with isocyanate during prepolymer formation and destroy the reaction chemistry. Phosphate buffer saline (PBS), ethylenediamine tetraacetic acid (EDTA), nutrient agar (media for bacterial growth), bacterial strains (Escherichia coli and Bacillus subtilis) were used for biological analysis. For the application of PUDs polyester-cotton or polycotton (PC) blend (50/50) was used.
Methodology
Sample codes and composition for the preparation of sodium alginate-based polyurethane dispersions (SA@PUDsAli).

IDPI: Isophorone diisocyanate; PEG: Polyethylene glycol; DMPA: Dimethylol propionic acid; SA: Sodium alginate; TEA: Triethyl amine.
Synthesis of NCO terminated polyurethane prepolymer
Reaction assembly for the synthesis of PUDs consisted of four-necked round bottom flask fitted with a mechanical stirrer, a reflux condenser, nitrogen inlet and outlet (for inert environment) and a dropping funnel (for adding reactants). The reaction assembly was kept in an oil bath for constant heating during reaction. Weighed amount of DMPA (0.95 mol) and PEG (1 mol for sample 1) were charged into the four-neck flask and heated in oil bath until it reached a temperature of 60℃ (for melting and complete mixing). After 30 min, IPDI (2 mol) (a cycloaliphatic isocyanate) was added dropwise with constant stirring along with 1–2 drops of DBTDL catalyst and the temperature of the reaction mixture was raised to 90℃. The stirring was carried out for 1 h to obtain NCO-terminated polyurethane (PU) prepolymer. Before adding the next reactant, to adjust the viscosity of prepolymer, 5–6 mL of MEK was added. In the meantime, temperature was also lowered to 55℃ and TEA (1 mol) was added to neutralize the acidic groups (–COOH) of DMPA keeping the stirring on for 45 min. At the end a neutralized PU prepolymer was obtained with NCO terminated ends (Figure 1).
Synthesis of sodium alginate-based polyurethane dispersion using cycloaliphatic isocyanate (IPDI) (SA@PUDsAli).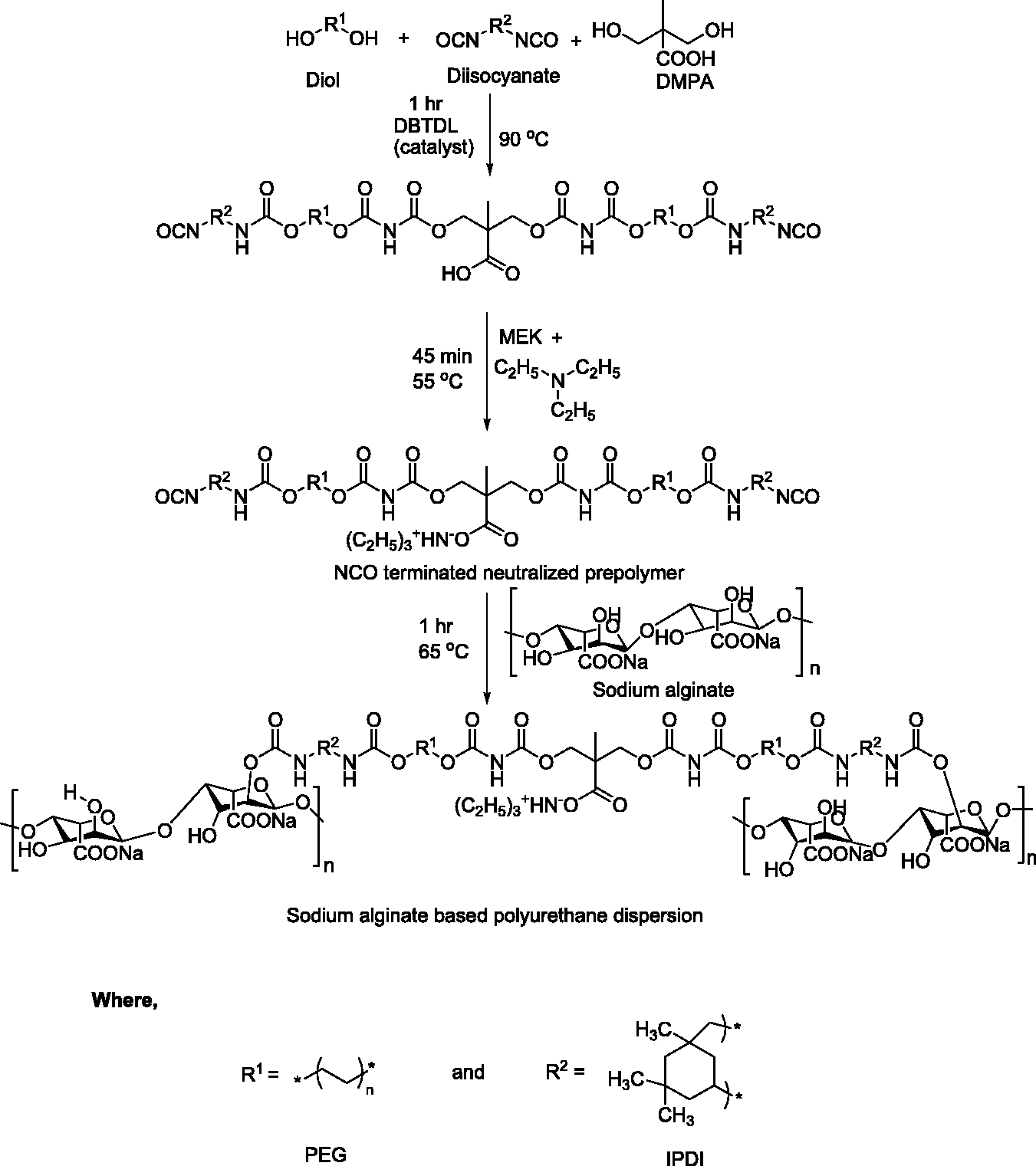
Addition of SA as chain extender and preparation of PUDs
Neutralized PU prepolymer was further reacted with SA (0.05 mol for sample 1) (chain extender). Calculated amount of SA was slowly added into the reaction flask in order to avoid the formation of gel at a temperature of 65–70℃. Stirring was continued to 2 h to ensure the completion of reaction. In the final step, measured volume of distilled water was added into the reaction mixture with constant vigorous stirring for 3 h at 25℃, in order to get a uniform PUD. Figure 1 represents the schematic pathway for the formation of PUD. The other entire samples were prepared following the same methodology by reacting the stoichiometric amounts as given in Table 1.
Physical and structural characterization
Structural characterization of prepared SA@PUDsAli was performed by Fourier transform infrared (FTIR) spectroscopy and proton nuclear magnetic resonance spectroscopy. Moreover, synthesized polyurethanes were physically examined for their appearance, stability and color [18].
Biological analysis of the synthesized PUDs
Antibacterial analysis
Two dilutions of synthesized PUDs were prepared, i.e. 5% and 10%. They were analyzed for their antibacterial activity by agar diffusion method against common human pathogenic bacteria including Bacillus subtilis (B. Subtilis) and Escherichia Coli (E. Coli) according to standard method [19]. Bacteria were grown on agar nutrient media in Petri plates which were then divided in suitable wells into which sample was placed. These plates were incubated at 37℃ for 24 h and the zone of bacterial growth inhibition was estimated in millimeters (mm) [20].
Blood hemolytic analysis
In order to investigate whether the samples are safe for applications inside human body, blood hemolytic analysis was performed using standard method American Society for Testing and Materials (ASTM) standards/ ISO 13485:2003 certified. All the samples were analyzed for their hemolytic activity using the method reported by Choi et al. [21] with a little modification. In short, fresh blood sample was taken in EDTA tube and centrifuged after adding PBS thrice to collect RBCs. After this 10 microliter of each sample was mixed with 90 microliter RBCs, centrifuged and absorbance of the supernatant was recorded in well-plate at 560 nm [21]. Blood hemolytic activity was calculated using formula as given below (equation (1))

Application of optimized sample on polycotton fabric
Polycotton blend (50/50) was used for dispersion application. Fabric was soaked in warm water (100℃) containing detergent for 30 min and then washed thoroughly with clean water in order to get rid of any impurity or dust particles. These fabrics were dried at ambient temperature before being coated with PUDs. Two dilutions (5% and 10%) of each sample were prepared and applied onto the fabric by pad dry cure method [18]. Drying of fabric after the application of PUDs was conducted at 80℃ for 30 min followed by curing for 5 min at 140℃, to achieve a uniform and firm coating on the fabric surface.
Analysis of treated polycotton fabric
Fabric sample after treatment with PUDs was analyzed by scanning electron microscopy (SEM) for morphological study and evaluated for tear strength, tensile strength and pilling resistance. Standard methods of tear strength (ASTM D- 1424/BS EN ISO 13937-2) [22], tensile strength (ASTM D-ISO 13934-2) [18] and pilling resistance (ASTM D-3514-02) [23] were used.
Results and discussions
Molecular characterization
FTIR Analysis of SA@PUDsAli
Spectrum of SA-based PUDs are shown in Figure 2 demonstrating major corresponding peaks of groups present in PUDs samples. While observing the FTIR spectra for six PUDs samples, a very weak peak (a slight bulging) of –NH stretching vibrations was observed in the range 3375.43 cm−1 to 3500 cm−1 as the urethane. Peak at 2850 cm−1 to 3000 cm−1 was attributed to the C–H stretching of –CH2 group. Absence of peak in the region 2200 to 2300 cm−1 indicated that all the –NCO groups have reacted. Band present around 1639.49–1637.56 cm−1 was an evidence of carbonyl C=O stretching of urethane (–NHCOO–) linkage [24]. Absorption peak in the range of 1462.04 cm−1 to 1458.18 cm−1 appeared due to C–H bending vibrations in –CH2 group. Stretching vibrations of C–C bond of –C(CH3)2 groups in IPDI exhibited peak in the range 1354.03 cm−1 to 1352.10 cm−1. Peak visible at 1052 cm−1 was allocated to the C–O–C group symmetric stretch in final PUDs samples (Figure 2).
FTIR spectra of PUDs samples (a) SA0.05@PUDAli (b) SA0.10@PUDAli (c) SA0.15@PUDAli (d) SA0.20@PUDAli (e) SA0.25@PUDAli. SA: sodium alginate, PUDs: polyurethane dispersions.
NMR spectroscopic analysis
Figure 3 represents the NMR spectrum of the polyurethanes synthesized using SA as chain extender. As the chemical composition of all the five compounds is similar having varying amount of SA and PEG, only one spectrum has been given to represent their structural character, assuming that concentration has no effect on signals (ppm values) in NMR spectrum. As shown by the Figure 3, proton labeled as H5-H9 (SA chain extender) exhibit signals in the range of 3.7–5.2 ppm [25]. Solvent peak (water) was observed near 4.7 ppm. Protons H1, H10 and H2 gave prominent signal at 3.5, 3.3 and 3.2 ppm, respectively, being deshielded due to the attachment of highly electronegative element (oxygen) in the immediate neighboring. The signals go on the upfield area, as the signaling group becomes less deshielded as in case of H4 (2.9 ppm) and H14 (2.1 ppm). Protons, H3, H12, H13 and H11 exhibited signals at 1.1, 1.0, 0.9 and 0.8 ppm, respectively, owing to the presence of least electron withdrawing groups in the neighborhood.
1H NMR spectrum of sodium alginate-based polyurethane dispersions using cycloaliphatic isocyanate (SA@PUDsAli).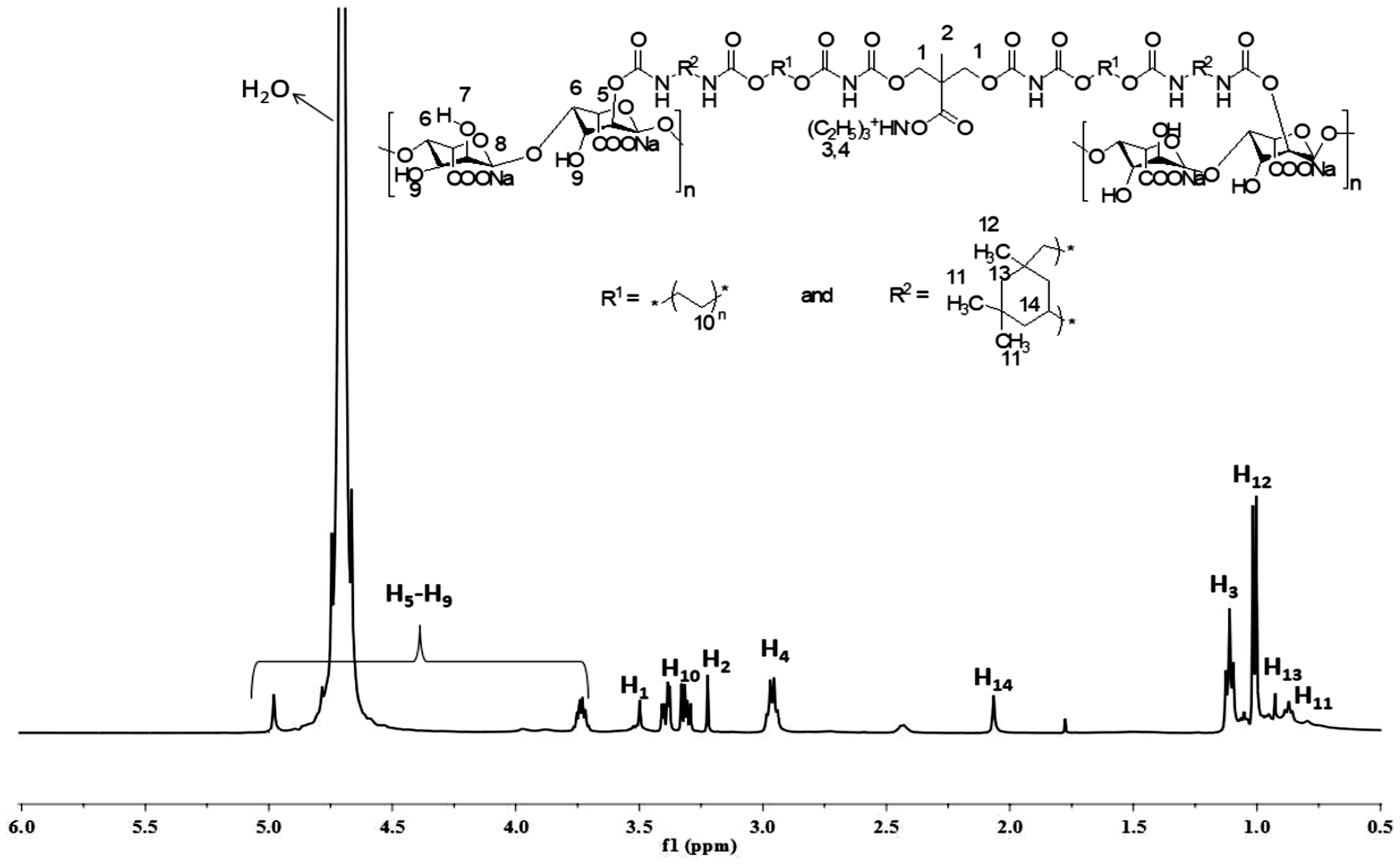
Physical characterization of SA@PUDsAli
Physical characteristics and sample codes of SA@PUDsAl

SA@PUDsAli: sodium alginate-based polyurethane dispersions using cycloaliphatic isocyanate.
While observing the appearance of PUDs, the samples were noted to be translucent and none of the samples was totally clear. There was a visual color variation of translucent yellow to a darker tinge as the SA contents changed from minimum (0.05 M) to maximum (0.25 M). It is due to the increased concentration of SA which imparted a darker tinge to the samples.
The stability investigations of the synthesized samples revealed them to be stable as they contain DMPA and TEA. DMPA is an internal emulsifier and helps in the uniform dispersion formation in aqueous media due to incorporated –COOH groups. These carboxylic groups are neutralized by TEA which further aided in the dispersion stability. PEG is also another stabilizing factor that was used as polyol, similar to the findings of in literature [26], where it was reported that using alkylene oxide polyol may enhance the shelf stability by reducing the interfacial tension of PUDs in water. In present study, PEG with molecular weigh 300 was used which resulted in small particle size in dispersion giving rise to high stability factor [27]. Studies have been reported to describe that macro diol affect the dispersion stability creating an impact of hydrophilicity [28].
Synthesized polyurethanes were kept for long time without disturbing at STP in order to check their stability period. It was noted that dispersions were stable for a maximum of 1 year (12 months). There was slight variation in stability of the samples, i.e. some of the samples were stable for more time span than 12 months but overall they followed the same trend of stability. The stability of PUDs owes to the presence of ionic units in the structural backbone imparted by carboxylic group in DMPA used as emulsifier and amino group of TEA used as neutralizing agent [7,29].
Biological analysis of synthesized SA@PUDsAli
Blood hemolytic activity of SA@PUDsAli
Blood hemolytic values of SA@PUDsAli.

SA@PUDsAli: sodium alginate-based polyurethane dispersions using cycloaliphatic isocyanate.
Antibacterial activity of SA@PUDsAli
Bacterial inhibition zone values of SA@PUDsAli.

SA@PUDsAli: sodium alginate-based polyurethane dispersions using cycloaliphatic isocyanate.
Distinguished antibacterial activity was exhibited by all the polyurethanes against both gram-positive (S. subtilis) and gram-negative bacteria (E. coli) as reported by Yagci et al. [32]. First sample SA0.05@PUDsAli gave minimum inhibitory effect with an inhibition zone of 28 mm (E. coli), 29 mm (S. subtilis) for 5% dilution and 31 mm (E. coli), 30 mm (S. subtilis) for 10% dilution. Sample SA0.10@PUDsAli had somewhat similar trend and represented inhibition up to 28 mm (E. coli), 30 mm (S. subtilis), 33 mm (E. coli) and 31 mm (S. subtilis) for 5 and 10% dilutions, respectively. The inhibition zone was almost in the similar range for the next three samples also as shown by data; in Table 4 increment in antibacterial activity at each step was due to the increasing SA contents that combat with microbes.
Mechanistically it can be said that presence of hydroxyl groups in SA might be responsible for the possible antimicrobial action. Maximum killing activity was observed when 0.25 moles SA was incorporated to the PU backbone and bacterial growth was inhibited in the vicinity of wells made for sample introduction to agar media [33]. The possible cause of antibacterial action might be due to the bacterial cell adhesion and ultimately entrapment by sticky SA incorporated PUDs, like being engulfed by it leaving no way for the passage of nutrients required for growth [34]. It is therefore inferred that the synthesized PUDs have very good antibacterial property and are promising candidates for antibacterial coating application.
Analysis of fabric after application of SA@PUDsAli
Tear strength analysis of fabric/ treated with SA@PUDsAli
Tear strength of PC fabric before and after the treatment with dispersions/ SA@PUDsAli.

SA@PUDsAli: sodium alginate-based polyurethane dispersions using cycloaliphatic isocyanate.
Tensile strength analysis of fabric/ treated with SA@PUDsAli
Tensile strength of PC fabric before and after the treatment with dispersions/ SA@PUDsAli.

SA@PUDsAli: sodium alginate-based polyurethane dispersions using cycloaliphatic isocyanate.
There are so many factors that may affect the tensile strength of the fabric including extent of crosslinking, attachment to the fabric, soft and hard segment contents in the dispersion applied on it [37]. All of these factors go on increasing as the amount of SA increases in the samples making the dispersion coating more entangled to the yarns by Van der Waals forces and hydrogen bonding formation [35]. These forces enable the formation of crosslinking of dispersion with fabric and make them stick together to bear more load and exhibit good to excellent tensile strength. On fabric treatment greater tensile strength of fabric coated with 10% dilution as compared to the 5% dilution is due to the more extent of penetration of sample in the yarns of fabric and strong film formation over the fabric surface [38].
Pilling resistance analysis of fabric/ treated with SA@PUDsAli
Pilling resistance of PC fabric before and after the treatment with dispersions/ SA@PUDsAli.

SA@PUDsAli: sodium alginate-based polyurethane dispersions using cycloaliphatic isocyanate.
Fabric after SA@PUDsAli treatment was more resistant towards pilling as compared to the control (untreated fabric). Fabric sample treated with SA0.05@PUDsAli had pilling resistance up to 4. It was further improved on increasing SA contents and reached a value of 4–5 for SA0.10@PUDsAli and SA0.15@PUDsAli applied fabric. Maximum value of pilling resistance was found to be 5 on application of samples SA0.20@PUDsAli and SA0.25@PUDsAli on PC fabric.
The enhancement in the pilling resistance on application of PUDs to the fabric was most probably due to the presence of dispersion layer on fabric surface which resisted in protruding out of yarns. Dispersion film on fabric was formed by polar interactions or hydrogen bonding resulting in more intact and smooth surface. Yarns were stuck together tightly and did not contribute in pilling by rupturing and this effect became more pronounced as the chain extender concentration, i.e. SA increased and reached a maximum pilling resistance value of 5 [35].
SEM analysis of fabric treated with SA@PUDsAli
SEM is used to view the surface morphology of sample using a focused beam of electrons. It is therefore performed in present study to check the surface features of fabric before and treatment with polyurethane dispersions under “field emission scanning electron microscopy” (FE-SEM). Figure 4 represents the SEM images of fabric samples treated with the 10% solution of each of the polyurethane dispersion synthesized using aliphatic isocyanates. Figure 4(a) shows the image of untreated fabric revealing that the surface does not have coating of any material and appear to be rough as compared to the other samples. Figure 4(b) to (f) represents the fabric sample finished with dispersion solutions and it is evident from the images that dispersion has been applied in an even manner and pressed firmly to the fabric surface making it smooth. This increased the tensile strength and reduced the chances of pilling formation as fibers now have very little tendency to protrude to make pilling.
SEM images of (a) untreated fabric and fabric samples treated with 10% solution of (b) SA0.05@PUDsAli (c) SA0.10@PUDsAli (d) SA0.15@PUDsAli (e) SA0.20@PUDsAli (f) SA0.25@PUDsAli. SA@PUDsAli: sodium alginate-based polyurethane dispersions using cycloaliphatic isocyanate.
Conclusions
Herein SA-based polyurethane dispersions with excellent antibacterial activity were synthesized. Antibacterial potential dispersions were synthesized by using varied concentration of SA from 0.05 M to 0.25 M. Other basic monomers used in the synthesis were IPDI, TDI, PEG and DMPA. After synthesis, biological testing of these materials was conducted. Blood hemolytic analysis data made it evident that the synthesized materials were not safe to apply as wound dressing coating inside human body and further investigation is needed to reach a non-hemolytic value. Strong antibacterial activity of synthesized dispersions make them potential materials to be applied in textile as antibacterial finishing. After morphological and biological characterization, dispersions were applied on polycotton blend fabric (50/50). Later on the treated fabric was tested for tear strength, tensile strength and pilling resistance to investigate the effect of dispersions over the finished fabric. Dispersion not only improved the tensile strength and pilling resistance but also affected the morphology of the fabric which was confirmed by SEM. On the other hand, a decline in tear strength of fabric was observed. While studying the physical parameters of the synthesized materials, it was observed that they were transparent yellow. Solid contents were estimated to be in the range of 37–41% and stability was found to be 1 year on average. It was therefore inferred that the synthesized PUDs were good candidate to be applied as antibacterial coating for textile materials.
Footnotes
Acknowledgement
Authors are thankful to Department of Chemistry, UAF, Pakistan and Higher Education Commission (HEC), Pakistan for providing facilities to accomplish the present project successfully.