Abstract
Converting waste fiber to high value-added carbonaceous materials has been considered as an effective and affordable route in response to the increasing volume of waste fiber in recent year. In this study, we are the first to prepare activated carbon powder derived from cashmere guard hair as a renewable waste protein fiber, using a chemical activation method at different impregnation ratios of K2CO3/cashmere guard hair char and activation temperatures ranging from 400℃ to 600℃. Characterization of the activated carbon powder was carried out by morphology study, specific surface area study, and adsorption study. Results have shown that the increase of the impregnation ratio and the activation temperature created more microporous structure in the activated carbon powder, and then increased the specific surface area of the activated carbon powder as well as the amount of methylene blue adsorbed. However, the carbon yield increases with the increase in the impregnation ratio of K2CO3/cashmere guard hair char and decreases with the increase in the activation temperature. The activated carbon powder, activated by the condition of 1:2 K2CO3/cashmere guard hair char impregnation ratio and 600℃ activation temperature, has a specific surface area of 764.86 m2g−1 and a carbon yield of 14.07 wt%. Compared to the activated carbon powder derived from fine merino wool fibers, the activated carbon powder derived from cashmere guard hair has higher carbon yield, surface area, and total pore volume, showing a superior adsorption performance.
Introduction
The demand for textile fibers is growing each year around the globe, and the life cycle of fibers has shortened, resulted that the volume of waste fiber has increased rapidly in recent years [1]. The incineration treatment of resulting waste fibers causes polluting the environment, increasing processing costs, and wasting resources. Therefore, the recycling of these fibers is becoming increasingly important. People focused on adopting reasonable methods to deal with different waste fibers.
Cashmere guard hair from the cashmere dehairing process has poor spinning property due to its coarse diameter, medullated fibers, and lack of crimp or curvature [2]. The ratio of cashmere guard hair to cashmere yield is approximately 0.7 to 1.2 for different breeds of cashmere goats. Therefore, annum yield of cashmere guard hair is about 20 kilotons. Total wool fiber yield per annum in the world is approximately 1.3 million tons. So the resource of waste animal protein fiber is vast. There are two ways to utilize waste animal protein fiber resource: physical recycling method and combined method of both physical and chemical means. The waste wool fibers were used for the preparation of adsorption reactor to remove the ethinylestradiol from domestic sewage [3] or for the preparation of usable textile after spinning and weaving [4]. The waste wool fibers were also used for the preparation of keratin [5,6], which is further used to produce keratin/PVA blended films [7], keratin/PEO blend nanofibers [8], and keratin sponge scaffolds [9]. Cashmere guard hair can be milled into fine powder and the powder was coated onto a polyester fabric in order to loading silver ion [10]. The resulting polyester fabric has the improved durability of antibacterial function against washing. Cashmere guard hair powder has superior adsorption performance towards Cr6+ compared to silk powder and wool powder [11]. Wen et al. processed merino wool into wool powder as sorbent materials for removing Co2+ ions from aqueous solution [12] and different dyes from dye solutions [13], which can be used to expand the utilization of waste wool fiber.
Wool fiber, having a high carbon content of approximately 46.53%, is a very suitable precursor for the preparation of activated carbon [14]. Activated carbon derived from natural resources, as a sorbent material, shows a good performance in the treatment of textile industry wastewater [15], and removal of NH4+–N [16] and 2, 4, 6-trichlorophenol [17] in polluted aqueous solution, and storage of industrial gas [18]. These activated carbon products are also used for storage of industrial gas [18], implement catalytic reaction [19], and prepare electrode materials [20,21]. Wool fiber-derived nitrogen-doped porous carbon prepared by molten salt carbonization method was used as electrode materials for supercapacitor [22]. The wool from worn-out clothes was converted to the high-performance hard carbons as high-performance anode for Na-ion batteries [23]. Converting wool fiber to high value-added carbonaceous materials has been considered as a novel route for disposing waste wool fiber. The preparing process of activated carbon from wool fibers affects its morphology and surface properties. Wen et al. [13] analyzed the difference of two classical methods to produce activated carbon from wool fibers: (1) chemical activation with H3PO4, ZnCl2, and K2CO3 and (2) chemical activation with K2CO3 and found that the latter method is better in its higher surface area compared to the former one. However, the available activated carbon derived from fine merino wool fibers is very expensive due to relatively high starting material cost. Moreover, until now no study on the morphology of activated carbon derived from different wools has been carried out. In this study, the preparation of activated carbon derived from cashmere guard hair was investigated in order to the maximization of resource utilization. Therefore, the focus of this research was to explore the feasibility of preparation of cashmere guard hair-based activated carbon activated by two-step chemical activation with K2CO3, which may find a new value-added application for cashmere guard hair. The effects of two important activated carbon preparation variables (impregnation ratio of K2CO3/cashmere guard hair char, activation temperature) were analyzed by investigating the activated carbon yield, its morphology structure, Brunauer–Emmett–Teller (BET) surface area, and adsorption amount of methylene blue.
Experimental
Preparation of activated carbon powder
Sample identifications and preparation conditions.

CGH-C: cashmere guard hair char; CGH-ACP: activated carbon powder derived by cashmere guard hair.
Characterization of activated carbon powder
Morphology study
Morphological features of cashmere guard hair char and activated carbon powder were observed by scanning electron microscopes (SEM, SNE-3000 M, South Korea SEC Co., Ltd and JCM-5000, Tokyo JEOL in Japan). Wen et al. [13] described the detailed operation of the JCM-5000. For the SNE-3000 M, an Mcm-100 spray coating apparatus was adopted to sputter-coat gold on SEM specimens by using a current of 38–39 mA for 3 min. The gold-coated samples were then transferred to the SEM specimen chamber and observed at an acceleration voltage of 30 kV under high vacuum.
Specific surface area study
The surface area and pore texture of the samples were analyzed by a Micromeritics Tristar II 3020 (Micromeritics, USA). The samples were dried in a vacuum oven at 200℃ for 24 h, and then degassed under nitrogen flow for at least 2 h under vacuum with a residual pressure lower than 10−3 Pa. The nitrogen sorption/desorption experiments were then carried out at 77.3 K. Based on the BET equation, the surface areas of the samples were derived by using nitrogen sorption/desorption isotherm.
Adsorption study
The absorption amount of methylene blue by activated carbon powder was used to further evaluate the activation effects. Activated carbon powder (0.1 g) derived from cashmere guard hair as absorbent was added into a 150-ml Erlenmeyer flask with 100 ml methylene blue solution (100 mg/l). The flask was shaken for 4 h at 25℃ and then kept at this temperature for 48 h. The residual of methylene blue in the filtrate after adsorption was analyzed by an ultraviolet spectrophotometer (UV-2450, Shimadzu Corp. in Japan) at a wavelength of 664 nm. The amount of methylene blue adsorbed by the activated carbon powder, 
Results and discussion
Morphology analysis
The SEM images of activated carbon powder under different impregnation ratios and activation temperatures are given in Figures 1 and 2. These SEM images display the formation of morphology evolution of cashmere guard hair chars affected by the impregnation ratio and the activation temperature. As can be seen from Figure 1, most of the cashmere guard hair char retained the fiber-shaped structure compared to the coarse cashmere guard hair, while the fiber-shaped structure among activated carbon powder derived from cashmere guard hair disappeared completely, which is different from the fine wool fiber-based activated carbon studied by Chen et al. [14]. The pore development in the cashmere guard hair char during pyrolysis and carbonization is affected significantly by the dosage of activation agent, K2CO3. Figure 1(b) shows that the cashmere guard hair chars were carbonized into compact pieces with no activated porous structure in the condition of no activated reagent. Increasing the impregnation ratio promotes the diffusion of K2CO3, KO, and CO2 molecules into the amorphous structure and thereby increases the K2CO3–C, KO–C, and CO2–C reactions, resulting in creating more pores in the activated carbon powder.
SEM images of cashmere guard hair chars and activated carbon powder under different impregnation ratio at activation temperature of 500℃: (a) cashmere guard hair chars; (b) K2CO3/wool char value of 0; (c) K2CO3/cashmere guard hair char value of 1:4; (d) K2CO3/cashmere guard hair char value of 1:2; (e) K2CO3/cashmere guard hair char value of 3:4; (f) K2CO3/cashmere guard hair char value of 4:4. SEM images of activated carbon powder under different activation temperatures (K2CO3/cashmere guard hair char impregnation ratio = 1:2): (a) 400℃; (b) 450℃; (c) 550℃; and (d) 600℃.

Figure 2(a) to (d) shows SEM images of activated carbon powder under different activation temperatures. These samples of activated carbon powder were prepared by the following conditions: 1:2 K2CO3/cashmere guard hair char impregnation ratio, 5℃/min heating rate, and 2 h activation time. The well-developed pores on the prepared activated carbon derived from the cuticle/cortex structure of wool fibers under higher activation temperature were observed. At 600℃ activation temperature, more activated carbon fragments derived from scale layer of fibers begun to form a porous structure. Higher temperature would improve the activity of the K2CO3–C reaction and K2O–C reaction as well as CO2–C reaction; therefore, a large numbers of new pores were created in the activated samples by continuous devolatilization of the chars and carbon burn off.
Yield analysis of activated carbon powder
The yield of activated carbon powder derived from cashmere guard hair, Y, is calculated based on equation (2)

In the process of carbonized and activated stage, the decomposition of activated agent, K2CO3, gives rise to K2O and CO2, and then K2O are reduced by carbon to give K and CO [25–27]. Meanwhile, the reaction of C–K2CO3 also occurred to form K, K2O, and CO [26,27]. The proposed comprehensive mechanism is as follows
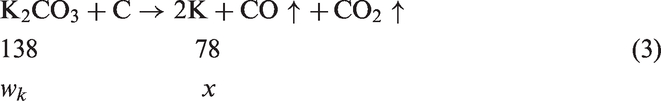
Therefore, the weight of K in the mixture of resulting activated carbon before rinsing is x = 
Substituting equation (4) into equation (2), the Y can be derived as

The effect of impregnation ratio of K2CO3/cashmere guard hair char and activation temperature on the yield of activated carbon powder is illustrated in Figure 3. In this study, the average yield of 68.64% for cashmere guard hair chars at the first stage of preparation of activated carbon powder was used to calculate the yield of activated carbon powder derived from cashmere guard hair. The curve of Figure 3 presents an S-like trend change, which can be fitted by S-shaped logistic function as below
Effect of impregnation ratio of K2CO3/cashmere guard hair char on the yield of activated carbon powder.

The correlation coefficient R2 of equation (6) is 0.97481. Rising impregnation ratio (K2CO3/cashmere guard hair char value from 0 to 1.0) leads to increasing the yield of activated carbon powder from 27.01% to 43.80%, which is consistent with Chen’s findings [14]. Figure 3 also reveals a general trend of fast increase for the yield of activated carbon powder when the impregnation ratio varies from 0.3 to 0.7. With the increase in the impregnation ratio, more carbon monoxide released by the reactions of K2CO3–C and KO–C can protect the carbon and volatile compounds from gasifying, burn off, and vaporizing, thereby resulting in increasing the yield of activated carbon powder although there is a weight loss of carbon due to increasing the dosage of K2CO3.
As shown in Figure 4, the plot of the yield against activation temperature gives a relationship of mono-exponential decay. Therefore, a mono-exponential decay model in equation (7) was employed to fit the experimental data, and the correlation coefficient R2 is 0.96879
Effect of activation temperature on the yield of activated carbon powder.

The higher activation temperature results in the lower yield. The yield of activated carbon powder decreases sharply when the activation temperature is beyond 550°X. It may be accounted for by the fact that the increase in temperature would release more volatiles as a result of intensifying the reaction rates of K2CO3–C, K2O–C, and CO2–C, causing yield decreased.
Specific surface area analysis
The specific surface area of resulting carbon powder samples under different impregnation ratios of K2CO3/cashmere guard hair char varying from 0.25 to 1 was analyzed, shown in Figure 5. The activation temperature is kept at 500℃. In this study, the BET specific surface area SBET for sample CGH-ACP-1 was not tested by the Micromeritics Tristar II 3020 due to its compact structure. Figure 5 shows that the SBET of activated carbon increases with the increase in impregnation ratio of K2CO3/cashmere guard hair char, which can be fitted by the linear model with equation (8)
Specific surface area of activated carbon powder under different impregnation ratios of K2CO3/cashmere guard hair char (activation temperature = 500℃).

Figure 6 depicts the curve of specific surface area of activated carbon powder against different activation temperatures. It can be seen that the activated carbon obtained under higher activation temperature has a relatively high SBET value, which can be described by the following equation
Specific surface area of activated carbon powder under different activation temperatures (K2CO3/cashmere guard hair char impregnation ratio = 1:2).

The correlation coefficient of this equation is greater than 0.99, indicating that the fitted model is appropriate for modeling the relationship between the SBET value and the selected activation temperature from 400℃ to 600℃. The SBET value increases sharply when the activation temperature is higher than 500℃ and the SBET of the carbon powder activated by 600℃ activation temperature reaches 764.86 m2/g. The activated carbon powder under high activation temperature has well-developed and interconnected pores validated in Figure 4, which results in higher SBET value.
The isotherms of N2 adsorption and desorption for activated carbon powders derived by cashmere guard hair via high impregnation ratios of K2CO3/cashmere guard hair char (CGH-ACP-5) and high activation temperature (CGH-ACP-8 and CGH-ACP-9) are shown in Figure 7. These curves have the similar change rule and they are Type I isotherm, showing the characteristic of microporous materials. Pore texture parameters and the yield of carbon materials prepared by different processes are given in Table 2. The 10 runs for each sample were used to determine the average value and standard deviation of the yield. The 3 runs for each sample were used to determine the average value and standard deviation of BET surface area and pore volume. It suggests that CGH-ACP-8 and CGH-ACP-9 have higher surface area and total pore volume than that of ACKC3 from 2-step chemical activation conditions, and the yield of ACKC3 is between CGH-ACP-8 and CGH-ACP-9, caused by the structure differences of cashmere guard hair and fine merino wool fiber. Compared to wool fiber-derived carbon via molten salt carbonization (WFC-MSC) prepared from molten salt carbonization method, CGH-ACP-9 has almost the same pore characteristics and the yield of carbon materials. CGH-ACP-8 has higher carbon yield, surface area, and total pore volume than that of CGH-ACP-5, indicating that the activation temperature has more significant activation effect on cashmere guard hair char than impregnation ratio of K2CO3/cashmere guard hair char. Results showed that when the impregnation ratio of K2CO3/cashmere guard hair char or the activation temperature is higher, the surface area and total pore volume of the obtained activated carbon powder are higher. However, the carbon yield will decrease when the activation temperature is higher. Taking all the factors into consideration, the impregnation ratio of 4:4 K2CO3/cashmere guard hair char and activation temperature of 550℃ are proposed to prepare activated carbon powder with higher carbon yield and surface area.
N2 adsorption/desorption isotherms of different active carbon powders. Pore characteristics and the yield of carbon materials prepared by different processes. ACKC: Activated carbon prepared by 2-steps K2CO3; WFC-MSC: Wool fiber-derived carbon via molten salt carbonization.

Adsorption of methylene blue
The adsorption of methylene blue by the activated carbon powder under different impregnation ratios of K2CO3/cashmere guard hair char ranging from 0 to 1 is presented in Figure 8. It can be seen from Figure 8 that the amount of methylene blue adsorbed by the activated carbon powder, Q (mg)/g, increases with the increase in impregnation ratio of K2CO3/cashmere guard hair char, which can be fitted by the mono-exponential model with a relatively high coefficient R2, being 0.96817, as illustrated by equation (10)
Adsorption properties of methylene blue for activated carbon powder under different impregnation ratios of K2CO3/cashmere guard hair char (activation temperature = 500℃).

The groups of activated carbon powder generated under higher impregnation ratio of K2CO3/cashmere guard hair char have higher surface area, which could be the main factor that had lead to the high Q value. The experimental Q value of CGH-ACP-2 for the K2CO3/cashmere guard hair char at impregnation ratio of 0.25 in this study is 13.75 mg/g, which is less than that of ACKC6 derived from fine merino wool fibers, while the Q value of CGH-ACP-3 for a K2CO3/cashmere guard hair char at impregnation ratio of 1:2 is 34.44 mg/g, which is higher than 28 mg/g of ACKC2 investigated by Chen et al. [14]. This phenomenon can be explained by the fact that the surface area of activated carbon powder derived from cashmere guard hair is larger than the activated carbon powder derived from fine merino wool fibers.
Figure 9 gives adsorption properties of methylene blue for activated carbon powder against different activation temperatures ranging from 400℃ to 600℃ when the impregnation ratio of K2CO3/cashmere guard hair char is 1:2, which can be fitted by the following equation with a high correlation coefficient of 0.99643
Adsorption properties of methylene blue for activated carbon powder under different activation temperatures (K2CO3/cashmere guard hair char impregnation ratio = 1:2).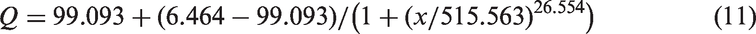

It can be observed from adsorption plots in Figure 8 that increasing activation temperature results in increasing the adsorption amount of methylene blue of activated carbon powder, and the Q value increases sharply when the activation temperature varies from 475℃ to 550℃ due to the high specific surface area, shown in Figure 6. The adsorption amount of methylene blue of CGH-ACP-9, 97.18 mg/g, is higher 2.5 times than 37.93 mg/g of ACKC3 derived from fine merino wool fibers [14] when the activation conditions are fixed as following: 1:2 K2CO3/cashmere guard hair char impregnation ratio, 600℃ activation temperature, 5℃/min heating rate, and 2 h activation time. This phenomenon also indicates that the adsorption property of activated carbon powder derived from cashmere guard hair is superior to that derived from fine merino wool fibers.
Conclusion
In summary, the cashmere guard hair was successfully converted to activated carbon powder by a chemical activation method with K2CO3. The morphology, carbon yield, and physical properties of activated carbon powder are significantly affected by the impregnation ratio of K2CO3/cashmere guard hair char and activation temperature. The fiber-shaped structure among activated carbon powder disappeared completely when the cashmere guard hair char was carbonized and activated. The high dosage of activation agent, K2CO3, and activation temperature can help to the pore development in the cashmere guard hair char during carbonization, resulting in creating more microporous structure in the activated carbon powder. The carbon yield increased with the impregnation ratio of K2CO3/cashmere guard hair char, while decreased exponentially with the increase in activation temperature from 400℃ to 600℃. The specific surface area of the activated carbon powder and its adsorption amount of methylene blue increased with the increase in impregnation ratio of K2CO3/cashmere guard hair char and activation temperature. When the impregnation ratio of K2CO3/cashmere guard hair char is 1:2 and the activation temperature is 600℃, the activated carbon powder derived from cashmere guard hair has a high specific surface area of 764.86 m2g−1 and a low carbon yield of 14.07 wt%. In order to obtain high specific surface area and yield of activated carbon powder, the impregnation ratio of 4:4 K2CO3/cashmere guard hair char and activation temperature of 550℃ are recommended.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to thank the Public Welfare Technology Application Project of Zhejiang Province (No. 2015C33039) and the Opening Project of Zhejiang Key Laboratory of Clean Dyeing and Finishing Technology (No. 1405) for their support. This research is also supported by an ARC Linkage project grant (LP120200536).
