Abstract
The aim of this study was to develop biodegradable medical nonwoven fabrics with added value, focusing on environmentally friendly natural substances for use in wound care. Propolis, a natural antibiotic, with confirmed antimicrobial and anti-inflammatory action was used. It was used in two formulations: ethanol extract of propolis and powder of native propolis. In order to obtain an environmentally friendly product, no chemical binder was used. Three modified procedures of applying propolis by immersing and scattering were used. Antimicrobial effect of the performed treatment was tested according to standard EN ISO 20645:2004 on three types of bacteria: Gram-positive Staphylococcus aureus (ATCC 6538), Gram-negative Escherichia coli (ATCC 11229) and Klebsiella pneumonia (ATCC4352). In this study, the microbial barrier properties of textiles for two bacterial endospores were investigated, with the aim of determining the microorganism mechanical permeability of textile. Fourier-transform infrared–attenuated total reflection was used to investigate the structural changes of Tencel nonwoven fabrics/propolis samples. On the basis of the results it can be concluded that the application of bee products, in this case of propolis in both formulations, on Tencel nonwoven fabrics provides satisfactory antimicrobial effects and an effective microbial barrier. Obtained results show that treatments 1 and 3 provide better antibacterial properties, indicating the further development potential of these treatments. AATCC Test Method 79, Absorbency of Textiles, was used to test absorption. The results show an increase in the absorption time of finished samples. The absorption time of finished samples can be reduced if they are stacked in a layered structure.
Keywords
Introduction
Traditional dressings, such as cotton gauze and bandages, are the most common products to treat wounds, providing a protective barrier against microorganisms. Likewise, cotton gauze has good drainage, flexibility and is easy to use in combination with drugs. However, a major problem with cotton gauze is its high tendency to dry, resulting in wound dehydration, which can inhibit the healing process. The consequence is that cotton fibres dry and stick to the wound because of exudate, which causes significant pain and trauma of the fragile epithelial tissue after removing the gauze [1,2]. Wound healing is a complicated process with haemostasis, neovascularisation and reepithelisation. Basic requirements of wound dressings are: (a) to protect the wound from external infections and to prevent bacteria from contaminating the wound; (b) to protect the wound and its environment from mechanical trauma and adverse climate (lack of moisture, uneven temperature in the wound, hypoxia, pH), which can be created by the wound dressing; (c) to absorb and to control exudate if necessary; (d) to improve patient comfort. Wound moisture is a necessary prerequisite for healing. All physical and biochemical processes in the wound are being catalysed by enzymes that are active only in moisture environment. This is also important for different growth factors, which are present in the wound immediately after injury – they cannot be active in dry environment. Except for the moisture, wound bed temperature plays a key role in wound healing. It has been demonstrated that when the temperature of the wound bed falls below the core body temperature, healing can be delayed due to lack of collagen deposition and a reduction in late-phase inflammatory cells and fibroblasts. During the dressing change, if the gauze is bonded to the wound, mechanical injury occurs, which is very painful to the patient. Often, it is necessary to put more layers to the dressing to improve absorption properties and to protect the wound from the environmental influences. Sometimes, these dressings become very bulky and noncomfortable for the patient [3]. Tencel is a branded lyocell fibre. Tencel is characterised by the ability of the fibres to absorb water, and water distribution is very uniform over the whole fibre cross section, making it less prone to the development of microorganisms. Cotton, which is most often used for wound care, keeps most of the water on the fibre surface, and is thus more susceptible to develop microorganisms, while Tencel is different and is being increasingly used in medicine and postoperative situations on the expense of cotton and other materials. Tencel does not release particles (textile dust) as opposed to cotton, which is prone to such shedding and is for this reason removed from medical applications [4]. A study by Diepgen and Schuster [5] showed a positive impact of Tencel on the care of patients with sensitive skin, even for the patients with atopic dermatitis or psoriasis. The research showed that Tencel is an acceptable fabric to be used in health care and medical environments, including postoperative ladies underwear [6].
Propolis (bee glue) is a resinous substance of bees Apis mellifera var. carnica, secreted by bud tissue and tree bark with which they defend themselves against harmful microorganisms. Bees collect it and mix with the secretion of jaw glands, modify it enzymatically so that the most important components, flavonoids, become pharmacologically active substances. Bees embalm all foreign substances in the hive. Propolis contains more than 300 components (plant resin, balsams, wax, pollen, organic acids, polyphenols, minerals, vitamins, terpenes, flavonoids, etc.) [7,8]. Propolis is one of the few natural remedies that has maintained its popularity over a long period of time. The use of propolis in wound healing has been known since ancient times. The chemical composition depends on the geographical and botanical origin. With regard to pharmacology flavonoids are the most important. They are plant pigments playing a protective role in plants. They are attributed many therapeutic effects such as antioxidant, antibacterial, antiviral, anti-inflammatory and antitumor. They are repeatedly mentioned as an aid in the treatment of various diseases because of their proven ability to ‘catch’ free radicals and the ability to inhibit specific enzymes [7,8]. It is used to treat dermatological, laryngological, gynecological disorders, neurodegenerative diseases, to heal wounds, burns and ulcers [9]. A study conducted by the Harvard School of Dental Medicine showed that propolis is effective against canker sores [10]. Sutt et al. [11] described the positive effect of alcoholic solution of propolis on the process of tissue regeneration. Scheller et al. [12,13] popularised the term ethanol extract of propolis (EEP). The study revealed that EEP solutions maintain antibacterial activity in an acidic or neutral pH range. Also, the use of EEP promotes the healing process of the damaged cartilage [12,13] and improves the ossification of bone defects [14]. It turned out that EEP supports the regeneration of dental pulp, reduces inflammatory and degenerative processes [15]. A study by Damyanliev et al. [16] revealed positive results in the propolis treatment of purulent surgical wounds. In several studies, the biological activity of propolis exhibited good results in burn wound healing [7,17,18].
Only a few studies dealt with eco-friendly natural antibacterial finish of cotton fabrics. In a 2009 study the antimicrobial activity of Aloe Vera, chitosan and curcumin on cotton, wool and rabbit hair were investigated [19]. Venkatrajah et al. [20] showed that cotton gauze with herbal extract has a positive effect on tissue regeneration. In a 2013 study by Sharaf et al. [21], propolis was used for the first time for the antibacterial finish of cotton fabric. The results showed excellent antibacterial properties.
Several studies have shown the successful use of EEP on a textile substrate, prepared as an emulsion. In a study from 2015, based on wound healing research on Wistar rats, cotton knitted fabric with newly developed emulsion based on propolis, beeswax and chitosan was used [22]. Cerempei et al. [23] in their research treated a 100% cotton knitted fabric with antimicrobial skin care emulsion in combination with EEP. The study of Vasilescu et al. [24] gives an overview of the burn wounds treatment, including special dressings used for enhancing epithelisation and for reducing the infection risk and surgical management. In a 2013 study by Abramiuc et al. [25], 100% cotton knitted fabric and propolis emulsion was used. Also, in 2013 and 2014 a study by Banupriya and Maheshwari [26,27] was published, in which the finish of a cotton fabric with antibacterial plant extracts (Michelia x Alba) is described where by good antibacterial properties compared to conventional finishing were achieved. Manuka honey, which inhibits the growth of bacteria and accelerates the healing process, is also used in wound care products [28]. The aim of the work was to create an environmentally friendly biodegradable gauze. Native propolis and biodegradable nonwoven fabric – Tencel® were used. Lyocell fibre is eco-friendly since products made from it can be recycled. Lyocell is biodegradable as it is a cellulosic fibre.
Methods and materials
Materials
Cotton gauze (100% cotton, 51.4 g/m2, warp fineness 13.8 tex, weft fineness 13.5 tex) and Tencel® nonwoven fabric (100% Lyocel, Lenzing, Austria) of mass per unit area 55.3 g/m2 (T50) and 30.8 g/m2 with a fineness of 1.7 dtex (T30) were used.
Propolis
As an antimicrobial agent propolis of original Croatian origin was used in two forms: 5.2% EEP produced by Apipharma d.o.o. (Zagreb, Croatia) and pure native propolis taken from the hive.
According to the literature [29], the characteristic constituents in temperate region propolis are flavonoids without B-ring substituents, such as chrysin, galangin, pinocembrin, pinobanksin. Caffeic acid phenethyl ester (CAPE) is a major constituent of temperate propolis with broad biological activities. Nine phenolic compounds in native propolis samples from the Croatia were detected. Among them chrysin was the most abundant (2478.5 µg/g propolis) [30].
Analysis of the EEP from Croatia showed that 72.2% of propolis samples contain galangin, 88.8% of samples contain kaempferol, naringenin and apigenin and 66.6% of samples contain caffeic acid. In all samples, pinocembrin was the dominant flavonoid. Chrysin was found in all propolis samples in a concentration ranging from 0.22% to 5.32%, Caffeic acid ranged from 0.27% to 2.67%, Chrysin was followed by naringenin, ranging from 0.22% to 2.41% [31].
Methods of material finishing
To obtain the antibacterial properties of the nonwoven fabric made of Lyocell fibres, three modified procedures of applying propolis without chemical binders and catalysts were used. The aim was to obtain an environmentally friendly and biodegradable Tencel gauze treated with a natural antibiotic. These are the results of the initial research aimed at the further development of the proposed procedures of applying natural substances to wound dressings in order to achieve the antibacterial effect.
Antibacterial treatment 1
To obtain environmentally friendly antibacterial properties of the Tencel nonwoven fabrics and cotton gauze, no binders and catalysts were used in the finishing process. The samples were boiled in an aqueous solution and NaCl for an hour (80g NaCl/1 l H2O; NaCl produced by Pag 91 d.o.o, Pag, Croatia). After boiling, the samples were rinsed in deionised cold water. Using the immersion method, the wet samples of cotton gauze and Tencel nonwoven fabrics were treated with 5.2% of EEP prepared by Apipharma d.o.o. After squeezing they were dried for 3 min at 140℃. Afterwards, the specimens were rinsed in deionised warm water (50℃) for 15 min and air-dried for 24 h.
Antibacterial treatment 2
In the second modified procedure of achieving antibacterial properties native propolis was taken from a hive. It was frozen and then pulverised (pounded in a mortar). The samples were boiled in an aqueous solution and NaCl for an hour (80g NaCl/1 l H2O). After boiling they were rinsed in deionised cold water. The scattering method was used to apply the prepared propolis powder (125 g/1 m2) to wet samples (after squeezing) of the Tencel nonwoven fabric in a uniform layer. The samples were dried at 140℃ for 3 min (Figure 1).
Schematic representation of treatment 2 (scattering method).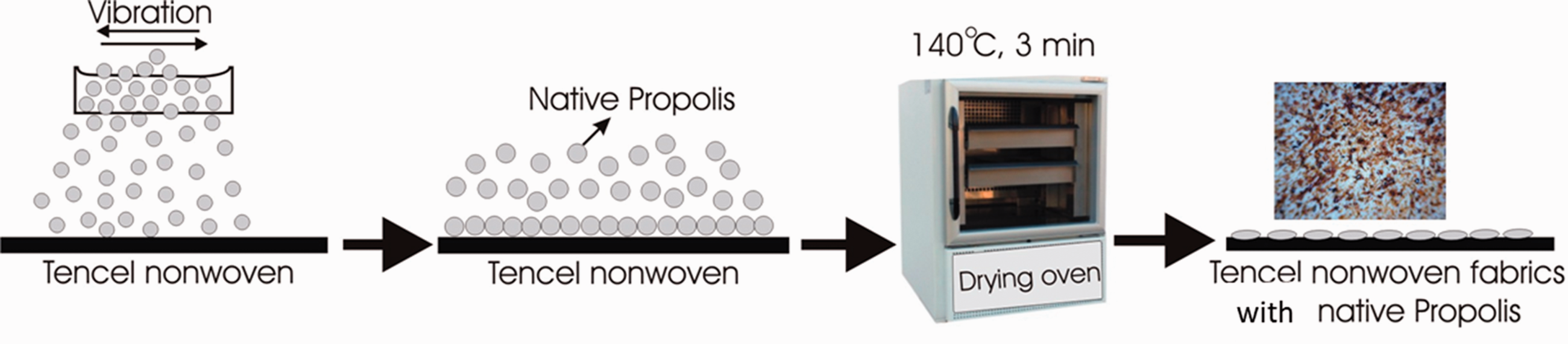
Antibacterial treatment 3
In the third modified procedure of achieving antibacterial properties native propolis was taken from a hive. It was frozen and then pulverised (pounded in a mortar). The samples were boiled in an aqueous solution and NaCl for an hour (80g NaCl/1 l H2O). After boiling they were rinsed in cold water. The scattering method was used to apply the prepared propolis powder (125 g/1 m2) to the wet samples of the Tencel nonwoven fabric in a uniform layer. The samples were dried at 140℃ for 3 min. After drying, the samples were inserted into the Meyer laboratory fusing machine (Maschinenfabrik Herbert Meyer GmbH, Rötz) at a temperature of 180℃ for 30 s and at a pressure of 2.5 bar (Figure 2).
Schematic representation of treatment 3.
Test for antibacterial effect
The test for antibacterial effect of the treated textile material was carried out in accordance with standard EN ISO 20645:2004 – Agar diffusion plate test. The effect of the antibacterial treatment was tested on three types of bacteria: Gram-positive Staphylococcus aureus (ATCC 6538) and Gram-negative Escherichia coli (ATCC 11229) and Klebsiella pneumoniae (ATCC 4352).
Qualitative method ISO 20645 is based on agar diffusion test. In qualitative testing, textile samples are laid on nutrient agar plates inoculated with the test bacteria. After incubation (24–48 h, at 37ºC, depending on the microorganism used), the plates are examined for bacterial growth directly underneath the fabric and around its edges (zone of inhibition). If the agent diffuses into the agar, the zone of inhibition becomes visible and its size provides some indication of the antimicrobial activity potency or the release-rate of the active agent. Although, the zone of inhibition does not necessarily imply that microorganisms have been killed, they might have only been prevented from growing [32].
Enterobacteriaceae such as E. coli and Klebsiella pneumoniae are the most common bacteria found in the chronic wound. We consider Escherichia coli as a representative of all enterobacteriaceae. On the contrary, the most common cause of acute wound infection is S. aureus, and therefore, we use this bacteria as a typical model bacteria.
Microbial barrier properties
Textile samples were prepared by fixing them on an O-ring device. In aseptic conditions, the spores were rubbed in equal motions on the front side of the tested samples (test field 78 cm2, 66 samples). The procedure was then repeated in the same order with the biological indicator stick reversed. A print was taken using a CT3P agar print plate (bioMe’rieux SA, Marcy I'Etoile, France), first from the back side, and then the front side, with a new plate. Agar plates were incubated for 72 h at 35 ºC, after which colony forming units (CFUs) were counted. This procedure was repeated following each treatment. This newly developed method of testing microbial barrier permeability in dry conditions involves directly rubbing the microorganisms onto the sterilised samples. Bacterial spores of the Bacillus genus Geobacillus stearothermophilus 105 (ATCC 12980, DSM 22) and Bacillus atrophaeus 106 (ATCC 49337, DSM 7264) were used as their primary purpose is to serve as a biological sterilisation control [33].
This work was specific because of the use of spores, which can survive in a dry environment, while suspensions of various types of microorganisms were used in similar evaluations. The use of a suspension moistens the fabric and the permeability is changed. With the use of spores, we were able to keep the fabric dry and test the mechanical permeability of microorganisms.
Absorbency test and thickness
Absorbency is one of several factors that influence textile processing such as fabric preparation, dyeing and the application of finishes. The suitability of a fabric for a particular use, as in the case of gauze, also depends on the ability and propensity of a fabric to take up water.
AATCC defines absorbency as the propensity of a material to take up and retain a liquid (usually water, in the pores and interstices of the material). In the AATCC test for absorbency (Method 79), a drop of water is allowed to fall from a fixed height of approximately 1.0 cm onto the surface of the sample, which is fixed firmly in an embroidery hoop, and the number of seconds required for the drop to be absorbed is noted. Absorbency is easily judged visually by the loss of specular reflection of the water drop. Time is recorded until the water drop is absorbed completely [34].
Material thickness was determined according to EN ISO 5084. The thickness of the fabric used was measured in 10 different areas of the sample at a pressure of 100 Pa (A = 5 cm2, F = 500 cN) [35].
Fourier-transform infrared spectroscopy
Fourier-transform infrared–attenuated total reflection (FTIR-ATR) was used in order to characterise the functional groups and analyse the structural changes of Tencel nonwoven fabric/propolis samples. The FTIR spectra were collected by FTIR spectrometer on a Perkin-Elmer Spectrum One with an ATR accessory in a transmittance mode at wavelength ranging from 4000 to 650 cm−1 with resolution of 4 cm−1.
Results with discussions
Results of mass per unit area and thickness change with regard to the treatment according to ISO standard.

SD: standard deviation; CV: coefficient variation (%); n = 10 ; EEP: ethanol extract of propolis.
Sample thickness was increased by applying an antibacterial agent. The highest increase was obtained after treatment 2 (scattering application). The reason for this was the application of propolis in the form of powder, which produces a dotted effect on the surface. Propolis particles on the surface increased the sample thickness, whereas in treatment 3 (scattering application) there was no dotted effect because so-called pressing was carried out after applying propolis powder and drying. In the fusing machine samples were subjected to a pressure of 2.5 bar for 30 s. After this process, propolis particles were spread over the Tencel nonwoven fabric. This procedure only slightly affected the thickness change in sample T30, while there was no thickness change in sample T50. Figure 3 shows a microscopic surface image of the Tencel nonwoven fabric to which the powder of originally Croatian native propolis was applied in two ways.
Microscopic surface images at 25 × magnification of the Tencel nonwoven fabric to which propolis was applied: (a) surface image after antibacterial treatment 2 (left – front side, right – back side); (b) surface image after antibacterial treatment 3 (left – front side, right – back side).
It is apparent that in both treatments an approximately uniform distribution of native propolis over the front side surface was obtained, while treatment 3 yielded a more uniform surface on the back side. In treatment 3 propolis spreading over the structure of the nonwoven fabric was visible, while in treatment 2 no spreading could be seen, but a protruding dotted effect was created.
An average of five readings was taken and expressed in seconds as the time required for the disappearance of a water drop placed on the fabric. The test results of the Tencel nonwoven fabrics in the absorbency test are shown in Figure 4 [34].
Absorbency test according to AATCC test method 79.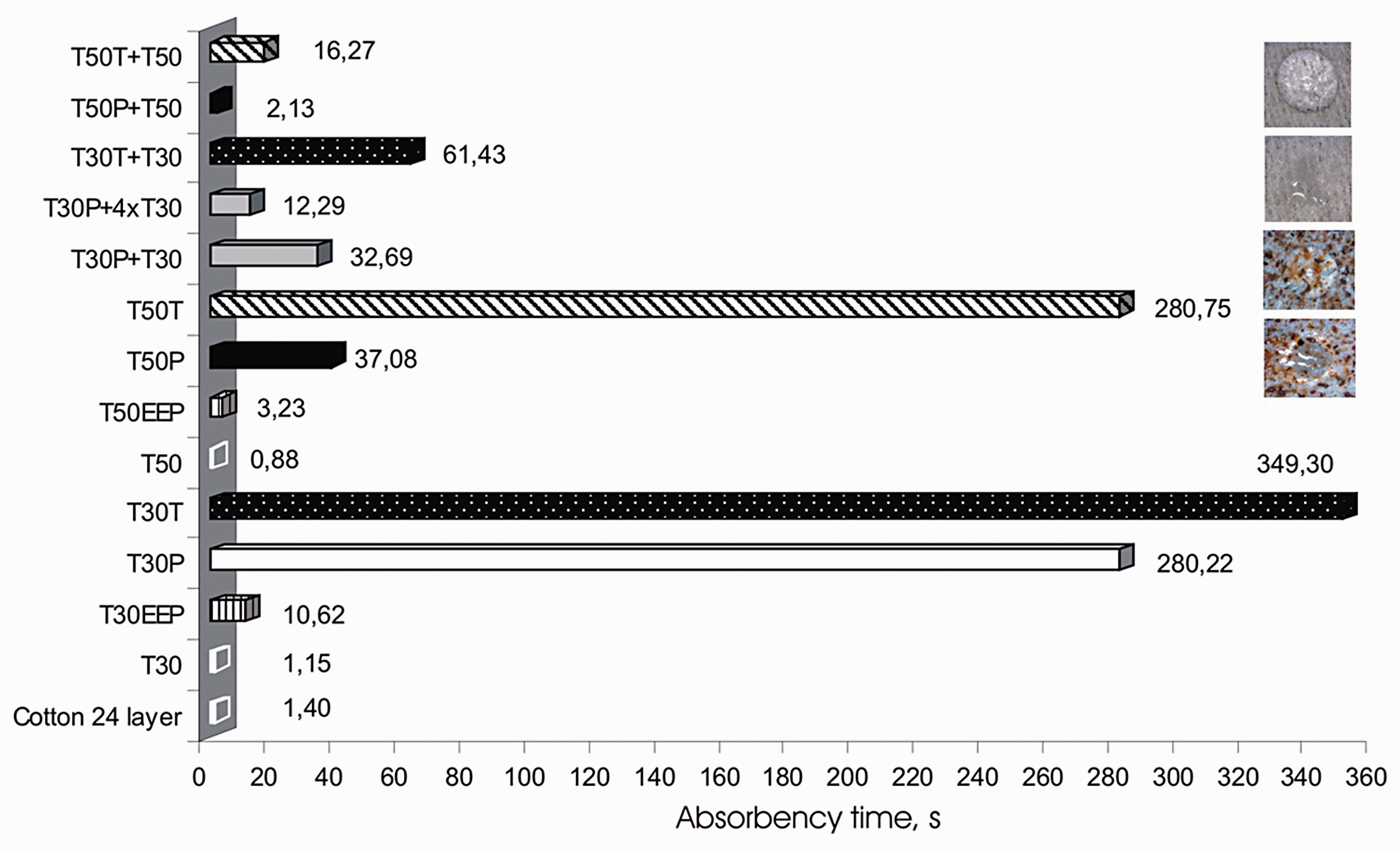
The time necessary to absorb water drops was completely determined by absorbency. Absorption time increased after the propolis finish of the textile substrate. The reason for increasing absorption time lies in the propolis composition. Propolis contains wax providing water-repellent effect. Since Tencel nonwoven fabrics are to be used for medical applications, primarily for wound care, it is essential to maintain absorbency, besides antimicrobial effects. The results show that absorption could be improved by increasing the number of layers (samples T50T + T50, T50P + T50, T30T + T30, T30P + T30, T30P + 4xT30). If the first layer of the Tencel nonwoven fabric with propolis was placed next to the wound, and then the second layer of the Tencel nonwoven fabric without finish was placed on the first layer, absorption time was reduced (Figure 5).
Layers position in material structure.
The absorption of the cotton gauze was impossible to test on one layer because water drops ran through to the other side. The results for the cotton gauze included 24 layers, which is the thickness applied in hospital treatment.
The results of the antibacterial effect of treatments are shown for Gram-positive bacteria S. aureus (ATCC 6538) and Gram-negative bacteria E. coli (ATCC 11229) and K. pneumoniae (ATCC 4352). To rate antibacterial effect, the inhibition zone and microscopic examination of the contact area under the sample were calculated. The inhibition zone size was calculated according to the following equation

The results of antibacterial effect are shown in Figure 6 and Tables 2 and 3. The result is the quality assessment of the antibacterial activity of the antibacterial agent applied to the textile substrate (EEP and native propolis).
Inhibition zone around the tested sample. Assessment results of bacteriostatic effectiveness. D is the total diameter of specimen and inhibition zone (mm); H is the inhibition zone (mm); d is the diameter of specimen (28 mm); EEP – treatment 1; P – treatment 2; T – treatment 3. EEP: ethanol extract of propolis. Assessment results of bacteriostatic effectiveness according to the standard for Gram-positive bacteria Staphylococcus aureus (ATCC 6538) [29]. EEP: ethanol extract of propolis. aThe growth of bacteria in the culture medium under the sample.


The results of the antibacterial effect of Gram-positive bacterium S. aureus (ATCC 6538) on the textile substrate are shown in Table 3. The results show that microbial resistance was achieved in samples Cotton EPP, T30EEP, T30T, T50EEP and T50T. The inhibition zone was determined, indicating a good antibacterial effect.
In the finished samples there was no inhibition zone. However, the microscopic analysis under the surface of the sample showed that the growth of Gram-negative bacteria was insignificant. In accordance with the standard EN ISO 20645: 1995 it can be concluded that the partial growth of bacteria indicates the bacteriostatic border effect. The microscopic analysis of the area under the sample surface indicated an unsatisfactory bacteriostatic effect in samples Cotton EEP, T50EEP, T50P for Gram-negative bacteria E. coli (ATCC 11229) and K. pneumoniae (ATCC 4352). In the case of samples Cotton EEP, the reason for an unsatisfactory effect of antibacterial effect could lie in testing only one layer of the cotton gauze having a distinctly loosely woven structure.
Usually, there is a difference in efficacy of different antimicrobial finishes to inhibit cell growth, which is explained by the difference in the structure of the bacterial cell wall (Gram-positive versus Gram-negative). Since the first listed bacteria (S. aureus) were Gram-positive, but E. coli and K. pneumoniae were both Gram-negative, here this explanation could be satisfactory. Different antibacterial agents have different targets for their antimicrobial action. It is well known for numerous antibiotics and some are more effective when applied to Gram-negative bacteria (third generation cephalosporins) and some are more effective to Gram-positive bacteria (penicillin) although they have similar structure (beta lactam). Only after phenotypic susceptibility testing, it is clear how some antimicrobials would work ‘in vitro.’ The only explanation for this action of propolis, to our knowledge, is a cell wall structure, but the cell wall structure itself is a very complex. Also, if we look at the difference in the cell wall structure, the Gram-positive cell wall had a thicker peptidoglycan layer (about 50 times thicker than the G-cell wall), but the Gram-negative cell wall consisted of one additional, outer membrane, which was not the case in Gram-positive bacteria [37].
Such results can be expected if the antimicrobial agent is covalently bonded to the textile substrate, which prevents its diffusion in agar, causing the zone of inhibition not to occur. There is no inhibition zone on the textile substrate with an inedible antimicrobial agent. Antimicrobial activity is possible only by direct contact where no growth under the sample occurs [38,39]. The literature review indicates that the propolis antibacterial activity is more pronounced on Gram-positive bacteria (Streptococcus and Staphylococcus) than on Gram-negative (Salmonella, E. coli and Pseudomonas). Biologically active propolis components are involved in antibacterial activity. Propolis inhibits the growth of bacteria in a way that damages their cytoplasm, cytoplasmic and cell membranes, causing partial bacteriolysis and inhibiting protein synthesis [40,41].
Tencel cellulose fibres are hygroscopic, water is absorbed into the fibre structure and that is the reason for a lower bacterial growth on Tencel [42]. Bacterial growth on Tencel can be limited completely, without any chemical additives. Bacterial growth on this material was considerably lower than in the case of synthetic fibres, where the number of bacteria grew by as much as 2000 times, as could be seen in the B. Redl study, Medical University Innsbruck [42].
As a disposable gauze was used in this study, wash fastness was unnecessary; this was the reason why no binders were used in impregnating fabric samples. By using this kind of impregnation only native products were used, resulting in environmentally friendly products. It is possible to arrive at a conclusion according to the literature that propolis provides a positive effect on the wound. Thus, in this case its release would be desirable.
Due to its composition propolis containing wax creates a necessary climate (continuous moistening, stable wound temperature) around the wound. When heated wax melts, it opens the pores of the gauze and provides greater permeability, but when it is cold, it returns to solid state, and as such it closes the pores and retains heat around the wound. The consistency of propolis depends on temperature conditions: it is solid and fragile in cold, and increasing the temperature to 30℃ becomes ductile and easy to form [43].
The first study dealing with propolis impregnation of bleached 100% cotton fabric was published by the author Sharaf. For the purposes of the study, Egyptian ethanol extract of propolis 1–5% and 5% different binders together with appropriate catalysts were used. The best results were obtained with glyoxal (3% propolis, 5% glyoxal) [21].
Test results of microbial barrier permeability for the tested semples after extreme contamination with bacterial spores Geobacillus stearothermophilus and Bacillus atrophaeus.

CFU: colony forming unit; n = 6.
The test of microbial barrier permeability to determine the microorganism mechanical permeability from the environment to a wound was conducted. The results are presented as the ratio of the number of bacterial colonies on the front side required to penetrate to the back side. Examination of the microbial barrier bandwidth for Cotton gauze samples, with and without EEP, on one layer and original package (24 layers), was carried out. Mechanical penetration of microorganisms from the environment to wound was lowest for Tencel T50T. Reason could be find in the process of applying native propolis to the sample. The propolis was applied by treatment 3 on the nonwoven fabric, where wax melting occurred. Melted wax sealed nonwoven fabric pores. The microbial barrier permeability of the Tencel samples, T30EEP, T30P and T30T, are nearly same because of low surface mass and larger surface pores of fabric. Comparing zero samples with all Tencel samples after treatment, mechanical bandwidth of microorganisms from the environment to a wound is reduced.
The FTIR provided information on chemical structures of Tencel nonwoven fabrics /propolis samples investigated in this work. The FTIR spectra of the blank cotton gauze and cotton EEP are presented in Figure 7. In both spectrum, it is possible to identify characteristic absorption bands of cotton due to cellulose macromolecule. The broad peak at a frequency of 3313 cm−1 due to the stretching vibration of the O–H bond [41]. The O–H stretching vibration sharpness (intensity) decreases in the cotton EEP due to the treatment of cotton fibres by 5.2% ethanol extract of propolis. The peak at 2896 cm−1 could be attributed to C–H stretching vibration. The presence of a band at 1605 cm−1 confirms the presence of the COO group. The peak at 1427 cm−1 was assigned to –CH2– CH2-scissoring, while the peak at 1314 cm−1 was attributed to OH bending vibration. The peaks at 1107 cm−1 and 1054 cm−1 corresponded to C–O–C stretching vibration and CH–O–CH2 stretching, respectively.
FTIR spectra of blank cotton gauze and cotton EEP.
Typical IR bands for pure propolis are shown in Figure 8, revealed bands at 3321 cm−1 (OH groups), 2924 cm−1 (aliphatic νCH2) and 2853 cm−1 (probably related to ethanol). The C = O stretching of flavonoids and lipids, found in propolis ethanolic extract is observed at 1692 cm−1. The peaks at 1630 cm−1 were the responses of the propolis component. The peaks at 1601 cm−1 and 1512 cm−1 related to an aromatic ring deformations and 1630 cm−1 to the stretching of C = O, C = C and asymmetric bending of N–H (flavonoids, amino acids) [44].
FTIR spectra of propolis.
FTIR was used to detect the functional groups and the effect of the treatment on propolis contained in Tencel nonwoven fabrics. It confirmed that propolis was in the nonwoven fabrics. As shown in Figure 9, T30 nonwoven fabric shows the absorption peaks attributed to the cotton (3321 cm−1, 1312 cm−1). The spectrum of T30EEP showed characteristic bands of propolis (e.g. 1631, 1601 and 1512 cm−1). When the Tencel nonwoven fabrics were subjected to treatment 2 (T30P sample) the peaks conected to propolis increased remarkably in intensity, possibly because most of the propolis were consumed by treatment 2 when compares to those of other two treatments.
FTIR spectra of Tencel nonwoven fabrics – T30.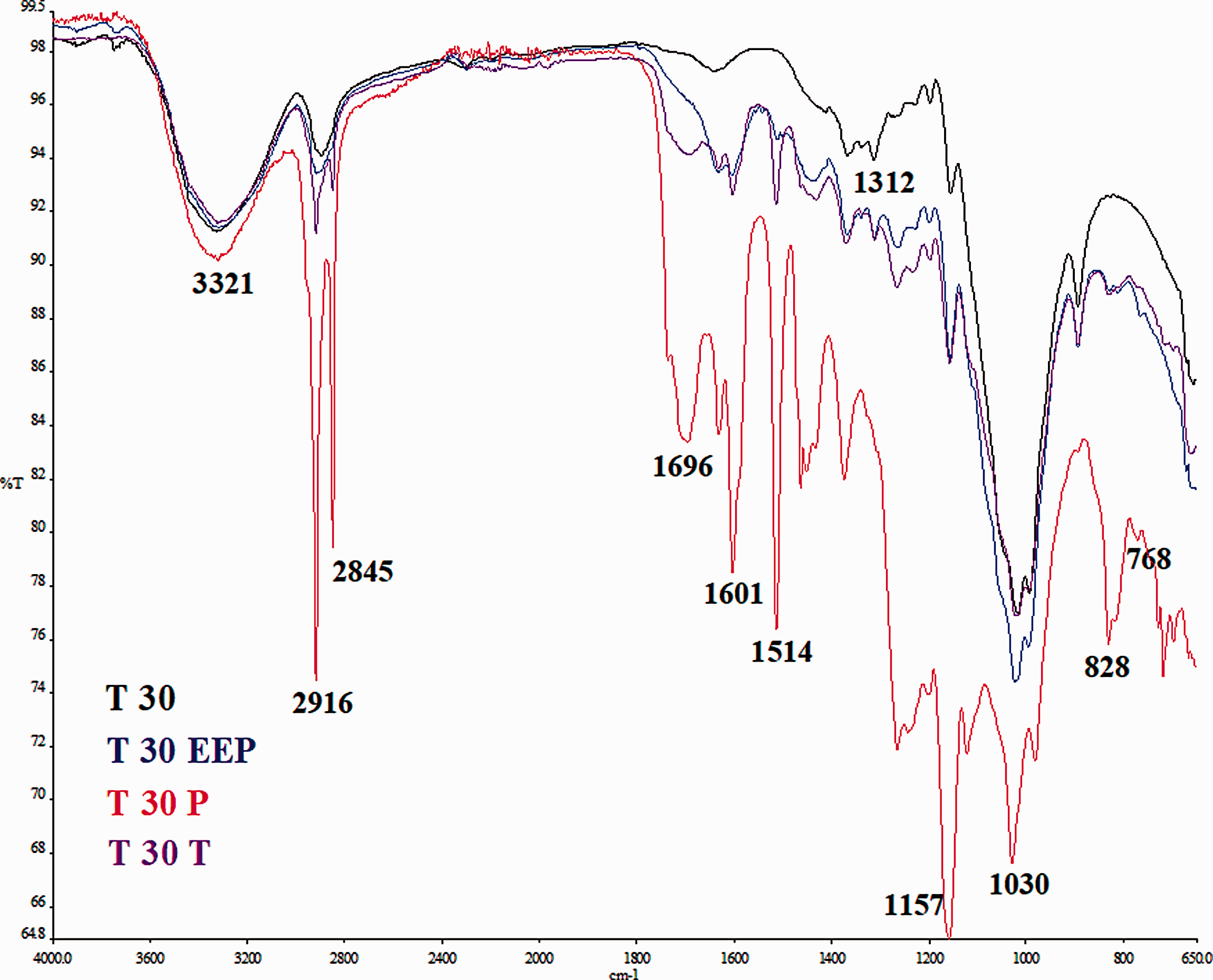
Figure 10 show the FTIR spectra in 4000–650 cm−1 region of Tencel nonwoven fabrics – T50, which were employed to characterised the structure of the nonwoven fabrics before and after treatments 1, 2 and 3. The characteristic absorption bands corresponding to propolis are observed at 1631, 1605, 1512 and from 1159 to 830 cm−1 in Tencel nonwoven fabrics T50 subjected to all three treatments. These absorption bands were in correspondence with the propolis content in the nonwoven fabrics and confirmed that propolis was in the nonwoven fabrics. The absorption band at 1601 cm−1 and 1512 cm−1 represented an aromatic ring deformation and 1631 cm−1 the stretching of C = O, C = C (flavonoids and lipids) and asymmetric bending of N–H (amino acids) [45]. The intensity of the absorption bands of propolis in the nonwoven fabrics subjected to treatment 2 (T50P) and treatment 3 (T50T) was increased, which may indicate a increase of propolis content in the nonwoven fabric after these two treatments in comparison with the untreated and T50EEP (treatment 1) samples.
FTIR spectra of Tencel nonwoven fabrics – T50.
Further research will be based on the development of proposed antimicrobial treatments aimed at the application of native propolis to obtain better anti-microbial properties on the textile substrate. When using EEP, it is assumed that immersion time of the Tencel nonwoven fabric in the EEA should be increased in order to increase antimicrobial effects. Also, when applying native propolis, the amount of applying propolis shall be optimised in order to increase absorption and antibacterial properties. We will try to extract wax from propolis in order to prevent the reduction of antimicrobial properties and to achieve higher propolis concentration. After the extraction of wax, we expect to see better absorption of propolis.
Conclusions
An innovative approach to this research is manifested in the application of environmentally friendly bee products (native propolis) in order to achieve the added value of medical textiles. The modified procedure of applying the antibacterial agent without the use of chemical binders and catalysts can be successfully used for the antibacterial treatment of cotton gauze and Tencel nonwoven fabric. Compared to the conventional methods of applying an antibacterial agent by exhaustion or impregnation, this treatment procedure is more favourable in ecological terms (reduced amount of chemicals, use of ecological antimicrobial agent – propolis and biodegradable Tencel nonwoven fabric). Applying treatments 1 and 3, better antibacterial properties were obtained, indicating the potential for further development of these treatments.
The results of testing microbial barrier permeability of medical textiles (Table 4) after extreme contamination with bacterial spores G. stearothermophilus and B. atrophaeus, showed that T50T provided better microbe barrier than other samples.
Better results for antibacterial effect and better absorption properties can be achieved by optimising the amount of applying native propolis and extending the time of immersing the textile substrate in EEP. Better absorption properties can also be achieved by combining layers of the treated textile substrate with the untreated layer of the textile substrate. From FTIR analysis it was found that T30P and T50T samples contain the largest content of propolis compared to the other investigated samples of Tencel nonwoven fabric.
Footnotes
Acknowledgement
The authors would like to express their gratitude to Dr Ksenija Varga for the samples of TENCEL® nonwoven fabric.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
