Abstract
Polyacrylonitrile nanofibrous membranes (n-PAN-FMs) with different fibre diameters were prepared using electrospinning process and modified with hydroxylamine hydrochloride. The modified n-PAN-FMs were further coordinated with Fe3+ ions to produce a series of the modified n-PAN-FMs Fe complexes, and the catalytic activity of which was investigated as the heterogeneous Fenton catalyst for the dye degradation. The effect of fibre diameter on modification and coordination of n-PAN-FMs as well as the catalytic activity of the resulting complexes was also examined. The results indicated that increasing fibre diameter proportionally enhanced the amidoximation degree of n-PAN-FMs. The modified n-PAN-FMs consisting of small fibre diameters easily coordinated with Fe3+ ions, especially at high temperature to form the complex with high Fe content at the same conditions. Besides, these complexes exhibited the catalytic performance for the dye degradation in the dark and under visible irradiation at a wide pH range. The complex with middle fibre diameter showed a stronger catalytic performance than the complexes with much bigger or smaller fibre diameter due mainly to its large specific surface area and the proper size pores among fibres as well as better water affinity.
Introduction
Recently, advanced oxidation processes (AOPs) have received a great deal of attention as the promising powerful methods to purify the organic dye-containing industrial wastewater [1–3]. Nanoscale materials are usually employed for improving the performance of the catalysts in these processes because of their larger specific surface area, which offers much enhanced and even new functions that cannot be achieved by bulk materials [4–6]. Although nano-TiO2 has become the most widely used photocatalyst for the dye degradation in the wastewater, it is difficult to recover the nanoparticle photocatalyst at the end of the reaction due to its small size and dilute concentration, thus re-contaminating the treated water [7–9]. To avoid the limitation, the photocatalyst in the form of nanofiber with the large aspect ratios (length-to-diameter) is used to enhance contact with pollutants because it is superior to particles as far as the recycling and aggregation are concerned. Electrospinning is a simple and versatile method for producing nanofibrous membranes from a variety of materials with fibre diameters ranging from several micrometers down to tens of nanometers [10–12]. Electrospun polyacrylonitrile (PAN) nanofibrous membranes have been of particular interests due to extraordinary properties such as small fibre diameters, extremely large specific surface areas, remarkably high porosity, as well as capabilities to control fibre diameter and interfibrous pore sizes among nanofibers [13]. Most importantly, PAN nanofibrous membrane generally exhibits good resistance to mineral acids, and is unaffected by common oxidising agents and sunlight [14]. However, it can be easily modified to introduce different functional groups due to its abundant nitrile groups [15–19]. Many efforts have been devoted to investigate the electrospun PAN nanofibrous membranes as the support substrates for the diverse functional materials such as titania [20], silver [21], palladium [22,23], CuS nanoparticles [24], β-cyclodextrin [25] and iron phthalocyanine [26] in different applications. On the other hand, our previous studies [14,27–29] have reported that the modified PAN conventional fibre was used as the support material for Fe3+ ions to prepare the heterogeneous Fenton catalyst for the dye degradation. Moreover, PAN nanofiber membranes can favor the catalytic activity of their Fe complex through large specific surface area and high porosity. However, there is no systematically comparative study on how the fibre diameter affects the catalytic activity of the modified PAN nanofibrous membrane Fe complex. In the present work, several PAN nanofibrous membranes with different diameters were firstly prepared using electrospinning technique, and then modified and coordinated with Fe3+ ions under the same conditions. The resulting complexes were used as the heterogeneous Fenton catalysts for the dye degradation. The effect of fibre diameter on modification, coordination of PAN nanofibrous membranes and catalytic activity of their Fe complexes was investigated for obtaining a highly active and low-cost catalyst composed of the appropriate diameter nanofibers.
Experimental
Materials and reagents
Commercial PAN yarns from Shanghai Sanli Wool Textile Company (Shanghai, China) were used in this study. Before the experiments, PAN yarns were firstly treated with an aqueous solution containing 1.0 g/L Na2CO3 and 2 g/L of soap at 80℃ for 30 min under constant stirring to remove all the impurities on the surface, and thoroughly rinsed with water and dried at room temperature. Their nitrogen, carbon and hydrogen contents were determined to be 22.97%, 66.31% and 5.51%, respectively by Vario EL III Elemental analyzer (Elementar Analysensysteme GmbH, Germany). Accordingly, the content of acrylonitrile groups in PAN yarn could be calculated to be 86.96%. N,N-dimethylformamide (DMF), NH2OH·HCl, FeCl3·6H2O and H2O2 (30%, w/w) were of analytical grade and used as received. An azo dye, Reactive Red 195 (abbr. RR 195) was used without purification, and its chemical structure was presented in our previous work [14]. Double-distilled and deionised water was used throughout the study.
Fabrication of PAN nanofibrous membranes
Specific parameters for electrospinning of n-PAN-FMs with varied Dm values.

Modification of n-PAN-FMs with NH2OH·HCl
The appropriate weights of NH2OH·HCl, NaOH and the dried n-PAN-FM were mixed with deionised water in a three-necked flask with a thermometer and agitator. The obtained mixture was heated and kept at 70℃ under stirring for a given time to prepare the amidoximated n-PAN-FM (denoted as AO-n-PAN-FM). Afterward, it was repeatedly washed with distilled water and dried in a vacuum oven and its degree of conversion from nitrile group to amidoxime group (CP%) was calculated from the mass gain [31].
Coordination of Fe3+ ions with AO-n-PAN-FMs
About 1.0 g of dried AO-n-PAN-FM was stirred in a 50 mL FeCl3 aqueous solution at 50℃ for 2 h. The resulting AO-n-PAN-FM Fe complex (denoted as Fe-AO-n-PAN-FM) was then filtered, washed with deionised water and dried under vacuum at 60℃. The residual concentration of Fe3+ ions in solution after reaction was determined using a VISTA MPX inductively coupled plasma optical emission spectrometer (ICP-OES, Varian Corp., USA) for calculating the Fe content (QFe) of the complex.
Surface and structural characterisation
Dm values of PAN nanofibrous membrane was characterised quantitatively by images analysis of scanning electron microscope (SEM) images. A field-emission scanning electron microscope (Hitachi S-4800, Japan) was used to obtain their SEM images, which were then imported into an image analyzer (Image-Pro Plus, Media Cybernetics Inc., USA) for measuring these fibre diameters and their distribution. Their specific surface area (SBET) was also determined by N2 adsorption using ASAP2010 surface area analyzer (Micromeritics, USA). In order to measure their porosities, PAN nanofibrous membranes were cut into squares (3 cm × 3 cm), the thickness of which was determined using a thickness gauge (Shanghai Chuanlu measure instruments Co., China). Its porosity (P) is expressed by the following equation [32]

Catalytic procedure and analysis
The catalytic activity of Fe-AO-n-PAN-FM was carried out in photoreaction system which consisted mainly of chamber, lamp, electromagnetic valve, relay, open Pyrex vessels and water bath, and its schematic diagram was presented in our previous study [14]. The light source inside the system used was a 400 W high-pressure mercury lamp (Foshan Osram Illumination Co., Foshan, China). A cut-off filter was used to ensure irradiation only by visible light (λ > 420 nm). The visible light intensity over the test solution surface was measured to be 8.42 mW/cm2 using an FZ-A radiometer (Beijing BNU Light and Electronic Instrumental Co., China), unless otherwise stated. About 0.2 g of Fe-AO-n-PAN-FM was immersed into 50 mL of test solution containing 0.05 mmol/L dye and 3.0 mmol/L H2O2 in the vessel. The vessel was exposed to the irradiation in photoreaction system and the temperature was kept at 25 ± 1℃. At different time intervals under irradiation, 1–2 mL of the test solution was sampled and analysed immediately using a UV-2401 Shimadzu spectrophotometer (Shimadzu Co., Japan) at the λmax of the dye used (522 nm). The decoloration percentage of the dye (D%) was calculated as follows

Results and discussion
Amidoximation of n-PAN-FMs with different fibre diameters
Five kinds of n-PAN-FMs with different Dm values were firstly prepared by optimising concentration of PAN solution, voltage and feeding rate, and Dm, P, θca and SBET of which were measured, respectively. Their amidoximation was performed with hydroxylamine hydrochloride for 60 min at 70℃ and pH=6, and the results are listed in Figure 1, Figure 2 and Table 2.
SEM images and diameter distributions of n-PAN-FMs with different Dm values (a) 156.7 nm, (b) 290.1 nm, (c) 512.2 nm, (d) 716.8 nm, (e) 842.7 nm. Effect of Dm on CP% value of n-PAN-FMs. P, θca and SBET values of n-PAN-FMs with different Dm values.


Figure 2 shows that the CP% values proportionally increase with Dm increasing, demonstrating that the larger Dm could highly enhance the amidoximation degree of n-PAN-FMs. The main reason is that as can be seen in Table 2 and Figure 1, PAN nanofibers with large diameters were randomly distributed to form many relatively bigger pores or channels between them inside the membrane, which also led to the increased surface roughness and decreased contact angle of the obtained n-PAN-FMs. This causes a better penetration and wetting of NH2OH aqueous solution into n-PAN-FMs, thus accelerating the amidoximation of their cyano groups. Although n-PAN-FMs containing thinner nanofibers have higher SBET values, small pore sizes between fibres limit contact of them with NH2OH, thus slowing down their amidoximation.
Coordination behavior of AO-n-PAN-FMs
The variation in Dm and P values of n-PAN-FMs after amidoximation.

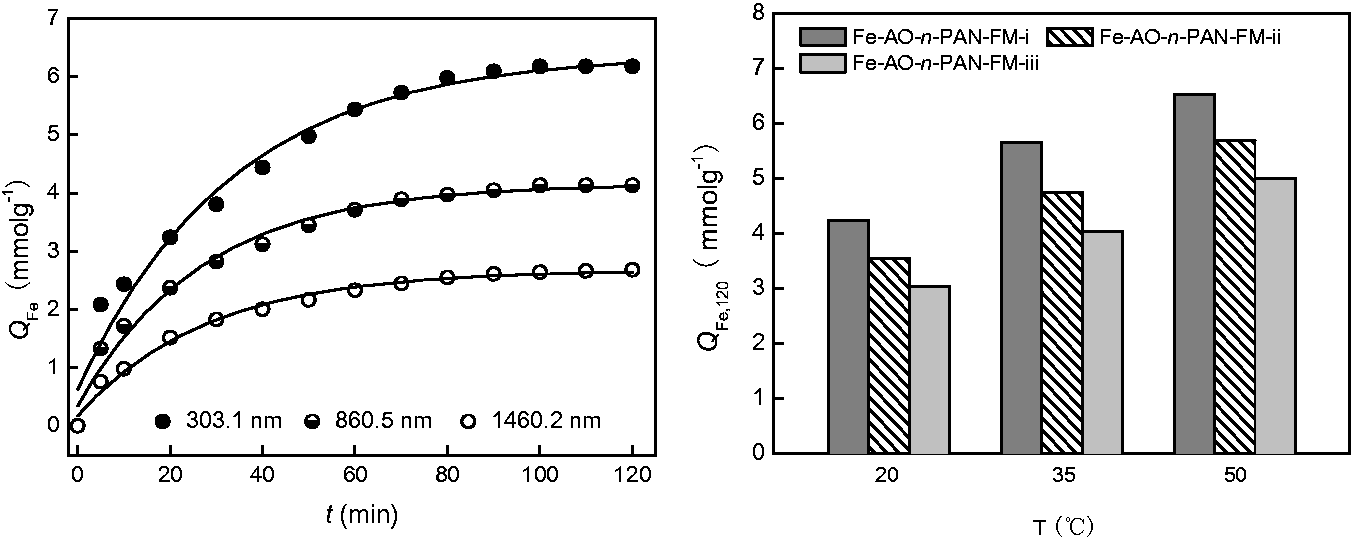
Coordination of Fe3+ ions with AO-n-PAN-FMs with different Dm values.
Figure 3 shows that the QFe values of the resulting Fe-AO-n-PAN-FMs gradually increase with prolonging reaction time. The Fe-AO-n-PAN-FM containing fibres with small diameter exhibits a higher QFe value at the given time. Moreover, elevation of reaction temperature also promotes the QFe values of the complexes. These results suggest that small fibre diameter, especially at high temperature favors the coordination of Fe3+ ions with the amidoximating groups of AO-n-PAN-FMs to form the complexes with higher QFe values. This is mainly owing to the large specific surface area and high porosity of AO-n-PAN-FMs with small fibre diameter, which enhance their affinity to Fe3+ ions through increasing the penetration and wetting of Fe3+ ions aqueous solution into nanofibrous membrane. In addition, Figure 4 and Table 4 present the FT-IR spectra of AO-n-PAN-FMs before and after Fe3+ ion coordination, as well as variation of characteristic adsorption peaks of their Fe complexes with different fibre diameters. The characteristic adsorption peaks of amidoxime group generally include 3000–3500 cm−1, 1646 cm−1, 1580 cm−1, 1244 cm−1 and 923 cm−1 [14,33]. These peaks are found to be less intensive and broader after Fe3+ ion coordination. It should be noticed from Table 3 that they shift to short or long position with increasing fibre diameter, respectively.
FT-IR spectra of (a) n-PAN-FM, (b) AO-n-PAN-FM, (c) Fe-AO-n-PAN-FM-i, (d) Fe-AO-n-PAN-FM-ii and (e) Fe-AO-n-PAN-FM-iii. The characteristic adsorption bands of amidoxime group and their attributions.
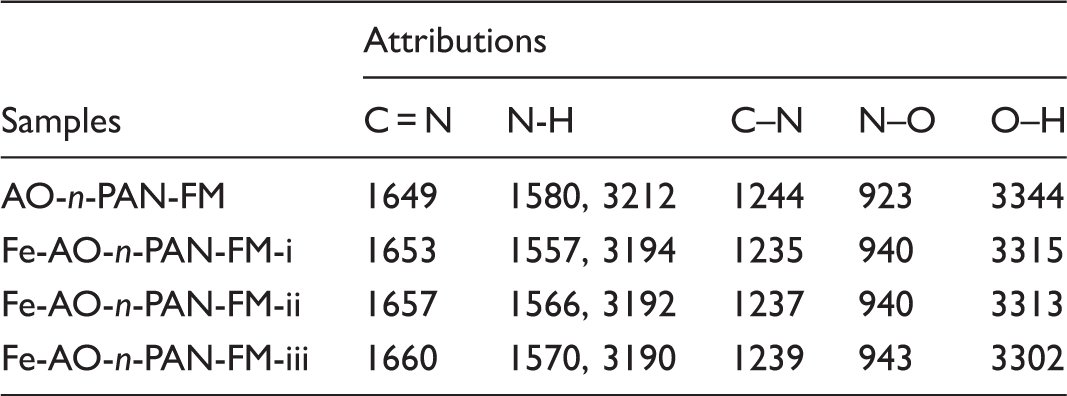
To further understand the coordination of Fe3+ ions with AO-n-PAN-FM, XPS and XRD spectra of AO-n-PAN-FM and its Fe complex are given in Figure 5(a) and (b). It can be seen that there are three peaks including C 1s, N 1s and O 1s in the XPS spectrum of AO-n-PAN-FM. New peaks at 710.8 and 723.9 eV corresponding to Fe 2p3/2 and Fe 2p1/2 appear in the XPS spectrum of Fe-AO-n-PAN-FM, implying the coordination of Fe3+ ions with AO-n-PAN-FM. Additionally, the two main characteristic peaks at 16.7° and 25.5° of n-PAN-FM were found to be much less intensive after the amidoximation in their XRD spectrum, which is similar to the result reported in our previous work [14]. This may be attributed to a possible reduction in the crystallinity and crystal size of PAN fibre during the reaction [33]. Both peaks almost disappear in the XRD spectrum of Fe-AO-n-PAN-FM, demonstrating that the coordination of Fe3+ ions further enhanced decrystallisation of n-PAN-FM.
XPS (a) and XRD spectra (b) of AO-n-PAN-FM and its complex.
Effect of fibre diameter on catalytic activity
Characteristic parameters of Fe-AO-n-PAN-FMs with different diameters.


Influence of fibre diameter on decomposition of RR 195 at different conditions (a) D% values during reaction, (b) D45% at different irradiation, (c) D45% at different pH, (d) D15% with different QFe level.
Figure 6(a) shows that D% value in case of only RR 195 within 30 min was less than 3%, indicating that little degradation of RR 195 occurred. D% value without Fe-AO-n-PAN-FMs increased slowly, which may be due to a limited oxidation of dye with H2O2 under light irradiation. While Fe-AO-n-PAN-FMs being used, D% values increased remarkably with the prolonging reaction time, and D% values within 30 min were higher than 80%, suggesting that Fe-AO-n-PAN-FMs could significantly enhance the oxidative decomposition of the dye with H2O2 in water under visible irradiation. It should be noticed in Figure 6(b) that highest D45% value was obtained in the case of Fe-AO-n-PAN-FM-ii. The dye degradation was conducted in the dark or under different visible irradiations (IA=8.58 mW/cm2, IB=10.66 mW/cm2 and IC=16.51 mW/cm2). Fe-AO-n-PAN-FMs exhibit the catalytic performance in the dark, and increasing light irradiation causes a gradual increment in their catalytic performance, thus accelerating the dye degradation. This is because they are active in the UV and visible regions [34]. When Fe-AO-n-PAN-FM is used as a catalyst, it reacts with H2O2 to generate hydroxyl radicals (•OH) by the reduction of Fe3+ ions to Fe2+ ions loaded on its surface in the dark or under visible irradiation. These hydroxyl radicals with higher oxidative power could degrade the dye molecules. A possible reaction process is described by Scheme 1.
Catalytic mechanism of Fe-AO-n-PAN-FM for degradation of dye molecule.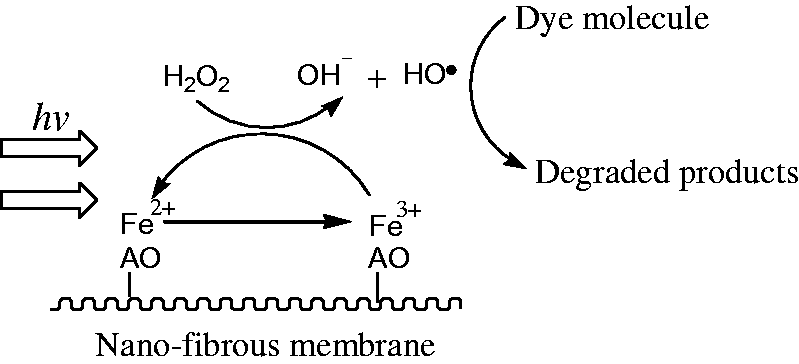
The three complexes could promote the dye degradation in a board pH range of 3–9 shown in Figure 6(c). Better degradation of the dye can be achieved in an acidic environment, especially at pH 6, which is attributed mainly to a faster decomposition of H2O2 into hydroxyl radicals and greater adsorption of the dye on the catalyst [35]. However, their catalytic activity dramatically decline in the alkaline pH level. Besides, it was clear from Figure 6(d) the dye degradation is faster when the complexes with high QFe values were used as the catalysts, which proposes that the catalytic activity Fe-AO-n-PAN-FMs is greatly dependent on their QFe value. The main reason is that high QFe value of the complex may cause more active sites on their surface, which can enhance the H2O2 decomposition and produce more hydroxyl in solution by the heterogeneous photo-Fenton reaction [36], which is responsible for the dye degradation.
Comparing D%, especially D45% values of the three complexes at the same condition in Figure 6, the catalytic activity is the highest for Fe-AO-n-PAN-FM-ii containing fibres with middle diameter. This is ascribed to the difference in the coordination structure and surface performance between these complexes. As seen from Table 5, P (%) and SBET values of the complexes decrease with increasing fibre diameter. The similar results are reported in the preparation of PAN and PET non-woven fabrics [37,38]. Higher P (%) and SBET values may be beneficial to the adsorption of the dye molecules from the solution into nanofibrous membranes, which promote the dye degradation since although a good heterogeneous catalyst should efficiently produce oxidising species, it is necessary to absorb the reactant molecules strongly enough for them to react [35]. On the other hand, non-woven fabrics can be viewed as a system of capillaries, and the walls of which are the fibres comprising the fabric. It is known that wetting requires penetration of liquid and displacement of the air occupying these capillaries. According to Laplace equation [39], the movement of liquid into capillaries is determined by the capillary pressure, which is highly dependent on diameter of capillaries inside fabric. White and Chen [40,41] reported that average diameter of capillaries is directly proportional to diameter of fibres comprising the fabric. Hence, in this work, it is believed that PAN nanofibers with small diameter showed a higher capillary pressure, thus leading to a rapid wetting and penetration of dye aqueous solution into the PAN nanofiber membrane, being responsible for the contact of membrane and dye molecules. However, small diameter of capillaries may slow down the movement rate of aqueous solution into the capillaries inside membrane [41], so that more time was needed to complete the wetting and penetration, which limited the contact between dye molecule and membrane. Accordingly, Fe-AO-n-PAN-FM containing small diameter of nanofibers hinder the penetration of dye solution to some extent, thus reducing the dye degradation. Consequently, lowest θca was observed for Fe-AO-n-PAN-FM-ii due to its middle fibre diameter and proper size of pores among fibres, which significantly enhances the dye degradation through better wetting of the membrane to increase the dye molecule adsorption on its interface. It is reasonable that Fe-AO-n-PAN-FM-ii containing fibres with middle diameter (612.3 nm) generated higher catalytic performance than the complexes with much bigger or smaller fibre diameter.
Conclusions
PAN nanofibrous membranes with different diameters were modified and coordinated with Fe3+ ions to prepare a series of heterogeneous Fenton catalysts for the dye degradation. Increasing the fibre diameter proportionally enhances their amidoximation. The obtained AO-n-PAN-FM consisting of fibre with small diameters easily coordinates with Fe3+ ions, especially at high temperature to form the complex with high QFe value at the same conditions. The resulting Fe-AO-n-PAN-FMs exhibit the catalytic performance in the dark, and which is significantly increased through introduction of visible irradiation or higher QFe value. Their catalytic activities are observed to be better in an acidic environment, especially at pH 6, however reduced in the alkaline pH level. The complex containing fibres with middle diameter shows a stronger catalytic performance than the complexes containing fibres with much bigger or smaller diameter. This is due mainly to the large specific surface area and the proper size pores among fibres, thus favouring the catalytic performance of the complex through the enhanced dye molecule adsorption on its surface.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Tianjin Municipal Science and Technology Committee for a Research Program of Application Foundation and Advanced Technology (11JCZDJ24600). This research was also supported in part by Innovation & Pioneering Talents Plan of Jiangsu Province (2015-340), National Key Technology Support Program (2015BAE01B03), Innovation Fund for Technology of China (14C26211200298), and Shaoxing Municipal Public-benefit Project (2014B70006).
