Abstract
The effect of alkali treatment on thermal, electrical and compressive behaviour of jute composite has been studied. The plain woven jute fabrics were manufactured using handloom. The manufactured fabrics were treated with alkali at the optimized treatment conditions of 5% NaOH for 4 h at 30℃ made into a composite of [0°]4 lay-up sequence by means of compression moulding technique using vinyl ester resin. The improvement in the crystallization of the alkali-treated jute fabric was characterized using differential scanning calorimetry and X-ray diffraction technique. The composites were characterized for compression strength, thermal conductivity and electrical resistance properties. It is observed from the results that the alkali-treated jute composites showed increased compression strength, electrical conductivity and thermal conductivity of the composites. This may be due to the better adhesion of the fabric–matrix interface with the removal of lignin and hemicelluloses that impart hydrophobicity on the fabric.
Introduction
The demand for the new advanced materials and environmental awareness is increasing. This gives an edge for the development of natural fibre reinforced composites used for wide range of applications. It can be used in advanced materials in cutting-edges ranges such as automotive, renewable energy, construction, aerospace and medical applications. The other great advantage of using the natural fibres as reinforcement is its easy availability, low cost, good electrical insulation, less hazardous to health, etc. But the major drawback with respect to the natural fibres is its low binding nature and water absorption that makes it a serious issue in using it for outdoor applications. The hydrophobic property of the composites can be improved by physical treatments such as cold plasma treatment, corona treatment and chemical treatment of the fibre with maleic anhydride, organosilanes, isocyanates, sodium hydroxide, permanganate and peroxide. Alkali treatment is one of the chemical method used to improve the adhesion between the natural fibre and matrix in a composite. The alkali treatment leads to the fibrillation by breaking down the fibre bundle into smaller fibres thereby develops a rough topography for better adhesion between the fibre and matrix. With the improved fibre–matrix adhesion, the mechanical properties also get increased [1]. The treatment of jute with alkali causes the disruption of the hydrogen bonding and removes a certain amount of lignin, wax, oils and hemicelluloses from the structure that improves the adhesion of the composite [2,3].
It is highly necessary to have better understanding on the thermal and mechanical behaviour in engineering an optimum design for a structure. The thermal conductivity of the composite depends on the nature of fibre and matrix, fibre–matrix interaction, volume fractions, thickness, orientation, interfacial bonding between fibre and matrix, etc [4]. Polymers are mainly used as insulating material in the product development. A lot of research has been carried out to improve the thermal conductivity of the polymer that helps in the higher dissipation of heat through the material. Due to the improvement in conductivity, the dielectric failures have been avoided and thereby the overheating of the product has been reduced. It is equally important to improve the electrical properties of the composite. The composite developed with outstanding dielectric properties have been used for engineering insulants in electrical and electronic products. The fibre reinforced composites can be used not only as insulators but also as an effective mechanical support for electric field carrying conductors. The jute fabric shows higher dielectric constant when compared to the glass fibre because of its hydrophilic nature of the composite [5]. The electrical resistivity has been improved by treating the fabric with various chemical treatments such as alkali, steric acid, peroxide, acetylation, permagnate, etc. The surface treatment of the fabric decreases the dielectric strength of the composite mainly due to the reduction in the number of hydroxyl groups in the structure [6].
The relative weakness in compression is the major limiting factor with respect to the application of any composite material. Hence, it is highly required to predict the compressive strength of the composite to develop a design for efficient and safe operation of the component [7]. It was also studied that the jute fabric has better compressive strength in addition to that of the tensile strength mainly due to its damping behaviour [8]. Considering the above discussion, the current study focusses, the study of compressive, thermal and electrical behaviour of the alkali-treated jute/vinylester resin composites.
Materials and methods
Jute fabric
Jute yarn (count – 8 lbs) procured from Shri Hanuman Traders Karur and Vinylester resin procured from Vasavibala resins, Chennai were used for the production of laminates. The procured yarn was made into a plain woven fabric of 16 EPI and 13 PPI with a GSM of 430. The fabric was then made into a composite with vinylester resin by compression moulding technique. The composite was made of four layers [0°]4 with a thickness of 4 mm and fibre volume of 41.63%. The laboratory reagents of sodium hydroxide and acetic acid were used for the chemical modification process.
Composite preparation
The composites used in this study were the unidirectional composites produced using the compression moulding technique. The fabric was preheated in an oven at 100℃ for 2.5 h to remove the moisture in the fabric. The vinylester resin, catalyst and accelerator were mixed in a weight ratio of 1:0.02:0.02, followed by a vacuum treatment for 3 to 5 min in order to remove the entrapped air bubbles. Each layer of fabric is then impregnated with the matrix material and stacked evenly on 30 cm × 30 cm frame of the compression moulding machine. The composites were made by pressing the material between the hot plates of a compression moulding press. It was then cured at room temperature for 24 h. Composites of stacking configurations [0°]4 were prepared and lay-up sequence is shown in Figure 1(a) and (b). The thickness of the composite was maintained at 4 mm.
(a) Lay-up configuration of [0°]4 structure composite and (b) lay-up sequence of [0°]4 composite.
Alkali treatment
Coded levels of three levels and three variables.
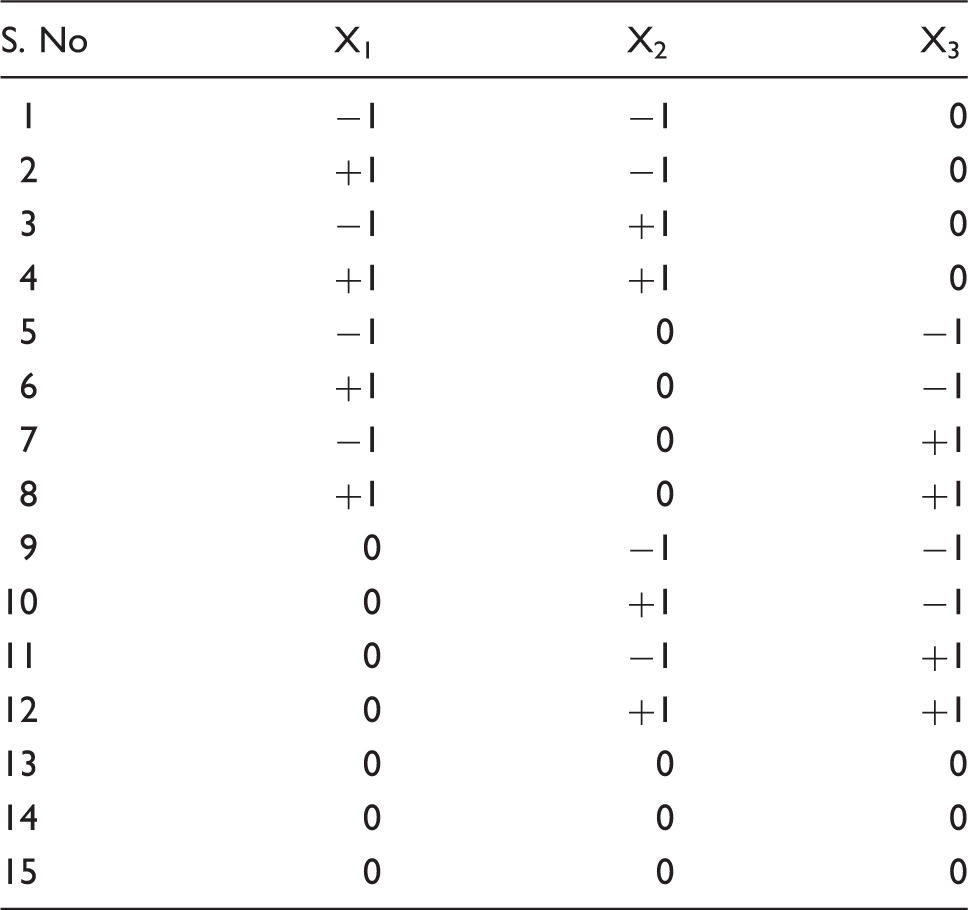
Actual values corresponded to coded values.

Dependent variable: Weight loss percentage, water absorbency rate. Independent variable: Time, temperature and concentration.
The alkali treatment was carried out by dipping the jute fabrics in 4%, 5% and 6% levels of sodium hydroxide (NaOH) solution for 2, 4 and 6 h at 20, 30 and 40℃ temperature. The treated fabrics were washed with distilled water including a few drops of acetic acid. It is further washed with fresh distilled water till the residual sodium hydroxide was removed from the fabric. The washed fabrics were oven dried at 60℃ for 24 h. The optimized condition of alkali treatment is found to be 5%, 4 h and 30℃ is used for the development of composites and they are characterised [9].
Material characterisation
Determination of lignin content
Lignin content of the jute fabric before and after plasma treatment was determined using TAPPI (Technical Association of Paper and Pulp Industries, New York) T-222-OS-74standard. Two grams of oven dried fibre sample was placed in a flask and 15 ml of 72% sulphuric acid was added. The mixture was stirred frequently for 2.5 h at 25℃ and 200 ml of distilled water were added to the mixture. Then the mixture was boiled for next 2 h and cooled. After 24 h, the lignin was transferred to the crucible and washed with hot water several times until it becomes acid free. The collected lignin was dried at 105℃ and cooled down in desiccators and weighed. The drying and weighing were repeated until constant weight to get the lignin weight.
Determination of hemicellulose content
The hemicellulose content of the jute fabric was determined using the TAPPI standard (2010).
Determination of holocellulose content
Three grams of oven dried fibre was weighed and placed in a flask. Then, 160 ml of distilled water, 0.5 ml of glacial acetic acid and 1.5 g of sodium chloride were added successively. The flask was placed in water bath and heated up to 75℃ for an hour. Then additional 0.5 ml of glacial acetic acid and 1.5 g of sodium chloride were added. The additions of acetic acid and sodium chloride were repeated 2 times hourly. The flask was placed in an ice bath and cooled down below 10℃. The holocellulose was filtered and washed with acetone, ethanol and water, respectively. Finally, the sample was dried in oven at 105℃ before weighed.
Determination of α-cellulose content
Two grams of oven dried holocellulose was placed in a beaker and 10 ml of sodium hydroxide solution (17.5%) was added. The fibres were stirred up by glass rod, so that they could be soaked with sodium hydroxide solution vigorously. Then sodium hydroxide solution was added to the mixture periodically (once every 5 min) for half an hour and the mixture temperature was kept at 20℃. About 33 ml of distilled water was added in the beaker and kept it for an hour. The holocellulose residue was filtered and transferred to the crucible and washed with 100 ml of sodium hydroxide (8.3%), 200 ml of distilled water, 15 ml of acetic acid (10%) and again water successively. The crucible with α-celluloses was dried and weighed.
Determination of hemicellulose content
The content of hemicelluloses of jute was calculated from formula below:
Hemicelluloses = Holocellulose content − α-cellulose content
X-ray diffraction (XRD) studies
XRD analyses of materials were performed at room temperature by using a Panalytical, Netherland & X’pert3 Powder (operating at 45 kV and 30 mA) with a Cu monochromatic radiation. The scans were achieved within a range from −40° to 220° (2θ) with a scanning step of 0.039°.
The crystallinity index (CI; Ic) was determined by using the equation below:

Atomic force microscopy (AFM)
AFM (NTMDT, NTEGRA Aura, and Russia) is used to measure the surface topography of the jute fabric. The imaging of jute was obtained using the multimode scanning with silicon Nitride cantilever under atmospheric conditions. Experiments were performed with a resonant frequency of 47–150 kHz. Root mean squares (RMS) roughness data were obtained by analysing topography images of specimens. Images of the jute fabrics were obtained at five different locations and the average RMS roughness was determined.
Pull out test
For characterization of the pull out of the fabric, the fabric was embedded in the matrix and the loading of the free ends. The loading of the sample leads to the stretching of the sample along the axis until fibre breakage in pull out tests. The force displacement curve has been recorded. The specimen is 4-mm thick and 25.4-mm wide with the embedded length of the 12.7 mm. Pullout tests were carried out using an Instron testing machine at a crosshead rate of 5 mm/min.
Compression strength
Compression behaviour of samples was carried out in a Zwick Roell Tensile Tester at a crosshead speed of 1.3 mm/min (ASTM D 695). The sample of 12.7 mm × 25.4 mm is placed between compressive plates parallel to the surface. The samples are then compressed at a uniform rate. The maximum load is recorded and compressive strength is thus determined.
Electrical resistivity analysis
Test of five samples is carried out on treated and untreated jute woven vinylester resin composites to determine the volume and surface resistivity (DIN 54345-1). The test is carried out in Fischer elektronik, Germany & Milli-TO 3, TE 50 with resistivity measuring range: 1 × 105 Ω resolution to 1.6 × 1015 Ω. The samples of 90 mm × 90 mm at equilibrium with standard atmospheric conditions are measured for electrical resistance between parallel electrodes by means of an electrical resistance meter.
Thermal conductivity analysis
Test of five samples is carried out on treated and untreated jute woven vinylester resin composites to determine the thermal conductivity (ASTM D 7340). The test is carried out in Lee’s Disc Apparatus by sandwiching the constant heat source between two identical metal cylinders mounted with their axes vertically. The specimen is placed on the top of the mounted upper cylinder and a third identical metal cylinder is placed over the specimen in such a way that all the cylinders and specimen are concentrically aligned. The heat is applied and the temperatures of the blocks are allowed to reach equilibrium. The thermal conductivity of the test specimen is then determined from the steady-state temperatures of the blocks, surface area of the blocks and specimen, and thickness of the specimen.
Differential scanning calorimetry (DSC) analysis
DSC measurement is performed using a simultaneous TG-DTA/DSC (NETZCH Germany & STA 449 F3 Jupiter) thermal analyser; a sample weight of 15–20 mg in an aluminium crucible and maximum height and diameter of 3 mm × 3 mm. It allows the measurement of mass changes and thermal effects between room temperature to 1550℃.
Results and discussions
Analysis of alkali-treated jute fabric
Characterisation of jute for chemical constituents
Chemical characterization for lignin and hemicellulose content of untreated and alkali-treated jute fabrics.

DSC analysis
DSC analysis was carried out for untreated and alkali-treated jute fabrics for determining its thermal behaviour. The thermal behaviour of the fabric is determined by the release and absorption of thermal energy by chemical reaction of jute constituents during heating.
Figure 2 shows the DSC of the treated and untreated jute fabric. An endothermic peak is observed in the range of 90–105℃ in both untreated and alkali-treated jute fabrics. This peak is mainly due to the evaporation of water absorbed in the jute fibres [11]. The broader hump or small peak observed at 296.3℃ in untreated and 296.7℃ in treated jute fabric is mainly due to the thermal degradation of hemicelluloses fraction present in jute. The endothermic peak at 363.2℃ is obtained for untreated jute fabric, whereas it has been shifted as exothermic peak at a temperature of 348℃ for treated fabrics. This shift of the nature of the peak may be because of the removal of non-cellulosic components such as hemicelluloses, pectin, etc., and decomposition of α-cellulose and lignin content during alkali treatment, leading to the cleavage of chemical linkage between the constituents [7,12]. The reduction in temperature requirement of 348℃ for treated fibres when compared to untreated of 363.2℃ is mainly due to the degradation of lignin and other cellulosic components already from the fibres by alkali treatment. The region between 150 and 250℃ shows no peak, which indicates the thermal stability of the jute fabric in the corresponding temperature range. The peak above 200℃ is associated with the destruction of hemicelluloses, cellulose and lignin content. From the analysis, it is evident that the alkali treatment removes the major constituents of hemicelluloses, lignin and cellulosic matters leading to the improvement in the hydrophobic nature of the fabric.
DSC analysis of untreated and alkali-treated jute fabrics.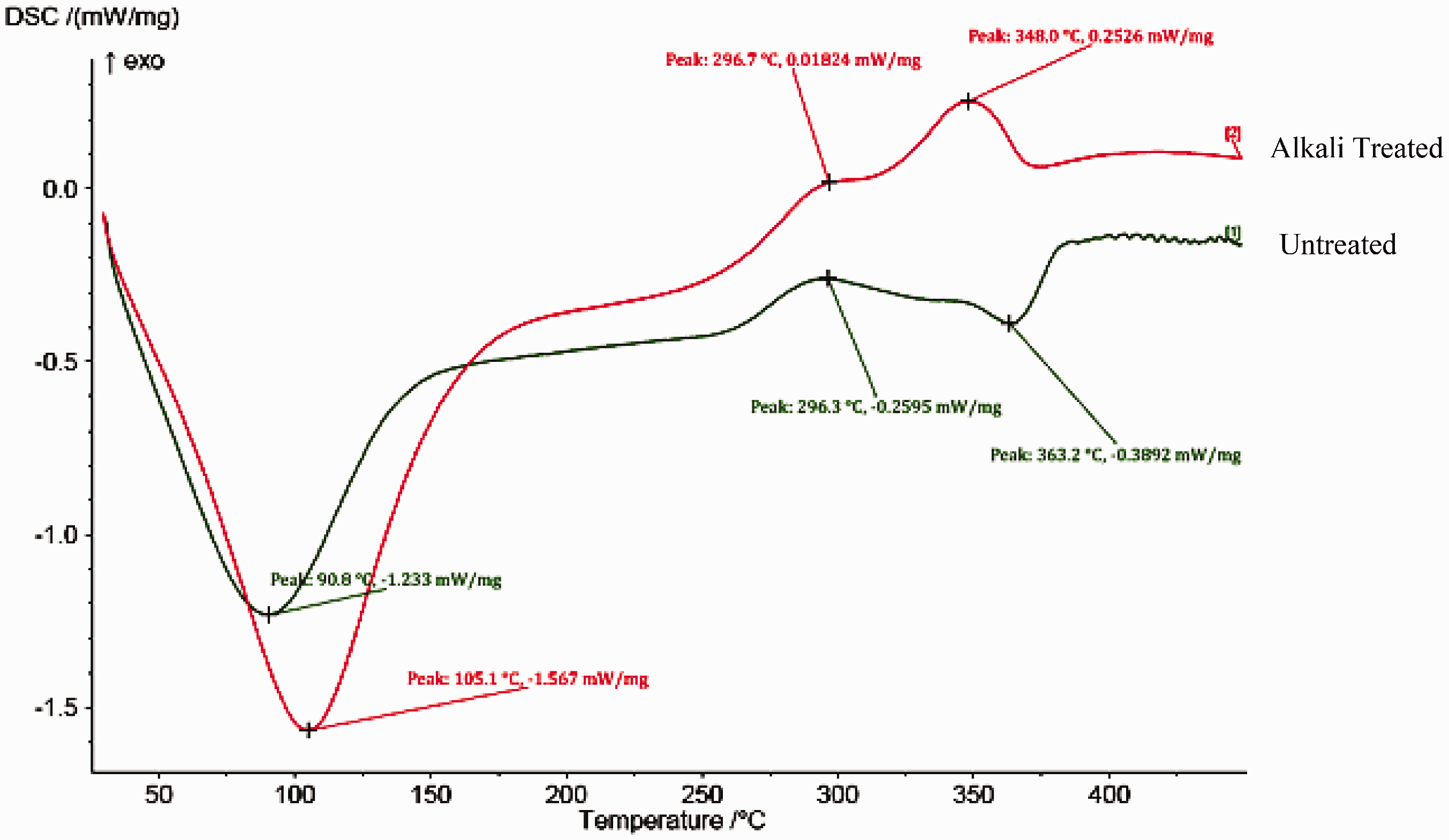
XRD studies
Jute consists of hydroxyl (–OH) and methylol groups as the major constituents and these groups creates a hydrogen bonding between –OH groups and adjacent jute polymeric groups that acts as a dominant structural force than van der Waals force. In addition, any cellulosic structure consists of crystalline, oriented amorphous, highly distorted crystalline and amorphous regions. Among these constituents, amorphous and highly distorted crystalline regions of the structure lies on the fringes of the crystallites coupled to adjacent crystallites by the oriented amorphous region that contains cellulose are highly reactive with the addition of alkali. From Figure 3 and Table 4, it is shown that peak 2θ of cellulose I of untreated jute has been shifted to that of cellulose II of alkali-treated jute fabric. With the alkali treatment, –OH present in the non-crystalline region causes the reduction in distortion and helps in the formation of crystallite size. The alkali, sodium hydroxide, reacts with the accessible hydroxyl group of the cellulosic structure of jute and forms traces of sodium salt of cellulose. The treatment of jute with alkali further leads to the processes such as micro-fibril swelling, crystalline areas disruption and new crystalline lattice formation. With these processes, the interfibrillar regions become less rigid and more rearrangement of fibres take place. It finally results in the agglomeration of chain or strong intermolecular hydrogen bonds with the conversion of cellulose I to cellulose II. The transformation of cellulose I to cellulose II indicates the removal of lignin that acts as a cementing material. The delignification of the cellulosic structure that interrupts the lamellar structure of the jute increases the ordered arrangement of the crystalline cellulose takes place.
XRD analysis of (a) alkali-treated and (b) untreated jute fabrics. XRD analysis of untreated and treated jute fabric.

It is also inferred from the CI value that it is slightly more for the alkali-treated fabric when compared to the untreated jute fabric, which indicates the increase in more orderly arrangement of cellulosic structure [13,14].
AFM
AFM topographical images of the jute fabric before and after modification are shown in Figure 4. These images have been used to visualize the surface roughness of the jute with modifications. The mean roughness of the alkali-treated jute is found to be 67.57 nm and untreated jute is found to be 28.99 nm. Compared with untreated, the alkali-treated fabric shows rougher surface due to the removal of the surface cementings. The alkali treatment removes the semicrystalline and amorphous portions such as hemicelluloses and lignin were removed. The treatment also cleans the surface impurities, wax and other natural oils from the fibrous structure. The treatment also causes the fibre fibrillation due to the axial splitting of the elementary fibres and thereby increases the effective surface area available for wetting by a matrix in a composite. This rougher surface increases the number of anchorage points for the interlocking of the resin to the fabric. This interlocking creates a better interfacial bonding between the fibre and matrix and gives better adhesion.
AFM images of untreated and alkali-treated jute fabrics.
Analysis of alkali-treated jute composite
Electrical properties
Electrical resistivity is an intrinsic property that quantifies how strongly a given material opposes the flow of electric current. The resistivity of the jute reinforced composite depends on various factors such as moisture content, crystallinity, composition, cellular structure, microfibrillar angle, etc. The volume and surface resisitivity of the sample gives the insulation quality of the given material.
Electrical resistivity of untreated and alkali treated jute composites.

In addition, the hydroxyl groups in the jute can absorb moisture and improves the conductivity of the composite. The reduction in the hydrophilic nature of the fabric in turn reduces the moisture absorption and number of polar groups in the system. With the alkaline treatment, the probability of interaction between the polar group present in jute and water molecules is reduced and destroys the reactive hydrogen bonding. This increases the hydrophobicity of the fabric and reduction in orientation polarization [16]. It is also inferred that with the improvement in the adhesion between the fabric and the resin results in compact packing of the fabric with the resin. This results in reduced porosity, voids and other irregularities of the composites, which in turn increases the volume resistivity and reduces the conductivity.
From Table 5, it is observed that the electrical resistivity of the composite is more when compared to the fabric. In the case of fabric composite, the gap between the warp yarns diminishes and provides more resistance for the charge particles to pass through the fabric. The electrical resistivity of the fabric composite is more when compared to fabric. This is mainly due to the conversion of point contact to flat circle contact by pressure application during compression moulding technique that is used for manufactuirng the composite. This in turn increases the hinderance of conducting path for the electrical particles to pass through the composite. This is also in line with the findings of Taipalu et al. [17]. It is also evident that with the application of resin during compression, the surface and volume resistivity increases mainly because the resin occupies the space between the yarns and pores in the structure. When compared to the surface resistivity, the volume resistivity of the composite is more especially in the alkali-treated composite because with the application of the resin, the internal orientation of the fabric remains constant. It is also due to that the availability of more surface area that leads to increased resin uptake and hence the resistivity increases [18].
Thermal properties
From Figure 5, it is observed that alkali-treated woven jute composite shows higher thermal conductivity than the untreated composite. This is mainly because, with the treatment of NaOH, the non-cellulosic material present in the jute structure has been removed. The cementing material in the jute structure has also been dissolved that leads to increase in crystallinity in the structure. The increased crystallinity creates better packing and heat flow in the structure. The surface area for adhesion on the fibre increases mainly due to the dissolution of lignin, hemicelluloses and other substances from the fibre structure. The increase in surface area results in more contact area between the fibre and matrix and less gap between them. Thereby, the flow of heat through the composite increases with reduced resisting energy.
Thermal conductivity of untreated and alkali-treated jute composites (W/mK).
It is also evident that with the removal of the various substances from the structure leads to more polarity on the fibre surface. It finally leads to higher polar–polar interaction between the fibre and matrix and thermal conductivity through the composite increases [19]. It is also evident that the removal of lignin from the jute structure due to alkali treatment leads to hydrophobicity of the structure in turn better adhesion with the vinylester resin making the composite more thermally conductive in nature.
Compressive behaviour
Figure 6 shows the compression strength of the treated and untreated jute composites. From the results, it is observed that the compression strength of the treated jute composite is slightly higher when compared to the untreated composite. This increase in compression strength with the alkali treatment is because of the removal of lignin and hemicelluloses that creates better wettability of the resin on to the fabric. The removal of waxy coating by alkali treatment increases the surface free energy, which favours interfacial strength between the fibre and matrix leading to improved compressive strength of the treated composite. In addition, treatment with alkali breaks down the fabric fibre bundles that results in more effective surface area available for contact with matrix polymers [20]. The removal of the surface constituents of jute leads to the increased surface roughness that creates more contact area between the fabric and resin. This increased adhesion between the fabric and resin leads to more resistance for the stress transfer from the matrix to the fabric leading to increased compression strength of the alkali-treated composite.
Compression strength of untreated and alkali-treated jute composites.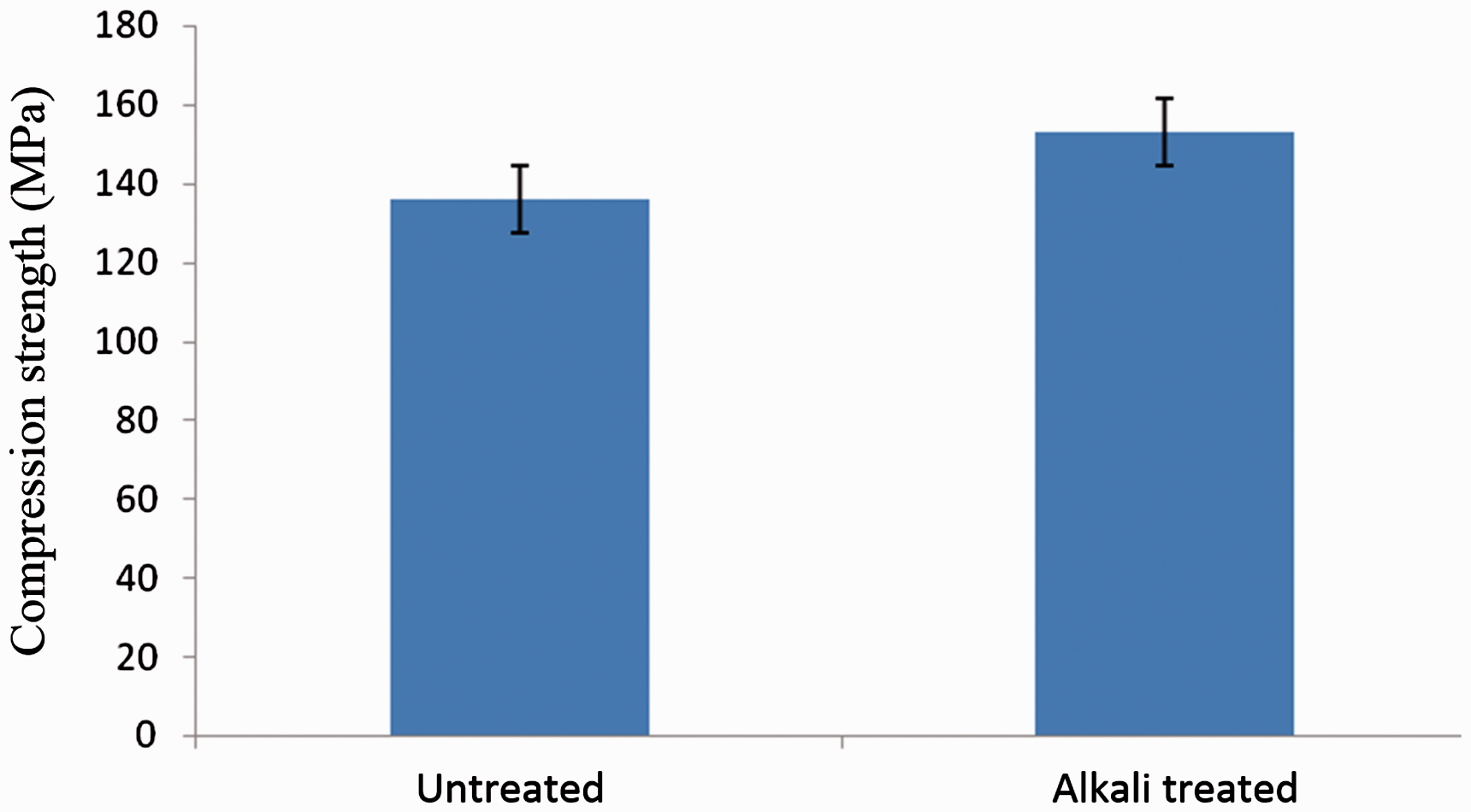
Pull out strength test
From Figure 7, it is observed that the pull out strength increases with the alkali treatment process. This shows that the alkali treatment increases the adhesion strength between the fabric and matrix. With the treatment of alkali, the fabric surface area increases with the removal of residues and chemical constituents such as hemicelluloses and lignin from the surface. It also creates cavity on the surface that allows the matrix to impregnate and better bonding between the fabric and matrix. In addition, with the removal of hemicelluloses and lignin it increases the hydrophobicity of the fabric and hence the adhesion between the fabric and resin improves.
Pull strength tests of untreated and alkali-treated composites.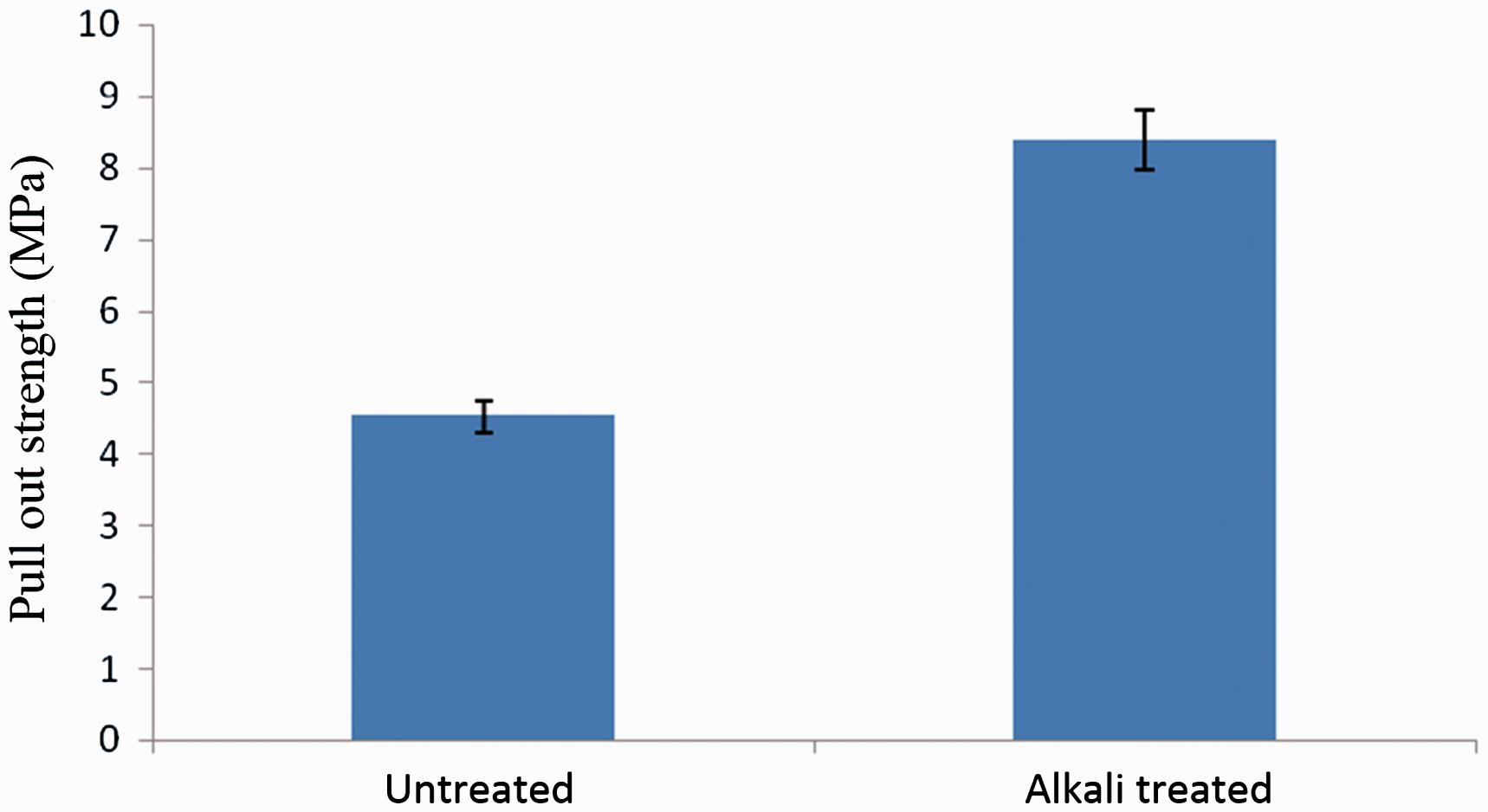
Conclusions
Jute fabrics were treated with NaOH and compressive, electrical and thermal behaviour of the composite were analysed. From DSC results, it was observed that the temperature required to degrade hemicelluloses and lignin is lower for untreated when compared to the treated one. This shows the reduction in the hemicelluloses and lignin content from the jute fabric with the alkali treatment. XRD study confirmed the improvement in the crystallinity of the treated jute due to the removal of hemicelluloses and lignin from the surface of the jute. From the AFM analysis, it is confirmed that the surface roughness of the fabric increases thereby the surface area for the resin bonding also increases. The composite prepared from the treated jute show improved electrical resistance due to the increased hydrophobicity and increased thermal conductivity due to the increased level of crystallinity that causes improved fibre alignment. Similarly, the treated jute composite shows higher compression strength due to increased adhesion between fabric and matrix with the removal of hydrophilic components from jute by alkali treatment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
