Abstract
Silk fabric was pretreated with 3-aminopropyltrimethoxysilane (silane) in supercritical carbon dioxide and then silver nanoparticles were synthesized on the pretreated silk fabric under microwave irradiation. The silver nanoparticle coated fabrics were characterized by Fourier transform infrared spectroscopy, scanning electron microscope, and ultraviolet–visible spectroscopy. Deposition rate and color characteristics of the silver coating of samples were investigated. The results show that silver-coated silk fabric with pretreatment of the silane via supercritical carbon dioxide possesses excellent ultraviolet protection, softness and the adhesive strength between silver coating and silk fabric is improved.
Introduction
Silver nanoparticles have been applied in many areas because of their conductive [1] and antibacterial properties [2], surface plasma resonance (SPR) [3], surface-enhanced Raman scattering (SERS) effects [4], chemical stability and thermal properties, photosensitivity [5], catalytic activity [6], and so on. These multiple properties make silver nanoparticles as a suitable candidate for fabrication of functional textiles with ultraviolet (UV) protection [7], antibacterial property [8], antistatic property [9], electrical conductivity [10], self-cleaning, electromagnetic shielding [11], and superhydrophobicity [12].
Silk produced by the silkworm has been conventionally used in textiles for thousands of years due to its inherently elegant lustre, great flexibility, environmental friendly, and excellent mechanical strength [13]. As a natural protein fiber, silk possesses a structure similar to human skin with smooth, breathable, soft, nonitching and antistatic characteristics. Therefore, silk is one of suitable materials for high-end clothing [14]. Nowadays, modification of silk fabric with silver nanoparticles has attracted much attention for various applications such as the medical, security and construction engineering, water technology, clothing industry, lightweight construction, and automotive industry [15].
However, silver coating has poor adhesion to textile surfaces because of the absence of chemical conjunction between silver coating and fibers. Recently, there has been growing use of organosilanes as coupling agents to improve the adhesion between metal nanoparticles and textile [16,17]. Generally, organosilanes are formed on substrates in anhydrous solvents such as toluene, ethanol, and acetone [18]. However, these solvents are not considered as “green” solvents.
In order to explore a proper method to solve the problem, researchers have been intense interested in the use of supercritical carbon dioxide (sc-CO2) as an environmentally friendly solvent for laboratory and industrial applications with the properties of inexpensive, nontoxic, inert, noninflammable, and recyclable in the last decades [19]. The use of sc-CO2 as a solvent to perform adsorption [20], impregnation [21], materials functionalization [22], and chemical reactions has received considerable attention as a viable and sustainable alternative to conventional liquid solvents. Particularly, sc-CO2 process is an attractive process for modification of textile materials because its unique properties under the supercritical state such as high density, low viscosity, low dielectric constant, high diffusivity, solubility, and zero surface tension [23]. Moreover, sc-CO2 have been both used as a solvent for infusing metal complexes into polymers and as plasticizer to soften the surface of the polymeric substrates and increase the adhesiveness between the metal layer and the polymeric substrates [24]. To the best of our knowledge, there is no report on the fabrication of organosilanes layers in sc-CO2 prior to synthesis of silver nanoparticle on textiles.
In this study, silk fabrics were pretreated with 3-aminopropyltrimethoxysilane in sc-CO2 and then silver nanoparticles were synthesized on the fabrics under microwave irradiation. The composition and chemical structure of silk fabrics modified with the silane were investigated. Deposits weight, surface morphology, and color characteristics of the silver nanoparticle coated silk fabrics were studied and washing fastness of silver-coated silk fabrics was evaluated.
Experimental
Materials
100% plain weave silk fabric in white color was used as the substrate. AgNO3 (>99%), trisodium citrate (≥99.0%), 3-aminopropyltrimethoxysilane (silane), and ethanol (99.7%) were purchased from Aladdin Reagent Co., Ltd. China. Carbon dioxide (99.99%) was used as received. All chemicals were analytical grade reagents and used without further purification.
Pretreatment of silk fabric samples
Prior to the deposition of silver nanoparticles, the silk fabrics were initially cleaned in ultrasonic baths with acetone, ethanol, and deionized water for 15 min, respectively. The supercritical pretreatment experiment was carried out using an sc-CO2 equipment (SFE System, Deyang, China). Experimental processes and apparatus employed are shown in Figures 1 and 2, respectively. An amount of 1 vol.% of 3-aminopropyltrimethoxysilane was dissolved in small amount of ethanol as co-solvent to increase the solubility of silane in sc-CO2 medium. The solution was then transferred into a solvent vessel. The cleaned fabrics were rolled and introduced into a reaction vessel. Carbon dioxide gas was then supplied via a high-pressure syringe pump and maintained at 10 MPa. After that, the device was sealed and liquid CO2 was added. The temperature was then increased gradually to 40℃. As the device was heated, the remaining CO2 was added to the system until the pressure was reached to 10 MPa. The treatment was kept at 10 MPa and 40℃ for 10 min. Finally, the system was slowly depressurized and allowed to cool down to room temperature. The silanized fabrics were collected for silver coating.
Schematic illustration of sc-CO2 pretreatment and silver nanoparticles coating on silk fabric. Schematic diagram illustrating of the sc-CO2 reaction: (A) preheater, (B) solvent vessel, (C) the silane, (D) reaction vessel, (E) silk fabric sample, (F) separation vessel, (3,5) barometer, (1,2,4,6,7) valves.

Silk fabrics coated with silver nanoparticles with modification of the silane via the traditional method were also prepared for comparison. For silk fabrics modified with the silane via the traditional method, the cleaned silk fabric samples were immersed into an 1 vol.% the silane ethanol solution at 40℃ for 2 h to form a self-assemble monolayer of the silane on the surface of the fabric. After silanization, the fabrics were dried in an air oven at 70℃ for 30 min.
Silver nanoparticles coating on the silane-modified silk fabrics
The silk fabrics modified with the silane were immersed in the mixture of aqueous solutions of AgNO3 (0.1 g, 200 mL) and trisodium citrate (1 wt%, 6 mL) under ambient condition. The reaction mixture was exposed to high intense microwave irradiation (800 W) in a domestic microwave oven operating in a cycling mode (on 10 s, off 5 s) to prevent boiling of solvent. The silver coating was carried out for 4 min. Silk fabrics were turned over every minute. Finally, the silk fabrics were rinsed with deionized water three times and dried at 70℃.
Characterization
FTIR was used to characterize the presence of chemical groups on the surface of the silane-modified silk fibers. The spectra were recorded on Nicolet 6700 for each measurement over the spectral range of 400–4000 cm−1 at resolution of 4 cm−1. The deposition rate (
Color measurements of the original and silver silk fabrics were conducted by a spectrophotometer (COLOR I5) under Pulsed Xenon D65 and processed using software with the small aperture. The pictures of the silver-coated silk fabrics were taken by a digital camera. Ultraviolet–Visible (UV–vis) transmittance spectra of silver nanoparticles coated silk fabrics were measured by a spectrophotometer (Shimadzu UV-2700) with wavelength changing from 200 nm to 800 nm. Softness of the original and silver-coated silk fabric pretreated with silane was evaluated by computer controlled stiffness tester (Model LLY-01).
Adhesion between the silver coating and the fabric were evaluated through washing fastness of silver-coated silk fabrics. According to the standard AATCC 61-2013 (Colorfastness to Laundering), the silver-coated silk fabrics were washed in 0.37% of detergent with 10 steel balls at 40℃ for 45 min then rinsed in deionized water twice and dried at 70℃. The color characteristics of silver-coated silk fabrics after washing were carried out by the spectrophotometer (COLOR I5) under Pulsed Xenon D65.
Results and discussion
FTIR spectra
FTIR spectra of the original and silver-coated silk fabrics without and with the silane pretreatment via traditional and sc-CO2 processes are shown in Figure 3. As shown in Figure 3(a), peak at 3300 cm−1 is ascribed to stretching vibration of N–H groups. Peak at 1650 cm−1 is attributed to stretching vibration of functional groups of amide. Peak at 1550 cm−1 is ascribed to deformation vibration of N–H in the molecular plane. The band of 1250 cm−1 is due to the stretching vibration of C–N. Deformation vibration of N–H out of the molecular plane is observed at 650 cm−1 [25]. These peaks are characteristics of silk protein. Compared with the FTIR spectrum of original silk fabric, characteristic peaks of silk fabric also can be detected on the silver-coated silk fabric. The silanol group of the silane forms an Si–O–Si bond with an –OH group on the silk fiber surface by dehydration condensation. After all of the –OH groups on the silk fiber surface are bonded with the silanol groups of the silane, no further silane molecules are deposited, and the silk fiber surface is covered by small amount of silane on the silk fabric. It can be seen from Figure 3(d) that –NH2 group appearing at 3200 cm−1 after modification of silane demonstrates that silane is successfully treated on the silk fabric. The peak of –NH2 is weak due to the low content of the silane on the silk fabric. However, both in Figure 3(c) and (e), the characteristic peaks of the silane cannot be detected in the spectra of the silver-coated silk fabrics modified with the silane via traditional and sc-CO2 processes by FTIR equipment due to relative small amount of the silane in the samples of silver-coated fabrics [26].
FTIR spectra of (a) original silk fabric, (b) silver-coated silk fabric without the silane modification, silver-coated silk fabric modified with the silane via (c) traditional process and silver fabric modified with silane via sc-CO2 process (d) before and (e) after silver coating.
Deposition rate
Effect of amount of AgNO3 on deposition rate
The effect of amount of silver nitrate on deposition rate of silver nanoparticles on silk fabrics is shown in Figure 4. Deposition rate of silver nanoparticles on the silk fabrics without and with silane via traditional and sc-CO2 processes are shown in Figure 4(a), (b), and (c), respectively. Regardless of the silk fabric without and with the silane modification, deposition rate of silver nanoparticles first increase and then decrease with the rise of the amount of silver nitrate. It can be seen that the rate rises with the increase of the amount of silver nitrate in the solution in the range of 0.02–0.08 g. Silver nitrate is the main contributor for the silver ions of the coating. When the amount of silver nitrate is small, the amount of silver nanoparticles reduced by sodium citrate is also small, causing the deposition rate to be small. As the amount of silver nitrate is increased, silver ions reduced by the electrons released increase and silver atoms which are deposited on the silk fabrics also increase. Therefore, the result shows that the deposition rate increases with the rise of the amount of silver nitrate. However, the sodium citrate is maintained and remains unchanged, and the electrons released from it gradually decrease; thereby the silver atoms reduced will decrease when concentration of silver ions reaches a maximum. Thus, the deposition rate decreases after the amount of silver nitrate are more than 0.08 g.
Effect of amount of AgNO3 of deposition rates of silver nanoparticles on the silk fabrics (a) without the silane, and with the silane via (b) traditional process and (c) sc-CO2 process.
For a given amount of silver nitrate and sodium citrate in the solution, deposition rate of the silver nanoparticles on the silk fabrics without the silane is the lowest; however, deposition rate of the silver nanoparticles on the silk fabrics modified with the silane via sc-CO2 process is the highest. Regardless of the conventional process or sc-CO2 process, deposition rates of silver nanoparticles on the silk fabrics with the silane are higher than that without the silane. It can be explained by that the amino groups at the ends of the silane molecules can easily and quickly trap silver ions from the bath. The trapped silver ions are immediately reduced by trisodium citrate in the solution and served as the seed for the continuous silver coating. As the reaction goes on, silver nanoparticles are continuously coated on the silk fabric and form the coating. Therefore, existence of the silane on the silk fabrics can speed up the reaction, resulting in increase of deposition rate and deposit weight. In addition, deposition rate of silver nanoparticles on silk fabrics modified with the silane via sc-CO2 is higher than that via conventional process. This may be explained by that the very low viscosity and null surface tension of sc-CO2 greatly accelerates the diffusion through the silk fibers in comparison with the traditional method. Therefore, more silane are uniformly distributed and tightly anchored on the silk fibers. Accordingly, deposition rate of silver nanoparticles on silk fabric via sc-CO2 is higher than that via traditional process.
Effect of concentration of trisodium citrate
Effect of concentration of trisodium citrate on deposition rate of silver nanoparticles on silk fabrics is illustrated in Figure 5. For silk fabric without and with the silane, deposition rate of silver nanoparticles first increases and then decreases with the rise of the concentration of trisodium citrate. The reducing power of the silver ions increases when the concentration of trisodium citrate is raised, i.e. the number of the neutral silver atoms reduced from the citrate ions increases causing the deposition rate to increase. However, when the reducing agent is in excess, the stability of the bath deteriorates or even decomposes, resulting in the decrease in the number of reduced neutral silver atoms and making the deposition rate drop.
Deposition rate of silver nanoparticles on the silk fabrics for varied concentration of trisodium citrate (a) without pretreatment of the silane, and pretreatment with the silane via (b) traditional process and (c) sc-CO2 process.
Surface morphologies
Surface morphologies of silver-coated silk fibers without and with modification of the silane via traditional process and sc-CO2 process are presented in Figure 6. As shown in Figure 6(a), only small amount of silver nanoparticles are dispersed on the silk fiber without the silane. However, it can be observed from Figure 6(b) and (c) that surface of the silver-coated silk fibers modified with the silane via traditional and sc-CO2 processes are both completely covered with aggregated silver nanoparticles. However, the surface of the silver-coated silk fibers modified with the silane via sc-CO2 process is rougher than that via traditional process. The result indicates that the much more and larger silver nanoparticles are deposited on the surface of silk fibers via sc-CO2 process. This is because that the silk with the silane via sc-CO2 can efficiently absorb silver atoms, which would result in fast crystal nucleation and growth. The migration and aggregation of silver particles are probably driven largely by the instability of silver atoms due to their high surface free energy. Their aggregation may produce thermodynamically stable particles with bigger sizes.
SEM micrographs of silver-coated silk fibers (a) without and with modification of the silane via (b) traditional process and (c) sc-CO2 process.
Color characteristics
Images of the silver-coated silk fabrics without and with the silane via traditional and sc-CO2 processes are presented in Figure 7. Colors of the silver-coated silk fabrics without the silane are yellow due to small amount silver deposition. Color of the silver-coated silk fabrics with the silane prepared via traditional process is brown and the color depths are higher than those without silane treatment. This may because the amount of silver nanoparticles on the silk fabric modified with the silane is more than that without modification of the silane. Furthermore, color of the silver-coated silk fabrics modified with the silane via sc-CO2 process is black.
Images of (a) silver-coated silk fabric, and silver-coated silk fabric pretreated with the silane via (b) traditional process and (c) sc-CO2 process.

UV–Vis transmittance
The UV–Vis spectra of original and silver-coated silk fabrics are illustrated in Figure 8. Original silk fabric has very strong transmittance in the UVA region (320–400 nm), weak transmittance in the UVB region (290–320 nm) and UVC absorption (220–290 nm). However, there is weak transmittance for silver-coated silk fabrics without the silane in the whole UV region. Silver-coated silk fabrics pretreated with the silane via sc-CO2 process show the least UV–Vis transmittance. This can be considered as a consequence of the most amount of silver nanoparticles deposit on the fabrics. During the silver coating process, the silver particles are deposited not only on the fiber surface but also in the spaces between the yarns. The coverage of the spaces can prevent the penetration of the UV irradiation through the fabric.
UV–Vis spectra of (a) original silk fabric, and (b) silver-coated silk fabric without and with pretreatment of the silane via (c) traditional process and (d) sc-CO2 process.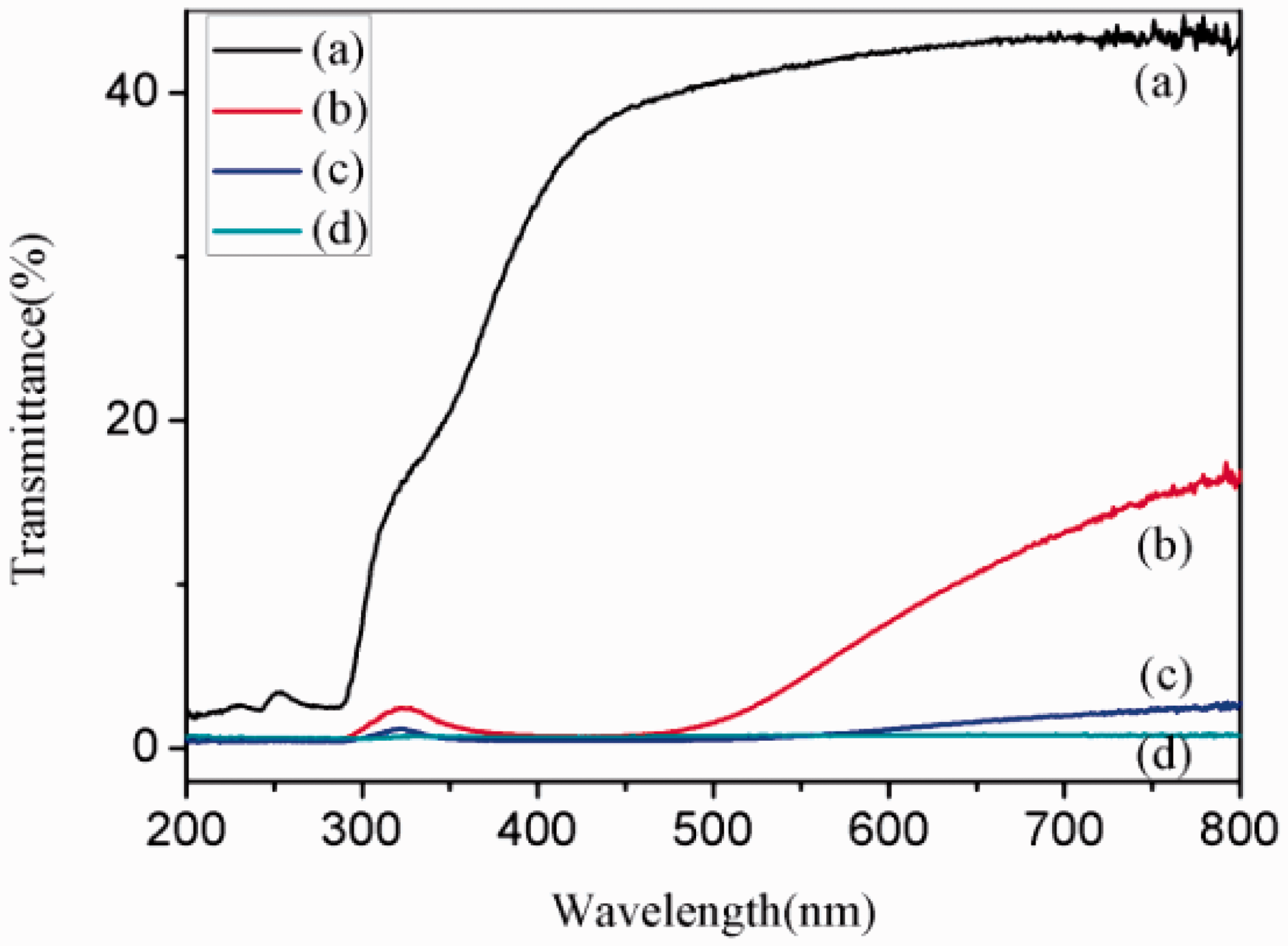
Color fastness
The adhesion of silver to silk fabric is one of the most important concerns to the silver coating on the silk fabric. A washing fastness test was employed to evaluate the adhesion of the silver coating on the silk fabric without and with the silane via traditional and sc-CO2 processes, and it was repeated five times on each specimen. The
Softness
The softness of silver-coated silk fabric modified with silane via traditional process and sc-CO2 process was evaluated by computer-controlled stiffness tester. Bending property has an important effect on the handle performance. Bending rigidity of samples was calculated based on the following equation

Bending rigidity of original silk and silver-coated silk modified with silane via traditional and sc-CO2 processes.

Conclusion
Silk fabric was pretreated by the silane with the assistance of sc-CO2, and then silver nanoparticles were deposited on the silk fabric under microwave irradiation. The amount of silver nanoparticles coating on the silk fabric pretreated with the silane via sc-CO2 process is much more effective than that via traditional process. UV–Vis transmittance of silver-coated silk fabric pretreated with the silane via sc-CO2 process is much lower than that original silk fabric. The results indicate that the silane pretreatment could provide silk fabric with an excellent protection of ultraviolet radiation. The silane pretreatment via sc-CO2 process significantly improves adhesive strength between the silver coating and the silk fabric. In summary, there is a potential application of pretreatment of the silane via sc-CO2 process in the microwave-assisted synthesis for the preparation of silver nanoparticles on silk fabric to improve the functional properties and adhesive strength.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by The National Natural Science Foundation of China (No. 51203099), The Science Foundation for Youth Scholars of Sichuan University, China (YJ2011020), and Ningbo Municipal Science and Technology Bureau (15H0640).
