Abstract
The technology of producing a magnetized polyester fabric for controlled and accelerated release of menthol in potential biomedical applications is reported in this paper. The magnetized polyester fabric is prepared in a facile hydrothermal process and subsequently modified with aminopropyltriethoxysilane, grafted with carboxylated β-cyclodextrin, and loaded with menthol under hydrothermal conditions. The microstructure, thermal and magnetic properties of both carboxylated β-cyclodextrins and magnetized polyester fabrics grafted with β-cyclodextrins are characterized by using field emission scanning electron microscopy, energy dispersive X-ray spectroscopy, X-ray diffraction, vibrating sample magnetometry, Fourier transform infrared spectroscopy, thermal gravimetric analysis, differential scanning calorimetry, and diffuse reflectance spectroscopy techniques. It is evident that the carboxylated β-cyclodextrin is synthesized and successfully grafted on the surface of the modified magnetic polyester fabric. Both hyperthermia effect and controlled release behavior of the magnetized polyester fabric loaded with menthol are measured under high-frequency alternating current magnetic fields. It is found that the menthol included in β-cyclodextrin could be released in a controlled and accelerated way under external alternating current magnetic fields.
Introduction
Controlled release of medical active ingredients (e.g. menthol) onto skin is desirable for medication and has many medical and healthcare applications. In current practice, these medical active ingredients are usually volatile and need to be hosted in an adsorbent substance such as β-cyclodextrin (β-CD) for gradual release of the ingredients when it is needed. β-Cyclodextrin is one of the cyclic oligosaccharides composed of seven
Functional polymeric nanofibers containing different cyclodextrin-inclusion complexes [10,11] and textiles grafted with β-CDs loaded with various drugs or fragrant [12] have been developed for the gradual release of active ingredients for the application of either skin disease treatments or odor control. For instance, β-CD grafted viscose fabric loaded with aescin formulations has been obtained by means of a two-step ultrasound-assisted reaction [13]. Cotton textiles containing β-CDs loaded with some natural active substances have been fabricated for control purposes [14–16]. However, the gradual release of ingredients from current β-CDs grafted textiles is not controllable. It is desirable for the release of active ingredients to be controllable in its amount when it is needed. There is little research currently on the actively controlled release of volatile compounds from textile products.
Magnetite micro- and nano-particles having superparamagnetism properties have been used in biomedical applications for the controlled release drugs and targeted cancer medication. The release of drugs incorporated with β-CD grafted magnetite can be triggered through either the hyperthermic effect of the magnetic particles in high frequency magnetic field (HFMF) or induction heat, and its amount is controllable by varying the intensity of HFMF or heat applied [17–19]. Aminopropyltriethoxysilane-coated magnetic Fe3O4 nanoparticles (APTES-MNPs) with β-CD were reported to have saturation magnetization value as high as 69 emug−1 and the outermost CD moieties to function as inclusion sites for drugs and biomolecules in magnetic drug delivery and bioseparation applications [20]. The silane coating outside the magnetic Fe3O4 cores provided amino group (─NH2) for linking the CD molecule, other silane coated magnetic nanoparticles with glycidylmethacrylate-grafted-maleated cyclodextrin composite hydrogel were used as a drug carrier for the controlled delivery of 5-fluorouracil [21]. However, the fibrous materials incorporated with magnetic particles grafted with β-CD loading with active ingredients for controlled release purposes have not been reported yet.
Menthol has been used as common cold medications like toothpastes, confectionery, cosmetics, insect repellent as well as components in traditional medicines for curing infections, insomnia, and irritable bowel syndrome [22,23]. It has been demonstrated that β-CD can form inclusion complexes with both enantiomers of menthol, resulting in the same stability constants for both complexes. The value of the stability constant is large enough to guarantee binding of highly volatile menthol inside β-CD cavity and small enough for a convenient release of the aroma from the host molecule [24,25].
In this paper, menthol is used as a model flavor molecule loaded onto a polyester fabric incorporated with β-CD grafted magnetic particles. It is expected that the magnetic particles deposited on the polyester fabric can serve as the hyperthermia-inducing media using the exothermic properties to raise the temperature of polyester fabric in HFMF for the controlled release of menthol for the treatment of cutaneous diseases.
In this research, different analytical techniques including field emission scanning electron microscopy (FESEM), energy dispersive X-ray (EDX) spectroscopy, X-ray diffraction (XRD), vibrating sample magnetometry, Fourier transform infrared (FTIR) spectroscopy, thermal gravimetric analysis (TGA), differential scanning calorimetry (DSC), and diffuse reflectance spectroscopy are employed to determine the structural changes of the resultant β-CD grafted magnetized polyester fabric loaded with menthol. The release of menthol from β-CD grafted magnetized polyester fabric is characterized by using colorimetric method. Both the hyperthermia properties and controlled release properties of the resultant polyester fabric are measured under external high-frequency alternating current magnetic fields.
Experimental
The technical route for the fabrication of magnetized polyester fabrics grafted with β-CD loaded with menthol is illustrated in Figure 1.
Schematic illustration of the technical route for the fabrication of magnetized polyester fabrics grafted with β-CD loaded with menthol.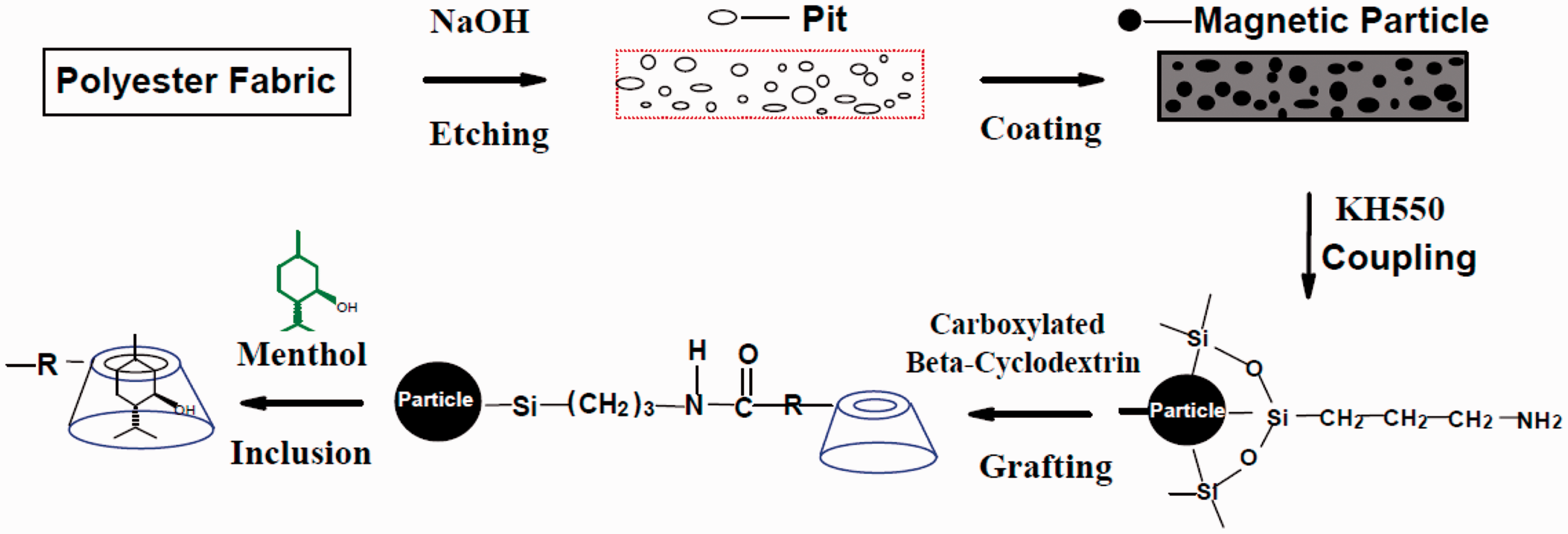
Materials and reagents
A plain woven fabric made of 100% polyethylene terephthalate fibers was provided by a local textile mill. The yarn counts are 7.3 tex identical in both warp and weft directions, and the numbers of threads in both warp and weft directions are 40 and 30 per centimeter, respectively. Deionized water was used throughout the experiments and the chemicals used in this research are of analytical grade without further purification, including ferric sulfate (Fe2(SO4)3, Tianjin Bodi Chemical Co., Ltd.), sodium thiosulfate (Na2S2O3·5H2O, Langfang Pengcai Fine Chemical Co., Ltd.), urea (CO(NH2)2, Beijing Kangpu Huiwei Technology Co., Ltd.), N,N-dimethylformamide (C3H7NO, Nanjing Chemical Reagent Co., Ltd), maleic anhydride (C4H2O3, Jining Baiyi Chemical Co., Ltd.), trichloromethane (CHCl3, Jiangsu Pengyu Chemical Co., Ltd.), menthol (C10H20O, CAS No. 89-78-1, Fortuneibo-Tech Co., Ltd.), 4-dimethylaminobenzaldehyde (C9H11NO, Guangzhou Boyang Chemical Industry Co., Ltd.), 98% concentrated sulfuric acid (H2SO4, Nanjing Senking Chemical Co., Ltd.), acetone (C3H6O, Nanchang Lanxiang Chemical Co., Ltd.), and anhydrous ethanol (C2H6O, Nanchang Lanxiang Chemical Co., Ltd.). β-Cyclodextrin (C42H70O35, CAS No. 7585-39-9) of food grade was obtained from Zibo Qianhui Biological Technology Co., Ltd. The silane coupling agent KH550 (γ-aminopropyltriethoxysilane, NH2(CH2)3Si(OC2H5)3) was purchased from Dow Corning (Shanghai) Co., Ltd.
Fabrication of magnetized polyester fabrics
A certain amount of ferric sulfate (0.48 g) was completely dissolved in 40 mL of deionized water at 60℃ with the aide of ultrasonic stirring (55 kHz, 100 W), followed by the addition of sodium thiosulfate. The molar ratio of sodium thiosulfate to ferric sulfate is 1:1. When the color of the suspension turned from dark purple to transparent, the pH value of the precursor suspension was adjusted between 1.8 and 2.5 ready to use by adding a certain amount of urea.
Alkali-etched polyester fabrics were made according to the following procedures. A piece of polyester fabric was immersed in a sodium hydroxyl aqueous solution of 100 g/L at a liquor ratio of 1:50 in 100℃ for 30 min, then successively washed with acetone, anhydrous ethanol and deionized water at 40℃ for 10 min, respectively.
About 1.2 g of the alkali-etched polyester fabric was added to the ferric precursor suspension in a Polytetrafluoroethylene (PTFE)-lined stainless steel autoclave. The autoclave was sealed and then heated up in a furnace up to 130℃ for 5 h, rotating at 10 revolutions per minute (r/min). After cooling down to room temperature, the magnetized polyester fabric was rinsed in deionized water without stirring, and then dried ready for further surface modification.
Modification of β-CD
About 5.68 g of β-CD was dissolved in 50 mL of 100% N,N-dimethylformamide solution at 80℃ under continuous stirring for 30 min, followed by the carboxylation reaction with 4.9 g of maleic anhydride for 12 h. Carboxylated β-CD was precipitated from the resultant suspensions by adding trichloromethane solution into it once the suspension was cooled down. The resultant carboxylated β-CD was collected using a vacuum filtration set-up, then thoroughly washed with acetone solution, and dried in a vacuum oven at 80℃ for 24 h.
Surface modification and grafting of carboxylated β-CD to magnetized polyester fabrics
The surface of magnetized polyester fabrics was first modified with silane coupling agent KH550 prior to grafting with carboxylated β-CD. Silane coupling agent water solution was made by adding coupling agent, KH550 (relative to the fabric weight 1%), to 200 mL of 10% ethanol water solution with continuous stirring for 40 min. The resultant magnetized polyester fabrics were immersed in the above KH550 solution for 30 min without stirring. Both sides of the saturated magnetized polyester fabrics were then irradiated with an ultraviolet light of main wavelength 365 nm for 30 min, respectively. Finally, the magnetized polyester fabrics modified with silane coupling agent KH550 were successively washed with acetone, anhydrous ethanol, and deionized water respectively, and dried in a vacuum oven at 60℃ for 3 h.
The magnetized polyester fabrics modified with KH550 were immersed in 80 mL of deionized water solution containing 1.5 g of carboxylated β-CD in a PTFE-lined stainless steel autoclave, which was gradually heated up to 120℃ at a speed of 3℃ per minute in a furnace. After 3 h of reaction in 120℃, the autoclave cooled down naturally. The magnetized polyester fabric grafted with carboxylated β-CD was obtained after the fabric was successively washed with acetone, anhydrous ethanol, and deionized water at 40℃ for 15 min, respectively, and dried at ambient temperature.
Inclusion of menthol
The magnetized polyester fabric grafted with β-CD was dipped in 80 mL of deionized water containing 0.17 g of menthol at 30℃ in a 100 mL PTFE-lined stainless steel autoclave. The autoclave was sealed and heated up to 120℃ in a furnace while rotating at 10 r/min. After 2 h of reaction at 120℃, the autoclave was cooled down naturally, and the magnetized polyester fabric loaded with menthol was successively rinsed with acetone, anhydrous ethanol and deionized water at 30℃ for 10 min, respectively, and finally dried at room temperature.
β-CD–menthol inclusion complexes were also obtained by using a hydrothermal process as described in literature [26]. A certain amount of carboxylated β-CD and menthol was added to 80 mL of deionized water at room temperature at the molar ratio of 1:1. The mixture solution was transferred to a 100 mL PTFE-lined stainless steel autoclave, which was sealed and heated up to 120℃ in a furnace for 4 h while rotating at 10 r/min. When the solution cooled down to room temperature, the white precipitant of β-CD–menthol inclusion complexes was obtained after being filtered, washed several times using anhydrous ethanol, and dried in a vacuum oven at 40℃.
Characterization techniques
All samples were purified with acetone, anhydrous ethanol, and deionized water prior to characterization. The morphologies of the resultant magnetized fabrics and β-CDs prepared at different stages were examined using a ZEISS EVO18 field emission scanning electron microscope. Energy dispersive X-ray analysis was conducted in an Oxford INCA Energy 350 EDX system attached to the field emission scanning electron microscope.
The crystal structure of the magnetite particles on the resultant magnetized polyester fabrics loaded with menthol was analyzed using a 7000S XRD spectroscopy diffractometer equipped with Cu Kα1 radiation (λ = 0.154056 nm, 40 kV and 40 mA) at a scan step of 0.02° and a scan rate of 5° per minute. The crystallite size of synthesized magnetic particles was calculated using Scherrer’s equation D = Kλ/ɛcosθ (where D is the diameter of a particle, λ is the X-ray wavelength, ɛ is the FWHM of the diffraction line, θ is the diffraction angle, and K is a constant of 0.89).
The chemical structures of the resultant magnetized fabrics and β-CDs prepared at different stages were determined via FTIR spectra analysis on a Perkin Elmer Frontier FTIR instrument in the range of 650–1800 cm−1 with a resolution of 4 cm−1 based on the attenuated total reflection mode.
The thermal properties of the resultant fabrics and β-CDs prepared at different stages were analyzed using thermogravimetry/derivative thermogravimetry (TG/DTG) analysis on a TGA Q500 instrument from 40℃ to 550℃ at a heating rate of 10℃ per minute and nitrogen flush rate of 20 mL per minute. The DSC analysis was performed on a DSC Q2000 instrument from 40℃ to 350℃ under the same conditions.
The changes of their optical properties of the resultant fabrics and β-CDs prepared at different stages were investigated on a PE LAMBDA 950 UV-VIS-NIR spectrophotometer equipped with a 150 mm integrating sphere at a scan rate of 120 nm per minute over the wavelength 200–800 nm.
Measurement of menthol contents
Two suspension solutions, solution A and solution B, were made first. Hundred milliliters of the suspension solution A was made by adding 0.5 g of 4-dimethylaminobenzaldehyde into 100 mL of sulfuric acid aqueous solution of 74% in concentration (prepared with the volumetric ratio of 98% sulfuric acid to deionized water at 1.6:1). A series of menthol aqueous solutions with various concentrations (1, 3, 4, 6, 8, and 10 g/L) were prepared by adding menthol of 0.1, 0.3, 0.4, 0.6, 0.8, and 1.0 g into 100 mL of a solution of the mixture of anhydrous ethanol and deionized water in the volume ratio of 1:1, respectively, which was denoted as solution B. One milliliter of menthol solution B was completely mixed with 5 mL of solution A. The mixed solution was heated in water bath at 100℃ for 2 min, then immediately cooled down. This mixed solution exhibited red grape color, and its absorbance at the maximum absorption wavelength of 550 nm was measured on a UV-1601 UV-VIS spectrophotometer (Beijing Beifen-Ruili Analytical Instrument (Group) Co., Ltd.). The reference solution is the solution of 1 mL of mixture of anhydrous ethanol and deionized water mixed with 5 mL of solution A. A calibration curve for menthol solution could thus be obtained from the plot of absorbance against concentrations of menthol in resultant solutions.
The magnetized polyester fabric loaded with menthol was kept in ventilated glass bottle and stored at constant air condition (temperature at 25 ± 3℃ and relative humidity of 65 ± 3%). The content of menthol loaded on the magnetized polyester fabric was obtained by first dissolving menthol loaded in the fabric of 5 g in 100 mL of deionized water at 100℃ and then collecting the distilled menthol from this water solution. The distillate menthol was then dissolved in anhydrous ethanol solution and mixed with the solution A. The concentration of the mixture solution could be obtained by measuring its UV-VIS absorbance against calibration curve. Thus, the dynamic release rate of menthol from the magnetized polyester fabric at room temperature was obtained through the measurement of the changes of the contents of the menthol loaded on the magnetized polyester fabric at regular time intervals. The percentage of menthol released from the resultant fabric at time t, Mt(%), is defined as the relative change of the menthol contents in the fabric in relation to its initial menthol concentration, as shown in equation (1) below:
The magnetic hyperthermia properties of the magnetized polyester fabric were measured using a GP-25 A high frequency magnetic induction heating power supply (Shaanxi Let Big Power Technology Co., Ltd.) with rated frequency of 70 kHz, and rated capacities of 1.5 kW and 0.6 kW, respectively. A piece of magnetized fabric sample (4.5 cm × 4.5 cm) in a glass dish was placed on the surface of its magnetic heating induction coil. The temperature of the fabric sample was monitored with a GM531 infrared thermometer (Shenzhen Benetech Co., Ltd.), and the dynamic temperature changes of magnetic fabrics were obtained from the average of dynamic temperature changes of three fabric specimens measured in this system. The schematic diagram is illustrated in the inset of Figure 8(b). The reusability of the magnetic polyester fabric loaded with menthol was also measured. The fabric sample was first heated in an oven at 100℃ for 1 h to release the menthol included within β-CDs, and re-loaded with menthol according to the method described in section “Inclusion of menthol.” The morphology and chemical composition of the magnetized polyester fabric re-loaded with menthol were examined by employing FESEM and EDX techniques, respectively.
Results and discussions
The morphology and chemical composition of resultant magnetized polyester fabrics loaded with menthol
The morphologies of polyester fabrics obtained at different stages are shown in the FESEM images in Figure 2. The surface of the original polyester fabric is clean and smooth without any other particulate substances observed (Figure 2a). After the polyester fabric is treated with alkali solution at 100℃ for 30 min, many tiny indentations are formed on fiber surface (not given). This is due to the hydrolysis of ester linkage. These small cavities play a positive role in anchoring the deposition of magnetic particles. The tensile properties of the polyester fabrics before and after alkali treatment are measured using a YG(B)026D-500 tensile tester according to GB/T3923.1-1997. The gauge length is 200 mm and the fabric width is 50 mm. The testing rate is 100 mm/min and the pretension is 2 N. The tensile strength of the fabrics in warp direction decreases from 633.2 N for untreated fabrics to 577.4 N for alkali treated fabrics, and in weft direction from 405.4 N to 367.0 N. The corresponding elongation decreases from 20.7% to 15.8% in warp direction and from 25.5% to 19.7% in weft direction, respectively. After the polyester fabrics are magnetized, a thin layer of granulates and agglomerated blocks of magnetic particles are deposited on the surface of the magnetized polyester fabrics. Many of granulates and agglomerates are in micrometer sizes, and are composed of submicrometer-sized particles less than 200 nm as shown in the upper right corner in the high-resolution FESEM image (Figure 2b). The magnetized fabrics modified with KH550 and grafted with β-CD are coated by a thick film of paste-like substances and scattered with some small particulates (Figure 2c). After the fabrics are loaded with menthol, no other distinct change is observed except the fabric surface becomes slightly smoother (Figure 2d).
FESEM images of polyester fabrics obtained at different stages of treatments: (a) original; (b) magnetized fabric; (c) modified with KH550 and grafted with β-CD; (d) loaded with menthol.
The composition analysis of these polyester fabrics obtained at different stages is shown in the EDX spectra in Figure 3, and the atom percentages of the elements of C, O, N, S, and other metal elements are detected. It is noticed that the atom percentages of both C and O elements in the original polyester fabric are 80.1% and 19.9%, respectively (Figure 3a), while additional elements of N (2.6%), Na (0.9%), S (5.4%), and Fe (41.3%) besides C (22.4%) and O (27.4%) are found in the surface of the magnetized polyester fabrics, indicating the existences of iron oxides. The element N, Na, and S are likely from the residues of the contamination of the precipitant of urea and reducing agent of sodium thiosulfate (Figure 3b). After the magnetized polyester fabrics are modified with KH550 and grafted with β-CD (Figure 3c), apart from the elements of C (51.4%), N (6.4%), O (27.1%), Na (0.7%), S (0.4%), and Fe (5.2%), a small amount of element Si (8.8%) is detected and this indicates the incorporating of Si from the silane coupling agent, KH550, into the fabrics. It is also noticed the significant increase of the contents of elements C and N and the marked decrease of element Fe in comparison with the element contents on the surface of magnetized polyester fabrics. It is thus reasonable to conclude that magnetic particles are covered by grafted β-CD, of which the additional C and N elements are detected from. The chemical compositions of the magnetized polyester fabric loaded with menthol in terms of atom percentages are C (68.8%), O (24.2%), Na (0.4%), Si (4.2%), and Fe (2.4%), respectively. This means that the magnetic particles, silane, and grafted β-CD deposited on the fabric surface are still detectable. However, the elements of N and S are not detected, this might be because the two elements from the contaminations are re-dissolved and washed away from the fabric during hydrothermal inclusion process and subsequently in washing process (Figure 3d).
EDX spectra of polyester fabrics obtained at different stages: (a) original; (b) coated with particles; (c) modified with KH550 and then grafted with β-CD; (d) loaded with menthol.
The morphology and chemical composition of β-cyclodextrin loaded with menthol
The morphologies of β-CDs modified at different processing stages are shown in their FESEM images in Figure 4, and their EDX spectra are also inserted in their corresponding FESEM images. The untreated β-CD agglomerates together to form larger clusters with irregular shape (Figure 4a). β-CD modified with maleic anhydride appears to be in the form of smaller powder particles (Figure 4b). After inclusion of menthol, the smaller β-CD particle with lamellar structure is formed (Figure 4c). It is also observed that only two elements, C and O, are detected for all of these samples. However, the atomic ratios of C to O in them are quite different. After treatment with maleic anhydride, the atomic content of C in the β-CD has a slight change from 63.5% to 64.3%, but increases significantly to 68.4% after subsequent inclusion with menthol. Meanwhile, the atomic content of O also slightly changes from 36.5% to 35.7% after being treated with maleic anhydride, then markedly decreases to 31.6% after following by inclusion with menthol. Thus, it is confirmed in the EDX elemental composition analysis that the changes of organic substances occur in β-CD after each chemical modifications. This is because β-CD has a cavity of approximately 7.0 Å in diameter and 7.5 Å in depth [4]. The cavity has a hollow hydrophobic interior and hydrophilic exterior and thus can act as a host to catch other small guest molecule chemicals inside it. The driving force for this host–guest complex formation is attributed to the hydrophobic and hydrophilic interactions [27].
FESEM images and EDX spectra of β-CDs obtained at different stages: (a) untreated; (b) carboxylated; (c) included with menthol.
Changes of characteristic groups in polyester fabrics and β-CDs analyzed using FTIR techniques
Polyester fabrics
The characteristic bonds of polyester fabrics obtained at different stages are shown in the FTIR spectra in Figure 5. The characteristic bands of the original polyester fabric appear in the FTIR spectrum at 1505 cm−1 (C─C stretching vibration of benzene ring skeleton, ν(C─C)), 1408 cm−1 (─CH2COCH2─ absorption band), 1339 cm−1 (CH2 wagging vibration, ωCH2), 1240, 1176, 1094, and 970 cm−1 (C─O stretching vibration, ν(C─O)), 1017 and 872 cm−1 (C─H out-of-plane bending vibration of benzene ring, γC─H), 847 cm−1 (CH2 rocking vibration, ρCH2), 792 and 723 cm−1 (CH out-of-plane bending vibration of benzene ring, γCH) cm−1. In addition, the carboxyl band at 1714 cm−1 (C═O stretching vibration, ν(C═O)) forms as a result of the hydrolysis reaction of the ester bond of polyester etched by alkali [28] It is also noted that, after the fabric is deposited with magnetic particles, the frequency of this band (C═O stretching vibration, ν(C═O)) slightly shifts to 1711 cm−1, and the ν(C─O) bands at 1240, 1094, and 970 cm−1 are also shifted to 1235, 1087, and 971 cm−1, respectively. After it is treated with KH550, four additional bands form in the fabric at 1195, 1080, 1008, and 693 cm−1 due to Si─O─Si groups in the silane coupling agent [29]. After the fabric is grafted with a thick layer of carboxylated β-CD in the fabric, two broad ν(C─O) bands at 1103 and 1003 cm−1 and two small bands at 752 and 691 cm−1 are noticed. In the magnetized polyester fabric loaded with menthol, the ν(C─O) bands shift to 1238, 1084, and 1017 cm−1 due to the existence of menthol [30].
FTIR spectra of polyester fabrics obtained at different stages.
β-Cyclodextrins and menthol
The interactions involved in the formation and stabilization of supramolecular assemblies with β-CD were investigated by using FTIR [31]. The FTIR spectra of β-CDs prepared at different stages as well as menthol are shown in Figure 6. It is apparent that the characteristic band at 1648 cm−1 in the FTIR spectrum of untreated β-CD is ascribed to the O─H in-plane bending vibration (β(O─H)), the band at 1153 cm−1 is attributed to the anti-symmetric glycosidic C─O─C stretching vibration (νa(C─O─C)) and the band at 1079 cm−1 corresponds to the C─O bending vibration (δ(C─O)). The bands at 1023 and 1000 cm−1 are assigned to the coupled C─C and C─O stretching vibrations (ν(C─C/C─O)). The band at 946 cm−1 corresponds to the α-1,4-bond skeleton vibrations of β-CD [18]. The band at 851 cm−1 is due to the glycosidic linkage of pyran rings. The bands at 755 and 705 cm−1 are caused by the vibrations of β-CD ring [19]. After treatment with maleic anhydride, the ν(C═O) band at 1723 cm−1 appears. The β(O─H) and δ(C─O) bands are shifted to 1634 and 1078 cm−1, while the ν(C─C/C─O) bands at 1023 and 1000 cm−1 are shifted to 1026 and 1003 cm−1, respectively. Meanwhile, the band at 851 cm−1 is shifted to 863 cm−1. After inclusion of menthol, being influenced by the absorption bands at 1105 and 1078 cm−1 (C─C stretching vibration, ν(C─C)), as well as 1045 and 1026 cm−1 (C─O stretching vibration, ν(C─O)) of menthol, the ν(C─C/C─O) bands at 1026 and 1003 cm−1 are slightly shifted to 1024 and 1002 cm−1, respectively. In addition, the β(O─H) band at 1648 cm−1 is shifted to 1643 cm−1 and a new band at 1101 cm−1 appears. Therefore, the formation of the inclusion complex of menthol and carboxylated β-CD is confirmed in the FTIR results.
FTIR spectra of β-CDs obtained at different stages and menthol.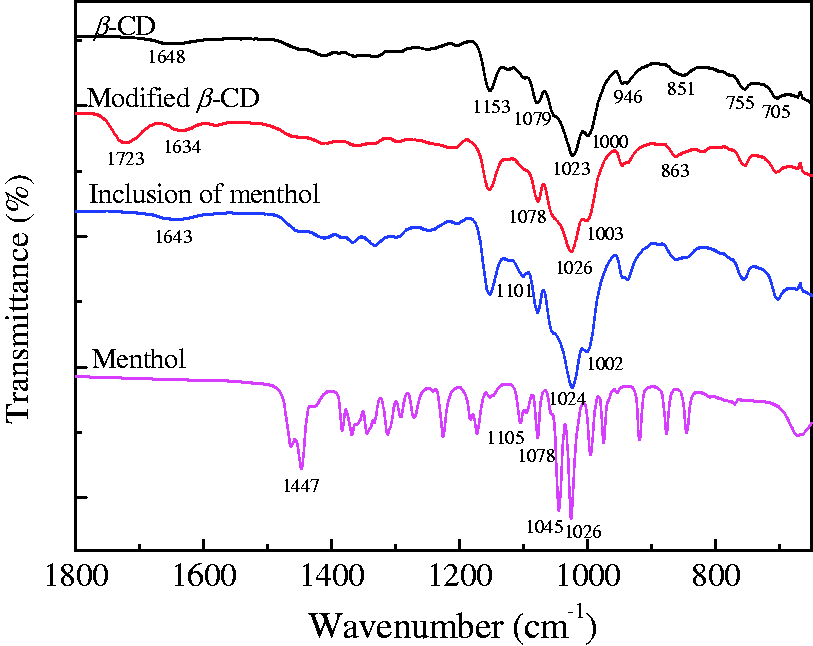
X-ray diffraction
The XRD pattern of the resultant magnetized polyester fabric loaded with menthol is shown in Figure 7. The diffraction peaks at 2θ=18°, 23°, and 26° are attributed to {010}, {#x02212;110}, and {110} planes of typical polyester polymer phase. A series of characteristic peaks at 2θ=30.4°, 35.7°, 37.3°, 43.3°, 53.7°, 57.2°, and 62.7° are noticed, which are ascribed to the planes of {220}, {311}, {222}, {400}, {422}, {511}, and {440} in the magnetite (Fe3O4) with a cubic spinel type structure (JCPDS card no. 19-0629) [32]. In addition, several small diffraction peaks at about 33.2°, 40.9°, 44.5°, and 64.8° are also detected, which are assigned to the planes of {104}, {113}, {202}, and {300} in the rhombohedral hematite (Fe2O3) (JCPDS card no. 33-0664) [33]. Thus, the coating of magnetic particles, which are mainly constituted with magnetite Fe3O4 together with a small amount of hematite Fe2O3, on the surface of polyester fabric is evident. The average crystallite size of the synthesized magnetic particle is 23.0 nm calculated by using Scherrer’s equation based on the width of the peaks at 30.4°, 35.7°, and 62.7°, which is far smaller than that measured from the FESEM image. However, the crystal structure of β-CD inclusion complex is not detected, this might be because of the relative small amount of β-CD and menthol with respect to other components.
XRD pattern of the resultant magnetized and menthol loaded polyester fabrics. (a) Magnetic hysteresis loop of the finally obtained polyester fabric loaded with menthol and (b) magnetocaloric heating curves of the magnetized polyester loaded with menthol under alternating current magnetic field at 70 kHz.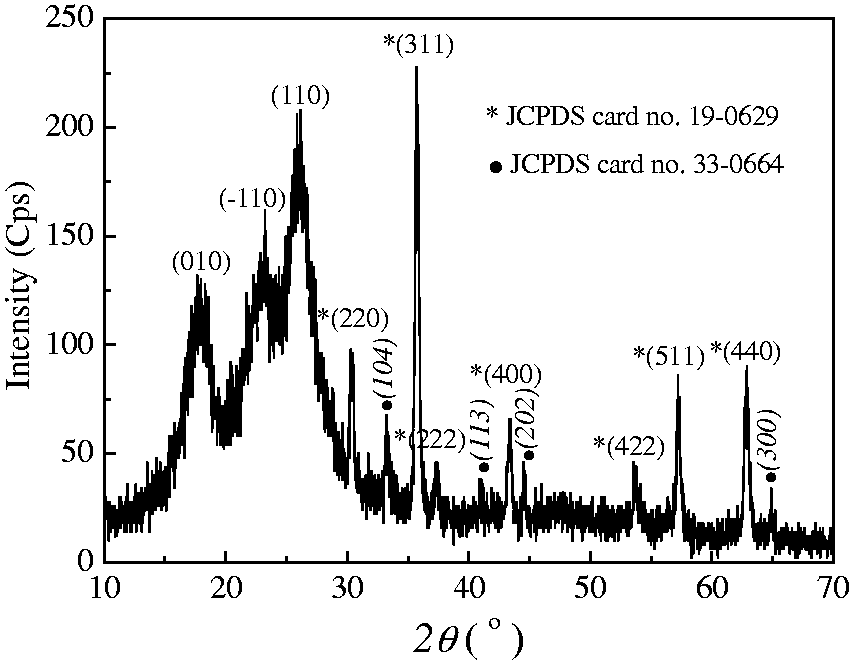
Magnetic properties and hyperthermia effect of magnetized polyester fabrics
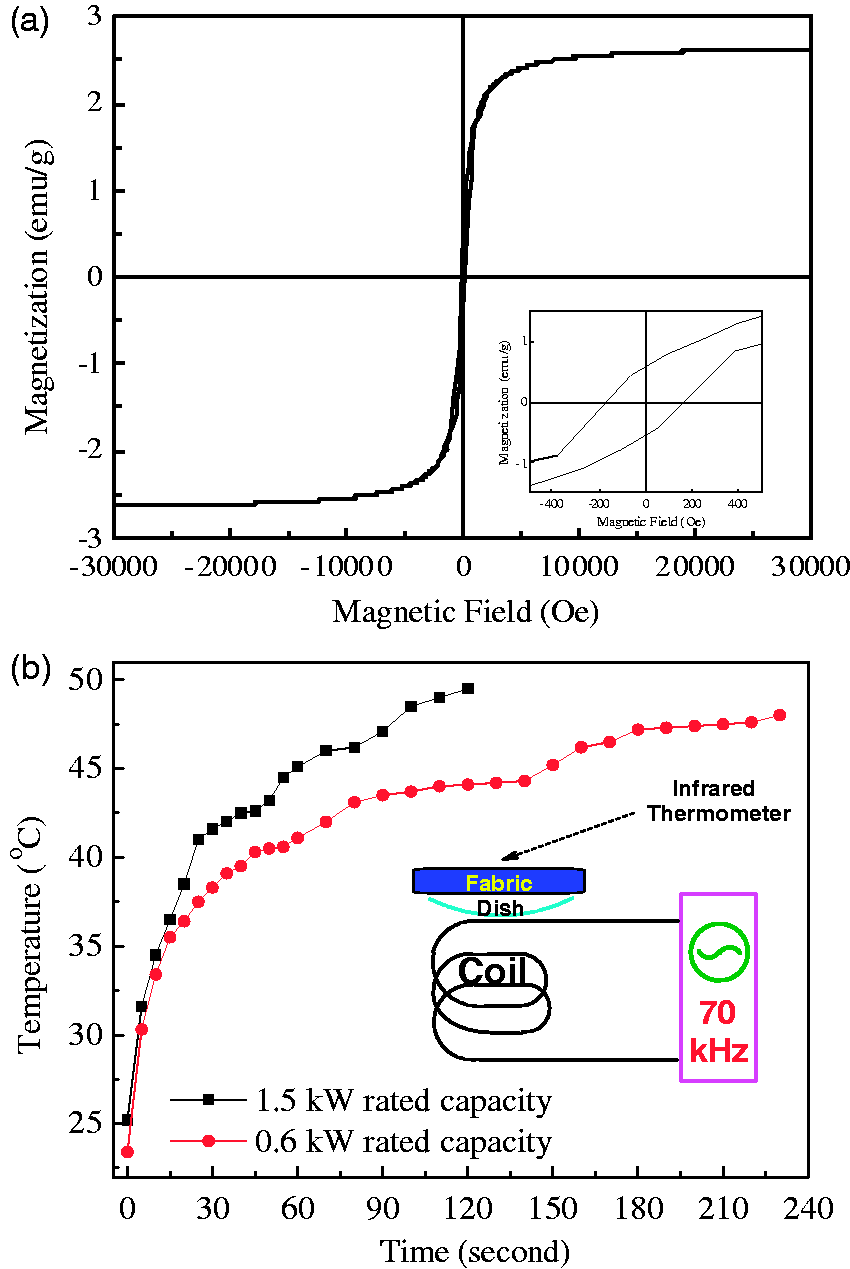
The hysteresis loop of the resultant magnetized polyester fabric loaded with menthol at room temperature is shown in Figure 8(a). It is evident that the magnetization sharply increases with the increases of the applied magnetic field intensity when it is smaller than 5000 Oe, and the magnetization hardly changes after the magnetic field intensity is greater than 10,000 Oe. The magnetized polyester fabric loaded with menthol has a saturation magnetization of 2.62 emu/g, and the coercivity force is determined as 0.56 emu/g and the remnant magnetization is calculated as 170 Oe from the inset in Figure 8(a).
The magnetocaloric heating curves of the magnetized polyester fabrics loaded with menthol under external high frequency alternating current magnetic field at 1.5 kW and 0.6 kW rated capacities are shown in Figure 8(b). It is clear that the temperature of the magnetized polyester fabric increases rapidly in the early stage of heating process (less than 30 s) under a specific alternating current magnetic field. After 90 s, the temperature of the magnetized polyester fabric increases slowly with the elapse of heating time. Under the magnetic field of 70 kHz in frequency, it takes 120 s for the magnetized polyester fabric to reach 49.5℃ when the magnetic field at the rated capacity of 1.5 kW, and it takes 230 s to rise to 48℃ when the magnetic field at the rated capacity of 0.6 kW. The heat generated by magnetic particles is attributed to hysteresis loss, Neel and Brownian relaxation mechanisms [34].
Thermal stabilities
Resultant polyester fabrics
The TG/DTG and DSC curves of the resultant polyester fabrics obtained at different stages are shown in Figure 9. It is noted from TG/DTG curves that when the original polyester fabric is coated with magnetic particles, the heat decomposition temperature decreases from 380.8℃ to 358.8℃. After the magnetized fabrics are modified with KH550, their heat decomposition temperature is further shifted to 372.7℃. After the resultant fabrics are grafted with carboxylated β-CD, their heat decomposition temperature has no change and kept at 372.7℃. When loaded with menthol, the heat decomposition temperature increases slightly to 374.8℃. The weight losses are 95.4%, 96.3%, 95.5%, 94.9%, and 90.2% for the original, magnetic particle coated, modified with KH550, grafted with β-CD, and menthol loaded polyester fabrics at the temperature of 550℃, respectively. The thermal decomposition process can be divided into two periods for all samples except for the magnetic particle coated polyester fabrics (see inset of Figure 9a). It is noticed from DSC curves that the endothermic peaks change a little, which are in the temperature range from 250.0℃ to 255.2℃. The reaction enthalpies are 60.5, 64.8, 51.5, 58.7, and 47.8 J/g for the original, magnetic particle coated, KH550 modified, β-CD grafted, and menthol loaded polyester fabrics, respectively. Moreover, a small endothermic peak at 20℃ with a corresponding reaction enthalpy of 5.97 J/g is observed in the final menthol loaded polyester fabric (see inset of Figure 9b), this suggests that the menthol molecules are included inside the β-CD cavity. As a summary, it is concluded that the menthol is loaded onto the magnetized polyester fabrics, and the successive fabric processing procedures only have slight effects on the thermal stability of polyester fabrics in comparison with original polyester fabrics.
TG/DTG (a) and DSC (b) curves of polyester fabrics obtained at different processing procedures.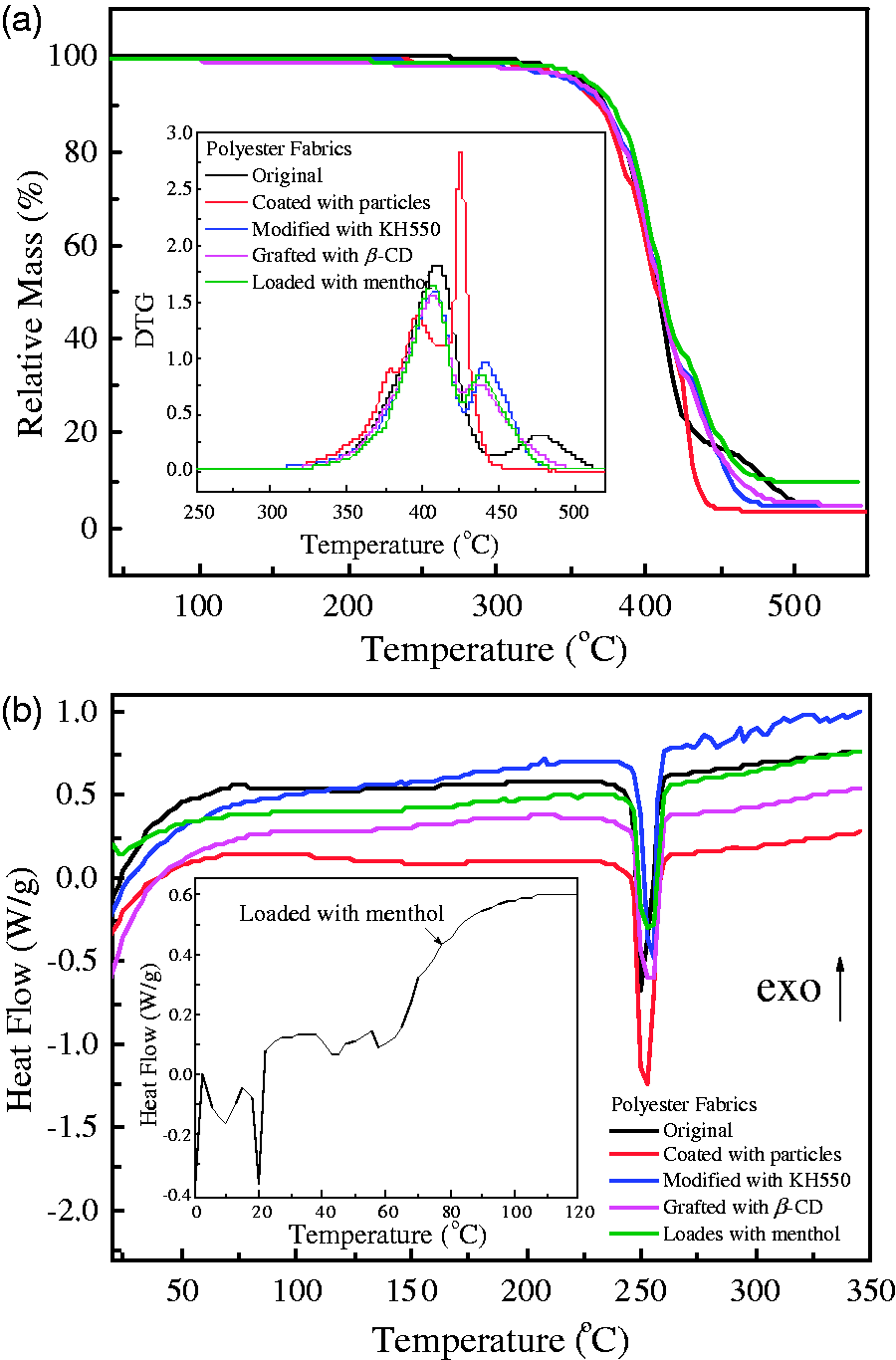
β-Cyclodextrins
The TG/DTG and DSC curves of β-CDs obtained at different stages are shown in Figure 10. It is clear from TG/DTG curves that there is a low initial decomposition temperature at 89.5℃ and a main decomposition temperature at 318.6℃ in the TG/DTG curve of the untreated β-CD. After treatment with maleic anhydride, the initial decomposition temperature nearly disappears and the main heat decomposition temperature decreases significantly from 318.6℃ to 262.7℃. After inclusion of menthol, the heat decomposition temperature increases to 307.5℃. The weight losses are 79.0%, 76.1%, and 87.5% for the untreated, carboxylation modified, and menthol included β-CDs at the temperature of 550℃, respectively. It is obvious from DSC curves that there are two endothermic peaks at 90.0℃ and 327.5℃ for the untreated β-CD, and the corresponding reaction enthalpies are 49.9 and 44.1 J/g. For the carboxylation modified β-CD, the initial endothermic peak shifts to 77.4℃, while the main endothermic peak disappears. After inclusion of menthol, its endothermic temperatures occur at 96.8℃ and 310.3℃, and the corresponding reaction enthalpies are 20.5 and 80.2 J/g. In brief, it is concluded that the thermal property of carboxylated β-CD included with menthol apparently changes.
TG/DTG (a) and DSC (b) curves of β-CDs obtained at different stages.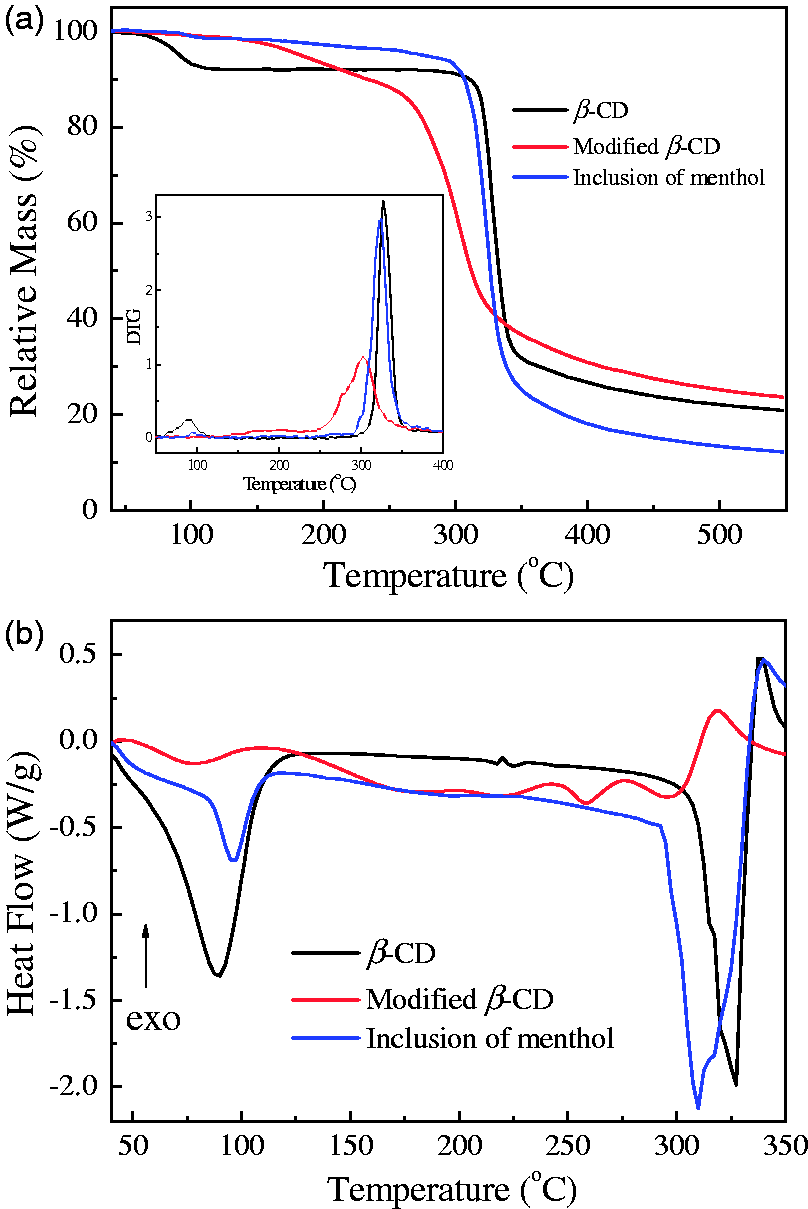
Optical properties
Polyester fabrics
The diffuse reflectance spectra of polyester fabrics obtained at different stages are shown in Figure 11. It is clear that the typical reflectance spectrum is noticed in the original polyester fabric, and the characteristic absorption peaks are observed at about 228 nm and 300 nm and there is a significant increase of reflectance in the range of wavelength between 300 nm and 400 nm. For the magnetic particle coated polyester fabrics, the reflectance is very small (less than 5%) in the whole range of wavelength between 200 nm and 800 nm owing to the deposition of black magnetic particles. After the fabric is treated with KH550, the reflectance gradually increases from around 5% to around 10% with the increases of wavelength across the whole wavelength (200–800 nm). After being grafted with carboxylated β-CD, the overall reflectance decreases slightly [35]. However, after being loaded with menthol, the reflectance increases to the similar level of the modified polyester fabrics.
Diffuse reflectance spectra of polyester fabrics obtained at different stages.
β-Cyclodextrins
It has been demonstrated that the UV and/or visible absorption spectrum of the guest molecules may be altered to some extent once the β-CD inclusion complex is formed [4]. The diffuse reflectance spectra of menthol and β-CDs obtained at different stages are shown in Figure 12. It is evident that when β-CD is modified with maleic anhydride, a significant red shift is noticed. After inclusion of menthol, a broad reflectance spectrum (230–380 nm) is observed, which is mainly influenced by the characteristic spectrum of menthol. This is ascribed to the partial shielding of the excitable electrons in the β-CD cavity [36].
Diffuse reflectance spectra of β-CDs obtained at different stages.
Measurement of menthol contents
Menthol is gradually released from the magnetized fabrics in ambient conditions (20℃, 65% relative humidity). The dynamic change of the percentage of menthol released from the magnetized polyester fabric in ambient conditions is shown in Figure 13(a). The relationship between the UV-VIS absorbance of menthol and its concentration of menthol is given in the inset of Figure 13(a). The average of menthol content remained in the magnetized polyester fabric is about 8% after 90 days of menthol release.
(a) Dynamic rate of menthol release from the resultant magnetized polyester fabric and (b) FESEM image of the magnetized polyester fabric loaded with menthol after five repeated inclusions.
It was found that the menthol residue in the magnetized polyester fabric was 7.9% after it was gradually released in ambient temperature for 90 days. For comparison purposes, a controlled accelerated release of menthol from the magnetized polyester fabrics under electromagnetic induced hyperthermia (see section “Magnetic properties and hyperthermia effect of magnetized polyester fabrics”) was also performed. When the magnetized polyester fabrics were heated up under electromagnetic induced hyperthermia at 1.5 kW rated capacity for 120 s, the menthol contents remained in the fabrics are 7.2%; when the fabrics were heated up at 0.6 kW rated capacity for 230 s, the residue menthol contents are 7.6%. It is evident that there is a controlled and accelerated release of menthol onto targeted surface when electromagnetic induced hyperthermia heating is introduced in the magnetized polyester fabrics.
After five cycles of repeated inclusion of menthol, the FESEM image of the magnetized polyester fabric loaded with menthol is shown in Figure 13(b). It is found that part of substances coated on the fabric surface fall off due to repeated hydrothermal treatments. It is indicated in the EDX results that the composition elements of the magnetized polyester fabrics loaded with menthol repeatedly are composed of elements C (64.1%), O (35.3%), Na (0.1%), Si (0.4%), and Fe (0.1%).
Conclusions
In this study, a technical route to fabricate magnetized polyester fabrics grafted with β-CD loaded with menthol mainly based on hydrothermal method is reported. The characteristics of the structure and properties of magnetized polyester fabrics prepared at various treatment stages are analyzed and discussed. The magnetic particles deposited on the fabric surface are primarily constituted with magnetite Fe3O4 and a small proportion of hematite Fe2O3. A thick layer of β-CD submicrometer-sized particles and membrane is coated on the surface of the magnetized polyester fabric. The magnetized polyester fabric exhibits a saturation magnetization of 2.62 emu/g, coercivity force of 0.56 emu/g, and remnant magnetization of 170 Oe. The magnetized polyester fabric loaded with menthol in carboxylated β-CD has demonstrated both apparent hyperthermia effect and the capability of controlled accelerated release of menthol on targeted surface; this fabric is able to be reloaded with menthol repeatedly.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study has been supported by the cooperative innovational center for technical textiles (Grant No. 2015-ZX21), Shaanxi Province of China. The corresponding author also acknowledges the Youth Leading Scholar Supporting Plan and the Innovation Fund of Postgraduate (No. cx201620) of Xi’an Polytechnic University.

