Abstract
Because of important potential and application prospect of aligned scaffolds in tissue engineering, it is necessary to prepare aligned scaffolds different from previous methods. We have prepared poly(lactic acid) fiber/polyurethane adhesive composite-aligned scaffolds by 1000 poly(lactic acid) melt parallel arrangement fibers and different polyurethane contents at 5, 10, 20, 25, and 30% separately. It can be found that polyurethane contents have great influence on bonding effect between fiber and adhesive, surface and cross-sectional morphology, thickness, weight, contact angle, stress and strain, pore diameter, porosity, pore interconnectivity, water absorption, and gelatin impregnation. The maximum of pore diameter and porosity of aligned scaffolds can be achieved to 64.24 µm and 66.67% by controlling poly(lactic acid) fiber parallel arrangement and polyurethane adhesive content. Moreover, the ultimate stresses of aligned scaffolds are 3.47 MPa along length direction and 1.02 MPa in width direction. Each composite-aligned scaffold has better fiber parallel arrangement, pore structure, and stress.
Introduction
Poly(lactic acid) (PLA) is a compostable and aliphatic polyester thermoplastic polymer that is derived from renewable resources, having high strength and stiffness but low impact strength. Hydrolysis of ester bond, temperature, moisture, and microbial environment plays an important role in PLA degradation [1]. PLA also has properties of nontoxic, processable, hydrophobic, and biodegradable [2]. Biocomposite consists of a biodegradable polymer as matrix material and fiber as reinforcing element [3]. Many PLA composite materials having good performances can be applied as porous scaffolds in tissue engineering. Synthetic graft PLA scaffolds manufactured by high pressure carbon dioxide mixing method can be used as a biological bone graft extender in impaction bone grafting [4]. PLA–poly(ethylene glycol)(PEG)/hydroxyapatite composites are used in bone tissue regeneration [5]. PLA/Ca-deficient hydroxyapatite nanocrystals prepared by solvent-cast technique are fit for bone-like substitutes. Polyglycolide/PLA composites made by melt processing with pores, discrete material regions, and biomimetic internal architectures suit for multitissue and structural tissue interface engineering [6]. PLA/calcium metaphosphate composite prepared by sintering method is applied in bone tissue engineering [7]. PLA/hydroxyapatite fibers (40–150 µm length and 2–10 µm diameter) ceramic–polymer composite biomaterials produced by hot pressing at 40 MPa and 180℃ [8] are used for bone tissue engineering. Poly(
Polyurethane (PU), a wide-ranging and modular-like polymer, is made from polyaddition of polyisocyanates and macropolyols in the polymer backbone. The polymer building blocks, the stoichiometry of isocyanate/OH components, and the reaction sequence have great influence on PU properties. Due to superior performances of PU, it has a very wide application range, i.e. foam, elastic fibers, thermplastic PU, spray elastomers, cast elastomers, synthetic leather, shoe sole, noise vibration harshness in automotive, integral foam, fiber composite materials, paints, coating, and adhesive [12]. PUs prepared from polyester polyols, aliphatic diisocyanates, and chain extenders, which have good mechanical properties, biocompatibility, and biodegradability can be used in biomedical fields [13]. pH-sensitive PUs can be used in intracellular multifunctional antitumor drug delivery [14], PUs synthesized with the process of microarray fabrication are applied in isolating human skeletal progenitor cells and augmenting skeletal cell growth [15], and block PUs based on poly(ε-caprolactone) (PCL) and PEG scaffolds are used in peripheral nerve regeneration [16]. PU can also be made for functional biomaterials [17], possessing good mechanical properties and bactericidal activity against Staphylococcus aureus and bacteriostatic activity against Escherichia coli [18], and fitting for a wide range of implantable and sterilized medical devices [19]. Other applications such as vascular prostheses [20], soft tissue repair and reconstructive procedures [21], new bone formation [22], bone glues [23], small vascular graft [24], nerve guidance with smoother surface [25], tissue regeneration [26], prosthetic heart valve leaflet [27], and implantable drug delivery [28]. Many PU composite materials can also be applied in medical aspects, i.e. PU/microsphere composite scaffolds [22], PU-hyaluronic acid copolymers [29], PU/liquid crystal composite membranes [30], PU/poly(lactic-co-glycolic) acid composite scaffolds [31], PCL–PU composite vascular scaffold [32], and PU/1,4-butanediisocyanate meniscal scaffold [33].
Scaffold chemistry, biofunctionalization, mechanics, and structure are key in determining cell response to scaffold designs [34]. As for medical composite scaffolds, porosity and pore diameter have great influence on in vitro and in vivo properties [35]. The interconnected macroporous network can allow for cell/tissue growth and flow transport of nutrients for in vitro and in vivo tissue [36]. The alignment degree is an important parameter to characterize degree of fiber parallel arrangement in scaffolds, which is also stated in the research of Nisbet [37], Guo [38], Ayres [39], and Lee et al. [40]. The direction of fibers arrangement in scaffolds can be referred to as alignment degree and it is a very important parameter. Aligned scaffolds can significantly induce the formation of muscle cell arrangement and muscular tube [41], guide cell bonding and migration direction in the same fiber arrangement direction [42], increase extracellular matrix [43], provide better biomimetic structure and mechanical environment in the body [43] in comparison with disordered scaffolds. The commonly used methods for preparing scaffolds consist of fiber bonding, solvent casting, particulate leaching, membrane lamination, and melt molding [44]. A successful scaffold is capable of creating porous structures to attain desired mechanical function, to transport impregnation and diffusion, to produce arbitrary and complex three-dimensional (3D) anatomical shape structure [45]. The thickness of scaffolds can affect cell viability and uniformity, and neocartilage formation [46]. Moreover, 3D sectional cell viability is obtained through thickness of polymer scaffolds from upper to lower surface [47]. The mechanical properties are important for porous scaffolds, especially for load bearing [48] in bone tissue engineering [35]. The surface property can influence the performance of a biomaterial in biological environment and the hydrophilicity of material is an important factor in cell adhesion and growth [49]. The water absorption is a direct parameter to express well-controlled pore architectures of scaffolds [50]. Hosseinkhani et al. [51] used gelatin and DNA to impregnate porous scaffolds, Costa et al. [52] studied the impregnation of acetazolamide and timolol in poly(glycolic acid) (PGA) fiber scaffold, and Stähli et al. [53] analyzed the impregnation time of scaffold with microporous and macroporous structure. So, it is necessary to measure the overall performance such as surface and cross-sectional morphology, alignment degree, chemical composition, pore diameter, porosity, thickness, weight, contact angle, water absorption, gelatin impregnation, and tensile property of aligned scaffolds.
Different from previous methods of preparing aligned scaffolds, in this paper, 1000 PLA melt fibers with better parallel arrangement first bonded by different PU contents at 5, 10, 20, 25, and 30% separately, then drying at 40℃ for 1 h with no pressure to prepare five various PLA/PU composite-aligned scaffolds. And the influence of PU content on internal structure and physical performance of PLA melt fiber-aligned scaffolds is studied for controlling fiber parallel arrangement, pore diameter, and porosity in the process of preparing scaffolds and meeting the performance requirements of ideal scaffolds in tissue engineering.
Materials and methods
Materials
Compared with other biodegradable synthetic polymers of PGA and PCL [54,55], it is easier to manufacture PLA melt fiber (Figure 1(a) to (d)) in practical spinning process. PLA melt fibers, with 54.77℃ glass transition temperature (Tg), 173.89℃ melting point temperature (Tm), and 47.17% crystallinity were manufactured by Donghua University (Shanghai, China). The fineness of PLA multifilament is 9.18 tex/72fiber and diameter of every PLA fiber in multifilament is 12.41 ± 0.89 µm (mean±standard deviation (SD)).
(a) PLA particles, (b) melt spinning process, (c) PLA melt fibers, (d) surface and cross-sectional morphology of PLA melt fiber, (e) fiber parallel arrangement, (f) PLA fibers bonded by PU adhesive, and (g) aligned scaffolds.
To achieve better orderly arrangement (Figure 1(e)), the PLA melt fibers with constant 1000 numbers were winded by YG086 electronic yarn measuring instrument under the condition of 100 r/min rotating speed and 100 cN tension. The YG086 electronic yarn measuring instrument was purchased from Changzhou First Textile Equipment Co., Ltd (Changzhou, China).
PU adhesive purchased from Hefei Huayue New Materials Technology Co., Ltd (Hefei, China) with 40% original content was used to prepare five different contents of PU at 5, 10, 20, 25, and 30%. 5, 10, 20, 25, and 30 mL 40% PU adhesives were first put into five 100 ml beakers, respectively, then 35, 30, 20, 15, and 10 ml purified water was mixed into five beakers, respectively, to prepare new adhesives. The beakers were kept in 03H-1 transfer magnetic stirrer for stirring 30 min to achieve homogeneous PU adhesive with 5, 10, 20, 25, and 30% for using.
Five brushes were, respectively, immersed in five different PU adhesives with 5, 10, 20, 25, and 30% content about 2 s first, and the brushes were taken out for brushing lightly on the surface of PLA fibers, respectively (Figure 1(f)). Then the previous steps were repeated for one time for performing better bonding effect between fibers. Five different wet-aligned scaffolds were manufactured at different PU contents of 5, 10, 20, 25, and 30% under the same condition of 1000 PLA fibers. Then five different wet-aligned scaffolds were cured at 40℃ for 1 h by DHG-9240A Electro-thermostatic blast oven (Shanghai Yiheng technology Co., Ltd, China) to achieve full drying and better bonding effect. The dried scaffolds (Figure 1 (g)) were removed from the oven for natural cooling at 27.3℃ room temperature and 81% humidity until completely cooled for experiment.
Methods
The ETD-2000 sputtering apparatus was used for gold plating of small samples 50 s for two times. Then the S-4800 (Hitachi, Japan) scanning electron microscopy (SEM) was carried out to observe the morphology of samples.
The value of alignment degree of PLA fibers in scaffolds can be measured by Image J software (National Institutes of Health, America) from SEM figures. A straight line was drawn parallel to x-axis along the horizontal direction of scaffolds. Then the angle formed between the fiber orderly arrangement direction and horizontal straight line direction, which can be used to express the alignment degree of fibers in scaffolds.
VECTOR 22 (Bruker Company, Germany) Fourier transform infrared spectroscopy (FT-IR), which had 4 cm−1 resolution, 32 s scanning time, and 4000–400 cm−1 wavelength range was applied to analyze the infrared spectrum of samples. The samples need to cut in a 1 cm×1 cm (length×width) patch and clamp it in the platform of laser tube for scanning.
Q20 (TA instrument, America) differential scanning calorimetry (DSC) was used to study the thermal properties of samples. The weight of samples is 4–6 mg, temperature range is 40–220℃, and the heating rate is 10℃/min. The TA Universal Analysis software was applied to analyze the change of Tg and Tm. The crystallinity of PLA can be calculated from the literature [56,57], where
The thermal degradation performance of samples was assessed by Q50 (TA instrument, America) thermogravimetric analyzer (TGA). The weight of samples in experiment is 4–6 mg, temperature range is 40–600℃, and the temperature ramp rate is 10℃/min in nitrogen (N2) atmosphere.
The static contact angle was measured by DSA100 type contact angle meter. Before experiment, the samples need to be cut in a size of 4 cm×2 cm (length×width) and then placed on the surface of the glass slide by double-sided adhesive tape to obtain the flat surface. The water droplets of 5 µl volume formed on the surface of samples were captured and calculated.
The thickness of scaffolds was measured by vernier caliper at five different positions to obtain mean value and SD.
BS 224S electronic balance manufactured by Dolly Scientific Instrument Co., Ltd (Beijing, China) with 0.1 mg–220 g test range was employed to measure the weight (g/cm2) of samples.
Liquid displacement method mentioned in Nazarov et al. [59] was applied to measure the porosity of scaffolds. Ethanol (C2H6O, molecular formula is 46.07) purchased from Sinopharm Chemical Reagent Co., Ltd (Shanghai, China) was used in the experiment. A total of 8–10 ml ethanol was first poured into a 100 ± 0.2 ml measuring cylinder and the value for V1 noted. The sample cut in a size of 1 cm×1 cm (length×width) was completely immersed in ethanol for 5 min and the value for V2 noted. Then the scaffold was gently removed from the measuring cylinder and the volume of residual ethanol for V3 noted. So, V2-V1 is the volume of scaffolds, V1-V3 is the pore volume of scaffold. The total volume of scaffold is
Image J software was applied to measure the vertical distance between two adjacent fibers, which could be considered as the pore diameter of scaffolds.
Pore interconnectivity of scaffolds can be analyzed directly from the cross-sectional morphology of scaffolds. First, the samples needed to be frozen in liquid nitrogen about 3 min and cut by blade along the longitudinal direction. Then a small piece of samples was used to observe the cross-sectional morphology of scaffolds.
Measuring method and calculation formula of water absorption can be referenced to previous literature, such as 1, 5,10, and 18 h absorption time in Yang et al. [60]. Five samples of each scaffold with size of 1 cm×1 cm (length×width) were measured in experiment for evaluating mean value. The wet weight (w2, g) of samples immersed in distilled water at 1, 2, 3, 4, 5, and 6 h was weighed. The value of water absorption percentage of scaffolds between dry weight (w1, g) and wet weight can be calculated by
Hundred milligram gelatin and 100 ml distilled water were first put in beaker and then kept them in drying oven at 60℃ and 1 h for complete dissolving. Then the beaker was kept in 03H-1 transfer magnetic stirrer for stirring 10 min to achieve uniform 1 mg/ml gelatin solution for using. Five samples of each scaffold with size of 1 cm×1 cm (length×width) were immersed in gelatin solution at 10, 20, 30, 40, 50, and 60 min to assess the results. The gelatin impregnation in scaffolds can be calculated from wet weight containing gelatin solution (w3, g), dry weight (w1, g), and volume of scaffold. The impregnation of scaffolds is
On the basis of references about stress [61] and strain [62], mechanical universal testing machine (Instron 5540A, America) was used to measure the tensile property of scaffolds at 100 mm/min tensile speed. The scaffolds were cut in a size of 2 cm×1 cm (length×width) by scissors and the clamping length was 1 cm×1 cm in experiment.
Statistical analysis
All values are carried out in accordance with mean ± SD in the form of writing, and the test number abbreviated to N.
Results
Surface morphology and alignment degree
It is indicated that the surface morphology from Figure 2(a) to (f) that these scaffolds have almost different pores on the surface of aligned scaffolds, suggesting the influence of PU adhesive content on surface morphologies of scaffolds is obvious. Before PU bonding, the smooth surface and parallel arrangement of PLA fibers can be easily seen from SEM figures, while it is impossible to completely see fiber morphologies after PU bonding. In addition, almost all PLA fibers are bonded together at different PU contents, and the bonding effect of fibers increases as PU content increases from 5 to 30%. It seems that the smaller pores and smoother surface are formed as the increasing of PU content, indicating the fibers in the scaffolds show a more and more close arrangement. In the process of experiment, the pore diameter and arrangement of fibers before bonding can be changed by any force. So, it is hard to obtain stable structure and performance without PU bonding for practical application in tissue engineering. It has no obvious difference of alignment degree of scaffolds before and after bonding from Figure 2(g), meaning that the PU adhesive content almost has no impact on alignment degree. So, scaffolds prepared by PLA fibers and PU bonding have better parallel arrangement. Chemical composition on the surface of aligned scaffolds at different PU contents obtained from SEM is shown in Figure 2(h), meaning nitrogen (N) shows an increasing trend while carbon (C) and oxygen (O) decrease as the increasing of PU content.
Cross-sectional SEM figure of (a) before PU bonding, and bonding at (b) 5%, (c) 10%, (d) 20%, (e) 25% and (f) 30% PU content, (g) alignment degree (N=10), and (h) chemical composition of aligned scaffolds prepared at different PU contents.
FT-IR, DSC, and TGA analysis
FT-IR spectra and peaks of PLA melt fiber-aligned scaffolds are shown in Figure 3(a). It can be found that there are four obvious peaks in different positions of FT-IR spectra curve, suggesting they have different changes. The spectra bands of PU at 3440 cm−1 of free N–H stretching vibration, 3348 cm−1 of hydrogen-bonded N–H, 1735 cm−1 of C=O urethane, 1550 cm−1 of N–H bending, and 1535 cm−1 of C–N are expressed in Vega-Baudrit et al. [63]. 2800–3000 cm−1 is CH stretching vibrations with antisymmetric and symmetric stretching modes of methylene groups, and 2795 cm−1 is O-CH2 stretching of PU [64].
(a) FT-IR spectra, (b) DSC thermograms, and (c) TGA curve of aligned scaffolds.
The peaks at 1725, 1463, 1370, 1174, and 1081 cm−1 are the characteristic structure of pure PLA [65]. Characteristic absorption bands at 1752, 1460, 1183, and 1090 cm−1 can be attributed to C=O stretching vibration, C–H deformation vibration, and C–O stretching vibration in PLA [66]. The experiments show that PUs have greater proportion of polyester polyol into the soft segment structure leading to higher degree of phase separation [67]. Phase separation of PU dispersions has correlation with thermal properties of PU adhesive composites. Peaks at 3742–3791, 2182–2388, 2016–2330, and 1918–2222 cm−1 in Figure 3(a) can be attributed to functional bands combination of PU adhesive and PLA melt fiber.
Tg = 53.14℃, Tm = 151.39℃, and 19.02% crystallinity of PLA membrane in Abdal-haya et al. [68], Tg = 60.4℃ and Tm = 165.1℃ of PLA electrospun fibrous mats in Shi et al. [69], Tg = 55.8℃ and Tm = 149.8℃ of PLA electrospinning membrane in Li et al. [70], Tg = 74℃ and Tm = 170℃ of PLA melt blowing nonwoven in Hammonds et al. [71], Tg = 64.3℃ and Tm = 172.4℃ of injection-molded neat PLA in Harris and Lee [1]. Tg = −52℃ and Tm = 49℃ of PU are obtained in Orgiles-Calpena's research [72], Tg = −14℃ and Tm = 47℃ of PU adhesive in Vega-Baudrit et al. [63]. Moreover, Tg can be increased by longer annealing time from 30 to 120 min at 90℃ and higher annealing temperature from 90 to 95℃ at 30 min annealing time to provide more chance for amorphous polymer molecular chains crystallizing [70].
DSC analysis of PLA melt spun fiber-aligned scaffolds.

TGA results of PLA melt spun fiber-aligned scaffolds.

Before 261℃, the curve of residual mass is basically parallel to x-axis, then the value of residual mass decreases sharply at 261.76–462.21℃ for decomposing the main chain of PLA, and finally it remains a straight line with no obvious weight loss as temperature is increasing from 462 to 600℃. In the initial temperature range, weight loss of scaffolds originates from the loss of moisture and trapped solvent [65]. In the process of temperature rising, the small molecules begin to volatilize readily and result in poorer thermal stability. Then the weight loss has a dramatic decrease mainly attributing to surface structural transformation of the crystal such as physical adsorption and chemical bonding, which is produced by the weak reaction between PLA fiber and PU bonding [65]. In Hammonds's study [71], the degradation of PLA melt blowing nonwovens begin at approximately 300℃, proceeds rapidly, and nears completion of mass loss at 375℃. It can be found that aligned scaffolds prepared with PLA melt fiber and PU adhesive in our experiment have higher temperature of 5% residual mass.
Pore diameter, porosity, and cross-sectional morphology
Three important parameters can be used to characterize the pore structure of aligned scaffolds, including pore diameter (Figure 4(a)), porosity (Figure 4(b)), and pore interconnectivity of cross-sectional morphology (Figure 4(c) to (g)). The results indicate that PLA fibers after bonding have smaller pore diameter in comparison with before bonding. Because of loose arrangement and easy deformation of PLA fibers before bonding, it is difficult to cut fibers by blade to achieve better cross-sectional shape and keep original pore characteristics. Only complete cross-sectional shape of PLA fibers after bonding shows in this paper. Results of fiber diameter, pore diameter, and porosity of aligned scaffolds are shown in Table 3. With the increasing of PU content in manufacturing PLA-aligned scaffolds under the same PLA fibers of 1000, the pore diameter, porosity, and visible pores between fibers present a decreasing tendency.
(a) Pore diameter (N = 10), (b) porosity (N = 3), and cross-sectional SEM of aligned scaffolds prepared at PU content (c) 5%, (d) 10%, (e) 20%, (f) 25%, and (g) 30%. And the change of (h) thickness and (i) weight, (N = 5). Results of fiber diameter, pore diameter, and porosity of aligned scaffolds.
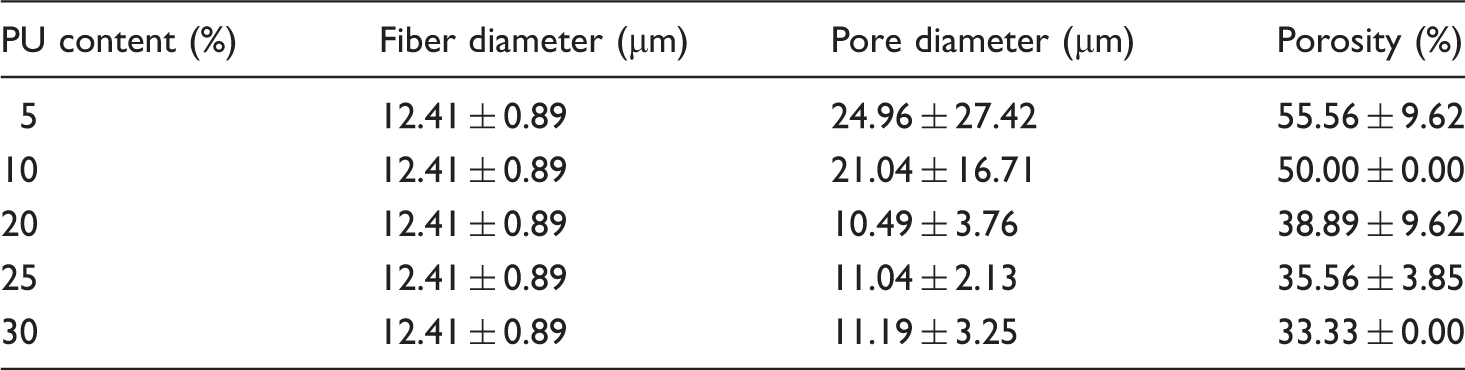
The circular cross-sectional shape of PLA fibers after bonding still maintains their original appearance, while it is difficult to observe smooth surface and single parallel arrangement. There seems to be that the PLA fiber lies in the central place and PU adhesive distributes surrounding the fiber to form a porous and composite structure. Combined with surface (Figure 2(a) to (f)) and cross-sectional morphology (Figure 4(c) to (g)) of aligned scaffolds, it is easy to see that the pore interconnectivity between PLA fibers and PU adhesive decreases as the adhesive content is increasing from 5 to 30%. PU adhesive is easy to enter into the pores between fibers and embed in the porous network in preparing composites. The reason is similar to Guo's research [73] that PEG was agglomerated in the SiO2 matrix network during the formation of the composite.
Thickness and weight
Effect of PU content on thickness and weight of aligned scaffolds shown in Figure 4(h) and (i) that the mean thickness increases from 0.05 to 0.14 cm, and mean weight increases from 0.04 to 0.071 g/cm2 as PU content increasing from 5 to 30%. Zero percent PU content in Figure 4(h) and (i) is the PLA fibers before bonding with no PU adhesive in scaffolds. It is easy to understand the more adhesive used to bond PLA fibers, the larger thickness and weight of adhesive forming on unit area of fibers. Weight of scaffold prepared at 5% PU bonding has no obvious linear increases because of inhomogeneous distribution of adhesive in scaffold, meaning the difference between test specimens exists in the same scaffold.
Contact angle
Results of contact angle.

Water absorption and gelatin impregnation
Water absorption (Figure 5(a)) of PLA melt fiber-aligned scaffolds increases sharply at first 0–1 h, then the value keeps a smaller increase at 1–2 h in comparing with values at first hour. After that, the water absorption has a relatively smaller change in a straight line basically parallel to the horizontal axis as time is increasing from 2 to 6 h. Due to larger porosity in scaffolds, the water absorption increases sharply at the beginning of 0–2 h time, then it is hard to absorb more water in saturated state as time is increasing. It can be found that scaffolds prepared with 5% PU content have larger water absorption than other scaffolds as a result of larger pore diameter and porosity. The values of water absorption at 1 h are similar to 100–150 and 250–300% of two PLA membranes [60].
Mean value of water adsorption (a) and gelatin impregnation (b) of aligned scaffolds (N = 5). Stress–strain curve of aligned scaffolds at length direction (c) and width direction (d).
Experimental volume of aligned scaffolds.

Tensile property
Stress and strain results of aligned scaffolds.

It can be explained that the stress of stretching PLA fibers in length direction is larger than width direction. With increasing of adhesive in composites, the interface bonding [76] between fiber and adhesive is closer, implying it is difficult to stretch scaffolds to fracture at such a tight structure. The result is also consistent with Wu's study that the stress of particles/matrix interface increases with a rise of filler content in composites [77]. The ultimate stress and strain of scaffolds prepared with 5–30% PU content and 1000 PLA fibers are 3.47 MPa and 1.89% along length direction, and 1.02 MPa and 25.69% in width direction.
Discussion
The purpose of this study was to prepare PLA/PU composite and porous-aligned scaffolds by 1000 PLA melt fibers with parallel arrangement and different PU contents at 5, 10, 20, 25, and 30% separately. As PU content increases from 5 to 30%, the bonding effect between fiber and adhesive, smooth surface, closely arrangement structure, thickness and weight, contact angle, and stress of aligned scaffolds shows an increasing trend. While the pore diameter, porosity, pore interconnectivity, water absorption, and gelatin impregnation of aligned scaffolds present a decreasing trend. The thermal stability shows a decreasing trend and PU adhesive content almost has no impact on alignment degree of PLA fibers in scaffolds.
The concept of aligned scaffolds was presented for the first time in 1991 [78], Rizvi et al. used silicone tubes filled with longitudinally aligned, small diameter collagen fibers to bridge a 10 mm gap created by transection of rat sciatic nerve. Fibroblasts and Schwann cells can migrate over aligned collagen fibers and improve tissue regeneration. The actin microfilaments were found to be distributed along the inner surface of the entire collagen gel cell membrane in either spherical shape at an initial stage of culture or elongated shape at a later stage [79]. Fibers mentioned in previous articles are directly used to prepare porous scaffolds in 2003. PCL fibers were first extruded and embedded in transparent, crosslinked poly(2-hydroxyethyl methacrylate) (pHEMA) gels, then the longitudinally oriented, fiber-free channels in the pHEMA gel were formed after process of sonicating pHEMA/PCL in acetone and completely dissolving PCL in composite [80]. Polypropylene microfibers were made by successive processes of heat drawing, fiber arraying by frame, and coating on one side by 25 mm thick adhesive layer [81]. Then Xu et al. [42] adopted electrospinning technology with rotating disk collector to prepare aligned nanofibrous scaffolds in 2004. It can be found that the porous structure of composite-aligned scaffolds of our study is similar to researches of Flynn et al. [80] and Neumann et al. [81], while they have different preparation methods. The pore diameter of PLA melt fiber composite-aligned scaffolds in our work is smaller than Neumann's paper [81], but more number of fibers and more stable structure.
Our work focuses on controlling fiber parallel arrangement in scaffolds and regulating pore diameter and porosity for cell growth under the condition of ideal pore structure. To achieve better parallel and orderly arrangement of fibers, PLA melt fibers were first winded under the condition of 100 r/min rotating speed and 100 cN tension. Then the brush was fully immersed in five different PU adhesives with 5, 10, 20, 25, and 30% content and gotten out for brushing lightly on the surface of PLA fibers, to prepare five scaffolds with different PU contents. After drying at 40℃ for 1 h, wet-aligned scaffolds were cured to achieve full drying and better bonding effect for use. The results indicate that three key steps including fiber parallel arrangement, bonding, and drying are important parameters for preparing aligned scaffolds by this method. Except using PLA melt fiber and PU adhesive in our experiment, other biodegradable and biocompatible fibers and adhesives can also be applied to prepare composite and porous-aligned scaffolds on the basis of this method for future research. Aside from melt spinning fibers, the dry spinning fibers, wet spinning fibers, and dry–wet spinning fibers with better biodegradable and biocompatible properties can also be applied to prepare aligned scaffolds. It can mix six different fibers with different proportions in one scaffold by winding process. In addition, the category and content of adhesive, and temperature, time, and pressure of drying process are significant parameters that cannot be ignored in manufacturing aligned scaffolds. It is necessary to control appropriate temperature for drying wet scaffolds without causing fiber shrinkage and deformation in the process of curing.
Conclusions
Five different PLA fiber/PU adhesive composite-aligned scaffolds were successively prepared by PLA melt fibers, PU adhesive with different contents and drying. The preparation of composite-aligned scaffolds in our work is different from previous literatures, but they have similar pore structure and fiber parallel arrangement. It can be found that each composite-aligned scaffold has better fiber parallel arrangement, pore diameter, porosity, and pore interconnectivity. The PU content has different influences on bonding effect between fiber and adhesive, alignment degree, surface and cross-sectional morphology, thickness, weight, contact angle, pore diameter, porosity, pore interconnectivity, water absorption, gelatin impregnation, stress and strain of aligned scaffolds. This preparation method for producing aligned scaffolds is simple, easy to operate, secure, environmental protection with no pollution, reproducible with better fiber parallel arrangement, and higher production efficiency for large-scale application. The textile fibers and some medical adhesives with better biodegradable and biocompatible properties can also be applied to prepare composite-aligned scaffolds by this method.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project is financially supported by China Postdoctoral Science Foundation (No. 2014M560481).
