Abstract
Novel flame-retardant back coating layer for historic textile fabrics was developed. Silica nanoparticles originated from agriculture waste rice husk were prepared through one pot thermal method. The morphological and structure properties of nanoparticles were studied. The silica nanoparticles were further impregnated with organic borate producing flame-retardant composite. The obtained composite incorporated with the binder by mechanical mixing providing flame-retardant coating paste. The coating paste spread on the back surface of textile fabrics. Varied compositions of nanoparticles, binder and organic borate were studied in the back coating layer. The flammability, thermal stability and mechanical properties of the blank and treated samples of linen fabrics as an inner support to the historical textiles were investigated. Flame retardancy of the back-coated linen samples has improved achieved high class of flame-retardant textile fabrics of zero rate of burning compared to 80.3 mm/min for blank. The synergistic effect of flame retardancy between nanoparticles and organic borate was investigated. The tensile strength of the flame retardant fabrics was enhanced by 27% and elongation was improved. The effect of industrial aging on the flame retardancy and mechanical properties of flame-retardant back coating textiles was studied.
Introduction
Textile materials constitute the majority of various market products due to their unique properties. However, the high combustibility of the material used in textile fabrics and fibers made them highly flammable and thermally less stable [1,2]. This defect will threaten the human life of textile consumers and reduce the lifetime display of historical textiles. Therefore, flame-retardant materials should be added to textile fabrics to reduce their flammability properties. Treatment of textile fabrics with these materials can be through different ways such as impregnation [3], back coating [4] and layer by layer self-assembly [5].Various materials have been used as flame-retardant to textile fabrics [2,6]. Recently, various nanomaterials have been used as flame retardants for textile materials [7,8]. TiO2 and ZnO nanoparticles have been used extensively in flame-retardant treatment of textile fabrics [3,9]. On the other hand, the historical importance of archaeological textiles in museums force the scientists to find a solution to various challenges, such as high combustibility, ease of ignition, light, relative humidity and temperature effects [10,11]. The protection of archaeological textiles against fire hazard is required to save the historical value of these rare types of textile fabrics. Interestingly, reducing the fire risk of archaeological textiles presents a preventive conservation way of display and storage of these historical materials. Linen fabrics are one of textiles used extensively in historic conservation, due to their unique properties, which used to support the historic textiles in restoration process. The restoration process is performed by fixing of historic textiles with linen fabric which used as a lining layer to historic textile in museum cabinet. In our previous studies, the flammability properties of different textile fabrics have been studied by the treatment of flame retardant as back coating to textiles [4]. Recently, we have investigated the flame retardancy, electrical conductivity and mechanical properties of cotton blend [12]. This is in addition to the flammability properties of polymer nanocomposites [13]. Our group has also been involved in the study of various nanomaterials [14–16]. Rice husk is considered as agricultural waste products. The abundance, low price and high yield of silica in rice husk encourage the scientists to produce cost-effective silica particles from rice husk for various applications [17]. Rice husk silica nanoparticles have been used in various applications [18]. This is in conjunction to the use of silica particles as flame-retardant fillers for polymeric materials [19]. On the other hand, borate materials have been used as flame-retardant materials for polymer composites [20]. In this study, we report for the first time the use of silica nanoparticles prepared from waste agriculture products such as rice husk (RH-SNP) along with organic borate in flame-retardant back coating formulations. Then the obtained composite used as back coating flame retardant layer to linen fabrics which used as an inner layer and support to the historical textiles. Considering it as first line of defense against fire hazard and provide strategy for historic textiles conservation. The effect of RH-SNP alone and in conjunction with organic borate on the flammability, thermal stability and mechanical properties of the treated samples was studied.
Experimental section
Materials
Linen fabrics were supplied by Egylan Co., second industrial zone, Alexandria, Egypt. Boric acid and chloroform were purchased from Sigma Aldrich, Germany. Diethylmalonate was supplied from Merck, Germany. Rice husk was bought from the market. The commercial binder used in the preparation of back coating paste was purchased from the market. Deionized (DI) was used water for washing.
Synthesis of diborate malonate
In a round-bottomed flask, add 1 mol of diethyl malonate with 2 mol of boric acid. Then, reflux at 120℃ for 2 h to obtain diborate malonate (DBM).
Synthesis of rice husk Silica nanoparticles (RH-SNP)
RH-SNP preparation was developed based on our previous report [21]. For synthesis, in clean crucible rice husk was heated in oven at 700℃ for 3 h at heating rate 20℃/min. The obtained silica was washed with DI water several times, followed by drying at 150℃. Afterwards, the silica particles were grinded using mortar for 1 h and finally sieved to obtain RH-SNP.
Preparation of back coating formulations
Flame-retardant back coating formulation compositions.

Thermal aging
The treated modern textile samples BC7 and BC8 (Table 1) were applied to industrial aging process. The samples were heated in an oven at 110℃ for 72 h which equal 25 years under normal temperature. [22]. Then, the flammability and mechanical properties were tested again.
Characterization
Fourier transform infrared (FT-IR) spectroscopy analysis was carried out using a Nicolet 380 spectrophotometer (Thermo Scientific). Thermogravimeteric analysis (TGA) was conducted using TGA 50 (TA Shimudzu, Inc.) from room temperature to 750℃ at a heating rate of 10℃ min−1 under nitrogen. Transmission electron microscope (TEM) images were taken using JEOL (JEM-1400 TEM). The mechanical properties were evaluated using tensile testing machine model H1-5KT/S. The flame retardancy measurements of the blank and different treated samples (rate of burning) were conducted using a Fire Testing Technology UL94 flame chamber according to modified ISO 3795 [4,12,23]. The rate of burning B in mm/min was calculated based on the following equation B = s/t × 60 where s is the damage distance (burnt distance) in millimeters and t is the time in seconds consumed for the burned distance.
Results and discussion
Structural characterization of diborate malonate
The DBM used in the synthesis of back coating flame-retardant formulations was synthesized and characterized. The structure of diborate malonate (DBM) was confirmed by FT-IR spectroscopy. Figure 1(a) shows the characteristic absorption band of boric acid, where the absorption peak was at 1406 cm−1 corresponds to asymmetric B–O stretching [24,25]. Furthermore, the peak situated at 1185 cm−1 is assigned to in plane B–O–H bending [24,26,27]. Moreover, the absorption band observed at 541 cm−1 was assigned to the in-plane O–B–O angle deformation mode [24,27]. The absorption band located at 642 cm−1 is related to out-of-plane OH deformation mode [27]. In addition, the bands observed at 822 and 3396 cm−1 correspond to out-of-plane BO3 angle deformation mode and O–H stretching vibration, respectively [24,25,27]. Figure 1(b) depicts the absorption bands of DBM where the strong peak observed at 1737 cm−1 corresponds to the C=O stretching and band situated at 2984 cm−1 is attributed to C–H stretching. Furthermore, the band detected at 3467 cm−1 corresponds to O–H stretching. This is in addition to the absorption band related to borate.
FTIR spectra of boric acid (a) and diborate malonate (b).
Structural characterization of rice husk silica nanoparticles (RH-SNP)
On the other hand, the RH-SNP used in the back coating layer was prepared through one-step thermal method. The prepared RH-SNP was further characterized using FT-IR spectroscopy, thermal gravimetric analysis and microscopic techniques. Figure 2(a) exhibit the characteristic absorption peaks of silica nanoparticle originated from rice husk (RH-SNP). The absorption band situated at 801 cm−1 corresponds to the bending vibration mode of the Si–O group and the peak observed at 464 cm−1 related to the rocking mode of the Si–O group. The strong absorption peak located at 1092 cm−1 is attributed to the asymmetrical stretching mode of the Si–O–Si group [28]. Furthermore, the absorption band found at 1635 cm−1 corresponds to the twisting vibration mode of the H–O–H group [28]. This is in addition to the absorption band observed at 3444 cm−1 which corresponds to stretching vibration mode of O–H [28]. The morphology of RH-SNP was also investigated. Figure 2(b) shows the TEM image of RH-SNP which reflects the average nanoparticle size of 150 nm that exists in aggregated form. Interestingly, the nanoparticle size of RH-SNP was confirmed through dispersion of the aggregated one by ultrasonication for 2 min as seen in the TEM image in Figure 2(c). However, The TGA graph of RH-SNP (Figure 2d) represents high thermal stability of RH-SNP with high purity of SNP up to 95%. The first mass loss below 400℃ could be due to the trapped moisture and some of remaining organic species. The second one in the temperature range 400–550℃ was due to the decomposition of carboneaous impurities adsorbed on RH-SNP surface after preparation. This confirmed the high purity of RH-SNP (95%) as shown in Figure 2(d).
FTIR spectra of RH-SNP (a), TEM images of RH-SNP (b), dispersed RH-SNP (c) and TGA thermogram of RH-SNP (d).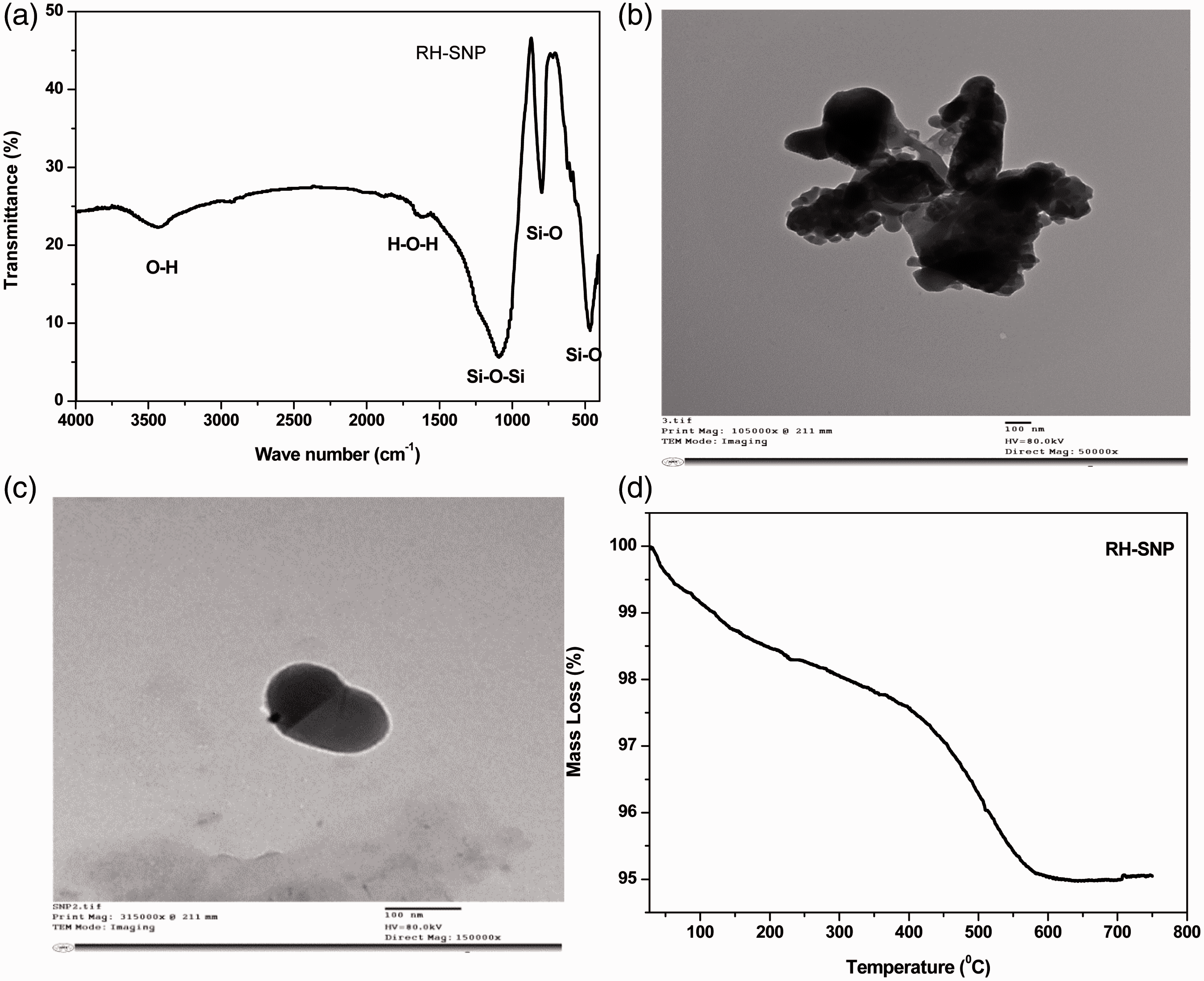
Preparation and characterization of flame retardant back coating textile fabrics
The back coating formulations based on RH-SNP and DBM were prepared through impregnation the RH-SNP with DBM (RH-SNP-DBM) followed by mixing with binder forming coating paste (RH-SNP-DBM-binder). Then, the paste was coated on the back surface of the linen fabrics as shown in Figure 3. The compositions of the back coating formulations were varied as tabulated in Table 1.
Schematic diagram representing the preparation of back coating flame retardant and coating on textile fabrics.
Thermal properties
The thermal stability of the blank and treated samples was analyzed using thermogravimeteric analysis. Figure 4(a) exhibits the thermogram of linen fabrics which decomposes in two steps. The first mass loss was below 100℃ which attributed to the trapped moisture. The second mass loss in the temperature range 300–500℃ corresponds to the decomposition of linen leaving char yield to 13.7%. Figure 4(b) shows the thermogram of BC4 which composed from linen treated with back coating layer (Table 1). The thermogram depicts three decomposition steps; the first one due to the trapped moisture which is below 100℃ and second decomposition step at 175℃ which is attributed to the decomposition of DBM. The third decomposition step which starts at 300℃ (onset decomposition temperature of linen) gives rise to char yield of 26%. Interestingly, the earlier mass loss of treated textiles than virgin one might be due to the decomposition of DBM and binder. It is important to note that, all the treated samples have the same thermal behavior (Figure 4) with similar mass loss steps. However, the char residue of the treated fabrics varied based on the mass of RH-SNP in the back coating layer. The char residue of BC7 sample reached up to 42.6% when the mass of RH-SNP was 50% of the back coating layer mass (Table 1 and Figure 4). This char layer retarded the further degradation of linen fabrics compared to blank one. It is important to note that, the samples used for thermal stability analysis were selected to reflect the effect of regular increase of RH-SNP mass in back coating layer on thermal stability behavior (Table 1). The morphology of linen fibers was investigated after back coating treatment. Figure 5(a) represents the microscopic image of cross section of linen yarn showing normal shape of untreated linen yarn. Figure 5(b) shows the image of cross section of treated linen yarn of BC7 which reflect little swelling after treatment. This might be due to the inclusion of the coating material with specially impregnated silica nanoparticles with organic borate (RH-SNP-DBM) inside the linen yarn.
TGA graphs of blank (a), BC4 (b), BC5 (c), BC6 (d) and BC7 (e). Microscopic image of cross section of untreated linen yarn (a) and treated yarn of BC7 (b).
Flammability and mechanical properties
The rate of burning of different textile samples.
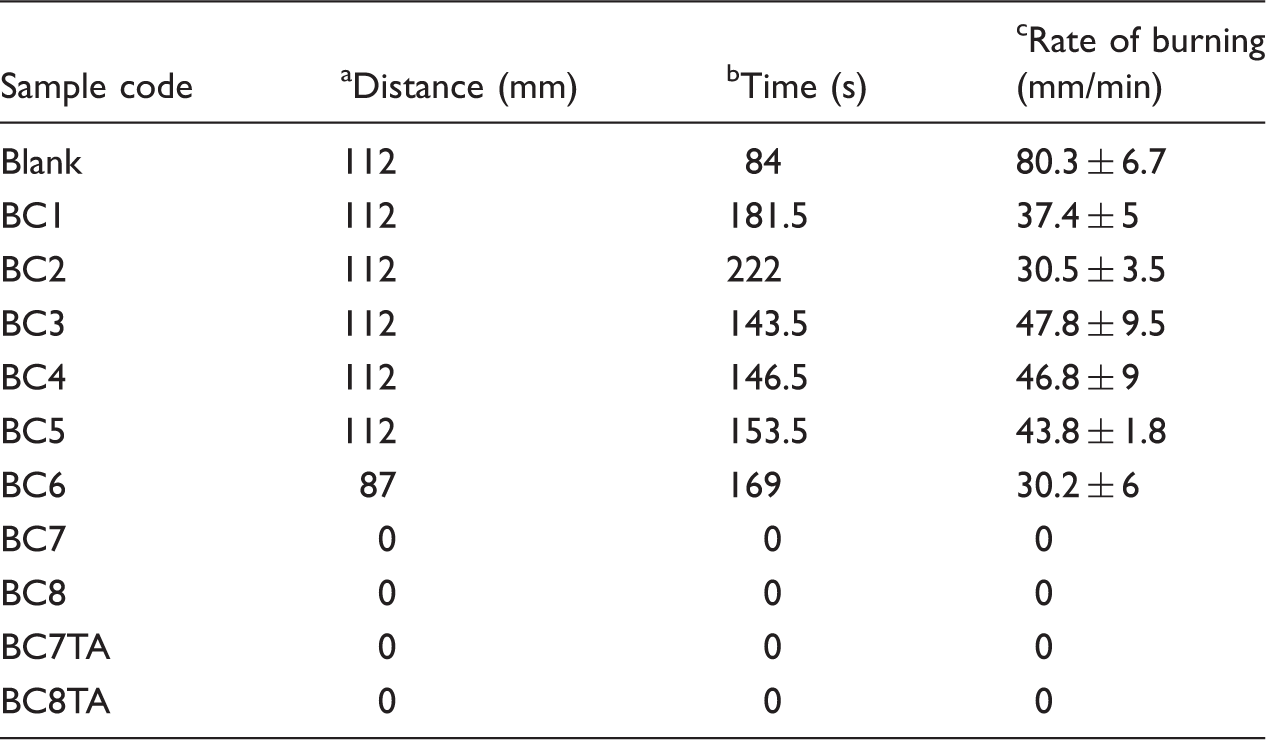
Average distance burned.
Average time used for burning.
Average rate of burning.

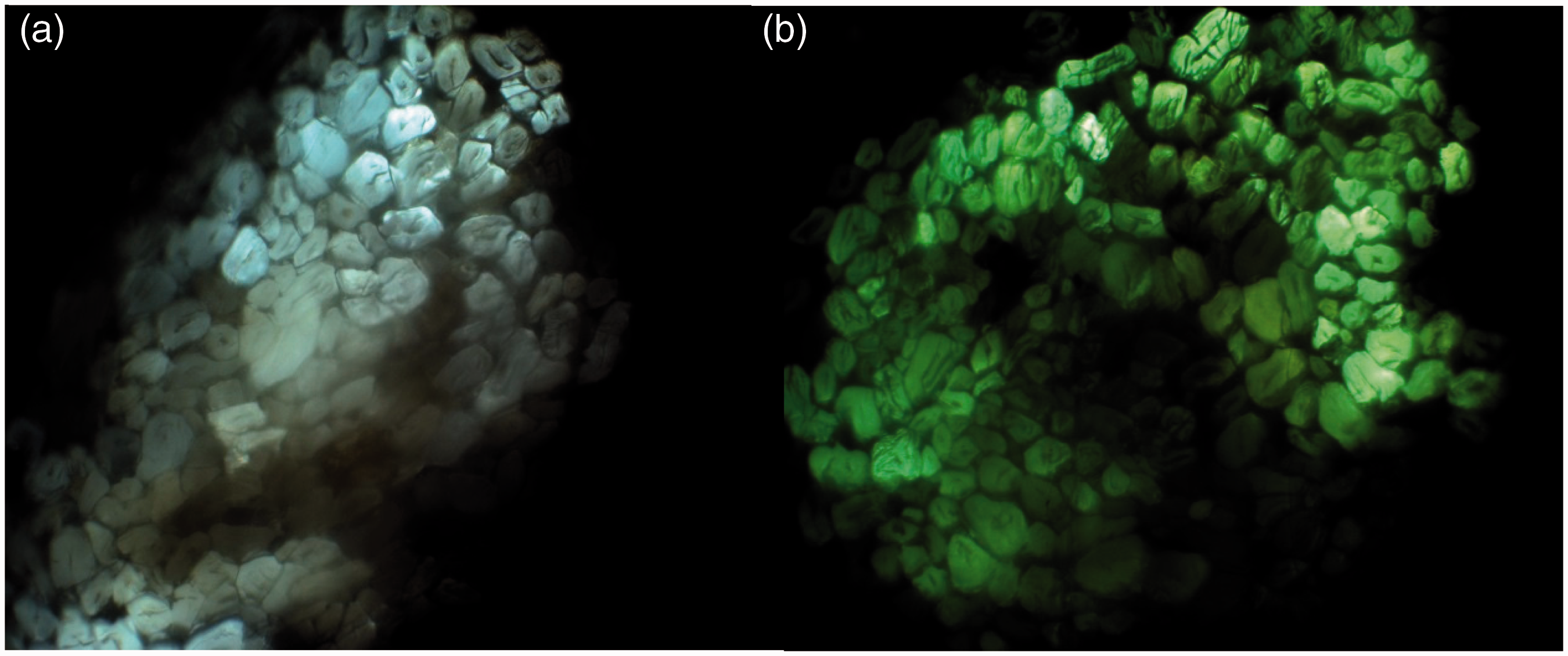
Digital photos of upper and back surface of BC7 (a, b) and BC8 (c, d) indicated the char layer formed.
So, the effective treated linen fabrics will significantly reduce the fire risk for historic textiles and prolong the display life time of these textiles in museum.
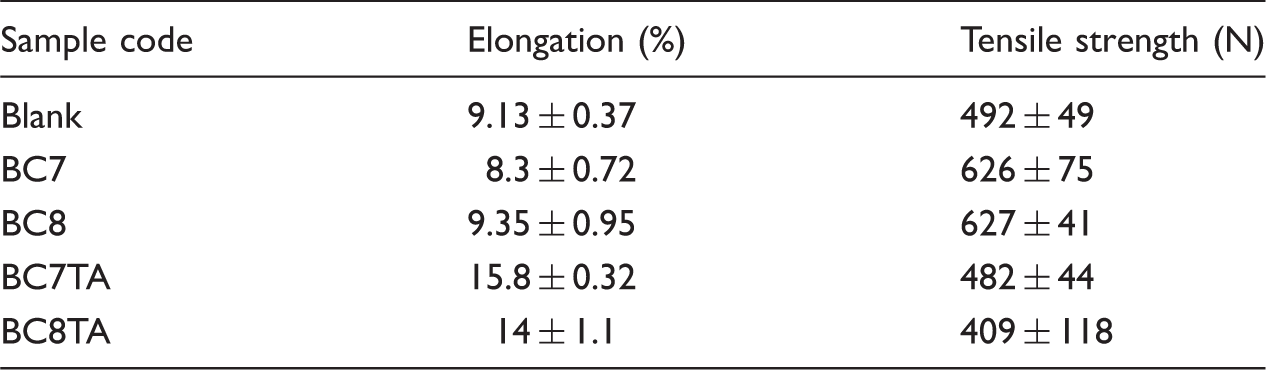
Mechanical Properties of blank, BC7, BC8, BC7TA and BC8TA.

Stress–strain curves of blank linen (a), BC7 (b), BC8(c), BC7TA (d) and BC8TA (e).
Conclusion
A smart and effective flame-retardant coating layer based on nanoparticles was developed. The coating layer was treated as back layer to the linen fabrics as an inner layer of historical textiles. Rice husk silica nanoparticles were prepared and incorporated in the back coating layer. The silica nanoparticles have been used alone or impregnated with organic borate in conjunction with binder in the back coating layer. Various compositions of flame retardant coating were studied. The rate of burning was significantly reduced and recorded zero for back-coated samples compared to 80.3 mm/min for blank. The flame retardancy and synergistic effect were investigated. Tensile strength and elongation were improved for treated samples compared to the blank one. Industrial thermal aging for the flame-retardant textile samples was studied. The flame retardancy properties were unaffected and elongation significantly improved after thermal aging.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
